Abstract
Autoinflammatory diseases are a genetically heterogeneous group of rheumatologic diseases that are driven by abnormal activation of the innate immune system. Patients present with recurrent episodes of systemic inflammation and a spectrum of organ-specific comorbidities. These diseases are mediated by the overproduction of various inflammatory cytokines, such as IL-1, IL-18, IL-6, TNFα, and type I interferon. Treatments with biologic agents that inhibit these cytokines have been very efficient in most patients. During the past 2 years, remarkable progress has been made in the identification of disease-associated genes owing mostly to new technologies. Next generation sequencing technologies (NGS) have become instrumental in finding single-gene defects in undiagnosed patients with early onset symptoms. NGS has advanced the field of autoinflammation by identifying disease-causing genes that point to pathways not known to regulate cytokine signaling or inflammation. They include a protein that has a role in differentiation of myeloid cells, a ubiquitously expressed enzyme that catalyzes the addition of the CCA terminus to the 3-prime end of tRNA precursors, and an enzyme that catalyzes the oxidation of a broad range of substrates. Lastly, newly described mutations have informed a whole new dimension on genotype-phenotype relationships. Mutations in the same gene can give rise to a range of phenotypes with a common inflammatory component. This suggests greater than anticipated contributions by modifying alleles and environmental factors to disease expressivity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Whole exome sequencing (WES) is at the height of discovery of disease-associated genes in patients with undiagnosed Mendelian genetic conditions in all fields of medicine. In the past, linkage-based genetic studies coupled with homozygosity mapping in consanguineous families, have enabled the molecular dissection of a number of human inherited disorders. At present time, sporadic patients and their unaffected parents (trios) or a group of unrelated patients with phenotypic similarities are subjected to WES in search of disease-associated genes. Discovery of causal gene in a single sporadic patient can pave the way for its confirmation in other patients with similar genetic condition or can inform targeted sequencing of candidate genes for polygenic diseases. Thus, genetic defects found in rare patients might facilitate studying more common polygenic human diseases by identifying subsets of patients with rare highly penetrant variants in multiple genetic loci. Studies of functional abnormalities linked to mutated proteins will help to better understand the molecular mechanisms of human health and disease and will ultimately provide more tailored therapeutic approaches [1].
March 2014: deficiency of ADA2 (DADA2)
Genetic studies
CECR1 disease-associated mutations were discovered by two independent groups that utilized whole exome sequencing to search for a causal gene in patients diagnosed with systemic inflammation and vasculopathy and/or necrotizing vasculitis Polyarteritis Nodosa (PAN) (Table 1). In one study, WES was performed on three unrelated patients from the outbred population and their unaffected parents [2]. Early onset symptoms of systemic inflammation and recurring stroke suggested genetic defect in a single gene. Assuming recessively inherited disease, the three patients shared novel and/or rare deleterious mutations in a single gene, CECR1. The second study benefited from the observation of patients with childhood-onset systemic and/or cutaneous PAN in the founder Georgian-Jewish and Turkish populations [3]. Homozygosity mapping coupled with exome sequencing led to the identification of a founder mutation, p.G47R, in the same gene CECR1. The allele frequency of p.G47R mutation is relatively high in Middle Eastern populations, while this variant is unreported in the European population. This variant has been recently identified in two affected siblings of Pakistani descent (unpublished data). Subsequent report has described another case with a fatal vasculopathy [4]. The p.R169Q mutation appears to be a founder mutation in the Dutch population [5, 6]. Patients with early onset recurrent stroke, neurologic manifestations and fever typically carry compound heterozygous mutations in CECR1. Structural variants and non-coding variants, i.e, splicing mutations should be considered in molecular diagnostics of patients with suspected ADA2 deficiency [2]. CECR1 is a polymorphic gene and the clinical significance of novel missense variants should be interpreted with great caution and in the context of family ancestry. Discovery of CECR1 diseases-associated mutations informed the screening of patients with Sneddon’s syndrome, a late-onset disease with similar clinical features [7]. CECR1 mutations might be relevant to a broader group of pathologies including the HHV-8-negative Castelman’s disease and complex forms of lacunar stroke and vasculitis [8].
Pathogenesis of STING-associated vasculopathy with onset in infancy (SAVI) and deficiency of ADA2 (DADA2). In both diseases, the endothelial cells of small vessels are affected. Pathological findings are particularly apparent in skin vessels and patients present with rashes and skin ulcerations. In addition, SAVI patients present with pulmonar features while DADA2 patients have neurological symptoms caused by lacunar strokes that result from occlusions in the deep brain. In SAVI, endothelial cell phenotype is mediated by the intracellular production of IFN-induced cytokines. In DADA2, endothelial cell activation and damage is caused by overproduction of pro-inflammatory cytokines that are produced by myeloid cells
Pathogenesis of DADA2
The CECR1 (cat eye syndrome chromosome region, candidate 1) gene encodes the adenosine deaminase 2 (ADA2) protein. ADA2 has partial homology to ADA1 protein. ADA1 and ADA2 function as intracellular enzymes to regulate the purinergic signaling pathway. The catalytic activity of ADA2 is significantly lower due to large differences in the arrangement of the substrate-binding pockets [9]. Inherited mutations in ADA1 result in severe combined immunodeficiency disease (SCID) caused by a defect in T and B-lymphocytes. In contrast, patients with mutations in ADA2 have only mild hypogammaglobulinemia due to a defect in terminal differentiation of B-cells [2]. The underlying cause of the immunological abnormalities in the B cells is not yet known.
Crystal structure of human ADA2 has revealed the presence of two adenosine deaminase growth factors (ADGF)-specific domains that mediate the protein dimerization and binding to the cell surface receptors [9]. Evidence that ADA2 functions as a growth factor comes from model studies in frog embryos and in Drosophila [10, 11]. CECR1/ADA2 homologues have not been found in mice, but they are present in many other organisms including Zebrafish. Transient knockdown with inhibitory antisense constructs (morpholinos) of the cecr1a paralog gene in Zebrafish causes intracranial bleeding in developing embryos [2]. Morphology of blood vessels in affected embryos is grossly intact, indicating that the cause is likely related to development of endothelial cells. The disruption of cecr1a expression in mpx-GFP transgenic fish causes a marked reduction in neutrophil numbers. ADA2 is known to induce T-cell-dependent differentiation of monocytes into macrophages [12]. These observations suggest a role for ADA2 in differentiation of endothelial and myeloid cells.
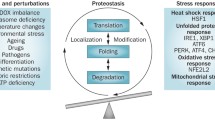
CECR1 disease-associated mutations are predicted to have deleterious effect on the protein stability, activity, and dimerization. The functional consequences of these mutations were confirmed in multiple ways: reduced expression of mutant ADA2 in cell lysates and supernatants of transfected cells, reduced ADA2 activity in patients’ blood and patients’ monocytes, and by biophysical analysis of mutant protein [2, 3]. The biochemical assay for measuring the ADA2 activity in blood can help establish the pathogenicity of novel CECR1 mutations. Lesional biopsies from affected skin and brain tissues display endothelial cell damage and activation along with substantial inflammatory processes. The inflammatory component of ADA2 deficiency is linked to a defect in macrophage differentiation (Fig.1, Table 2). A high ratio of M1 (pro-inflammatory) vs. M2 (anti-inflammatory) macrophages may cause an overproduction of proinflammatory cytokines that inflict damage to blood vessels [2]. Activated neutrophils may also contribute to the inflammatory process [13]. Deficiency of ADA2 protein can be replenished with plasma transfusions or recombinant ADA2 protein, pending its kinetics properties. HSCT transplantation or gene therapy may offer an alternative treatment option for severe cases [5, 6, 14].
August 2014: STING-associated vasculopathy with onset in infancy (SAVI)
Genetic studies
Whole exome sequencing of a single patient and her unaffected parents led to the identification of a de novo mutation, p.N154S, in the TMEM173 gene, which encodes the stimulator of interferon genes (STING) (Table 1) [15]. The index patient presented early onset symptoms of systemic inflammation, cutaneous rash, and pulmonary manifestations. Candidate gene screening in patients with similar features identified mutations in five other sporadic cases. Altogether, three de novo deleterious missense mutations, (p.V147L, p.N154S, p.V155M,) were described in six patients with severe vasculopathy. Subsequent study showed that the p.V155M mutation has highly variable expression and that it can be associated with a phenotypically different disease reminiscent of systemic lupus erythematosus [16]. One mutation, p.V147L, was suspected to be mosaic with a variable prevalence in different cell types [15]. Pathogenic variants are clustered in exon 5 of TMEM173, and they reside close to the STING dimerization site. These mutations likely reinforce the stability of the STING dimmer and thus may mimic the effect of ligand binding. The patients reported in the first study are of diverse ancestry, which is consistent with de novo origin of their causal variants. The second study highlights that in families with dominantly inherited traits, the possibility of reduced penetrance should not be ignored. Germline and somatic mutations in TMEM173 might be explored in other types of late onset idiopathic cutaneous vasculopathies and SLE-like phenotypes.
Pathogenesis of SAVI
STING (also known as MITA, MYPS, ERIS) is a type I IFN stimulator that is essential for host defense against viral and bacterial infection [17]. TMEM173 is ubiquitously expressed with high presence in myeloid, natural killer and T cells, endothelial cells, alveolar pneumocytes and macrophages, and bronchial epithelium. STING is a key signaling molecule in the innate immune response to cytosolic double stranded-DNA (dsDNA) [18, 19] STING binds to dsDNA either directly or through second messengers referred to as CDNs (cyclic dinucleotides) that are produced in response to pathogen-associated DNA in the cytosol [17]. Activation of STING, by means of TBK1 and IRF-3 phosphorylation, induces expression of type I interferons that are essential for successful pathogen elimination. Sting-deficient mice develop normally but are extremely susceptible to viral infections as they lack the ability to upregulate Ifn-beta [20]. Recent study has shown that interaction of the NLR and STING pathways is important for fine-tuning host responses to intracellular DNAs [21].
The overexpression of TMEM173 missense mutations in HEK293T cells linked disease-associated mutations to increased IFNB1 activity [15, 16]. Patients’ cells show constitutive, i.e., ligand-independent activation of the STING-IFNβ pathway. This has been demonstrated by the strong interferon response-gene expression signature in patients’ cells, elevated blood levels of interferon-induced cytokines, and the constitutive phosphorylation of STAT1 in mutant cells [15]. All together, these data provide convincing evidence that TMEM173-associated mutations are gain of function [15, 16]. Patients’ skin biopsy samples show widespread small-vessel vasculopathic changes, occlusions, and inflammation. In contrast to ADA2, STING is expressed in endothelial cells and bronchial epithelium; thus, the pathological changes in blood vessels and lung are due to intrinsic defects in these cells (Fig.1). Stimulation of human primary endothelial cells (HUVAC) with the STING ligand cGAMP resulted in increased expression of genes that mediate inflammation and apoptosis [15]. As a result, activated endothelial cells are more susceptible to apoptosis. Higher rates of spontaneous cell death were also observed in patients’ monocytes and T cells. Treatment of patients’ cells with JAK inhibitors suppressed phosphorylation of STAT1 and downregulated the transcription of IFN-regulated genes. This data suggest that the JAK inhibition should be explored in treatment of these patients [15].
September 2014: TNFRSF11A-associated hereditary fever disease
Genetic studies
TNFRSF11A (RANK, PDB, ODFR) is the signaling receptor with role in osteoclast differentiation and bone remodeling [22, 23]. RANK-ligand (RANKL) mediates essential signal for osteclastogenesis [24]. Mutations at this gene have been associated with familial expansile osteolysis, autosomal recessive osteopetrosis, and Paget disease of bone [25]. Although these patients are not known to have fever, study in mice and rats showed that RANK/RANKL have a key role in thermoregulation in rodents via the COX2-PGE [2] /EP3R pathway [26]. Recently, a de novo genomic duplication containing the TNFRSF11A gene was reported in a patient with complex phenotype including neonatal onset of systemic inflammation and congenital abnormalities [27]. The TNFRSF11A gene was noted as one of the 30 genes that reside in the 10-Mb genomic duplication. Candidate gene screening identified two additional patients (mother and daughter) with a novel heterozygous 1-bp deletion (p.Met416Cysfs*110) in exon 9 of TNFRSF11A (Table 1). Mutated protein lacks a C-terminal part of the intracellular domain. Disease symptoms are similar to patients with TNF receptor-associated periodic syndrome (TRAPS). None of the three patients presented with osteopetrosis or osteolysis.
Pathogenesis of disease
The protein encoded by the TNFRSF11A gene is a member of the TNF-receptor superfamily. The RANK receptor is known to induce the activation of NF-kappa B and MAPK8/JNK and to regulate the interaction between T cells and dendritic cells [28]. Rank-deficient mice have profound osteopetrosis resulting from a block in osteoclast differentiation, and they lack peripheral lymph nodes [29].
The pathogenesis of TNFRSF11A-associated disease is unclear (Table 2). The TNFRSF11A gene duplication suggests a gain of function in the pathway, while the heterozygous frameshift mutation is more consistent with a haploinsufficiency or a dominant negative effect. Expression of the mutant protein in patients’ leucocytes was normal. Transient expression of mutant TNFRSF11A (frameshit mutation) in HEK293T cells failed to activate NF-kB signaling suggesting a functional impairment of the mutant protein [27]. However, this experiment does not, by itself, prove a causal role in the disease pathogenesis.
September 2014: NLRC4-associated inflammatory diseases (SCAN4; NLRC4-MAS; NLRC4-FCAS)
Genetic studies
Gain of function mutations in the NLRC4 (IPAF; CARD12) gene have been associated with an autoinflammatory syndrome that can manifest either as a life-threatening disease suggestive of macrophage activation syndrome (MAS) or a milder phenotype resembling familial cold autoinflammatory syndrome (FCAS) [30–32] (Table 1). Exome sequencing was performed independently in two sporadic patients (trios) with early onset fever, failure to thrive, rash, joint pains, and elevated inflammatory markers, including hyperferritinemia. One patient presented with severe gastrointestinal inflammation [31]. Two novel causal mutations, p.T337S and p.V341A, were identified in NLRC4. The mutations are located in a highly conserved HD1 region of the NLRC4 nucleotide-binding domain (NBD), and they are predicted to disrupt the ability of NLRC4 to maintain itself in an auto-inhibited state. Another pathogenic variant in NLRC4, p.H443P, was identified in a Japanese family with milder symptoms including cold-induced rash, fever, and arthralgia. This mutation also resides in the NBD domain of NLRC4 but is associated with a phenotypically different illness. Similar pleiotropy has been described for NLRP3-associated diseases. The clinical variability of NLRP3- and NLRC4-associated diseases is partly related to the genotype. For example, some missense mutations are likely to maintain mutant proteins in the constitutively active state, while other variants require environmental triggers such as cold and stress to cause the inflammasome activation. Further studies are necessary to explore full spectrum of monogenic inflammasome-related diseases.
Pathogenesis of disease
The NLRC4 (IPAF, CARD12) inflammasome initiate inflammation in response to bacterial ligands (flagellin) [33, 34]. In contrast to NLRP3, NLRC4 is highly expressed in intestinal epithelial cells and macrophages where it functions to provide immune defense against Gram-negative bacteria including Salmonella [35–37]. The NIAP/NLRC4 epithelium-intrinsic inflammasome drives expulsion of infected cells to restrict the pathogen’s intraepithelial proliferation. The intestinal epithelial cells of the Nlrc4 −/− mice exhibit elevated Salmonella burdens at early stages of infection [35]. The activity of NLRC4 might be exacerbated early in life by newly acquired microflora, and as host-microbe symbiosis matures, the inflammasome activity declines. That may explain the symptoms of early onset enterocolitis in one family and resolution of GI symptoms in two surviving patients [31]. The mutant NLRC4 is constitutively active: spontaneous ASC oligomerization and caspase-1 cleavage was observed in cells transfected with mutant construct and in unstimulated patient’s cells [30, 31]. As expected, mutant myeloid cells produce high levels of casp-1 mediated cytokines IL-1β, and IL-18. Activated macrophages appear to be the main source of IL-18 [30]. Mutant macrophages are more susceptible to spontaneous cell death (pyroptosis). LPS-primed and infected patient’s macrophages showed more cell death despite impaired cytokine production [31]. This suggests that the balance of cytokine production vs. pyroptosis in mutant cells may influence the clinical variability of NLRC4-associated phenotypes (Table 2).
The Japanese group used a mouse model to corroborate on the pathogenicity of the single FCAS-associated mutation in their study [32]. Transgenic mice that express the mutant Nlrc4 developed dermatitis, swollen joints, bone erosions, and splenomegaly. Inflammatory symptoms were exacerbated with exposure to cold. The inflammatory phenotype induced by the mutant Nlrc4 protein is IL-1β and IL-17A dependent. Treatment with IL-1 and IL-17 inhibitors reduced the number of infiltrating neutrophil while the depletion of lymphocytes did not rescue the phenotype.
One patient with MAS-like presentation was treated with an IL-1 inhibitor and this normalized acute phase reactants, serum IL-1 levels but not serum IL-18 levels [30].
September 2014: TRNT1 deficiency
Genetic studies
Periodic fever syndrome manifesting with a variable phenotype of congenital sideroblastic anemia, B cell immunodeficiency, and developmental delay has been termed SIFD [38]. Symptoms occur during early infancy. Neurological symptoms and metabolic abnormalities are notable features in multiple cases, suggesting mitochondrial involvement. In the first clinical report, 7 out of 11 children died due to multi-organ or cardiac failure. Consanguinity was noted in three patients suggesting recessive disease inheritance. Bone marrow transplantation corrected hematological and immunological manifestations in one patient. The SIFD syndrome is caused by biallelic mutations in the TRNT1 gene [39, 40]. Combination of linkage analysis, consanguinity mapping, and exome sequencing led to identification of 10 missense, 3 frameshift, and 3 splice variants in this gene. Pathogenic variants are either unreported in public databases or very rare, which is consistent with recessive inheritance for rare diseases. Missense mutations affect evolutionarily conserved residues, and some are clustered in the active site of TRNT1 (Table 1).
Pathogenesis of disease
TRNT1 encodes the ubiquitously expressed CCA-adding enzyme that is essential for template-independent maturation of nuclear and mitochondrial transfer RNAs [41]. The complete deficiency of TRNT1 is most likely embryonic lethal given the unique function of this enzyme. Deficiency of the TRNT1 homologue in budding yeast showed that the disease-associated mutations result in partial loss of function of TRNT1 and have variable degrees of enzyme activity [39]. The enzyme deficiency in CCA1 deletion yeast strain was fully restored with wild-type human TRNT1 and partially rescued by human mutant proteins. Expression of mutant protein was variably reduced in patients’ fibroblasts. Knockdown o f TRNT1 in patients’ fibroblasts has been linked to impaired mitochondrial translation [40]. Molecular mechanism of sideroblastic anemia, inflammation, and immunodeficiency are still largely unknown (Table 2).
January 2015: monogenic form of systemic juvenile idiopathic arthritis
Genetic studies
Systemic-onset juvenile idiopathic arthritis (SoJIA) is an inflammatory disease of unknown cause [42]. Recent studies suggested that SoJIA might be linked to abnormalities in the innate immune system [43, 44]. SoJIA is typically considered a polygenic disease, and candidate-gene association studies reported many risk alleles. Combination of linkage analysis, homozygosity mapping, and WES led to identification of a homozygous founder missense mutation, p.C284R, in exon 4 of the LACC1 gene in 5 consanguineous families with 13 affected patients from the Saudi Arabia [45]. All patients presented early onset symptoms including characteristic quotidian fever, evanescent rash, and symmetrical polyarthritis. Nine patients had persistent systemic inflammation. Patients were treated with a combination of NSAID, steroids, and biologic therapies, but none of the children achieved complete remission. The causal variant affects evolutionarily conserved residue and is predicted deleterious. This mutation is unreported in public databases and is not found in over 2,000 Arab controls. Recessive disease inheritance suggests a loss of function mutation; however, this has not been proved by functional experiments (Table 1).
Pathogenesis of disease
Molecular function of the LACC1 (Laccase domain-containing 1) gene is largely unknown. It belongs to a family of Laccases, which are multi-copper oxidoreductases that catalyze the oxidation of a variety of phenolic and non-phenolic compounds. Laccases are present both in prokaryotes and eukaryotes, but there are no published model studies to shed light on the protein function. Allele variants in the LACC1 gene have been strongly associated with susceptibility to leprosy in multiple populations [46–48]. These data indirectly imply that the LACC1 gene might have a role in the regulation of the innate immune responses.
Conclusion
The emerging genetic technologies complemented by the development of public databases of human variation can allow discovery of disease causal genes in sporadic and unrelated patients. At present time, allele frequencies of suspected pathogenic variants can be interrogated in more than 60,000 exomes derived from multiple populations. Candidate variants should be always evaluated in the context of patient’s ancestry. Ex vivo studies in primary patients’ cells and model organisms corroborate genetic evidence for pathogenicity. Finally, collaborations may save time and money; having two pathogenic variants in a candidate gene is always more assuring than having a single mutation.
References
Ombrello MJ, Sikora KA, Kastner DL (2014) Genetics, genomics, and their relevance to pathology and therapy. Best Pract Res Clin Rheumatol 28:175–189
Zhou Q et al (2014) Early-onset stroke and vasculopathy associated with mutations in ADA2. N Engl J Med 370:911–920
Navon Elkan P et al (2014) Mutant adenosine deaminase 2 in a polyarteritis nodosa vasculopathy. N Engl J Med 370:921–931
Garg N et al (2014) Novel adenosine deaminase 2 mutations in a child with a fatal vasculopathy. Eur J Pediatr 173:827–830
van Montfrans J, Zavialov A, Zhou Q (2014) Mutant ADA2 in vasculopathies. N Engl J Med 371:478
Van Eyck L, Liston A, Meyts I (2014) Mutant ADA2 in vasculopathies. N Engl J Med 371:478–479
Bras J, Guerreiro R, Santo GC (2014) Mutant ADA2 in vasculopathies. N Engl J Med 371:478–480
Van Eyck L, Liston A, Wouters C (2014) Mutant ADA2 in vasculopathies. N Engl J Med 371:480
Zavialov AV, Yu X, Spillmann D, Lauvau G, Zavialov AV (2010) Structural basis for the growth factor activity of human adenosine deaminase ADA2. J Biol Chem 285:12367–12377
Iijima R et al (2008) The extracellular adenosine deaminase growth factor, ADGF/CECR1, plays a role in Xenopus embryogenesis via the adenosine/P1 receptor. J Biol Chem 283:2255–2264
Dolezal T, Dolezelova E, Zurovec M, Bryant PJ (2005) A role for adenosine deaminase in Drosophila larval development. PLoS Biol 3:e201
Zavialov AV et al (2010) Human adenosine deaminase 2 induces differentiation of monocytes into macrophages and stimulates proliferation of T helper cells and macrophages. J Leukoc Biol 88:279–290
Belot A et al (2014) Mutations in CECR1 associated with a neutrophil signature in peripheral blood. Pediatr Rheumatol Online J 12:44
Van Eyck L Jr et al (2015) Hematopoietic stem cell transplantation rescues the immunologic phenotype and prevents vasculopathy in patients with adenosine deaminase 2 deficiency. J Allergy Clin Immunol 135:283–287, e5
Liu Y et al (2014) Activated STING in a vascular and pulmonary syndrome. N Engl J Med 371:507–518
Jeremiah N et al (2014) Inherited STING-activating mutation underlies a familial inflammatory syndrome with lupus-like manifestations. J Clin Invest 124:5516–5520
Burdette DL et al (2011) STING is a direct innate immune sensor of cyclic di-GMP. Nature 478:515–518
Barber GN (2014) STING-dependent cytosolic DNA sensing pathways. Trends Immunol 35:88–93
Burdette DL, Vance RE (2013) STING and the innate immune response to nucleic acids in the cytosol. Nat Immunol 14:19–26
Ishikawa H, Barber GN (2008) STING is an endoplasmic reticulum adaptor that facilitates innate immune signalling. Nature 455:674–678
Zhang L et al (2014) NLRC3, a member of the NLR family of proteins, is a negative regulator of innate immune signaling induced by the DNA sensor STING. Immunity 40:329–341
Boyce BF, Xing L (2007) The RANKL/RANK/OPG pathway. Curr Osteoporos Rep 5:98–104
Walsh MC, Choi Y (2014) Biology of the RANKL-RANK-OPG system in immunity, bone, and beyond. Front Immunol 5:511
Soysa NS, Alles N, Aoki K, Ohya K (2012) Osteoclast formation and differentiation: an overview. J Med Dent Sci 59:65–74
Whyte MP (2006) Paget's disease of bone and genetic disorders of RANKL/OPG/RANK/NF-kappaB signaling. Ann N Y Acad Sci 1068:143–164
Hanada R et al (2009) Central control of fever and female body temperature by RANKL/RANK. Nature 462:505–509
Jeru I et al (2014) Brief Report: Involvement of TNFRSF11A molecular defects in autoinflammatory disorders. Arthritis Rheumatol 66:2621–2627
Anderson DM et al (1997) A homologue of the TNF receptor and its ligand enhance T-cell growth and dendritic-cell function. Nature 390:175–179
Dougall WC et al (1999) RANK is essential for osteoclast and lymph node development. Genes Dev 13:2412–2424
Canna SW et al (2014) An activating NLRC4 inflammasome mutation causes autoinflammation with recurrent macrophage activation syndrome. Nat Genet 46:1140–1146
Romberg N et al (2014) Mutation of NLRC4 causes a syndrome of enterocolitis and autoinflammation. Nat Genet 46:1135–1139
Kitamura A, Sasaki Y, Abe T, Kano H, Yasutomo K (2014) An inherited mutation in NLRC4 causes autoinflammation in human and mice. J Exp Med 211:2385–2396
Franchi L et al (2006) Cytosolic flagellin requires Ipaf for activation of caspase-1 and interleukin 1beta in salmonella-infected macrophages. Nat Immunol 7:576–582
Vance RE (2015) The NAIP/NLRC4 inflammasomes. Curr Opin Immunol 32C:84–89
Sellin ME et al (2014) Epithelium-intrinsic NAIP/NLRC4 inflammasome drives infected enterocyte expulsion to restrict Salmonella replication in the intestinal mucosa. Cell Host Microbe 16:237–248
Zhao Y et al (2011) The NLRC4 inflammasome receptors for bacterial flagellin and type III secretion apparatus. Nature 477:596–600
Nordlander S, Pott J, Maloy KJ (2014) NLRC4 expression in intestinal epithelial cells mediates protection against an enteric pathogen. Mucosal Immunol 7:775–785
Wiseman DH et al (2013) A novel syndrome of congenital sideroblastic anemia, B-cell immunodeficiency, periodic fevers, and developmental delay (SIFD). Blood 122:112–123
Chakraborty PK et al (2014) Mutations in TRNT1 cause congenital sideroblastic anemia with immunodeficiency, fevers, and developmental delay (SIFD). Blood 124:2867–2871
Sasarman F, et al (2015) The 3' addition of CCA to mitochondrial tRNASer(AGY) is specifically impaired in patients with mutations in the tRNA nucleotidyl transferase TRNT1. Hum Mol Genet
Xiong Y, Steitz TA (2006) A story with a good ending: tRNA 3'-end maturation by CCA-adding enzymes. Curr Opin Struct Biol 16:12–17
Cimaz R, Von Scheven A, Hofer M (2012) Systemic-onset juvenile idiopathic arthritis: the changing life of a rare disease. Swiss Med Wkly 142:w13582
Pascual V et al (2008) How the study of children with rheumatic diseases identified interferon-alpha and interleukin-1 as novel therapeutic targets. Immunol Rev 223:39–59
Pascual V, Allantaz F, Arce E, Punaro M, Banchereau J (2005) Role of interleukin-1 (IL-1) in the pathogenesis of systemic onset juvenile idiopathic arthritis and clinical response to IL-1 blockade. J Exp Med 201:1479–1486
Wakil SM et al (2015) Association of a mutation in LACC1 with a monogenic form of systemic juvenile idiopathic arthritis. Arthritis Rheumatol 67:288–295
Wong SH, Hill AV, Vannberg FO, India-Africa-United Kingdom Leprosy Genetics, C (2010) Genomewide association study of leprosy. N Engl J Med 362:1446–7, author reply 1447-8
Zhang FR et al (2009) Genomewide association study of leprosy. N Engl J Med 361:2609–2618
Sales-Marques C et al (2014) NOD2 and CCDC122-LACC1 genes are associated with leprosy susceptibility in Brazilians. Hum Genet 133:1525–1532
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is a contribution to the Special Issue on The Inflammasome and Autoinflammatory Diseases - Guest Editors: Seth L. Masters, Tilmann Kallinich and Seza Ozen
Rights and permissions
About this article
Cite this article
Aksentijevich, I. Update on genetics and pathogenesis of autoinflammatory diseases: the last 2 years. Semin Immunopathol 37, 395–401 (2015). https://doi.org/10.1007/s00281-015-0478-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00281-015-0478-4