Abstract
Purpose
Mandibular condyle development can be evaluated with radiographic evaluation of subchondral cortical bone. The aim of this study is to investigate mandibular condyle cortication (MCC), articular eminence cortication (AEC) and mandibular cortical index (MCI) according to age and gender on CBCT, and relationship between each other.
Methods
CBCT scans of 520 patients (312 male and 208 female, age range 7–84) were retrospectively investigated to evaluate MCC, AEC and MCI. MCC and AEC were examined in sagittal section and MCI in panoramic reformatted image. MCC and AEC were classified as Type I, Type II and Type III according to density difference between the cortical bone enclosing condyle and articular eminence. Categorical variables were tested by Chi square.
Results
When MCC, AEC and MCI were compared, a significant relationship was detected (p < 0.05). According to age, there was a significant difference between MCC, MCI and AEC types with each other (p < 0.05). No significant discrepancy was found between gender and MCC, AEC and MCI (p > 0.05).
Conclusions
This study evaluated the distribution of MCC, AEC, MCI according to age and gender and detected that these cortications were correlated. This result may be caused by anatomical proximity bone components and functional stimuli. Knowledge of these cortications can be important for accurate diagnosis of TMJ disorders and may also be helpful for prediction of osteoporotic changes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The bone components of the temporomandibular joint (TMJ) are mandibular condyle, glenoid fossa of the temporal bone, and fibrocartilage articular disc is located between these structures [8, 26]. The growth and development of the mandible are important factor contributing to maturation of the mandibular condyle [16]. The cortical border of condyle, glenoid fossa and articular eminence commence to appear at approximately 18 years of age [26]. The remodeling of the mandibular condyle leads to differences in the mineralization of the cortical bone of the condyle [20]. The subchondral cortex of condyle is mainly involved for shock absorption, and force transmission to the mandibular ramus [19, 25]. In addition, hormones, genetic, environmental and ethnic factors can affect the development of TMJ components and TMJ diseases [5]. The condyle and the articular eminence morphology are affected from each other, and the other factors such as age, craniofacial development, function and the condyle adapts to the changes in the articular eminence and articular disc [12, 29]. Besides, some pathological conditions may cause thinning or loss of mandibular condyle, articular eminence and glenoid fossa cortication [26].
Changes in the cortical layers of the mandible may develop due to factors such as age, mineral loss and osteoporosis. MCI is a strong radiographic indicator for osteoporosis [7, 13, 27]. Since the mandibular inferior cortex can demonstrate the mineral status of the buccal cortical structure, it has been determined suitable for the examination of the mineral structure of the skeleton. The inferior cortex can be evaluated from the distal of the mental foramen according to the mandibular cortical index (MCI) classification [13].
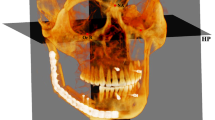
The condyle development can be examined with radiographic evaluation of the subchondral cortical bone [16]. Magnetic resonance imaging (MRI), computed tomography (CT), arthrography, cone-beam computed tomography (CBCT) can be used for visualization of the TMJ [6, 9, 14]. CBCT, which provides low-dose and high-resolution images of head and neck structures, ensures images to be scanned in multiplanar sections, to obtain three-dimensional images, to be reconstructed without magnification, and modeling images with software [5, 9]. CBCT is more reliable than two-dimensional imaging methods and images are attained with higher accuracy [10]. The bone changes in the condyle morphology can be analyzed in three dimensions using CBCT [11].
The development of mandible, condyle and articular eminence are interrelated. Therefore, the cortication of the mandibular condyle (MCC), articular eminence (AEC) and MCI may be expected to be correlated. In the literature, there are quite limited studies that classified the degrees of the MCC [5, 16]. However, to the best of our knowledge, there is no publishing study to categorize the AEC and examine the relationship between AEC with MCC and MCI. Similar to MCI, AEC and MCC may also be useful in assessing the quality of the related bone and predicting osteoporotic changes. The aim of this study was to investigate the MCC, AEC and MCI according to age and gender on CBCT images, and the relationship between each other.
Methods
In this retrospective study, the images taken with Planmeca Promax 3D (Helsinki, Finland) CBCT device in the tomography archive of University Faculty of Dentistry, Dentomaxillofacial Radiology Department were used. Before the study, ethical approval was obtained from University Ethics Committee (Protocol No: 2019/29). Multiplanar images were obtained from 16 × 9, 16 × 16 FOV (field of view) with 0.4 mm3 voxel size and 1 mm slice thickness. The cases with bone-related metabolic disease (osteopetrosis, paget, etc.), systemic, inflammatory, degenerative joint disease, fracture line in the maxillofacial region or any lesion were excluded from the study. CBCT scans of 520 patients (312 male and 208 female) between the ages of 7 and 84 were investigated.
Image analysis
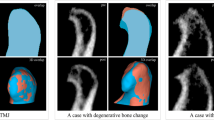
Romexis software (Helsinki, Oy, Finlandiya) was used to analyze the images. The right and left MCC and AEC were evaluated on the sagittal section where the condyle was most clearly seen and most intense was selected as the reference for each image. The whole images were assessed and categorized according to the difference of the density between cortical bone enclosing the condyle and articular eminence and the bone areas around these structures. As similar in the classification of MCI used for the evaluation of bone density and quality, MCC and AEC were classified into three different types by the new method we developed (Figs. 1, 2):
Type I: The condylar and articular eminence surfaces are appeared to be similar or higher density compared to enclosing cortical areas.
Type II: The condylar and the articular eminence surfaces are appeared to be lower density than the enclosing structures.
Type III: The condyle and articular eminence have no cortication.
We analyzed the MCI according to the classification of Klemetti [13], which describes the morphology of mandibular cortical bone depending on the lacuna or porosity on panoramic radiography. In the analysis of mandibular cortical bone morphology, panoramic reformatted image of CBCT was used because it was thought to be more reliable and useful than panoramic radiographs [9]. The inferior mandibular cortical bone was evaluated bilaterally from the distal of the mental foramen and MCI is divided into three groups (Fig. 3): C1, endosteal margin of the cortex is even and sharp; C2, endosteal margin shows semilunar defects exhibiting one or three layers of endosteal cortical residues; C3, endosteal cortical layer contains many cortical residues and appears porous. MCI was examined visually from both sides, and the more deteriorated cortex was used.
All CBCT images were examined by two maxillofacial radiologists, one with 7 years of experience (EDY) and the other is research assistant (CB). To determine the reliability of intra- and inter-observer calibration, 20% of the images were randomly selected and assessed by the observers 2 weeks after the initial evaluation.
Statistical analysis
The intraclass correlation coefficient was used to calculate inter- and intra-observer agreement. SPSS software (version 20.0) (Armonk, NY, IBM) was used for data analysis. The Shapiro–Wilk test was used to examine the normal distribution of numerical data. Independent t test was applied to compare the numerical values of two groups with normal distribution. Categorical variables were made by Chi square test. Two-way anova test was used to examine the effect of two independent variables. Significance was accepted when p value < 0.05.
Results
The coefficient of intra- and inter-observer reliability for all the assessments was found to be excellent (0.88 and 0.91, respectively). A total of 520 CBCT images of 312 (60%) males and 208 (40%) females (with a mean age of 40.43 ± 17.28 and 39.44 ± 17.43, respectively) were investigated retrospectively. The distribution of MCC and AEC according to age and gender in both sides was demonstrated in Table 1. 321 (61.73%) cases were found as Type I MCC, 127 (24.43%) were Type II, and 72 (13.84%) were Type III in the right condyle (Table 1). On the left side, Type I MCC was determined at 44.31 ± 14.09 years, Type II at 41.75 ± 17.90 years, and Type III at 19.66 ± 12.58 years (Table 1). There was no significant difference between the MCC and gender in both sides (p > 0.05) (Table 1). In the assessment of right AEC, it was detected 207 (39.81%) Type I, 262 (50.39%) Type II and 51 (9.80%) Type III (Table 1). The mean age for AEC on the right side was 42.72 ± 15.78 for Type I, 40.19 ± 17.52 for Type II, and 28.29 ± 17.83 for Type III (Table 1). No significant discrepancy was detected between AEC and gender in both sides (p > 0.05) (Table 1).
Distribution of the mean age for each MCC and AEC type was indicated in Table 2. A significant discrepancy was found among MCC and AEC types by age on both sides (p < 0.05). There was a significant difference between left MCC and right AEC Type III and both Type I and Type II by age (p < 0.001), but there was no significant difference between Type I and Type II (p > 0.05) (Table 2).
The distribution of MCI according to age and gender is shown in Table 3. 300 cases (57.69%) were C1, 198 cases (38.07%) were C2, and 22 cases (4.23%) were C3. There was a significant discrepancy between MCI types and age (p < 0.001). The mean age was 33.33 ± 14.82 for C1, 47.52 ± 16.09 for C2 and 64.09 ± 9.42 for C3. No significant difference was determined between the MCI and gender (p > 0.05) (Table 3).
The comparison of MCC and AEC on the right and left sides was demonstrated in Table 4. A significant relationship was found in total and both genders on the right and left sides (p < 0.001). On the left side, in males and total, when the AEC was Type I, Type II and Type III, the most prevalent MCC type was Type I, Type II and Type III, respectively. In women, when AEC was Type II, the most frequent MCC type was Type I and when the MCC was Type III, the most common AEC type was Type II.
The comparison of MCC and AEC with MCI on the right and left sides was indicated in Table 5. A significant association was detected between MCC and MCI in total and males (p < 0.05), but there was no significant discrepancy in females (p > 0.05). In total and males, when the MCI type was C2, the most prevalent MCC type was Type I. When the MCC was both Type II and Type III, the most common MCI type was C1. In the comparison of AEC and MCI, there was a significant relationship on the left side (p < 0.05), but no significant difference was in both genders on the right and left sides (p > 0.05). On the left side, when the AEC was Type I, Type II and Type III, the most common MCI type was C1 (Table 5).
Discussion
In this study, the cortication of mandibular condyle, articular eminence and MCI were evaluated according to age and gender on CBCT images and the relationship between these three parameters was investigated. We found a statistically significant relationship between MCC, AEC and MCI. There are quite limited studies in the literature on cortication of TMJ bone components and only the condyle cortication was assessed [5, 16]. To the best of our knowledge, this is the first CBCT study to examine MCC, AEC and MCI, and the relationship between them.
Various radiographic techniques, such as panoramic [1, 3], MRI [22], CT [4] are available for imaging TMJ components. Liu et al. [17] used panoramic radiography in their studies on TMJ maturity, but Morimoto et al. [18] evaluated cortical bone formation in mandibular condyles with MRI. The development of cross-sectional imaging by tomography and CT are more advanced technique than traditional methods [21]. However, CBCT has a higher diagnostic sensitivity for bone changes in TMJ than CT, and allows the evaluation of condyle morphology [21]. For this reason, we used CBCT to examine the cortication of condyle and articular eminence in this study.
Lei et al. [16] have classified the cortical bone formation of the condyle as follows; complete formation, partial formation or no cortical bone. Bayrak et al. [5] divided MCC into three groups; Type I, there is no cortication in the condyle; Type II, the condyle superior surface is less dense than surrounding structures; Type III, the condyle surface is similar to or higher than the surrounding cortical areas. We categorized MCC and AEC, as similar in the classification of MCI.
The development of the articular eminence is based on the functional stimulus from the condyle which subsequently may cause variations in the depth of glenoid fossa. The cortices can remain unclear till adulthood [26]. Lei et al. [16] notified in their CBCT study that subchondral cortical bone formation in the condyle begins at the age of 13–14 years and completes at 22 years in males, and starts at 12–13 years and completes at 21 years in females, and is usually not seen before this age. According to Arnett and McLaughlin [3], if there is no cortication in the condyle, this refers to the immature condyle, and the onset of cortication is about 15–16 years old. The morphological formation of the articular eminence inclination and height is completed about 20 years of age [16].
In adults, the joint surfaces of the condyle, articular eminence and the glenoid fossa contain a cortical border, but they are not observed before approximately 18 years of age [26]. Consistent with this, in the present study, Type III MCC, which has no cortication, was observed in men at 18.50 ± 11.25 years in the right and 18.52 ± 11.15 years in the left, and it was also observed in women at 19.39 ± 12.14 years in the right and 21.59 ± 14.73 years in the left. However, in the study of Bayrak et al. [5], Type I MCC, which has no cortication, was detected in males at 14.14 ± 2.3 years in both right and left sides, and in females, 13.06 ± 2.19 years in the right and 13.01 ± 2.15 years in the left. The mean age for all three types of MCC was younger in both genders in the study of Bayrak et al. [5] than in this study. These differences may be due to wider age range of our study sample.
Bayrak et al. [5] concluded that Type I and Type III MCC was detected in older ages in males than in females, and Type II was similar in all cases. Lei et al. [16] determined that the full development of condyle occurred in females 1 year earlier than males. In the current study, no significant discrepancy was found between MCC and genders. It is thought that these different results may be caused by factors such as parafunctional habits, condylar resorption, hormones (especially the effect of oestrogen on cartilage and bone metabolism) and ethnical changes.
When the mean age for MCI was examined, Ledgerton et al. [15] reported that the C1 category was observed in the younger age group, the C3 was in the older age group and the C2 was in all age groups. They also indicated that C3 demonstrates a higher risk of osteoporosis than C1. Yalcin et al. [28] concluded in the patients with scleroderma that the mean age for C3 was determined at advanced ages than C1. Uysal et al. [24] stated that C1 decreased while age increased and C3 was seen in the oldest age group. They also notified that C1 and C3 were found to be significantly higher in females than in males. In the present study, there was no significant difference between MCI and gender, but a significant difference was found between MCI types and age. In accordance with the literature, we concluded in both males and females, C1 were seen at younger ages, while C3 was observed at older ages.
When we compared the MCC with AEC, a significant relationship was found between them. The mean ages for Type I and Type II MCC and AEC were similar, while Type III AEC was detected at advanced ages than Type III MCC in both genders and two sides. The reason for this may be that the subchondral articular eminence formation begins to occur at advanced ages than MCC. However, since there is no other publishing study about AEC, the results could not be discussed. Further studies are needed in this area.
There was a significant relationship between MCC, AEC and MCI. In the comparison of AEC with MCI, a significant relationship was detected on the left side, but not on the right side. Parafunctional activity may be the cause of different results on the right and left sides. When the mean ages for MCC, AEC and MCI were examined, MCC and AEC Type I were seen at older ages than C1 category, while Type II and Type III were observed at younger ages than C2 and C3. The reason for this is that the completion of the subchondral cortical formation of the condyle and articular eminence is considered to be at advanced ages than the mandible.
Based on the results of this study, even if the cortications of articular eminence and mandibular condyle do not appear completely and clearly on panoramic images, MCI may also be a radiological marker for AEC and MCC in cases where CBCT is not available. Furthermore, when there is insufficient determination of MCI due to the overlapping of the buccal and lingual cortices on the panoramic image [23], AEC and MCC may also be an indicator for MCI. In addition, CBCT images from the small field of view are recommended for osseous assessment of TMJ. However, in such case, the mandible will not be present, and it will not be possible to examine MCI. Therefore, evaluation of MCC and AEC may be useful for osteoporotic changes. In a study conducted by Alkhader et al. [2], it is considered that the measuring bone density of mandibular condyle is useful in predicting patients at risk of osteoporosis.
Considering the fact that MCI is a strong indicator of osteoporosis, the results of this study may be scientific evidence for further investigations related with the effect of AEC and MCC on osteoporosis prediction, but further research with multiple groups (osteoporosis, osteoarthritis, etc.) is needed to correctly present study results.
One limitation of this retrospective study was that the medical history, systemic diseases or TMJ internal derangements of the patients were unknown. Therefore, future studies either prospective, or including a particular group of diseases that affect cortications may be planned. In addition, the number of subjects can be increased and the cases can be divided into age groups.
Conclusion
In this study, we investigated the distribution of MCC, AEC and MCI according to age and gender and found that these cortications were correlated. This result may be caused by anatomical proximity of the mandible with the bone components of TMJ, functional stimuli, and the relation of development the TMJ bone components. Knowledge of cortications of articular eminence and condyle can be important for accurate diagnosis and appropriate treatment planning for TMJ disorders and may also be helpful for prediction of osteoporotic changes, similar to MCI. Future studies can be performed prospectively or with larger sample size.
References
Ahmad M, Hollender L, Anderson Q, Kartha K, Ohrbach R, Truelove EL et al (2009) Research diagnostic criteria for temporomandibular disorders (RDC/TMD): development of image analysis criteria and examiner reliability for image analysis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 107:844–860
Alkhader M, Aldawoodyeh A, Abdo N (2018) Usefulness of measuring bone density of mandibular condyle in patients at risk of osteoporosis: A cone beam computed tomography study. Eur J Dent 12:363–368
Arnett GW, McLaughlin RP (2004) Diagnosis the case. In: Arnett GW, McLaughlin RP (eds) Facial and dental planning for orthodontists and oral surgeons, 1st edn. Mosby, California, pp 145–146
Bag AK, Gaddikeri S, Singhal A, Hardin S, Tran BD, Medina JA et al (2014) Imaging of the temporomandibular joint: an update. World J Radiol 6:567–582
Bayrak S, Halicioglu S, Kose G, Halicioglu K (2018) Evaluation of the relationship between mandibular condyle cortication and chronologic age with cone beam computed tomography. J Forensic Leg Med 55:39–44
Brooks SL, Brand JW, Gibbs SJ, Hollender L, Lurie AG, Omnell K-Å et al (1997) Imaging of the temporomandibular joint: a position paper of the American Academy of Oral and Maxillofacial Radiology. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 83:609–618
Devlin H, Karayianni K, Mitsea A, Jacobs R, Lindh C, van der Stelt P et al (2007) Diagnosing osteoporosis by using dental panoramic radiographs: the OSTEODENT project. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 104:821–828
Gallo LM (2005) Modeling of temporomandibular joint function using MRI and jaw-tracking technologies–mechanics. Cells Tissues Organs 180:54–68
Gupta M, Mishra P, Srivastava R, Jyoti B (2015) Cone beam computed tomography: a new vision in dentistry. Digit Med 1:7–16
Honey OB, Scarfe WC, Hilgers MJ, Klueber K, Silveira AM, Haskell BS et al (2007) Accuracy of cone-beam computed tomography imaging of the temporomandibular joint: comparisons with panoramic radiology and linear tomography. Am J Orthod Dentofacial Orthop 132:429–438
Ikeda K, Kawamura A (2009) Assessment of optimal condylar position with limited cone-beam computed tomography. Am J Orthod Dentofacial Orthop 135:495–501
Katsavrias EG (2006) Morphology of the temporomandibular joint in subjects with Class II Division 2 malocclusions. Am J Orthod Dentofacial Orthop 129:470–478
Klemetti E, Kolmakov S, Kröger H (1994) Pantomography in assessment of the osteoporosis risk group. Scand J Dent Res 102:68–72
Larheim TA (1995) Current trends in temporomandibular joint imaging. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 80:555–576
Ledgerton D, Horner K, Devlin H, Worthington H (1999) Radiomorphometric indices of the mandible in a British female population. Dentomaxillofac Radiol 28:173–181
Lei J, Liu MQ, Yap AU, Fu KY (2013) Condylar subchondral formation of cortical bone in adolescents and young adults. Br J Oral Maxillofac Surg 51:63–68
Liu YY, Wang H, Yang ZY, Ba K, Li MX, Liu L (2010) The correlation between temporomandibular joint maturity and second molar root development in adolescents. Int J Stomatol 37:154–156
Morimoto Y, Konoo T, Tominaga K, Tanaka T, Yamaguchi K, Fukuda J et al (2007) Relationship between cortical bone formation on mandibular condyles and alternation of the magnetic resonance signals characteristic of growth. Am J Orthod Dentofacial Orthop 131:473–480
Renders GAP, Mulder L, van Ruijven LJ, van Eijden TMGJ (2007) Porosity of human mandibular condylar bone. J Anat 210:239–248
Renders GAP, Mulder L, van Ruijven LJ, van Eijden TMGJ (2006) Degree and distribution of mineralization in the human mandibular condyle. Calcif Tissue Int 79:190–196
Rozylo-Kalinowska I, Orhan K (2019) Imaging of the Temporomandibular Joint. In: Rozylo-Kalinowska I, Orhan K (eds) Cone beam computed tomography (CBCT) in TMJ imaging, 1st edn. Springer Nature, Switzerland, pp 125–131
Sano T, Westesson PL, Larheim TA, Takagi R (2000) The association of temporomandibular joint pain with abnormal bone marrow in the mandibular condyle. J Oral Maxillofac Surg 58:254–257
Secgin CK, Gulsahi A, Yavuz Y, Kamburoglu K (2019) Comparison of mandibular index values determined from standard panoramic versus cone beam computed tomography reconstructed images. Oral Surg Oral Med Oral Pathol Oral Radiol 127:257–264
Uysal S, Çağırankaya BL, Hatipoğlu MG (2007) Do gender and torus mandibularis affect mandibular cortical index? A cross-sectional study. Head Face Med 3:37
van Eijden TMGJ, van der Helm PN, van Ruijven LJ, Mulder L (2006) Structural and mechanical properties of mandibular condylar bone. J Dent Res 85:33–37
White SC, Pharoah MJ (2014) Oral radiology: principles and interpretation. In: White SC, Pharoah MJ (eds) Cone beam computed tomograph: anatomy, 7th edn. Mosby, St. Louis, pp 214–228
White SC, Taguchi A, Kao D, Wu S, Yoon D, Suei Y et al (2005) Clinical and panoramic predictors of femur bone mineral density. Osteoporos Int 16:339–346
Yalcin ED, Avcu N, Uysal S, Arslan U (2019) Evaluation of radiomorphometric indices and bone findings on panoramic images in patients with scleroderma. Oral Surg Oral Med Oral Pathol Oral Radiol 127:23–30
Zain-Alabdeen EH, Alsadhan RI (2012) A comparative study of accuracy of detection of surface osseous changes in the temporomandibular joint using multidetector CT and cone beam CT. Dentomaxillofac Radiol 41:185–191
Funding
No funding resource is associated with this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
Formal consent is not required.
Ethical approval
This retrospective study was approved by the Ethical Committee of Gaziantep University (Decision No: 2019/29) and conducted in the Dentomaxillofacial Radiology Department of Gaziantep University, Faculty of Dentistry.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yalcin, E.D., Bozan, C. Relationship between mandibular condyle and articular eminence cortication with mandibular cortical index on cone-beam CT. Surg Radiol Anat 42, 515–522 (2020). https://doi.org/10.1007/s00276-019-02385-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-019-02385-7