Abstract
We compared the use of manual compression and Angio-Seal and Starclose arterial closure devices to achieve hemostasis following common femoral artery (CFA) punctures in order to evaluate safety and efficacy. A prospective nonrandomized, single-center study was carried out on all patients undergoing CFA punctures over 1 year. Hemostasis was achieved using manual compression in 108 cases, Angio-Seal in 167 cases, and Starclose in 151 cases. Device-failure rates were low and not significantly different in the two groups (manual compression and closure devices; p = 0.8). There were significantly more Starclose (11.9%) patients compared to Angio-Seal (2.4%), with successful initial deployment subsequently requiring additional manual compression to achieve hemostasis (p < 0.0001). A significant number of very thin patients failed to achieve hemostasis (p = 0.014). Major complications were seen in 2.9% of Angio-Seal, 1.9% of Starclose, and 3.7% of manual compression patients, with no significant difference demonstrated; 4.7% of the major complications were seen in female patients compared to 1.3% in males (p = 0.0415). All three methods showed comparable safety and efficacy. Very thin patients are more likely to have failed hemostasis with the Starclose device, although this did not translate into an increased complication rate. There is a significant increased risk of major puncture-site complications in women with peripheral vascular disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Percutaneous endovascular interventions are increasingly performed in an effort to avoid surgery. The growing success of interventions and requirement for day case or outpatient procedures has led to a concomitant rise in the use of arterial closure devices to achieve hemostasis and allow early mobilization following arterial punctures. When compared to manual compression, several studies have confirmed patient comfort, reduced time to achieve hemostasis, reduced time to ambulation, and early discharge [1–5].
The safety and efficacy of the Angio-Seal is well documented [6–8] and this device has been used in our department for several years. However, some problems have been reported in the literature, of which acute femoral artery occlusion, infection, and distal embolization are the most serious. More recently, the Starclose was introduced to our practice. There have only been two published studies assessing the use of Starclose. This clip-based device remains outside the vessel lumen and theoretically should not cause intraluminal problems [9, 10]. No studies have been published comparing these two closure devices; therefore, in an attempt to inform our practice, we prospectively collected data using these devices and manual compression.
The Angio-Seal STS Plus (St. Jude Medical) is composed of a collagen sponge and a specially designed polymer anchor connected by a self-tightening suture. All of the components are absorbable. The device seals and sandwiches the arteriotomy between its two primary members: the anchor and collagen sponge. Hemostasis is achieved primarily by the mechanical means of the anchor–arteriotomy–collagen sandwich, which is supplemented by the coagulation-inducing properties of the collagen. The device is contained in a delivery system that stores and then delivers the absorbable components to the arterial puncture. Immediate repuncture of the artery is feasible and the components are said to absorb within 3 months, leaving no residual material.
The Starclose (Abbott Vascular Devices) features a star-shaped Nitinol clip approximately 4 mm in diameter that is positioned against the outside arterial wall at the puncture site and then released. The device has small pins that grab the arterial tissue from the outside, then folds inward, causing it to pucker and seal the puncture, providing an extravascular closure that does not enter the vessel lumen. The clip applier is attached to the introducer sheath, which is inserted following the primary procedure. The clip is then advanced down the inside of the sheath. The vessel locator button is depressed. The device is apposed against the arteriotomy with gentle traction. The thumb advancer slides forward, splitting the sheath as the clip is advanced to the arteriotomy. While maintaining tension, the trigger button is depressed, deploying the clip. The clip applier and sheath are withdrawn.
Materials and Methods
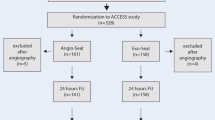
A prospective single-center nonrandomized trial was carried out at our institution from February 2005 to February 2006. Data were collected on a total of 429 common femoral artery punctures carried out during this period using either an antegrade or retrograde technique. Hemostasis was achieved by either manual compression or Angio-Seal or Starclose arterial closure devices. All arterial punctures were performed by specialist registrars and consultant vascular radiologists. Manual compression was performed by all grades. Arterial closure devices were inserted only by radiologists who had undergone appropriate training in the use of the device and were certified by the manufacturers of the devices.
A record was made of the type of puncture (retrograde versus antegrade), the procedure performed, the number of needle punctures, sheath size, and choice of closure method and any reasons directing this choice. Patient characteristics such as diabetes, renal failure, visible femoral calcification, very thin or obese patients, and the use of anticoagulation were also recorded. Difficulties or complications in deployment, as well as complications after deployment, the nature of these complications, and any treatment that ensued were recorded. Inpatients were reviewed at 24 h. Day cases were reviewed at 6 h or immediately prior to discharge and given a contact telephone number in case of problems. Any complications after this period were identified and fed back to us by the patients directly or via the original referring clinicians. All patients with successful device closure were mobilized after 2 h bed rest, whereas those requiring manual compression were mobilized after 6 h bed rest.
Certain patients were excluded from the use of an arterial closure device. These included procedures where anything less than a 6F catheter or sheath had been used, as both closure devices are 6F, puncture of a poor quality common femoral artery (caliber less than 6 mm/heavily calcified), superficial femoral artery (SFA) puncture or puncture adjacent to the femoral bifurcation, puncture of a graft, imminent surgical arteriotomy or interventional procedure at the site of puncture, previous surgery in the groin, and preexisting hematoma. In all other cases, the type of closure device selected depended on operator preference, as some had not been trained in the use of the Starclose but became trained during the course of the study.
Complications are classified according to the Society of Interventional Radiology (SIR) guidelines. Minor complications are those that required no treatment. Major complications are defined as those requiring treatment and further hospitalization, those requiring an unplanned increase in level of care, those with permanent adverse sequelae, and those resulting in death [11, 12]. This study did not evaluate the departmental time saved by the use of these devices, patient comfort, or cost-effectiveness. Statistical analysis was performed using Fisher’s exact test and the χ2 test with Yates analysis. A p-value of less than 0.05 was deemed to be statistically significant.
Results
A total of 426 femoral arterial punctures were carried out during the study period. One hundred sixty-seven Angio-Seal (39.2%) and 151 Starclose (35.4%) devices were used to achieve hemostasis. In 108 cases (25.4%), manual compression was applied. The type of procedure carried out (diagnostic or interventional) and the patient demographics are summarized in Table 1.
The events in the two closure device groups were divided into four categories on an intention-to-treat basis and those in the manual compression group were divided into minor and major complications. These are shown in Table 2.
Immediate deployment of the selected closure device failed (group 1) with successful subsequent manual compression in 10 Angio-Seal (5.9%) and 11 Starclose (7.3%) devices, demonstrating no significant difference. In the Angio-Seal group, the sheath could not be advanced over the guidewire on six occasions. In two of these, the guidewire was recorded as being buckled. In a further four patients, the reason for failure is not known. In the Starclose group, one patient had oozing around the sheath prior to deployment, which caused a hematoma and the device sheath could not be passed. In four patients, the delivery device could not be advanced and the device had to be withdrawn undelivered. In one of these cases, it was later recognized that the metal delivery system had been bent. In six patients, the reason for failure is not known.
There was a significant difference (p < 0.005) between the devices in group 2, where deployment was apparently successfully but hemostasis was not achieved, with four Angio-Seal (2.4%) versus 18 Starclose (11.8%) devices. In all cases, manual compression was immediately applied successfully with no complications. It is documented that two patients from the Starclose group were confused, uncooperative, and would not lie still, and in one case, a 7F sheath had been used. The majority of these cases were also recorded in the beginning of the study. The characteristics of the 18 Starclose patients are shown in Table 3. From these data, a significant number of very thin patients failed to achieve hemostasis (p = 0.014). None of the other characteristics achieved statistical significance.
Minor complications (group 3) were seen in seven patients in the Angio-Seal (4.2%) group, eight in the Starclose (5.3%) group, and four in the manual compression (3.7%) group. These comprised small, tender hematomata requiring no intervention. One patient in the Angio-Seal group complained of pain in the right iliac fossa, but no underlying cause for this was found. In the manual compression group, there were two small pseudoaneurysms that required no treatment and discharge was not delayed.
Major complications (group 4) occurred in five patients (2.9%) in the Angio-Seal group, three patients (1.9%) in the Starclose group, and four patients (3.7%) in the manual compression group. This was not statistically significant (p = 0.72). In the Angio-Seal group, one patient developed a pseudoaneurysm requiring thrombin injection, which delayed discharge by 48 h. One patient developed an asymptomatic common femoral artery occlusion on the side of the Angio-Seal that was discovered the following week when intervention was carried out to treat the contralateral side. The initial angiogram showed narrowing at this site. At the time of the repeat angiogram, this had progressed to a complete occlusion. No treatment was carried out, as the patient was asymptomatic and there was established collateral formation. The third developed a common femoral vein thrombosis on the side of the Angio-Seal deployment and presented a week later with an edematous leg. The diagnosis was confirmed on ultrasound and anticoagulation was required. A fourth patient developed a massive retroperitoneal hematoma that was demonstrated on computed tomography (CT). This required surgical evacuation but no repair of the puncture site, as bleeding had ceased. The fifth patient in this group attended for treatment of a stenosis detected on duplex graft surveillance 10 weeks following angioplasty of a stenosis in a distal femoral vein graft. A filling defect was identified on the angiogram performed by a contralateral femoral puncture. This was presumed to be due to the Angio-Seal anchor and was balloon dilated satisfactorily with no significant residual stenosis on duplex follow-up. (Figs. 1 and 2).
In the Starclose group, one patient developed a pseudoaneurysm that required thrombin injection. One patient developed a large hematoma requiring blood transfusion, surgical evacuation, and ICU admission. The patient subsequently made a good recovery. The third patient had a diagnostic renal transplant angiogram with an apparently uneventful post procedural period and returned to the referring hospital. Two days later, he developed an ischemic lower limb on the side of the Starclose deployment. Magnetic resonance angiography (MRA) demonstrated an external iliac artery occlusion. (Figs. 3 and 4). The patient required a femoro-femoral crossover graft and made a good subsequent recovery.
In the manual compression group, one patient developed a large hematoma that delayed discharge by 3 days, two patients developed pseudoaneurysms requiring thrombin injection, and one patient with a large groin hematoma died the next day from an unrelated upper gastrointestinal hemorrhage. The patient characteristics of all of those developing minor and major complications were analyzed and none of these were shown to be statistically significant. These figures are shown in Table 4. However, a significant number of female patients were found to develop major complications and this is shown in Table 5.
Discussion
Manual compression has long been the principal method for achieving arterial puncture-site hemostasis [13]. A variety of arterial closure devices have been introduced with the aim of reducing time to hemostasis, time to ambulation, patient discomfort, and improving patient throughput. Reducing time to ambulation and therefore discharge has become increasingly important with rising pressure to perform procedures on an outpatient basis. In addition, the use of closure devices can be helpful in the hypertensive and anticoagulated patient and those who are unable to lie flat for prolonged periods of time [14]. There are a range of arterial closure devices that achieve hemostasis based on three primary mechanisms: using collagen plugs (Angio-Seal, Vasoseal, Duett), suture-based devices (Perclose), and clips (Starclose) or staples (Vascular Closure System). It is incumbent on those using such devices to ensure that they are at least as safe as manual compression with regard to puncture-site complications and that there is no long-term disadvantage to their use.
The reported incidence of arterial puncture-site minor and major complications following manual compression range from 1.5% to 9 % [15, 16]. The standards of the Royal College of Radiologists (RCR) of the United Kingdom recommend that for interventional procedures, major complications should not exceed 4.0% for puncture-site hematoma and 0.5% for puncture-site occlusion [17]. In series using large sheath sizes (up to 11F), multiple antiplatelet agents with heparin, and stent placement, the complication rates ranged from 5.9% to 17% [18]. Predictors of puncture-site complications include advanced age, female gender, renal failure, lower-extremity vascular disease, shock, longer time to sheath removal, intervention within 24 h of thrombolytic therapy, stent implantation, higher heparin doses, and concomitant placement of venous sheaths [19, 20]. It should be noted, however, that most of the published data on complication rates are from the field of cardiology with associated use of antiplatelet and thrombolytic therapy during procedures. A variety of clinical trials and observational studies show conflicting results, with some showing superiority, inferiority, and no difference when vascular closure devices are compared with manual compression [21]. In spite of the escalating use of these devices in the treatment of peripheral vascular disease, there is a relative paucity of data in the radiology literature.
Angio-Seal has been on the market since the early 1990s and its use and complication rates are well documented. The advantages of Angio-Seal include the ease and speed of deployment and achieving hemostasis with a low complication rate [22]. The disadvantage is the presence of the intravascular suture and anchor, which can act as a nidus for platelet aggregation, the possibility of intravascular deployment of the collagen plug causing femoral artery occlusion, and the risk of distal embolization of the anchor. Common femoral artery occlusion secondary to the use of Angio-Seal has been reported [5, 23, 24]. In one study, 5 out of 742 (0.7%) patients receiving Angio-Seals developed acute femoral artery occlusion requiring urgent vascular surgery [25]. The potential for distal embolization of the anchor emphasizes the need to ensure that vascular surgeons are aware of the presence of the anchor in patients undergoing subsequent surgical procedures so that the retaining suture is not cut [26]. Failure of the Angio-Seal anchor to resorb with an associated vigorous foreign body reaction resulting in delayed occlusion 3 months after deployment has also been reported [27]. The published occlusion or embolization rates for the Angio-Seal device (0–1.4%) were similar to the Vasoseal (0–1.6%) device, which is another closure device employing a collagen plug [18].
The Starclose device is a relative newcomer on the scene. It was the introduction of this device that stimulated this study, as we wished to determine whether there were differences that might affect our practice. In the two published studies of Starclose to date, no major complications have been reported [9, 10]. The main advantage of the Starclose device is that it is entirely extraluminal. The nitinol clip remains extra-arterial and the endothelium is not exposed to foreign material. Although the clip remains in the patient permanently, Nitinol is a highly biocompatible alloy and not expected to cause any adverse sequelae. This is supported by extensive data and experience accumulated over many years with staples, stents, and clips [28].
Our results show a low rate of deployment failure with no significant difference between the two devices. However, failure to achieve hemostasis using the device alone occurred significantly more with the Starclose than the Angio-Seal (11.8% versus 2.4%). This might in part be due to a learning curve, as most of these events were observed in the initial period of data collection and this occurred less frequently as operator confidence increased. In training departments, this element of a learning curve is inevitable. Further analysis of this subgroup demonstrated a significant number of very thin patients. Moderate tract ooze requiring additional compression has been reported with this device in 40% of patients [10]. In all of these patients, the arteriotomy sites had been sealed and the ooze resulted from the surrounding capillaries and arterioles adjacent to the arteriotomy.
From a safety point of view, the most important group is those developing major complications. This study shows no significant difference in the major complication rates among the three groups, which range from 1.9% for Starclose, 2.9% for Angio-Seal to 3.7% for manual compression. Our major complication rate of 2.9% for Angio-Seal is comparable to the reported rates in the literature, which ranges from 0% to 3.6% [29–34]. The major complication rates for all three groups fall within the RCR guidelines. There was no significant difference in the documented patient characteristics in those developing complications between the two closure devices. All five major complications in the Angio-Seal group were in female patients. In the Starclose group, two were male and one female. In the manual compression group, there were three female patients and one male patient. Overall, there is a significantly higher incidence of major complications in all groups combined in female (4.7%) versus male patients (1.3%) (p = 0.0415). The increased risk of severe access complications in female patients following the use of collagen-based vascular closure devices has been reported [35].
The most surprising complication in the Starclose group is the patient who developed arterial occlusion after deployment, as the extraluminal nature of this device should preclude such a complication. Review of the arteriograms showed no abnormality of the artery at the puncture site and no arterial dissection. The implication is that the Nitinol clip must have caused luminal narrowing leading to thrombosis.
Overall, we noted a significant increased risk of major puncture-site complications in women with peripheral vascular disease. Analysis of our data suggests that very thin patients are more likely to have failed haemostasis with the Starclose device, although this did not translate into an increased complication rate. This study demonstrates that the Angio-Seal and Starclose devices are safe for introduction into peripheral vascular practice, allowing rapid mobilization without an increase in the major puncture-site complication rate.
References
Muller DW, Shamir KJ, Ellis SG, et al. (1992) Peripheral vascular complications after conventional and complex percutaneous coronary interventional procedures. Am J Cardiol 69:63–68
Fram DB, Giri S, Jamil G, et al. (2001) Suture closure of the femoral arteriotomy following invasive cardiac procedures: A detailed analysis of efficacy, complications, and the impact of early ambulation in 1,200 consecutive, unselected cases. Cathet Cardiovasc Diagn 53:163–173
Baim DS, Knopf WD, Hinohara T, et al. (2000) Suture-mediated closure of the femoral access site after cardiac catheterization: results of the suture to ambulate and discharge (STAND I and STAND II) trials. Am J Cardiol 85:864–869
Rinder MR, Tamirisa PK, Taniuchi M, et al. (2001) Safety and efficacy of suture mediated closure after percutaneous coronary interventions. Cathet Cardiovasc Intervent 54: 146–151
D?Souza S (2004) Closure devices: indications and results. In: Wyatt MG, Watkinson AF (eds) Endovascular Intervention: Current Controversies. TFM Publishing. Shrewsbury, pp 205–216
Kussmaul WG, Buchbinder M, Whitlow PL, et al. (1995) Rapid arterial hemostasis and decreased access site complications after cardiac catheterization and angioplasty: Results of a randomized trial of a novel hemostatic device. J Am Coll Cardiol 25:1685–1692
Applegate RJ, Grabarczyk MA, Little WC, et al. (2002) Vascular closure devices in patient treated with anticoagulation and IIb/IIa receptor inhibitors during percutaneous revascularization. J Am Coll Cardiol 40:78–83
Duffin DC, Muhlestein JB, Allisson SB, et al. (2001) Femoral arterial puncture management after percutaneous coronary procedures: A comparison of clinical outcomes and patient satisfaction between manual compression and two different vascular closure devices. J Invasive Cardiol 13:354–362
Hermiller J, Simonton C, Hinohara T (2005) Clinical experience with a circumferential clip-based vascular closure device in diagnostic catheterization. J Invasive Cardiol 17:504–551
Ruygrok PN, Ormiston JA, Stewart JT, et al. (2005) Initial experience with a new femoral artery closure device following percutaneous coronary intervention with glycoprotein IIb/IIa inhibition. Cathet Cardiovasc Interv 66:185–191
Sacks D, Marinelli DL, Martin LG, et al. (1997) Reporting standard for clinical evaluation of new peripheral arterial revascularization devices. J Vasc Intervent Radiol 8:137–149
Leoni CJ, Potter JE, Rosen MP, et al. (2001) Classifying complications of interventional procedures: A survey of practicing radiologists. J Vasc Intervent Radiol 12:55–59
Semler HJ. (1985) Transfemoral catheterization: mechanical versus manual control of bleeding. Radiology 154:235
Macdonald S, Thomas SM, Cleveland TJ, et al (2002) Outpatient vascular intervention: A two-year experience. Cardiovasc Intervent Radiol 25:403–412
Popma JJ, Satler LF, Pichard AD, et al. (1993) Vascular complications after balloon and new device angioplasty. Circulation 88:1569–1578
Nasser TK, Mohler ER, Wilensky RL, et al. (1995) Peripheral vascular complications following coronary interventional procedures. Clin Cardiol 18: 609–614
The Royal College of Radiology. Standards in vascular radiology. BFCR 99:9–40
Hoffer EK, Bloch RD (2003) Percutaneous closure devices. J Vasc Intervent Radiol 14:865–886
Mandak JS, Blankenship JC, Gardner LH (1998) Modifiable risk factors for vascular access site complications in the IMPACT II trial of angioplasty with versus without eptifibatide. J Am Coll Cardiol 31:1518–1524
Piper WD, Malenka DJ, Ryan TJ (2003) Predicting vascular complications in percutaneous coronary interventions. Am Heart J 145:1022–1029
Aggarwal K, Murtaza M (2004) Vascular closure device complications: The case is not closed yet. J Invasive Cardiol 16(5):251
Castriota F, Tarantino F, Troiani E, et al. (1998) Femoral arterial hemostasis using the Angio-Seal system after coronary and vascular percutaneous angioplasty and stenting. J Invasive Cardiol 10:464–469
O?Sullivan GJ, Buckenham TM, Belli AM (1999) The use of the Angio-seal haemostatic puncture device in high-risk patients. Clin Radiol 54:51–55
Abando A, Hood D, Weaver F (2004) The use of the Angioseal device for femoral artery closure. J Vasc Surg 40:287–290
Carey D, Martin JR, Moore CA, et al. (2001) Complications of femoral artery closure devices. Cathet Cardiol Intervent 52:3–7
Henry M, Amor M, Allaoui M, et al. (1995) A new access site management tool: The Angio-Seal hemostatic puncture closure device. J Endovasc Surg 2:289–296
Shaw JA, Gravereaux EC, Winters GL, et al. (2003) An unusual cause of claudication. Cathet Cardiol Intervent 60:562–565
Caputo RP, Ebner A, Grant W, et al. (2002) Percutaneous femoral arteriotomy repair: Initial experience with a novel staple closure device. J Invasive Cardiol 14:652–656
Park Y, Roh HG, Choo SW, et al. (2005) Prospective comparison of collagen plug (Angio-Seal) and suture-mediated (the Closer S) closure devices at femoral access sites. Korean J Radiol 6:248–255
Eidt JF, Habibipour S, Saucedo JF, et al. (1999) Surgical complications from hemostatic puncture closure devices. Am J Surg 178:511–516
Aksoy M, Becquemin JP, Desgranges P, et al. (2006) The safety and efficacy of Angioseal in therapeutic endovascular interventions. Eur J Vasc Endovasc Surg 32(1):90–93
Shammas NW, Rajendran VR, Alldredge SG, et al. (2002) Randomized comparison of Vasoseal and Angioseal closure devices in patients undergoing coronary angiography and angioplasty. Cathet Cardiovasc Intervent 55:421–425
Bown MJ, Blanshard KS, Cutress ML, et al. (2002) Off-license use of Angio-Seal arterial puncture closure device. Eur J Vasc Endovasc 24:372–373
Shrake KL (2000) Comparison of major complication rates associated with four methods of arterial closure. Am J Cardiol 85:1024–1025
Eggebrecht H, von Birgelen C, Naber C, et al. (2004) Impact of gender on femoral access complications secondary to application of a collagen-based vascular closure device. J Invasc Cardiol 16:247–250
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ratnam, L.A., Raja, J., Munneke, G.J. et al. Prospective Nonrandomized Trial of Manual Compression and Angio-Seal and Starclose Arterial Closure Devices in Common Femoral Punctures. Cardiovasc Intervent Radiol 30, 182–188 (2007). https://doi.org/10.1007/s00270-006-0226-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-006-0226-1