Abstract
We report a case of painful perineal metastasis from urinary bladder carcinoma in a 73-years-old woman, treated with CT-guided radiofrequency ablation (RFA). The pain was immediately relieved and follow-up at 1 and 6 months showed total necrosis of the mass. One year later, the patient has no pain and her quality of life is improved.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Percutaneous radiofrequency ablation (RFA) under CT guidance is a minimally invasive technique that has been used for over a decade for the treatment of primary and secondary liver tumors [1, 2]. It is a low-cost method that provides treatment on an outpatient basis. (Some centers performing RFA require patients to stay overnight in the hospital.) This procedure has been rapidly adopted and, in some cases, it is chosen over surgery because it requires less resources, time, recovery and cost, but especially because it can provide complete tumor eradication in properly selected candidates and may improve patients’ prognosis. It reduces morbidity and mortality and provides amelioration of the patient’s quality of life. The complications are minimal in experienced hands [3–5].
In recent years RFA has been used for the treatment of lung [6, 7], renal [8, 9], brain [10], bone [11–13], prostate [14] and breast [15] primary or metastatic tumors with promising results [3].
As RFA has also been used successfully for the treatment of painful osteoid osteomas [11] and bone metastases [12] we decided to use it for the treatment of a painful perineal metastasis from urinary bladder carcinoma.
Case Report
A 73-year-old woman presented to our hospital complaining of severe pain at the perineum, with no response to analgesic medication. The patient suffered from urinary bladder carcinoma and had undergone bladder excision 5 years previously, followed by chemotherapy.
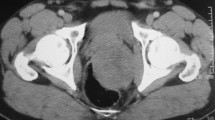
After the clinical examination, a dual-phase abdominal CT scan revealed a soft tissue mass in the perineal fat adjacent to the right ischium, with no bone involvement (Fig. 1A). Because of the patient’s medical history and the CT results we decided, in consultation with her physician, to proceed to core needle biopsy of this pelvic lesion under CT guidance (informed consent was obtained).
A Soft tissue mass in the perineal fat (arrows), adjacent to the right ischium. B CT-guided placement of RFA needle (curved arrow) at the center of the lesion (arrows). C Contrast-enhanced CT scan immediately after the RFA session shows no contrast uptake by the lesion (arrows). D Contrast-enhanced CT scan at 12 month follow-up shows “cystic” transformation and a decrease in size (arrows) of the treated metastasis.
The histologic result showed “metastasis from bladder carcinoma.” Surgical excision and radiation therapy was recommended. Since the patient refused to undergo the recommended therapy, we suggested percutaneous CT-guided RFA as alternative treatment. She accepted and we proceeded to ablation after we obtained her informed consent.
RFA was carried out by a consultant radiologist specializing in biopsies and RFA. Prior to RFA, the patient had screening blood tests that included measurements of the international normalized ratio (INR), partial thromboplastin time (PTT) and platelet count.
The day of the session, 45 min before the procedure, analgesic medical treatment was administered (as the whole procedure was under local anesthesia): one pill of 3 mg bromazepan (Lexotanil, Roche) per os and 0.05 g pethidine hydrochloride intramuscularly.
Using spiral CT (Picker 5000, Philips Medical Systems, The Netherlands) we started the preprocedural CT scan with 5 mm contiguous slices. The patient was placed in the prone position. Once the inlet was chosen we performed local anesthesia (15 ml of 2% lidocaine hydrochloride). We removed the anesthetic needle and cleaned the skin with povidone iodine 10% in preparation for the insertion of the RFA needle. The depth from the skin to the edge of the lesion was calculated from the appropriate CT image. After the patient’s preparation had been completed, one dispersive electrode was applied to her skin.
We used an electrosurgical generator (Electrotom HiTT 106, Berchtold Holding, 78505 Tuttlingen, Germany) and a HiTT (high-frequency induced thermotherapy) needle applicator, perfusable with 0.9% NaCl solution (EZ 703-20: outer diameter 2.0 mm, shaft length 150 mm, electrode length 20 mm). Controls were set according to the manufacturer’s instructions.
The device was inserted from the inlet in a stepwise fashion, while the position of the tip was controlled repeatedly with three contiguous 5 mm CT images. After confirming that the tip of the needle was centered at the lesion (Fig. 1B) we connected the dispersive electrode and the needle to the generator.
RFA energy (50 W), was applied for 10 min, according to the manufacturer’s instructions for the management of this type and size of lesion. When the ablation was finished we checked the necrosis of the ablated lesion with dual-phase spiral CT after intravenous contrast medium administration (Iomeron 300, Bracco, Milan, Italy) (Fig. 1C).
The patient was hospitalized for 24 hr with no complications. Next day she went home with instructions. She had immediate relief of her pain symptoms. The follow-up at 1 and 6 months showed total necrosis of the mass. One year later (Fig. 1D) the patient has no pain and her subjective quality of life has been improved.
Discussion
Cancer is one of the leading causes of death. A great number of patients with cancer develops metastatic disease and over 50% of them have poorly controlled pain [16, 17]. As pain control in these patients is often difficult to achieve, they have a poor quality of life. Many different therapies (chemotherapy, hormonal therapy, radiation therapy, radioisotope therapy and surgery) in conjunction with analgesic medication have been used in an attempt to obtain pain relief. Gangi et al. [18] used percutaneous CT-guided alcohol injection for treatment of painful bone metastases.
According to the literature, percutaneous CT-guided RF ablation can be used as an alternative method for metastatic neoplasm treatment and may provide an alternative to conventional therapies for pain control [11, 12, 19].
The successful therapy of these tumors and the ability to control pain with RFA encouraged us to try this low-cost and minimally invasive method for the treatment of a painful perineal metastasis.
The treated lesion was not infiltrating the rectum. Dual-phase CT scan and colonoscopy revealed no sign of rectal wall involvement. If the metastasis had been in contact with the bowel, a protective technique such as saline injection between the target area and the rectum wall could have been useful in order to avoid possible complications such as bowel necrosis through the heat or fistula formation [20].
Our patient refused radiation therapy and surgical resection. She also could not receive chemotherapy because of poor therapeutic effect and the response to analgesic medication was poor.
It was remarkable that her pain improved immediately after the RFA session. At 1, 6 and 12 month follow-ups she remained free of pain.
In conclusion, we believe that RFA treatment of painful metastatic disease is a promising minimally invasive low-cost method that can provide palliation of painful metastatic lesions. It might be an option for patients who are not candidates for surgery or conventional therapies.
References
M Friedman I Mikityansky A Kam SK Libutti MM Walther Z Neeman JK Locklin BJ Wood (2004) ArticleTitleRadiofrequency ablation of cancer Cardiovasc Intervent Radiol 27 427–434 Occurrence Handle10.1007/s00270-004-0062-0 Occurrence Handle15383844
A Siperstein A Gotomirski (2000) ArticleTitleHistory and technological aspects of radiofrequency thermoablation Cancer J 6 S293–S301
DE Dupuy SN Goldberg (2001) ArticleTitleImage-guided radiofrequency tumor ablation: Challenges and opportunities. Part II J Vasc Interv Radiol 12 1135–1148 Occurrence Handle1:STN:280:DC%2BD3MrjvFWqtw%3D%3D Occurrence Handle11585879
T Livraghi L Solbiati MF Meloni et al. (2003) ArticleTitleTreatment of focal liver tumors with percutaneous radio-frequency ablation: Complications encountered in a multicenter study Radiology 226 441–451
JP McGahan GD Dodd SuffixIII (2001) ArticleTitleRadiofrequency ablation of the liver: Current status AJR Am J Roentgenol 176 3–16 Occurrence Handle11133529
DE Dupuy WW Mayo-Smith GF Abbott et al. (2002) ArticleTitleClinical applications of radio-frequency tumor ablation in the thorax Radiographics 22 IssueID[Special issue] S259–S269 Occurrence Handle12376615
L Thanos S Mylona M Pomoni et al. (2004) ArticleTitlePrimary lung cancer: Treatment with radio-frequency thermal ablation Eur Radiol 14 897–901 Occurrence Handle10.1007/s00330-003-2189-9 Occurrence Handle1:STN:280:DC%2BD2c7ps1GktQ%3D%3D Occurrence Handle14666377
DA Gervais FJ McGovern RS Arellano et al. (2003) ArticleTitleRenal cell carcinoma: Clinical experience and technical success with radio-frequency ablation of 42 tumors Radiology 226 417–424
D Schultze CS Morris AD Bhave et al. (2003) ArticleTitleRadiofrequency ablation of renal transitional cell carcinoma with protective cold saline infusion J Vasc Interv Radiol 14 489–492 Occurrence Handle12682207
EM Merkle JR Shonk L Zheng et al. (2001) ArticleTitleMR imaging-guided radiofrequency thermal ablation in the porcine brain at 0.2 T Eur Radiol 11 884–892 Occurrence Handle10.1007/s003300000626 Occurrence Handle1:STN:280:DC%2BD38%2FhtVKhtA%3D%3D Occurrence Handle11372628
DI Rosenthal FJ Hornicek M Torriani et al. (2003) ArticleTitleOsteoid osteoma: Percutaneous treatment with radiofrequency energy Radiology 229 171–175
MR Callstrom JW Charboneau MP Goetz et al. (2002) ArticleTitlePainful metastases involving bone: Feasibility of percutaneous CT- and US-guided radio-frequency ablation Radiology 224 87–97
JK Erickson DI Rosenthal DJ Zaleske et al. (2001) ArticleTitlePrimary treatment of chondroblastoma with percutaneous radio-frequency heat ablation: Report of three cases Radiology 221 463–468
C Selli CA Scott F Garbagnati et al. (2001) ArticleTitleTransurethral radiofrequency thermal ablation of prostatic tissue: A feasibility study in humans Urology 57 78–82
K Dowlat D Robinson B Schwartzberg (2001) ArticleTitleImage-guided percutaneous breast cancer ablation meeting at the American Society of Breast Surgeons Am J Surg 182 429–433 Occurrence Handle10.1016/S0002-9610(01)00768-1 Occurrence Handle1:STN:280:DC%2BD3Mnps1Glsg%3D%3D Occurrence Handle11720686
CS Cleeland R Gonin AK Hatfield et al. (1994) ArticleTitlePain and its treatment in outpatients with metastatic cancer N Engl J Med 330 592–596 Occurrence Handle10.1056/NEJM199403033300902 Occurrence Handle1:STN:280:ByuC3svhsVU%3D Occurrence Handle7508092
A Jacox DB Carr R Payne et al. (1994) ArticleTitleNew clinical-practice guidelines for the management of pain in patients with cancer N Engl J Med 330 651–655 Occurrence Handle10.1056/NEJM199403033300926 Occurrence Handle1:STN:280:ByuC3svhs1M%3D Occurrence Handle7508094
A Gangi B Kastler A Klinkert et al. (1994) ArticleTitleInjection of alcohol into bone metastases under CT guidance J Comput Assist Tomogr 18 932–935 Occurrence Handle1:STN:280:ByqD2M7ot1M%3D Occurrence Handle7962803
GM Vanderschueren AH Taminiau WR Obermann et al. (2002) ArticleTitleOsteoid osteoma: Clinical results with thermocoagulation Radiology 224 82–86
BS Kapoor DW Hunter (2003) ArticleTitleInjection of subphrenic saline during radiofrequency ablation to minimize diaphragmatic injury Cardiovasc Intervent Radiol 26 302–304 Occurrence Handle10.1007/s00270-003-2704-z Occurrence Handle14562984
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thanos, L., Mylona, S., Kalioras, V. et al. Palliation of Painful Perineal Metastasis Treated with Radiofrequency Thermal Ablation. Cardiovasc Intervent Radiol 28, 381–383 (2005). https://doi.org/10.1007/s00270-004-9250-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-004-9250-1