Abstract
Vigilance is commonly used to assess anti-predator behavior; however, the majority of studies assess only high-cost vigilance, which interrupts any other activities. Low-cost vigilance, by comparison, allows animals to be vigilant while engaged in other activities, thereby reducing the cost of vigilance. Here, we investigate the use of high- and low-cost vigilance in relation to environmental and temporal factors in a wild population of the bat-eared fox (Otocyon megalotis), a small mesopredator, using behavioral observations of eight habituated animals. We hypothesized that vigilance would increase with tall, dense vegetation, high wind speeds, and low ambient light, and that vigilance would decrease in the presence of conspecifics and over the period of a single observation session. Overall, bat-eared foxes spent significantly more time engaged in low-cost vigilance than in high-cost vigilance. High-cost vigilance increased with vegetation height, in the presence of adult conspecifics and in winter. We suggest these effects are due to impeded lines of sight with increases in vegetation height, increased competition in the presence of conspecifics, and increased social interactions in winter. Low-cost vigilance was highest at the start of observational sessions, and we suggest that this is due to observer effects, as foxes demonstrated increases in both high- and low-cost vigilance specifically towards observers during this time. This study demonstrates that foxes are highly dynamic in their vigilance behavior. Due to the low predation pressure in the study area, the use of mostly low-cost vigilance may be sufficient in this environment, and thus foxes can allocate more time to other activities. Our results indicate that, in this environment, vigilance is most likely driven by factors other than predation risk, but that foxes may still respond to certain cues of predation risk. Ultimately, in areas of low predation risk, species susceptible to predation may retain responses to cues of risk, but adapt behaviors to reduce the associated costs.
Significance statement
While studies examining perceived risk in herbivores frequently assess vigilance, the examination of vigilance is largely under-investigated in mesopredators. Here, we present findings from one of the first studies to examine high- and low-cost vigilance in a small mesopredator. We demonstrate that in an area with minimal predation pressure low-cost vigilance is more intensively used and that despite the absence of predator sightings, ambush predators may still pose a perceived threat to foxes. Ultimately, when predation pressure is low, social interactions and competition may be some of the costliest activities. Results further indicate that while this small canid habituates quickly to observer presence, the initial presence of observers cannot be considered truly neutral.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Foraging theory predicts that animals strive to find the optimal balance in the trade-off between foraging success and predator avoidance (Lima and Dill 1990). Typically, this trade-off can vary depending on food type and availability (Beauchamp 2009), predation risk (Lima and Bednekoff 1999), social dynamics (Elgar 1989; le Roux et al. 2009), vegetation type (Tchabovsky et al. 2001; Devereux et al. 2006), and abiotic factors such as ambient illumination, temperature and wind speed (Elgar 1989; Carr and Lima 2010; Prugh and Golden 2014). Both vegetation type and abiotic variables can impede visual and auditory senses and have been shown to be important in shaping perceived risk (Biebouw and Blumstein 2003; Hayes and Huntly 2005; Prugh and Golden 2014).
Vigilance levels continue to be a common way of assessing perceived risk (le Roux et al. 2009; Périquet et al. 2010). However, most studies focus on high-cost vigilance, or vigilance that interrupts other activities such as foraging (Lima and Bednekoff 1999). A small number of studies have additionally investigated low-cost vigilance, which enables other behaviors to continue while an animal is scanning for potential threats (Unck et al. 2009; Périquet et al. 2012). Being able to remain vigilant while engaged in other behaviors considerably reduces the cost of vigilance, as there is no temporal trade-off between the two behavior types (Illius and Fitzgibbon 1994; Bednekoff and Lima 2005). High-cost vigilance, however, has been shown to be more important in areas of high predation risk (Unck et al. 2009).
Mesopredators occupy a trophic position in which they are not only predators, but are also prey to or killed by larger predators (Ritchie and Johnson 2009). Therefore, mesopredator vigilance patterns might be affected by perceived predation risk, however studies remain rare, and almost no research exists on high- and low-cost vigilance in mesopredators (but see le Roux et al. 2009). For social mesopredators, individuals must balance detection of predators and prey as well as finding mates or conspecifics.
Bat-eared foxes (Otocyon megalotis, “foxes” hereafter) are small (3–5 kg), socially monogamous, largely nocturnal mesopredators that feed predominantly on termites, but also on other insects, rodents and fruits (Nel 1990; Maas and Macdonald 2004). Foxes have both diurnal and nocturnal predators, including terrestrial species such as black-backed jackal (Canis mesomelas), and avian threats, such as martial eagle (Polemaetus bellicosus; Malcolm 1986), and are common victims in interspecific killings (Palomares and Caro 1999). Previous studies have shown that foxes exhibit anti-predator responses in terms of social behavior (e.g. mobbing) and home-range use (Kamler et al. 2012, 2013), but their vigilance patterns remain unclear (Welch et al. 2017).
This study aims to investigate high- and low-cost vigilance in foxes, specifically focusing on factors potentially influencing vigilance: perceived predation risk, social factors, and observer effects. If perceived predation risk has an effect, we expect vegetation height and density (impaired visibility in dense and high vegetation; Devereux et al. 2006; Embar et al. 2011; Emerson et al. 2011), moon illumination (impaired visibility during new moon nights; Biebouw and Blumstein 2003) and wind (impaired hearing with high wind speeds; Hayes and Huntly 2005) to have an effect on vigilance. Social factors can also influence vigilance, whereby the presence of conspecifics has been demonstrated to either increase or decrease individual vigilance, due to competition (Shrader et al. 2007) and the ‘dilution effect’ (Delm 1990) respectively. Bat-eared foxes are known as social foragers (Lamprecht 1979), and we would therefore expect that the presence of conspecifics should lower individual vigilance rather than increasing it. Observers have been shown to influence perceived risk in even habituated animals (Nowak et al. 2014), and in the case of foxes, the presence of observers appears to reduce perceived risk (Welch et al. 2017). However, fine-scale analysis of vigilance patterns might reveal changes throughout the observational period, and foxes should be expected to gradually habituate to the presence of an observer over the course of an evening.
Specifically, we predicted that vigilance would increase (1) in higher, denser vegetation, (2) in high wind conditions and (3) in lower levels of ambient light. As foxes are social foragers, we hypothesized that vigilance would decrease (4) in the presence of conspecifics and (5) at times of the year when social interactions were more frequent. In addition, we expected that observer effects would be evident in (6) a gradual decrease in observer-directed vigilance over the course of a single observation session.
Methods
Study site and species
Behavioral observations were conducted at the Kuruman River Reserve (KRR, 28° 59′ S, 21° 49′ E) in the southern Kalahari Desert, Northern Cape, South Africa, on a wild but habituated (prior to the start of this study) population of foxes. The climate in the region is distinguished by a hot, wet summer period (October–April) and cold, dry winters (May–September). The average annual rainfall for this area is 264 mm (2010–2015, KRR weather station). Based on the descriptions of Low and Rebelo (1996), the vegetation classification for this area is Kalahari Thornveld, and consists of three principal habitats, namely dune areas comprised of undulating dunes and perennial grasses (Aristida, Eragrostis, Schmidti and Stipagrostis spp.), flat river terraces with two predominant species, perdebos (Galenia africana) and driedoring (Rhigozum trichotomum), and dry riverbed, which is predominantly open habitat, interspersed with small camel thorn (Acacia erioloba) and black thorn (Acacia mellifera) bushes.
Foxes are predominantly nocturnal and are relatively common in the study area (several individuals seen on a daily basis). Most large predators were historically extirpated from the area. Black-backed jackals and caracals (Caracal caracal) remain in the area, but are hunted on the surrounding farmlands. Thus, predation pressure in the study area was low, and black-backed jackals and caracals are rarely seen at the study site (3 per annum and 2 per annum respectively, based on unpublished data from 2015) despite a large sampling effort (Kalahari Meerkat Project 2017). Recorded mortality events of study individuals was as a result of disease (1 confirmed case, 2 suspected). Another predominant cause of death in the area is due to road accidents (RJW, pers. obs.). We recorded no mortality events due to predation.
Behavioral data
Foxes were habituated to observers and followed on foot from approximately 1–2 m while collecting behavioral data (c.f. le Roux et al. 2009). Foxes were collared and observers radio-tracked individuals to find their location prior to behavioral investigations. Observers followed each fox once a week for a period of 2 h (“follows” hereafter). Monthly ‘all-night follows’ were done where reiterative 2-h follows were conducted throughout the night with 1-h rest periods in between. A total of 164 follows were completed on eight adult foxes (five males, three females) between August 2015 and April 2016. On average, 18 follows were conducted per individual (range 6–48). Follow data were collected directly on a Samsung handheld tablet using the program Cybertracker (http://www.cybertracker.org).
To assess anti-predator behavior, we conducted 10-min vigilance focals (“focals” hereafter; c.f. Favreau et al. 2014) before and after every follow. A total of 270 focals were completed for the aforementioned eight foxes between August 2015 and April 2016. On average, 34 focals were conducted per individual (ranging from 6 to 77). Vigilance focal data were collected using voice recordings that were later transcribed and collated. Focals focused solely on vigilance behavior (bout rate and duration). The possible cause of vigilance (observer, social i.e. other foxes in the vicinity, potential predator, or unknown), the presence of conspecifics (no conspecifics, habituated individuals, unhabituated individuals or pups), and the type of vigilance (high cost—individuals stopped any other activity to be vigilant; low cost—vigilance while engaged in other activities like movement or foraging) were recorded (c.f. Unck et al. 2009). For this study, all high-cost vigilance events interrupted foraging, and low-cost vigilance occurred simultaneously with movement behaviors predominantly, but also marking behaviors, grooming and consuming large prey items. All food-motivated vigilance events (i.e. clearly listening/looking for food) were discarded. Additionally, focals were assigned a type, either ‘pre’ or ‘post’ follow. Pre-focals were recorded at the beginning of observation periods before the follow (approximately 1830 h–2000 h) and post-focals were recorded after follows. On nights where data were collected throughout the night, all vigilance focals after the first follow were categorized as ‘post’. Despite differences in the time of night at which they were recorded, post-focals did not differ in proportion of time vigilant (Kruskal-Wallis: χ2 = 0.450, df = 3, P = 0.930) or rate of vigilance (χ2 = 1.415, df = 3, P = 0.702) and therefore were pooled as post-focal data. Focals shorter than 5 min were discarded from analyses. During the focals, foxes were in constant line of sight and if they were out of sight for more than 1 min, the focal was restarted. It was not possible to record data blind because our study involved focal animals in the field.
Vegetation effects
In order to assess the effects of vegetation height and density on vigilance, vegetation data were collected for every vigilant event and additionally for non-vigilant events (when foxes were foraging). Vegetation density was categorized into ‘sparse’ (bare ground or roots of perennial species more than 1 m apart) or ‘dense’ (roots of perennial species less than 50 cm and up to 1 m; c.f. Périquet and le Roux 2017). Vegetation height was categorized into ‘low’ (lower than the eyeline of a moving fox) or ‘high’ (higher than the eyeline of a moving fox). Vegetation data were collected during focals for each vigilant event. For non-vigilant events, five randomly selected non-vigilant events were selected post-hoc from follow data associated with the focal (i.e. on the same night). These points had associated GPS locations (recorded during the follow) and these locations were visited the following day to describe vegetation using the methods above.
Abiotic factors
All focals were assigned environmental variables (moon illumination index and wind speed) and a temporal variable (season). A moon illumination index (0–1) was obtained from http://aa.usno.navy.mil/data/docs/MoonFraction.php for our study location. For each focal, we assigned a moon illumination index equal to the moon illumination if the moon was above the horizon at the time of the observation and zero otherwise. Nights with full cloud cover were removed from analyses, as this could have confounded moon illumination. Wind speeds (0–6 m/s) were extracted from data collected every hour at a weather station on site. Seasons were categorized as spring (September–November), summer (December–February), autumn (March–May) and winter (June–August).
Statistical analyses
All analyses were performed in R (R Core Team 2016), using the lme4 package (Bates et al. 2015). Data exploration was carried out and model assumptions verified by following the protocol described in Zuur et al. (2010) and Zuur and Ieno (2016). Graphs were created using the ggplot2 package (Wickham 2009) in R and report actual data rather than predicted model results. All graphs show mean and standard error unless stated otherwise.
Behavioral data
To investigate whether foxes spent a greater proportion of their vigilance time invested in low-cost vigilance, we used a chi-square goodness of fit test. Expected proportions were based on the assumption that vigilance would be divided evenly between low- and high-cost vigilance.
Vegetation effects
To assess how the probability of high- and low-cost vigilance changed with vegetation height and density, we used generalized linear mixed effects models (GLMMs) with binomial error distribution and a logit link (logistic regression) with the response variable being set to vigilant (1), or not vigilant (0). Vegetation height (high or low) and vegetation density (dense or sparse) were set as main effects. Individual fox was included as a random effect.
Abiotic factors
We used GLMMs with a binomial error distribution and a logit link to investigate how time spent in high-cost vigilance (as a proportion of focal time) varied with moon illumination, wind speed, season and focal type. Individual fox was included as a random effect. Interactions between variables were investigated, however these were non-significant and therefore removed from models. The analyses were repeated for low-cost vigilance. We used GLMMs with a Poisson error distribution and a log link to investigate how the rate of high- and low-cost vigilance varied with the same abiotic factors as already described. To correct for over-dispersion an observation level random effect was added (Elston et al. 2001; Harrison 2014).
Social effects
To compare how proportion of time spent in high- and low-cost vigilance, and high- and low-cost vigilance rate varied with the presence of conspecifics, we selected a random sample of 20 vigilance focals with no conspecifics present and compared them to vigilance focals with habituated conspecifics present (n = 16), unhabituated conspecifics present (n = 14) and pups present (n = 21), using a Kruskal-Wallis test (as data were not normally distributed). Post-hoc analyses were applied using Wilcoxon rank-sum tests for each pairwise combination and a Bonferroni correction was applied.
Observer-directed vigilance
We used GLMMs with a binomial error distribution and a logit link to investigate how both high- and low-cost observer-directed vigilance (as a proportion of focal time) varied with focal type including individual fox as a random effect. We used GLMMs with a Poisson error distribution and a log link to investigate how the rate of high- and low-cost observer directed vigilance varied with focal type including individual fox as a random effect. To assist with over-dispersion an observation level random effect was added (Elston et al. 2001; Harrison 2014).
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Results
Behavioral data
A total of 270 vigilance focals were recorded, including 140 pre-focals and 130 post-focals. Within these focals, 1526 vigilant events were documented, to which 63% the cause was unknown, 11% of events were caused by conspecifics, 23% were caused by human observers and the remaining 3% were caused by non-threatening heterospecific species (e.g. common duiker, Sylvicapra grimmia). Foxes spent a significantly higher proportion of time invested in low-cost vigilance than in high-cost vigilance (χ2 = 6.76, df = 1, P < 0.01), with high-cost vigilance accounting for only 37% of time spent vigilant.
Vegetation effects
The probability of engaging in high-cost vigilance decreased in low vegetation, but vegetation density had no effect (Table 1). Neither vegetation height nor density had an effect on the probability of engaging in low-cost vigilance (Table 1).
Abiotic factors
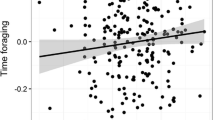
Moon illumination and wind speed had no effect on the proportion of time spent in either high- or low-cost vigilance (Table 1). Season had no impact on the proportion of time spent in low-cost vigilance, but the proportion of time spent in high-cost vigilance was lower in spring than in winter (Table 1). The proportion of time spent engaged in low-cost, but not high-cost, vigilance was greater for pre-focals when compared to post-focals (Table 1; Fig. 1).
Moon illumination and wind speed had no effect on high- or low-cost vigilance rate. Season had no effect on low-cost vigilance rate, but high-cost vigilance rate was lower in spring, summer and autumn than in winter (Table 1, Fig. 2). High-cost vigilance rate was not affected by focal type, but the rate of low-cost vigilance was greater for pre-focals (0.0075 ± 0.0009, mean ± standard error) when compared to post-focals (0.0030 ± 0.0005; Table 1).
Social effects
The proportion of time invested in high- (χ2 = 6.606, df = 3, P = 0.856) and low- (χ2 = 5.033, df = 3, P = 0.169) cost vigilance did not vary among different social contexts. However, high-cost vigilance rate varied significantly between different social contexts (χ2 = 14.64, df = 3, P < 0.01), being highest in the presence of habituated conspecifics (0.011 ± 0.006) vs. no conspecifics (0.003 ± 0.005; W = 277, df = 34, P < 0.001), and vs. pups (0.002 ± 0.005; W = 262, df = 35, P < 0.01, Fig. 3a). There was no difference in low-cost vigilance rate among the different social contexts (habituated conspecifics 0.002 ± 0.008, unhabituated conspecifics 0.002 ± 0.003, pups 0.008 ± 0.012 and alone 0.005 ± 0.015, χ2 = 3.425, df = 3, P = 0.331, Fig. 3b).
Bat-eared fox a high-cost vigilance rate and b low-cost vigilance rate in the presence and absence of conspecifics. Plots represent medians with interquartile range, whiskers represent 1.5 × interquartile range and outliers are points outside this range, Y axes are truncated so data are presented within text
Observer-directed vigilance
The proportion of time engaged in high-cost observer-directed vigilance did not vary between the two focal types. High-cost observer-directed vigilance rate was higher in the pre-focal (Table 1; Fig. 4). The proportion of time engaged in low-cost observer-directed vigilance was higher in the pre-focal (0.0080 ± 0.0015 vs 0.0026 ± 0.0013; Table 1), as was low-cost observer-directed vigilance rate (Table 1; Fig. 4).
Discussion
Our results showed that bat-eared foxes invested most of their vigilance effort into low-cost vigilance. High-cost vigilance has been shown to be more important in areas where perceived risk is greater (Unck et al. 2009), and therefore the use of predominantly low-cost vigilance may indicate that perceived risk is generally low in this environment, as suggested by the absence of any recorded mortalities due to predation. Due to low predation pressure, low-cost vigilance may be sufficient, and thus foxes can allocate more time to foraging. Foxes are unlikely to have lost appropriate anti-predator behaviors altogether, despite low predation pressure, and the use of high-cost vigilance under certain situations (in tall vegetation, in the presence of adult conspecifics, during winter and towards observers at the start of observation sessions) may, however, suggest these factors are associated with greater perceived risk.
Tall vegetation significantly increased the probability of high-cost vigilance, but not of low-cost vigilance, which suggests bat-eared foxes do not associate areas of tall vegetation with sites of refuge, as found for other fox species (Hall et al. 2013). As vegetation height categories were associated with line of sight, we consider that greater vigilance in higher vegetation is due to obscured line of sight and thus difficulty in detecting potential dangers, and similar patterns have been shown in other studies (Bednekoff and Blumstein 2009). Increased vigilance in tall vegetation suggests that, despite the lack of potential predator sightings, ambush predators may still pose as a perceived threat to foxes, or that foxes still respond to certain cues of predation risk, despite predators being largely absent. Previous studies have highlighted that even after the loss of predators, prey may still retain appropriate anti-predator behaviors (Blumstein 2006). Increases in vigilance with vegetation height may also have been associated with the presence of hidden conspecifics.
Previous studies have reported that both moon illumination and wind speed affect perceived risk of predation in certain species e.g. tammar wallabies (Macropus eugenii, Biebouw and Blumstein 2003) and brush-tailed rock-wallabies (Petrogale penicillata, Carter and Goldizen 2003). Additionally, recent research suggests that foxes rely on hearing as their predominant sense (Renda and le Roux 2017), but that wind speed does not influence their ability to detect prey (S. Renda et al. unpubl. data). We therefore suggest the detection of predators may similarly be based on auditory processes but that the generally low wind speeds (0–6 km/h) recorded during our observations were not enough to significantly impair their capability of detecting potential predators.
High-cost vigilance rate, but not low-cost, was influenced by social factors and foxes were more vigilant in the presence of habituated conspecifics than they were when foraging alone or with pups. Previous work has highlighted that increased vigilance in the presence of conspecifics may be related to competition over food and conspecific monitoring (Beauchamp 2001, 2009; Lung and Childress 2007; Shrader et al. 2007), and social vigilance has been highlighted to be particularly pertinent in areas of low predation pressure (Hirsch 2002). Lower levels of vigilance in the presence of pups suggests that predation risk may not be the biggest driver of vigilance in areas with low predation pressure. If predation risk had posed a considerable threat, we would have expected increased vigilance in the presence of pups as demonstrated in previous studies (Lung and Childress 2007; Lashley et al. 2014). We suggest that pups did not compete over food with adults, as adults regularly provisioned them with food, and their survival influences adult fitness (Trivers 1972). Additionally, our results showed vigilance was greater in winter. We suggest this may be related to increased social interactions during this time (R. De Bruin et al. unpubl. data). Bat-eared foxes are monogamous and pair bonding occurs during winter months (Nel 1984), with pairs foraging together to facilitate mate guarding (Wright et al. 2010). We therefore suggest that foxes may perceive greater resource competition during this time and increase vigilance in the presence of conspecifics for mate guarding/seeking extra pair mating, or possibly due to increased competition in the presence of conspecifics (Beauchamp 2001). Unfortunately, as most partners were unhabituated at this study site, pairs were rarely observed foraging together in our presence making the validation of this hypothesis difficult with our data. We therefore recommend that future studies test this idea with pairs of habituated foxes. Furthermore, our results indicated that winter was the only season where high-cost was greater than low-cost vigilance and we suggest this is due to greater day-time activity and foraging during this season (Lourens and Nel 1990). Our results indicate that mate association or competition may be some of the costliest activities in an environment with low predation pressure.
Greater low-cost vigilance was observed during the pre-focal (at the start of observation sessions, before the follow) compared to post-focals. Other studies have indeed reported that time of day or night changes in vigilance patterns can be attributed to predation risk in birds, in areas where predation risk is evident (McNamara and Houston 1986), and in marsupials, in areas where predation risk is not evident e.g. quokkas (Setonix brachyurus, Blumstein et al. 2001). At our study site, large predators are absent, and jackals and caracals are persecuted in the surrounding areas, therefore presence is extremely rare. This implies that the observed increased vigilance early in the night may be related to fox cautiousness towards observers. Both high- and low-cost observer-directed vigilance was greater during the pre-focal than the post-focal, indicating that our initial presence certainly had an effect on foxes. The presence of observers close to habituated animals is said to be neither positive nor negative, but studies have highlighted that observer effects may indeed occur, albeit subtle and difficult to detect (McDougall 2012; Nowak et al. 2014). Therefore, the general increase in low-cost vigilance observed in pre-follow focals may have been an artifact of our presence, as foxes may have been generally wary in our presence initially and subsequently relaxed with time (McDougall 2012). Previous studies have demonstrated that the presence of observers on habituated animals may or may not directly alter the behaviors being assessed (Crofoot et al. 2010; McDougall 2012), and this is something that should be considered when interpreting the results from this study.
In conclusion, our study reveals that in areas with low predation pressure, vigilance behavior is concentrated towards low-cost vigilance, with the use of high-cost vigilance under certain conditions. This study demonstrates that vigilance can indeed be investigated in habituated animals, but that care must be taken to ensure that data collection commences at a point when animals are fully relaxed around human observers. Previous studies have demonstrated that species can retain appropriate anti-predator behaviors long after the loss of predators from an ecosystem (Blumstein 2006), and thus vigilance is unlikely to be completely lost in an environment with low (but possible) predation risk. Mesocarnivores (and prey in general), in areas of low predation risk, may retain responses to certain cues of risk, but may adapt behaviors to reduce the associated costs. Future studies should aim to assess the use of high- and low-cost vigilance for foxes in areas with greater predation risk to assess whether the partitioning of time to each type of vigilance differs from this study, and evaluate how high- and low-cost vigilance may vary with spatio-temporal factors. We conclude that the drivers of vigilance in areas with low predation risk may be more related to factors other than predation risk, such as the presence of conspecifics and observers, but that foxes may still respond to certain cues of predation risk.
References
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48
Beauchamp G (2001) Should vigilance always decrease with group size? Behav Ecol Sociobiol 51(1):47–52. https://doi.org/10.1007/s002650100413
Beauchamp G (2009) How does food density influence vigilance in birds and mammals? Anim Behav 78(2):223–231. https://doi.org/10.1016/j.anbehav.2009.04.029
Bednekoff PA, Blumstein DT (2009) Peripheral obstructions influence marmot vigilance: integrating observational and experimental results. Behav Ecol 20(5):1111–1117. https://doi.org/10.1093/beheco/arp104
Bednekoff PA, Lima SL (2005) Testing for peripheral vigilance: do birds value what they see when not overtly vigilant? Anim Behav 69(5):1165–1171. https://doi.org/10.1016/j.anbehav.2004.07.020
Biebouw K, Blumstein DT (2003) Tammar wallabies (Macropus eugenii) associate safety with higher levels of nocturnal illumination. Ethol Ecol Evol 15(2):159–172. https://doi.org/10.1080/08927014.2003.9522680
Blumstein DT (2006) The multipredator hypothesis and the evolutionary persistence of antipredator behaviour. Ethology 112(3):209–217. https://doi.org/10.1111/j.1439-0310.2006.01209.x
Blumstein DT, Daniel JC, McLean IG (2001) Group size effects in quokkas. Aust J Zool 49(6):641–649. https://doi.org/10.1071/ZO01032
Carr JM, Lima SL (2010) High wind speeds decrease the responsiveness of birds to potentially threatening moving stimuli. Anim Behav 80(2):215–220. https://doi.org/10.1016/j.anbehav.2010.04.021
Carter K, Goldizen AW (2003) Habitat choice and vigilance behaviour of brush-tailed rock-wallabies (Petrogale penicillata) within their nocturnal foraging ranges. Wildlife Res 30(4):355–364. https://doi.org/10.1071/WR02095
Crofoot MC, Lambert TD, Kays R, Wikelski MC (2010) Does watching a monkey change its behaviour? Quantifying observer effects in habituated wild primates using automated radiotelemetry. Anim Behav 80(3):475–480. https://doi.org/10.1016/j.anbehav.2010.06.006
Delm MM (1990) Vigilance for predators: detection and dilution effects. Behav Ecol Sociobiol 26:337–342
Devereux CL, Whittingham MJ, Fernández-Juricic E, Vickery JA, Krebs JR (2006) Predator detection and avoidance by starlings under differing scenarios of predation risk. Behav Ecol 17(2):303–309. https://doi.org/10.1093/beheco/arj032
Elgar MA (1989) Predator vigilance and group size in mammals and birds: a critical review of the empirical evidence. Biol Rev 64(1):13–33. https://doi.org/10.1111/j.1469-185X.1989.tb00636.x
Elston DA, Moss R, Boulinier T, Arrowsmith C, Lambin X (2001) Analysis of aggregation, a worked example: numbers of ticks on red grouse chicks. Parasitology 122(Pt 5):563–569
Embar K, Kotler BP, Mukherjee S (2011) Risk management in optimal foragers: the effect of sightlines and predator type on patch use, time allocation, and vigilance in gerbils. Oikos 120(11):1657–1666. https://doi.org/10.1111/j.1600-0706.2011.19278.x
Emerson SE, Brown JS, Linden JD (2011) Identifying Sykes’ monkeys’, Cercopithecus albogularis erythrarchus, axes of fear through patch use. Anim Behav 81(2):455–462. https://doi.org/10.1016/j.anbehav.2010.11.018
Favreau F-R, Goldizen AW, Fritz H, Blomberg SP, Best EC, Pays O (2014) Within-population differences in personality and plasticity in the trade-off between vigilance and foraging in kangaroos. Anim Behav 92:175–184. https://doi.org/10.1016/j.anbehav.2014.04.003
Hall LK, Day CC, Westover MD, Edgel RJ, Larsen RT, Knight RN, McMillan BR (2013) Vigilance of kit foxes at water sources: a test of competing hypotheses for a solitary carnivore subject to predation. Behav Process 94:76–82. https://doi.org/10.1016/j.beproc.2012.12.007
Harrison XA (2014) Using observation-level random effects to model overdispersion in count data in ecology and evolution. PeerJ 2:e616. https://doi.org/10.7717/peerj.616
Hayes AR, Huntly NJ (2005) Effects of wind on the behaviour and call transmission of pikas (Ochotona princeps). J Mammal 86(5):974–981. https://doi.org/10.1644/1545-1542(2005)86[974:EOWOTB]2.0.CO;2
Hirsch BT (2002) Social monitoring and vigilance behavior in brown capuchin monkeys (Cebus apella). Behav Ecol Sociobiol 52(6):458–464. https://doi.org/10.1007/s00265-002-0536-5
Illius AW, Fitzgibbon C (1994) Costs of vigilance in foraging ungulates. Anim Behav 47(2):481–484. https://doi.org/10.1006/anbe.1994.1067
Kalahari Meerkat Project (2017) Volunteer FAQ. Nirvana and Wordpress, http://kalahari-meerkats.com/kmp/
Kamler JF, Stenkewitz U, Klare U, Jacobsen NF, Macdonald DW (2012) Resource partitioning among cape foxes, bat-eared foxes and black-backed jackals in South Africa. J Wildl Manag 76:1241–1253
Kamler JF, Stenkewitz U, Macdonald DW (2013) Lethal and sublethal effects of black-backed jackals on cape foxes and bat-eared foxes. J Mammal 94(2):295–306. https://doi.org/10.1644/12-MAMM-A-122.1
Lamprecht J (1979) Field observations on the behaviour and social system of the bat-eared fox Otocyon megalotis Desmarest. Ethology 49:260–284
Lashley MA, Chitwood MC, Biggerstaff MT, Morina DL, Moorman CE, DePerno CS (2014) White-tailed deer vigilance: the influence of social and environmental factors. PLoS One 9(3):e90652. https://doi.org/10.1371/journal.pone.0090652
le Roux A, Cherry MI, Gygax L, Manser MB (2009) Vigilance behaviour and fitness consequences: comparing a solitary foraging and an obligate group-foraging mammal. Behav Ecol Sociobiol 63(8):1097–1107. https://doi.org/10.1007/s00265-009-0762-1
Lima SL, Bednekoff PA (1999) Back to the basics of antipredatory vigilance: can nonvigilant animals detect attack? Anim Behav 58(3):537–543. https://doi.org/10.1006/anbe.1999.1182
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68(4):619–640. https://doi.org/10.1139/z90-092
Lourens S, Nel JAJ (1990) Winter activity of bat-eared foxes Otocyon megalotis on the Cape West coast. S Afr J Zool 25(2):124–132. https://doi.org/10.1080/02541858.1990.11448200
Low AB, Rebelo AG (1996) Vegetation of Southern Africa, Lesotho and Swaziland. Department of Environmental Affairs and Tourism, Pretoria
Lung MA, Childress MJ (2007) The influence of conspecifics and predation risk on the vigilance of elk (Cervus elaphus) in Yellowstone National Park. Behav Ecol 18:12–20
Maas B, Macdonald DW (2004) Bat-eared foxes ‘insectivory’ and luck: lessons from an extreme canid. In: Macdonald DW, Sillero-Zubiri C (eds) The biology and conservation of wild canids. Oxford University Press, Oxford, pp 227–242. https://doi.org/10.1093/acprof:oso/9780198515562.003.0014
Malcolm JR (1986) Socio-ecology of bat-eared foxes (Otocyon megalotis). J Zool 208:457–467
McDougall P (2012) Is passive observation of habituated animals truly passive? J Ethol 30(2):219–223. https://doi.org/10.1007/s10164-011-0313-x
McNamara JM, Houston AI (1986) The common currency for behavioral decisions. Am Nat 127(3):358–378. https://doi.org/10.1086/284489
Nel JAJ (1984) Behavioural ecology of canids in the South-Western Kalahari. Koedoe 27:229–235
Nel JAJ (1990) Foraging and feeding by bat-eared foxed Otocyon megalotis in the southwestern Kalahari. Koedoe 33:9–16
Nowak K, le Roux A, Richards SA, Scheijen CPJ, Hill RA (2014) Human observers impact habituated samango monkeys’ perceived landscape of fear. Behav Ecol 25(5):1199–1204. https://doi.org/10.1093/beheco/aru110
Palomares F, Caro TM (1999) Interspecific killing among mammalian carnivores. Am Nat 153(5):492–508. https://doi.org/10.1086/303189
Périquet S, le Roux A (2017) Seasonal patterns of habitat selection in the insectivorous bat-eared fox. Afr J Ecol published online. https://doi.org/10.1111/aje.12492
Périquet S, Valeix M, Loveridge AJ, Madzikanda H, Macdonald DW, Fritz H (2010) Individual vigilance of African herbivores while drinking: the role of immediate predation risk and context. Anim Behav 79(3):665–671. https://doi.org/10.1016/j.anbehav.2009.12.016
Périquet S, Todd-Jones L, Valeix M, Stapelkamp B, Elliot N, Wijers M, Pays O, Fortin D, Madzikanda H, Fritz H, Macdonald DW, Loveridge AJ (2012) Influence of immediate predation risk by lions on the vigilance of prey of different body size. Behav Ecol 23(5):970–976. https://doi.org/10.1093/beheco/ars060
Prugh LR, Golden CD (2014) Does moonlight increase predation risk? Meta-analysis reveals divergent responses of nocturnal mammals to lunar cycles. J Anim Ecol 83(2):504–514. https://doi.org/10.1111/1365-2656.12148
R development core team (2016) R: a language and environment for statistical computing, version 3.0.2. R Foundation for Statistical Computing, Vienna http://www.R-project.org
Renda S, le Roux A (2017) The sensory ecology of prey detection in the bat-eared fox (Otocyon megalotis). Behaviour 154(2):227–240. https://doi.org/10.1163/1568539X-00003419
Ritchie EG, Johnson CN (2009) Predator interactions, mesopredator release and biodiversity conservation. Ecol Lett 12(9):982–998. https://doi.org/10.1111/j.1461-0248.2009.01347.x
Shrader AM, Kerley GIH, Kotler BP, Brown JS (2007) Social information, social feeding, and competition in group-living goats (Capra hircus). Behav Ecol 18(1):103–107. https://doi.org/10.1093/beheco/arl057
Tchabovsky AV, Krasnov BR, Khokhlova IS, Shenbrot GI (2001) The effect of vegetation cover on vigilance and foraging tactics in the fat sand rat Psammomys obesus. J Ethol 19(2):105–113. https://doi.org/10.1007/s101640170006
Trivers RL (1972) Parental investment and sexual selection. In: Campbell B (ed) Sexual selection and the descent of man. Aldine-Atherton, Chicago, pp 136–179
Unck CE, Waterman JM, Verburgt L, Bateman PW (2009) Quantity versus quality: how does level of predation threat affect Cape ground squirrel vigilance? Anim Behav 78(3):625–632. https://doi.org/10.1016/j.anbehav.2009.05.028
Welch RJ, Périquet S, Petelle MB, le Roux A (2017) Hunter or hunted? Perceptions of risk and reward in a small mesopredator. J Mammal 98(6):1531–1537. https://doi.org/10.1093/jmammal/gyx100
Wickham H (2009) ggplot2: elegant graphics for data analysis. Springer-Verlag, New York. https://doi.org/10.1007/978-0-387-98141-3
Wright HWY, Gray MM, Wayne RK, Woodroffe RB (2010) Mating tactics and paternity in a socially monogamous canid, the bat-eared fox (Otocyon megalotis). J Mammal 91(2):437–446. https://doi.org/10.1644/09-MAMM-A-046.1
Zuur AF, Ieno EN (2016) A protocol for conducting and presenting results of regression-type analyses. Method Ecol Evol 7(6):636–645. https://doi.org/10.1111/2041-210X.12577
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Method Ecol Evol 1(1):3–14. https://doi.org/10.1111/j.2041-210X.2009.00001.x
Acknowledgments
This research was supported by The National Research Foundation (NRF) who awarded grants to AlR (Thuthuka Grant-TTK1206041007) and Fredrik Dalerum (NRF Grant No. 90491). Logistical support and access to conduct research on the reserve was granted by the Kalahari Research Trust, Kuruman River Reserve and Kalahari Meerkat Project (supported by ERC Grant No. 294494 to T. H. Clutton-Brock since 1 July 2012). We thank additional members of the bat-eared fox research team (S. Renda, E. Karslake and K. Jumbam) for help collecting data, R. J. Wasserman for reviewing earlier drafts and two anonymous reviewers for providing useful comments to improve the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All protocols were evaluated by The University of the Free State’s ethical committee and were in accordance with The University of the Free State’s ethical standards protocol under the ethical clearance number 11/2013. The Department of Nature Conservation approved all fieldwork under permit number 476/2/2013.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by C. Soulsbury
Electronic supplementary material
ESM 1
(CSV 15 kb)
Rights and permissions
About this article
Cite this article
Welch, R.J., le Roux, A., Petelle, M.B. et al. The influence of environmental and social factors on high- and low-cost vigilance in bat-eared foxes. Behav Ecol Sociobiol 72, 29 (2018). https://doi.org/10.1007/s00265-017-2433-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-017-2433-y