Abstract
Loud, low-frequency traffic noise can mask songbird vocalizations, and populations of some urban songbird species have shifted the frequency of their vocalizations upward in response. However, the spectral structure of certain vocalization elements may make them resistant to masking, suggesting that species that use these notes could be more successful in areas with high levels of traffic noise. To test this idea, we recorded Carolina chickadees (Poecile carolinensis), whose calls feature “D” notes with an overtone spectral structure, along a traffic noise gradient in Durham and Orange Counties, North Carolina, USA. Frequency parameters of “D” notes did not change with noise level suggesting the possibility that these notes can be communicated effectively in noise, but further investigation is needed to test this hypothesis directly. In addition, we performed a playback experiment demonstrating how the use of spectrograms to measure note frequencies is unreliable, especially when recordings are made in noisy areas. We used an alternative method based on the predictable frequency structure of “D” notes. Our experiment is one of few that address the effects of urban noise on calls produced by both sexes as opposed to song produced only by males during the breeding season. Understanding how vocalizations with different spectral structures may be affected differentially by traffic noise will increase our ability to predict how the expansion of noisy areas may impact songbird community composition in the future.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The increased ambient noise usually associated with urban spaces is a feature of human-dominated environments that can influence animal behavior (reviewed in Ditchkoff et al. 2006). In particular, high levels of noise can reduce the effectiveness of vocal communication, causing urban animals to compensate in some way (reviewed in Rabin and Greene 2002; Patricelli and Blickley 2006; Warren et al. 2006; Brumm 2013). Urban gray squirrels (Sciurus carolinensis), for example, rely more on visual signals (such as tail flagging) than auditory signals (such as alarm calling) when compared to their rural counterparts (Partan et al. 2010). Modified signal production in noisy environments has been observed across a range of taxa, from anurans to marine mammals (reviewed in Brumm 2013).
Noise is a particular challenge for urban populations of species that use vocal signals as the principal method of interspecific communication. Songbirds are one such group of species. Traffic noise in particular has acoustic characteristics that can mask or otherwise interfere with the perception of songbird vocalizations (Brumm and Slabbekoorn 2005). Traffic noise is typically low-frequency and has greatest amplitude between 1 and 4 kHz, with much of its energy concentrated below 1 kHz (Sandberg 1987; Rochat 2004; Fig. 1). When a bird’s song or call falls within this range, the noise could potentially mask it (depending on the signal-to-noise ratio), reducing the distance over which it can be heard (i.e., the signal’s active space; Marten and Marler 1977; Wiley and Richards 1982). Possibly due to noise masking, songbird abundance is lower in areas with high levels of traffic noise (Reijnen et al. 1995; Rheindt 2003; Proppe et al. 2013). In addition to reducing songbird abundance, noise also appears to influence vocal production.
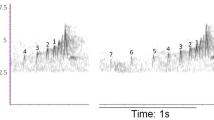
Interference of traffic noise on spectral measurements of “D” notes. Spectrograms of two chick-a-dee calls, comprised of tonal introductory notes and “D” notes. One call was recorded in a low noise area (top), and all overtones of “D” notes are visible. However, a call recorded in high-amplitude low-frequency traffic noise (bottom) has noise obscuring the lower-frequency overtones. Components 1 and 2 are the two fundamental frequency bands produced by the two sides of the syrinx; component 1 is the minimum frequency
Songbirds in noisy urban areas often produce vocalizations at higher frequencies than birds in quiet conditions, a response that has been observed in a large number of species (reviewed in Brumm and Zollinger 2013; Gil and Brumm 2014). This could be due to selection for higher vocalizations in noisy areas. However, this frequency modification has been observed to be a behavioral response in certain species, with birds switching to higher-frequency vocalizations in response to noise (Verzijden et al. 2010; Proppe et al. 2011; Goodwin and Podos 2013; Potvin and Mulder 2013). By shifting the frequency of notes upward, less of the notes’ bandwidth overlaps with traffic noise. This potentially allows more of the note to be clearly discriminated by the receiver, and Pohl et al. (2012) demonstrated that upward frequency shifts do in fact increase great tits’ (Parus major) ability to detect songs in the context of urban noise. However, not all birds in noisy areas perform a frequency shift (Hu and Cardoso 2010; Hanna et al. 2011; Dowling et al. 2012; Ríos-Chelén et al. 2013), and responses to noise can vary among closely related, co-occurring species (Francis et al. 2011).
Additionally, release from masking is not the only possible explanation for the observation of higher frequencies in urban areas. Producing louder vocalizations also improves signal transmission in noise (Nemeth and Brumm 2010), and studies of many bird species have found amplitude and frequency to be positively correlated (Osmanski and Dooling 2009; Schuster et al. 2012; Nemeth et al. 2013; see Discussion). However, if vocal constraints or very loud noise make it impossible to improve signal-to-noise ratio via increased amplitude, frequency shifting could be another potential strategy.
Even if upward frequency shifts ameliorate the problem of noise masking from traffic, they may not be required in all cases. No previous studies that have documented a frequency shift have explicitly studied notes composed of harmonics or overtones. These notes may be less susceptible to the masking effects of traffic noise; however, since the higher-frequency components of these notes are perceptible even if the lower-frequency components are masked. An example is the “D” note of the chickadee (genus Poecile). “D” notes are part of the chick-a-dee call and consist of evenly spaced overtones that suggest a harmonic series (Fig. 1). Experiments have shown, however, that the overtones in “D” notes are not harmonics but instead are a series of sum and difference frequencies resulting from cross-modulation of two sounds produced by either side of the syrinx (Nowicki and Capranica 1986a, b). The first two (lowest) spectral components are thought to be the two fundamental frequencies produced by the two sides of the syrinx (Figs. 1 and 2), and the regular spacing among overtones is determined by the frequency difference between the first two components (Nowicki and Capranica 1986a, b). Chickadees can vary the frequency structure of their “D” notes (Nowicki 1989). They might achieve this by changing the frequency difference between the first two spectral components, thereby changing the difference between all spectral components (Nowicki 1989).
Illustration of how max and peak frequencies were measured using amplitude spectra. The horizontal dotted line indicates the cut-off point used in our measurements, 36 dB below the peak amplitude of the call. Peaks below the frequency of component 1 are background noise. Each call was high-pass filtered at 1105 Hz before measurements were taken. This call is shown without filtering to show the relative noise level
Nowicki (1989) found that the spacing of “D” note overtones is the most salient feature of the chick-a-dee “D” note, conveying key information about flock identity. Black-capped chickadees (Poecile atricapillus) vary the spacing of overtones in “D” notes and through mutual imitation flockmates converge on a common spacing of overtones while other characteristics of the “D” note (such as bandwidth and note duration) do not converge to the same extent (Mammen and Nowicki 1981; Nowicki 1989). Because much of the information contained in “D” notes is believed to be encoded in the spacing of overtones, Nowicki (1989) surmised that the “D” note within the chick-a-dee call of the black-capped chickadee could be less affected by the effects of environmental attenuation because the key information (the overtone spacing) is available across the bandwidth of the “D” note. That is, the spacing can often still be perceived even if some frequencies of the note are imperceptible due to attenuation or degradation.
Taking Nowicki’s (1989) logic one step further, we surmise that the communication of “D” notes should be possible despite the masking effects of low-frequency noise (Cynx and Shapiro 1986). “D” notes typically have a bandwidth extending above 4 kHz (McLaren 1976; Ficken et al. 1978, 1994; Hailman 1989; Bloomfield et al. 2004, 2005) leaving the higher-frequency overtones unmasked by low-frequency noise. Thus, receivers should be able to discern flock identity from the unmasked overtone spacing of notes even if the lower frequencies are masked.
In a first step toward addressing this hypothesis, we conducted a field study to examine the relationship between traffic noise and acoustic features of “D” notes in the chick-a-dee call produced by wintering Carolina chickadees (Poecile carolinensis). We investigated whether Carolina chickadees, like previously studied bird species, raise the lowest frequencies of notes within their calls when vocalizing in noise. The closely related black-capped chickadee has been shown to sing at higher frequencies in noise (Proppe et al. 2011; Goodwin and Podos 2013), so we presume that Carolina chickadees can modify their calls in a similar manner. We would not expect to see this shift in acoustic frequency, however, if receivers can discern flock identity from the higher, unmasked overtones in “D” notes. While chickadee songs have been previously studied, frequency modification of chickadee calls in response to noise has not been tested.
During the early stages of this project, we observed that loud traffic noise often obscured the lower frequencies of chick-a-dee calls in our recordings. Noise masking of the spectral components of vocalizations grows with growing noise amplitude, making it increasingly difficult to make accurate measurements of minimum frequency (Zollinger et al. 2012). Measurement error of minimum frequency that increases with increasing noise could lead to the conclusion that minimum frequency increases as noise increases, even if no such relationship exists. We performed a playback experiment that tested our ability to make accurate measurements of acoustic features of calls recorded in noise using a common measurement method—cursor measurements made from spectrograms. In addition, because of the predictable frequency structure of chick-a-dee calls, we were able to use an alternative method for determining each call’s minimum frequency.
Methods
Description of the chick-a-dee call
The chick-a-dee call is common to all chickadee species (genus Poecile) and functions as the primary method of maintaining group cohesion (Smith 1972; Gaddis 1979; Hailman 1989; Lucas and Freeberg 2007; Krams et al. 2012). These calls are used year-round by both males and females in a variety of contexts related to assembly or arousal, including announcing an individual’s position to flockmates and conveying information about food resources or predators (Brewer 1961; Bloomfield et al. 2005; Templeton et al. 2005; Freeberg 2008; Mahurin and Freeberg 2009; Soard and Ritchison 2009; Bartmess-LeVasseur et al. 2010). There are functional analogs of the chick-a-dee call in many parid species (chickadees, tits, and titmice) with similar acoustic structure (Hailman 1989).
The Carolina chickadee’s chick-a-dee call spans a frequency range of 1.5–9 kHz, with the lower range of the call falling within the masking range of traffic noise (Bloomfield et al. 2005; Fig. 1). The calls contain a variety of note types in addition to overtonal “D” notes, including tonal “introductory notes” which appear as rapid frequency-modulated tonal sweeps, and “C” notes, which are considered “noisy” and contain both tonal and overtonal elements (Bloomfield et al. 2005).
Recording chick-a-dee calls
We recorded the calls of individual Carolina chickadees in November 2011–January 2012 at various sites in Durham and Orange Counties, North Carolina, USA. To sample calls produced in a wide range of traffic noise levels, we recorded at sites in a variety of settings based on their proximity to roads: far from any roads on forested nature trails, along low-traffic residential roads, and in wooded areas adjacent to major highways. We visited each site only once. Because birds were not banded and thus were not individually recognizable, we chose recording sites located at least 400 m from each other to minimize the chance of recording the same bird more than once. This range is sufficient to prevent sampling overlap between nearby flocks (Mahurin and Freeberg 2009; Bartmess-LeVasseur et al. 2010; Freeberg 2012). In addition, we did not record more than two birds at any site, to avoid recording the same bird twice.
We recorded individual chickadees that were calling spontaneously between 0600 and 1100 h, and at ambient temperatures between 5 and 19.5 °C. We identified calling birds as we walked along sidewalks or paths and recorded individuals that we were able to approach within 10 m using a Marantz PMD 670 digital recorder (sampling rate: 44.1 kHz, resolution: 16 bit) and a Shure SM-57 microphone mounted in a Sony PBR-330 parabola. We recorded birds perched no higher than 10 m from the ground. We recorded each individual until it flew beyond 20 m away or stopped vocalizing.
Immediately after recording a bird, we measured the ambient noise level (dB SPL) directly below the perch using a VWR Traceable Type 2 sound level meter (set to A-weighting, Lo range, slow response) held at a height of 1.1 m and facing the nearest road. We recorded the noise level every 20 s for 2 min and averaged the six measurements to obtain an estimate of the site’s ambient noise level. We also recorded air temperature at the site using a Holdpeak 816A ambient air thermometer.
Measuring acoustic characteristics of field-recorded “D” notes
We measured characteristics of “D” notes for all calls using Signal 4.0 software (Engineering Design, Belmont, MA, USA). Each call was high-pass filtered at 1105 Hz, as in Freeberg (2012), in order to remove some of the low-frequency background noise (primarily from traffic) without removing any part of the call. To minimize bias, all note measurements were made blind to the noise level in which the bird was recorded.
We made note measurements for all birds from which we recorded at least two calls containing “D” notes (n = 23). For each “D” note, we measured the frequencies of three spectral parameters: peak frequency, maximum frequency, and minimum frequency. Peak and maximum frequencies were measured using amplitude spectra (Zollinger et al. 2012; Fig. 2). We defined peak frequency as the frequency of the peak of greatest amplitude within the call (after filtering at 1105 Hz, the peak frequency was always part of the call, never the noise) and the maximum frequency as the highest frequency peak with amplitude greater than −36 dB relative to the peak amplitude (Nowicki 1989; Bloomfield et al. 2005; Fig. 2). Measurements from all of a bird’s recorded notes were averaged to yield one value for each parameter.
Before making “D” note measurements from our recordings, visual inspection of note spectrograms and amplitude spectra revealed that in many cases, moderate to high levels of low-frequency traffic noise obscured the lowest overtones, including but not limited to the fundamental frequencies, of “D” notes (Fig. 1). The difficulty of making accurate measurements of lower-frequency vocalizations recorded in noise has been noted previously (Nemeth and Brumm 2009; Zollinger et al. 2012) and we surmised that noise masking would interfere with our ability to accurately measure the frequency of the lowest components of “D” notes. Specifically, the problem lies in the fact that as traffic noise level increases, noise masking spreads to higher frequencies obscuring increasingly higher frequency overtones. This means that the lowest overtones detectable for measurement would increase with increasing noise level, creating the appearance of a positive relationship between minimum frequency and noise that, in fact, may not exist.
Because the fundamental frequencies were masked by noise, we calculated the minimum frequencies of “D” notes based on overtone spacing. The consistent frequency difference between overtones (hereafter “overtone interval”) in “D” notes allowed us to calculate the minimum frequency of each note, even if it was not identifiable on the amplitude spectrum due to noise. This is because the interval between the first two frequency components (which typically occur at about 1680 and 2015 Hz for Carolina chickadees; Bloomfield et al. 2005; Fig. 1) is reflected in the interval of all higher components (Nowicki 1989). Therefore, if the frequency of at least one overtone and the overtone interval of a note are known, it is possible to calculate the expected frequencies of the note’s other overtones.
Using this rationale, we calculated estimated values for each “D” note’s minimum frequency by subtracting its overtone interval from the note’s peak frequency. Peak frequency was chosen as the starting point for our calculations because this peak (the peak of highest amplitude) always appeared clearly and unambiguously on the amplitude spectrum. Four overtone intervals, located between the five adjacent peaks of highest amplitude on the note’s amplitude spectrum (the peak frequency was always included), were averaged to yield an overtone interval value for that note. From each note’s peak frequency, we subtracted the note’s overtone interval value until we obtained a value that fell within the published range for the minimum frequency of Carolina chickadee “D” notes (average 1851 Hz ± 163 Hz (SD), range 1688–2014 Hz; Bloomfield et al. 2005), plus or minus 10 Hz. Therefore, the calculated minimum frequency always fell between 1678 and 2024 Hz. This calculation led to an unambiguous estimate of minimum frequency—there was never a case in which our calculation for a particular note yielded more than one value within this range.
We also recorded the number of “D” notes in each call and calculated the duration of each individual “D” note using spectrograms scaled to −24 dB relative to peak amplitude.
Playback test: low-frequency traffic noise and accurate note frequency measurements
The practice of using spectrograms to measure note frequencies has long been criticized for producing inaccurate results (see Greenewalt 1968; Beecher 1988; Zollinger et al. 2012), but the practice persists. It is particularly problematic when used to measure frequencies of calls recorded in noise. Many previous studies that have reported finding a correlation between noise and song minimum frequency used measurements from spectrograms, which raises questions about the validity of the results (discussed in Zollinger et al. 2012). We conducted a playback experiment to test our ability to accurately measure the minimum frequency of calls recorded in noise using spectrograms. We played synthesized chick-a-dee calls (Fig. 3) through a loudspeaker at sites with a range of traffic noise levels, and recorded them using the same equipment we used to record wild chickadee calls. This experiment mirrored the way that we recorded the calls of wild chickadees; but by recording synthesized calls with known minimum frequency, we could test whether loud traffic noise affected our ability to make accurate measurements.
Spectrograms showing synthesized “D” notes played back and recorded at varying noise levels. Synthesized “D” notes for playbacks had a known minimum frequency and lacked background noise. a Four synthesized “D” notes based on the acoustic features of a natural note recorded in a quiet area. The minimum frequency of the note, as measured using cursor placement on a spectrogram, is 1.93 kHz. b Playback of the synthesized note shown in A in a quiet area. The minimum frequency detectable using cursor placement is 1.93 kHz. c Playback of the synthesized note shown in A in a noisy area with low-frequency overtones masked by noise. The minimum frequency detectable using cursor placement is 3.27 kHz. The measurement error of minimum frequency for this note in a noisy area is 1.75 kHz. Although the notes are identical to those recorded in B, the notes in C appear to have a higher minimum frequency. Note that our spectral analysis of “D” notes was done using amplitude spectra, not spectrograms (see Methods)
We synthesized five chick-a-dee calls with different minimum frequencies (1300–2980 Hz) using Signal 4.0 software. Each call was constructed based on the spectral structure of a “D” note recorded in the local chickadee population. The frequencies of the lowest spectral components, the relative sound energies of the overtones, and the overtone intervals all mimicked the natural call on which it was based. Constructing “D” notes rather than using the original recorded calls allowed us to create playback files that had excellent signal-to-noise ratio (free from attenuation and from other environmental sounds such as other birds, traffic noise, and wind) and had clearly discernable spectral components in the lower frequencies. We then played the calls at ten sites characterized by varying levels of background noise (40.3–65.0 dB (A)). At each site, we played the five synthesized calls using a Marantz PMD 670 digital recorder connected to an AV70 Powered Partners loudspeaker. Recordings were made in flat open areas, free of dense vegetation or trees that might affect sound transmission, and ground cover was either grass or leaves (not concrete). The speaker was elevated so that its center was 1.1 m above the ground to minimize “ground effects” (Wiley and Richards 1982). We played calls at a 65–75 dB, SPL measured at 1 m from the speaker using a VWR Type 2 sound level meter (set to A-weighting, Lo range, slow response). We recorded the synthesized calls at a distance of 10 m from the speaker, and made ambient noise level measurements using the previously described protocol. We made minimum frequency measurements of “D” notes from spectrograms scaled to −24 dB relative to the note’s peak amplitude, and calculated measurement error for each note (error = measured frequency − known frequency).
Analysis
To assess the effects of traffic noise on wild Carolina chickadee calls, we examined the relationships between call parameters and traffic noise using linear regression models for each parameter in JMP 10.0.0 (copyright © 2012 SAS Institute Inc.), including temperature as a covariate in addition to noise as well as their interaction. The residuals of all models were tested for normality using Shapiro-Wilk tests, and for homogeneity of variances using Breusch-Pagan tests. If the interaction between noise and temperature was not significant, it was dropped from the model. There were three recording sites at which we recorded two birds (at all others, only one bird was recorded). Because recording took place in the winter, two birds recorded at the same site were likely members of the same flock (Brewer 1961; Smith 1972), and since other chickadee species show within-flock vocal convergence (Mammen and Nowicki 1981), measurements taken from birds recorded at the same site are potentially not independent. We therefore averaged the values for these pairs of birds, essentially treating them as subsamples. To guard against the effects of small sample sizes, we also performed Spearman’s rank correlations between call parameters and noise and call parameters and temperature.
Our playback test aimed to determine if traffic noise impeded our ability to accurately measure the minimum frequency of “D” notes using spectrograms, and whether noise level and the known frequencies of synthesized notes were predictors of measurement error. We analyzed correlations between these variables using Spearman’s rank correlation tests in R Statistical Computing software (R Foundation for Statistical Computing, Vienna, Austria, 2013).
Results
“D” notes
We obtained field recordings of the calls of 23 birds, recorded in noise levels varying from 37.3 to 69.3 dB. On average, we obtained 2.75 calls per bird (range 2–6; average number of “D” notes per call 3.9; range 1–9). Calculated minimum frequency ranged from 1744 to 1901 Hz with an average of 1828 Hz ± 10.4 (SE). Peak frequency ranged from 3887 to 4955 Hz with an average of 4234 ± 55.7 (SE). Maximum frequency ranged from 5795 to 8031 Hz with an average of 6666 Hz ± 110 (SE) Overtone interval ranged from 396 to 539 Hz with an average of 448 Hz ± 10.2 (SE).
The acoustic properties of “D” notes did not change as noise level became greater—linear models revealed no relationships between traffic noise level, temperature, or any of the acoustic parameters that we measured (maximum or peak frequency, overtone interval, or number of notes per call; Table 1), or estimated (minimum frequency). In addition, none of the Spearman’s rank correlations between call parameters and noise or between call parameters and temperature were significant.
Playback test
In our playback test using synthesized “D” notes of known minimum frequency (Fig. 3), we found that traffic noise level was a strong predictor of error when measuring minimum frequency; as traffic noise increased, error increased (r = 0.594, n = 50, p < 0.001). Specifically, as noise level increased, we ascribed a higher minimum frequency to playback calls than was the actual minimum frequency because noise at lower frequencies masked the lowest frequency on amplitude spectra. The known minimum frequency did not significantly predict measurement error (r = −0.274, n = 50, p = 0.055), and the interaction of traffic noise and true minimum frequency was also not significant (r = −0.047, n = 50, p = 0.745).
Discussion
The results of our playback test using synthesized “D” notes of known minimum frequency are key because they indicate the potential for measurement error that could suggest a false relationship between note frequency and noise. Measurement error of minimum frequency that increases with increasing noise can lead to the conclusion that minimum frequency increases as noise increases even if no such relationship exists. Indeed our own recordings of chickadee calls at various noise levels exemplify this problem. Had we simply used spectrograms to measure “D” note minimum frequency, our measurements of “D” notes would have led us to conclude that there existed a positive relationship between minimum frequency and noise, whether one existed or not. Our work underscores the importance of using amplitude spectra rather than spectrograms when making note frequency measurements (Greenewalt 1968; Zollinger et al. 2012).
Studies that used amplitude spectra to measure note frequencies still found that some species respond to noise by increasing vocalization frequency (Verzijden et al. 2010; Proppe et al. 2012). In our analysis of the “D” notes of Carolina chickadee calls, we found no significant relationships between traffic noise level and “D” note minimum frequency, peak frequency, maximum frequency, duration, overtone interval, or the number of notes per call. Thus, the birds in our study did not produce “D” notes within their calls at higher frequencies in noise as has been observed in the songs of other songbird species.
Our experimental results are supported by research on acoustic perception in birds and other taxa. First, with regard to perception of notes with harmonic structure, several studies suggest that the perceived frequency of harmonic tones (similar to the overtone spectral structure of “D” notes; Nowicki 1989) is based on pitch periodicity (the relative spacing of harmonics or overtones) and not on the tone’s fundamental frequency (primates, Plomp 1967; Tomlinson and Schwarz 1988; cats, Chung and Colavita 1976; Heffner and Whitfield 1976; songbirds, Cynx and Shapiro 1986; rodents, Shofner 2011). For example, Cynx and Shapiro (1986) showed that starlings (Sturnus vulgaris) do not discriminate between a harmonic sound and that same sound with the lowest-frequency harmonics removed, suggesting that the sounds are perceived similarly regardless of whether the lowest frequencies can be heard. Another recent study showed that chinchillas (Chinchilla laniger) are capable of distinguishing among harmonic tones with different fundamental frequencies in the presence of low-frequency masking noise, an ability that persists even when the fundamental frequencies are removed (Shofner 2011). Our study suggests that songbirds may have a similar ability to discriminate among notes with harmonic or overtonal structure, such as “D” notes, even in the presence of low-frequency urban noise. If these kinds of notes are indeed perceived as previous studies suggest, then masking of the lowest overtones by noise may not pose a significant problem for the communication system. Thus, the use of “D” notes in flock-specific contact calls by chickadees might offer these species an advantage in adapting to noisy urban life. Further studies are needed to test explicitly whether species that use harmonic or overtonal notes have a fitness advantage in urban areas.
Alternative hypotheses
Although our results suggest that “D” notes are not strongly compromised by urban noise and thus may be beneficial in noisy areas, we must consider other factors that could account for the absence of a correlation between the minimum frequency and traffic noise. One possibility is that Carolina chickadee “D” notes occur above a certain “frequency threshold,” above which the masking effects of noise are less severe. Dowling et al. (2012) found that species whose songs had average minimum frequencies at or above 1.85 kHz (calculated from songs recorded in non-urban areas) showed no correlation between minimum frequency of song and noise. The average minimum frequency of “D” notes produced by Carolina chickadees (in relatively quiet areas) is 1.85 kHz (Bloomfield et al. 2005); thus, “D” notes may be above the frequency range that is severely impacted by noise. That said, other studies suggest that noise can impact songs produced at higher frequencies [e.g., chiffchaffs, average minimum frequency about 3 kHz: Verzijden et al. (2010)].
Another possible explanation for the observed lack of correlation between noise and “D” note frequencies comes from a recent study in which passerine species with learned vocalizations showed stronger relationships between noise level and frequency than species with innate vocalizations (Ríos-Chelén et al. 2012). The authors hypothesized that this was observed because there is more room for plasticity in the learned vocalizations. Chick-a-dee calls are not learned, perhaps explaining the observed lack of correlation between note frequency and noise. However, calls of other species have been shown to be modified in response to environmental conditions (Leonard and Horn 2008). In addition, chickadees are able to modify acoustic features of the chick-a-dee call, in particular the spacing of overtones in “D” notes, to enable flock call convergence (Nowicki 1989). Thus. vocal plasticity and the ability to learn and produce new call characteristics are present in adult chickadees, suggesting the ability to modify calls in response to environmental factors such as noise.
Does frequency modulation truly provide benefits to urban songbirds?
Whether or not frequency shifts provide a communication benefit is debated. Nemeth and Brumm’s (2010) model predicts that a difference of 205 Hz (Slabbekoorn and den Boer-Visser 2006) would produce a 9–10 % increase in communication distance and a 19–21 % increase in the active space of great tit songs, independent of amplitude changes. Although increased amplitude has much greater positive effects on communication distance (47–48 %) and active space (115 %), increased frequency nonetheless has a separate effect. Whether its effect is large enough to influence fitness is still unknown.
The Lombard hypothesis (LH) suggests that increases in amplitude are much more effective at improving transmission in noisy environments than frequency shifts, and suggests that frequency changes are merely a side effect of birds singing louder (Nemeth and Brumm 2010). Humans, for example, speak at a higher pitch as a side effect of increasing vocal amplitude (Traunmüller and Eriksson 2000). When exposed to broadband white noise, budgerigars (Melopsittacus undulatus) and tinamous (Eudromia elegans) produce calls at a higher peak frequency, even though calls remain masked by the white noise (Osmanski and Dooling 2009 and Schuster et al. 2012, respectively). The authors concluded that the increase in frequency was a by-product of increased amplitude. However, the LH does not always apply when amplitude and frequency are coupled. Blackbirds (Turdus merula) in noisy areas sing song types with higher-pitched elements, but these elements are naturally produced at higher amplitude than other elements, so this cannot be attributed to the Lombard effect (Nemeth et al. 2013). To test whether the LH explains songbird response to urban noise, Potvin and Mulder (2013) exposed silvereyes (Zosterops lateralis) to both high-frequency and low-frequency noise. Though silvereyes increased the amplitude of their vocalizations in both treatments, they decreased the frequency of vocalizations made in high-frequency noise. Thus, frequency modification in response to noise may not always be a side effect of increased amplitude.
Future directions
Further investigation is needed to provide support for the hypothesis that notes with harmonic or overtone structure (hereafter “harmonic-like”) are adaptive in a noisy world. Our study hints at this possibility, but carefully controlled laboratory studies, in which vocalizations can be recorded with minimal background noise, are essential. Our playback experiment highlighted the difficulty of making accurate measurements in heavy background noise, which prevented us from comparing “D” notes to the other tonal notes within the call. This comparison would have allowed us to test whether “D” notes are produced differently than tonal notes in noise within the same call, an outcome we might expect if tonal notes are compromised by noise to a much greater degree than “D” notes. A useful focus for future laboratory studies would be species whose harmonic-like notes have a minimum frequency within the range that has shown the greatest response to noise in tonal notes, roughly between 1 and 3 kHz (Hu and Cardoso 2010; Verzijden et al. 2010; Dowling et al. 2012).
Another avenue for future study is to investigate whether the use of harmonic-like notes can confer fitness benefits in areas of high traffic noise. Some songbird species do experience lower reproductive success in areas with high industrial noise (Habib et al. 2007) or traffic noise (Halfwerk et al. 2011). Reijnen et al. (1995) found that near roads, breeding densities of many songbird species decrease by up to a third. This reduction was partially explained by the amount of traffic noise from the road, leading them to hypothesize that difficulty communicating could be driving songbirds away from the area. If harmonic-like notes can be effectively communicated in spite of traffic noise, then species that use them could show differential abundance and breeding success near roads when compared to species that do not. Breeding success could be particularly affected if a species’ songs (in contrast to calls) contain harmonic-like notes.
The overwhelming majority of previous research on the effects of noise on avian vocalizations has dealt with bird song. Our study is one of the few to explicitly examine how urban noise affects the frequency of songbird calls (Potvin et al. 2011; see also Hu and Cardoso 2010). The few previous studies found that nestling tree swallows (Tachycineta bicolor) produce begging calls at higher frequencies when exposed to ambient white noise (Leonard and Horn 2008), and demonstrated the Lombard effect in the alarm calls of urban noisy miners (Manorina melanocephala; Lowry et al. 2012). Bird song has been heavily studied because it is a dual-function signal operating in both mate attraction and territory defense contexts. Bird calls, which are used in the contexts of maintaining contact with conspecifics and advertising food resources or alarm (among other things), are “neglected orphans” in comparison (Marler 2006, p. 31). Communication via calls is critical to survival, so it is important to understand how noise can affect species dynamics and fitness by masking these important social signals. Further, in most species, only male songbirds sing but both sexes produce calls, making the impacts of noise relevant for communication in both sexes. In sum, further research is necessary to determine how noise masking of calls might differentially affect bird species.
Building greater understanding about how the spectral structure of vocalizations can buffer communication against noise will help us to understand the impact of anthropogenic noise on avian communities. In turn, this may help us to anticipate how the constant expansion of noisy areas will affect species composition in the future and to predict which species might be most at risk from a growing road network.
References
Bartmess-LeVasseur J, Branch CL, Brownin SA, Owens JL, Freeberg TM (2010) Predator stimuli and calling behavior of Carolina chickadees (Poecile carolinensis), tufted titmice (Baeolophus bicolor), and white-breasted nuthatches (Sitta carolinensis). Behav Ecol Sociobiol 64:1187–1198
Beecher MD (1988) Spectrographic analysis of animal vocalizations: implications of the ‘uncertainty principle’. Bioacoustics 1:187–208
Bloomfield LL, Charrier I, Sturdy CB (2004) Note types and coding in parid vocalizations II: the chick-a-dee call of the mountain chickadee (Poecile gambeli). Can J Zool 82:780–793
Bloomfield LL, Phillmore LS, Weisman RG, Sturdy CB (2005) Note types and coding in parid vocalizations III: the chick-a-dee call of the Carolina chickadee (Poecile carolinensis). Can J Zool 83:820–833
Brewer R (1961) Comparative notes on the life history of the Carolina chickadee. Wilson Bull 73:348–373
Brumm H (ed) (2013) Animal communication and noise. Animal signals and communication 2. Springer, Berlin
Brumm H, Slabbekoorn H (2005) Acoustic communication in noise. Adv Stud Behav 35:151–209
Brumm H, Zollinger SA (2013) Avian vocal production in noise. In: Brumm H (ed) Animal communication and noise. Animal signals and communication 2. Springer, Berlin, pp 187–227
Chung DY, Colavita FB (1976) Periodicity pitch perception and its upper frequency limit in cats. Atten Percept Psychophysiol 20:433–437
Cynx J, Shapiro M (1986) Perception of missing fundamental by a species of songbird (Sturnus vulgaris). J Comp Psychol 100:356–360
Ditchkoff SS, Saalfeld ST, Gibson CJ (2006) Animal behavior in urban ecosystems: modifications due to human-induced stress. Urban Ecosyst 9:5–12
Dowling J, Luther D, Marra P (2012) Comparative effects of urban development and anthropogenic noise on bird songs. Behav Ecol 23:201–209
Ficken MS, Ficken RW, Witkin SR (1978) Vocal repertoire of the black-capped chickadee. Auk 95:34–48
Ficken MS, Hailman ED, Hailman JP (1994) The chick-a-dee call system of the Mexican chickadee. Condor 96:70–82
Francis C, Ortega C, Cruz A (2011) Different behavioural responses to anthropogenic noise by two closely related passerine birds. Biol Lett 7:850–852
Freeberg TM (2008) Complexity in the chick-a-dee call of Carolina chickadees (Poecile carolinensis): associations of context and signaler behavior to call structure. Auk 125:896–907
Freeberg TM (2012) Geographic variation in note composition and use of chick-a-dee calls of Carolina chickadees (Poecile carolinensis). Ethology 118:555–565
Gaddis P (1979) A comparative analysis of the vocal communication systems of the Carolina chickadee and tufted titmouse. Ph.D. thesis, University of Florida
Gil D, Brumm H (2014) Acoustic communication in the urban environment: patterns, mechanisms, and potential consequences of avian song adjustments. In: Gil D, Brumm H (eds) Avian urban ecology. Oxford University Press, Oxford, pp 69–83
Goodwin SE, Podos J (2013) Shift of song frequencies in response to masking tones. Anim Behav 85:435–440
Greenewalt CH (1968) Bird song: acoustics and physiology. Smithsonian Institution Press, Washington, DC
Habib L, Bayne EM, Boutin S (2007) Chronic industrial noise affects pairing success and age structure of ovenbirds Seiurus aurocapilla. J Appl Ecol 44:176–184
Hailman JP (1989) The organization of the major vocalizations in the Paridae. Wilson Bull 101:305–343
Halfwerk W, Holleman LJM, Lessells CM, Slabbekoorn H (2011) Negative impact of traffic noise on avian reproductive success. J Appl Ecol 48:210–219
Hanna D, Blouin-Demers G, Wilson DR, Mennill DJ (2011) Anthropogenic noise affects song structure in red-winged blackbirds (Agelaius phoeniceus). J Exp Biol 214:3549–3556
Heffner HE, Whitfield IC (1976) Perception of the missing fundamental by cats. J Acoust Soc Am 59:915–919
Hu Y, Cardoso GC (2010) Which birds adjust the frequency of vocalizations in urban noise? Anim Behav 79:863–867
Krams I, Krama T, Freeberg TM, Kullberg C, Lucas JR (2012) Linking social complexity and vocal complexity: a parid perspective. Philos Trans Roy Soc B 367:1879–1891
Leonard ML, Horn AG (2008) Does ambient noise affect growth and begging call structure in nestling birds? Behav Ecol 19:502–507
Lowry H, Lill A, Wong BBM (2012) How noisy does a noisy miner have to be? amplitude adjustments of alarm calls in an avian urban adapter. PLoS ONE 7:e29960
Lucas JR, Freeberg TM (2007) ‘Information’ and the chick-a-dee call: communicating with a complex vocal system. In: Otter KA (ed) Ecology and behaviour of chickadees and titmice: an integrated approach. Oxford University Press, Oxford, pp 199–213
Mahurin EJ, Freeberg TM (2009) Chick-a-dee call variation in Carolina chickadees and recruiting flockmates to food. Behav Ecol 20:111–116
Mammen DL, Nowicki S (1981) Individual differences and within-flock convergence in chickadee calls. Behav Ecol Sociobiol 9:179–186
Marler P (2006) Bird calls: their potential for behavioral neurobiology. Ann N Y Acad Sci 1016:31–44
Marten K, Marler P (1977) Sound transmission and its significance for animal vocalization I: temperate habitats. Behav Ecol Sociobiol 2:271–290
McLaren MA (1976) Vocalizations of the boreal chickadee. Auk 93:451–463
Nemeth E, Brumm H (2009) Blackbirds sing higher-pitched songs in cities: adaptation to habitat acoustics or side-effect of urbanization? Anim Behav 78:637–641
Nemeth E, Brumm H (2010) Birds and anthropogenic noise: are urban songs adaptive? Am Nat 176:465–475
Nemeth E, Pieretti N, Zollinger SA, Geberzahn N, Partecke J, Miranda AC, Brumm H (2013) Bird song and anthropogenic noise: vocal constraints may explain why birds sing higher-frequency songs in cities. Proc R Soc Lond B 280:1754–1766
Nowicki S (1989) Vocal plasticity in captive black-capped chickadees: the acoustic basis and rate of call convergence. Anim Behav 37:64–73
Nowicki S, Capranica RR (1986a) Bilateral interaction in vocal production of an oscine bird sound. Science 231:1297–1299
Nowicki S, Capranica RR (1986b) Bilateral syringeal coupling during phonation of a songbird. J Neurosci 6:3595–3610
Osmanski MS, Dooling RJ (2009) The effect of altered auditory feedback on control of vocal production in budgerigars (Melopsittacus undulatus). J Acoust Soc Am 126:911–919
Partan SR, Fulmer AG, Gounard MAM, Redmond JE (2010) Multimodal alarm behavior in urban and rural gray squirrels studied by means of observation and a mechanical robot. Curr Zool 56:313–326
Patricelli GL, Blickley JL (2006) Avian communication in urban noise: causes and consequences of vocal adjustment. Auk 123:639–649
Plomp R (1967) Pitch of complex tones. J Acoust Soc Am 41:1526–1533
Pohl NU, Leadbeater E, Slabbekoorn H, Klump GM, Langemann U (2012) Great tits in urban noise benefit from high frequencies in song detection and discrimination. Anim Behav 83:711–721
Potvin DA, Mulder RA (2013) Immediate, independent adjustment of call pitch and amplitude in response to varying background noise by silvereyes (Zosterops lateralis). Behav Ecol 24:1363–1368
Potvin DA, Parris KM, Mulder RA (2011) Geographically pervasive effects of urban noise on frequency and syllable rate of songs and calls in silvereyes (Zosterops lateralis). Proc R Soc Lond B 278:2464–2469
Proppe DS, Sturdy CB, St. Clair CC (2011) Flexibility in animal signals facilitates adaptation to rapidly changing environments. PLoS ONE 6:e25413
Proppe DS, Avey MT, Hoeschele M, Moscicki MK, Farrell T, St. Clair CC, Sturdy CB (2012) Black-capped chickadees Poecile atricapillus sing at higher pitches with elevated anthropogenic noise but not with decreasing canopy cover. J Avian Biol 43:325–332
Proppe DS, Sturdy CB, St. Clair CC (2013) Anthropogenic noise decreases urban songbird diversity and may contributes to homogenization. Glob Chang Biol 19:1075–1084
Rabin LA, Greene CM (2002) Changes to acoustic communication systems in human-altered environments. J Comp Psychol 116:137–141
Reijnen R, Foppen R, Braak CT, Thissen J (1995) The effects of car traffic on breeding bird populations in woodland. III Reduction of density in relation to the proximity of main roads. J Appl Ecol 32:187–202
Rheindt FE (2003) The impact of roads on birds: does song frequency play a role in determining susceptibility to noise pollution? J Ornithol 144:295–306
Ríos-Chelén AA, Salaberria C, Barbosa I, Macías Garcia C, Gil D (2012) The learning advantage: bird species that learn their song show a tighter adjustment of song to noisy environments than those that do not learn. J Evol Biol 25:2171–2180
Ríos-Chelén AA, Quirós-Guerrero E, Gil D, Garcia CM (2013) Dealing with urban noise: vermilion flycatchers sing longer songs in noisier territories. Behav Ecol Sociobiol 67:145–152
Rochat JL (2004) Transportation noise issues. In: Kutz M (ed) Handbook of transportation engineering. Vol. 1000. McGraw-Hill, New York, pp 19.1–19.15
Sandberg U (1987) Road traffic noise—the influence of the road surface and its characterization. Appl Acoust 21:97–118
Schuster S, Zollinger SA, Lesku JA, Brumm H (2012) On the evolution of noise-dependent vocal plasticity in birds. Biol Lett 8:913–916
Shofner W (2011) Perception of the missing fundamental by chinchillas in the presence of low-pass masking noise. J Assoc Res Otolaryngol 12:101–112
Slabbekoorn H, den Boer-Visser A (2006) Cities change the songs of birds. Curr Biol 16:2326–2331
Smith ST (1972) Communication and other social behavior in Parus carolinensis. Nuttall Ornithological Club, Cambridge, MA
Soard CM, Ritchison G (2009) Chick-a-dee calls of Carolina chickadees convey information about degree of threat posed by avian predators. Anim Behav 78:1447–1453
Templeton CN, Greene E, Davis K (2005) Allometry of alarm calls: black-capped chickadees encode information about predator size. Science 308:1934–1937
Tomlinson RWW, Schwarz DWF (1988) Perception of the missing fundamental in nonhuman primates. J Acoust Soc Am 84:560–565
Traunmüller H, Eriksson E (2000) Acoustic effects of variation in vocal effort by men, women, and children. J Acoust Soc Am 107:3438–3451
Verzijden MN, Ripmeester EAP, Ohms VR, Snelderwaard P, Slabbekoorn H (2010) Immediate spectral flexibility in singing chiffchaffs during experimental exposure to highway noise. J Exp Biol 213:2575–2581
Warren PA, Katti M, Ermann M, Brazel A (2006) Urban bioacoustics: it’s not just noise. Anim Behav 71:491–502
Wiley RH, Richards DG (1982) Adaptations for acoustic communication in birds: sound transmission and signal detection. In: Kroodsma DE, Miller EH (eds) Acoustic communication in birds vol. 2. Academic, New York, pp 131–181
Zollinger SA, Podos J, Nemeth E, Goller F, Brumm H (2012) On the relationship between, and measurement of, amplitude and frequency in birdsong. Anim Behav 84:e1–e9
Acknowledgments
We thank Ed Ibarguen and the Washington Duke Inn and Golf Club for providing transportation between sites during data collection, and Sara Childs and the Office of the Duke Forest for permitting access to the Duke Forest for field recordings. We thank Susan Peters for the help in all aspects of the project, especially her help in synthesizing notes for playback and suggesting improvements to the manuscript. Thanks to Stephen Nowicki and his lab at Duke University for their encouragement and suggestions, the S.P.I.C.E. Lab at the University of Central Florida for editing a draft of the manuscript, and Mohamed Noor at Duke University for his prompt and thoughtful feedback on an early draft of the manuscript. Duke University provided logistical and financial support. The project was funded by a Duke University Undergraduate Research Support Grant to MKG.
The Duke University Institutional Animal Care and Use Committee approved the project under Protocol No. A237-11-09.
Ethical standards
The experiments performed here comply with the current laws of the United States of America.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by H. Brumm
Rights and permissions
About this article
Cite this article
Grace, M.K., Anderson, R.C. No frequency shift in the “D” notes of Carolina chickadee calls in response to traffic noise. Behav Ecol Sociobiol 69, 253–263 (2015). https://doi.org/10.1007/s00265-014-1838-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-014-1838-0