Abstract
Biparental incubation is frequent among shorebirds and is expected when the survival prospects of offspring increase relative to uniparental incubation. To understand why this occurs, it is important to identify the factors that constrain uniparental incubation. It is assumed that birds choose nesting sites that provide an appropriate microclimate for incubation. Many shorebirds nest in sites with no or little cover, where ambient temperatures at ground level might be >50°C during very hot days. Shorebirds nest in exposed sites because predation risk on incubating adults is higher in covered sites. In hot environments, incubating shorebirds might experience heat stress in exposed sites, and this may compromise nesting success if adults are unable to attend their nests continuously, limiting the possibilities of uniparental incubation and thus the expression of a sexual conflict over incubation. The operative temperatures of Kentish plovers (Charadrius alexandrinus) were recorded in exposed and covered sites, and the thermal behaviour and incubating tactics of pair members were studied in a hot environment. During the hottest part of the day, there was a difference of 10–15°C in the operative temperatures of plovers between covered and exposed sites. Plovers in covered sites did not exhibit any thermoregulatory behaviour indicative of thermal stress, probably because the thermal range encountered by them in such places during most of the daytime was close to the thermo-neutral zone. The frequency with which plovers in exposed sites exhibited thermoregulatory behaviour was related to ambient temperature. Under very hot conditions, incubating birds were probably unable to maintain homeostasis for long periods and pair members resorted to shortening incubation bouts. Female Kentish plovers mainly incubate in the daytime and males during the night. However, the probability of diurnal incubation by males increased with ambient temperature in exposed nests, but not in covered ones. In fact, the frequency of participation in diurnal incubation by males was greater in exposed than in covered sites, suggesting that the participation of males in diurnal incubation may be related to the inability of females to stay at the nest during long periods when the ambient temperature is high. Even after resorting to shortened incubation bouts, the plovers may be unable to attend their nests continuously during heat waves, and the nests may be deserted. The propensity of plovers to desert their nests was affected by proximity to water, with nests located close to water being deserted less frequently. It seems likely that susceptibility to thermal stress changed in relation to proximity to water because in sites close to water it was possible to belly-soak, which would allow a more continuous nest attendance. Therefore, despite the adoption of behavioural solutions to face heavy heat loads, nesting success was vulnerable to these solutions because heat stress during extended periods may constrain parental nest attendance, and this may limit the opportunities for sexual conflicts over incubation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Habitat choice may affect metabolic processes through the effects that microclimate, and in particular ambient temperature, exert on physiological capacity, so that thermoregulatory costs may be an important factor influencing habitat selection by animals (Huey 1991; Pianka 1994). Most studies that consider the thermal consequences of nest-site placement have indeed found that nests are located in the more thermally favourable sites (review in Walsberg 1985), which would reduce thermoregulatory costs (Thomas and Maclean 1981; Wolf and Walsberg 1996; Schreiber 2002). In this respect, shorebirds provide an interesting study-case, since they are ground-nesting and many species nest in hot environments with little or no cover, where temperatures at ground level may be >50°C during the hottest parts of the day (Purdue 1976; Grant 1982; Ward 1990). The reason why some bird species nest in exposed sites, where temperatures may be very high, is that predation on incubating adults is higher in covered than in exposed sites (Jehl and Mahoney 1987; Wiebe and Martin 1998; Amat and Masero 2004). If the high temperatures experienced by incubating shorebirds overwhelm their thermoregulatory capabilities, placing nests in thermally stressful sites may constrain nest attendance, which may then affect nesting success (Grant 1982; Zerba and Morton 1983; Jehl and Mahoney 1987; Wiebe and Martin 1998). Indeed, very high temperatures, although infrequent, may cause massive nest failures in some ground-nesting species (Salzman 1982).
Thermoregulation often involves both physiological and behavioural adjustments (Whittow 2002). Small birds have some physiological mechanisms that favour body-water economy in hot environments, such as hyperthermia (Weathers 1981; Tieleman and Williams 1999). However, if ambient temperature is too high, the physiological mechanisms may be insufficient, and to mitigate thermal stress, incubating birds in hot environments may then deploy several behavioural responses to either reduce heat gain, or facilitate evaporative cooling (Grant 1982; Kalsi and Khera 1986; Ward 1990; Downs and Ward 1997; Brown and Downs 2003). In extreme situations, for example, nesting shorebirds have been observed belly-soaking (i.e. wetting their ventral plumage), which has been interpreted as a mechanism to dissipate body temperature (Maclean 1975; George Kainady and Al-Dabbagh 1976; Grant 1982; Kalsi and Khera 1986). Nevertheless, the ability to adopt some of these strategies may depend on nesting circumstances, which may affect nesting success. For instance, birds nesting far from water cannot belly-soak (Bergstrom 1989), which may constrain the time that birds are able to remain at nests during hot conditions. This inability of birds to remain at nests during long periods under hot conditions may mean that the eggs reach lethal temperatures (Maclean 1967; Grant 1982), and nest desertion should be higher in sites far from water.
Kentish plovers (Charadrius alexandrinus) are small shorebirds that readily nest on exposed sites in hot environments (Grant 1982; Page et al. 1985; Fraga and Amat 1996). This species is striking because both pair members participate in parental activities until one of them, usually the female, deserts from brood care some days after hatching (Lessells 1984; Székely 1996; Amat et al. 1999b). Although there may be a sexual conflict over parental care (Székely et al. 1999; Kosztolányi et al. 2003), biparental incubation is expected if the survival prospects of offspring increase relative to uniparental incubation. Kentish-plover nests are usually incubated by females during daytime and by males at night (Nakazawa 1979; Fraga and Amat 1996; Kosztolányi and Székely 2002). However, heterogeneity of environmental temperatures could produce variation in the time that females may devote to diurnal incubation by limiting the duration of incubation bouts. This may be so under very hot conditions, when incubating plovers may be unable to maintain homeostasis for long periods, thus compromising nest attendance, and pair members may then resort to shortening incubation bouts, with males increasing their participation in diurnal incubation (Purdue 1976; Nakazawa 1979; Kalsi and Khera 1986; Ward 1990; Fraga and Amat 1996). Therefore, abiotic factors may constrain uniparental incubation, limiting the possibilities for a sexual conflict over incubation.
Although the thermal environment is thought to limit incubation activity in shorebirds, few studies have examined how environmental temperatures affect the behaviour of adults at their nests (Grant 1982; Downs and Ward 1997; Brown and Downs 2003). In this paper, we used observational and experimental data to determine whether, by using behavioural strategies, Kentish plovers are able to resolve the conflicting demands (nest attendance vs nest desertion) that they should experience when incubating their eggs in a hot environment. To this aim, we first characterised the operative temperatures in covered and exposed sites, to show whether nesting in exposed sites places incubating plovers under heat stress. Operative temperature is defined as the temperature experienced by an animal as a result of the sum of air temperature and a temperature increment or decrement that subsumes radiative and convective factors (Bakken 1976). Secondly, we studied the behavioural thermoregulatory strategies of the plovers according to nest cover and ambient temperatures. Thirdly, we determined whether the ability to adopt behavioural strategies to cope with heat stress is an important determinant of nesting success. We predicted that those factors that limit the ability of the plovers to maintain homeostasis will predispose them to desert their nests during stressful thermal conditions.
Methods
The study was conducted at Fuente de Piedra lake (1,354 ha), in Málaga province, southern Spain (37°06′N, 4°45′W), during the breeding seasons of 1991/1999. The water level in the lake varies both within and between breeding seasons (see Fraga and Amat 1996 for a description of the site and breeding biology of the species). In Fuente de Piedra, about 70% of the nests are in sites with very little or no cover. In this lake, when nests of Kentish plovers are in covered sites, they are usually placed under Arthrocnemum bushes. Modal clutch size is 3 and incubation lasts 27 days.
Nest sites and bird trapping
The nests were allocated to two categories according to their cover: “exposed” or “covered” depending on whether they were viewed by the researcher >75% or ≤75%, respectively, of the sites from 1 m around the nests (for details, see Amat et al. 1999a; Amat and Masero 2004). Adults were captured at nests using walk-in traps. Plovers were sexed based on their dimorphic plumage. Nest were revisited every 3–6 days until the eggs hatched, or the nests were depredated or deserted. The time of day at which captures were made was also recorded. Traps were placed on nests arbitrarily according to time of day.
For shorebirds, one way to alleviate heat stress is belly-soaking (Maclean 1975). Because of the variations in the water levels of the lake, the opportunities for belly-soaking may be limited when water levels are low, and this may preclude a continuous nest attendance by adults when ambient temperatures are very high, so that embryos could reach lethal temperatures (Grant 1982). It was determined whether the probability of nest desertion was affected by proximity to water, by comparing the fate of nests that were close to water (there was some water source <200 m from nests while they were active) and nests far from water (≥200 m). From this analysis, depredated nests were excluded. Although we set an arbitrary distance of 200 m, we think that our comparison is rather conservative, as we did not find evidence of belly-soaking for individuals nesting >50 m from a water source. In addition, based on observations at nests, birds moved to water sources >200 m from nests mainly by flying, and heat produced as a by-product of flight would exacerbate the thermal stress (e.g. Davies 1982; Thomson et al. 1998), thereby constraining the advantages of belly-soaking. Distances of nests to the nearest water source were estimated by pacing if they were <300 m; otherwise, nests were allocated to a category of ≥300 m.
Temperatures
Air temperature (T a), egg temperature (T egg) and operative temperature (T e) (Bakken et al. 1981; Bakken 1992) were measured with copper-constantan thermocouple probes connected to Omega OM-550 dataloggers (Omega Engineering, Stamford, Conn.) during 1997–1999. Air temperature was measured 5 cm above ground level (T a5) with a 20-gauge probe located about 1 m from the nesting scrape. This probe was not shaded and its tip was covered with white reflective tape (Ward 1990). To record T egg, we removed an egg from the nest and substituted it with a Kentish-plover egg filled with plaster-of-paris, into which a 36-gauge probe had been inserted. The original egg was placed in a nearby nest and returned to its nest once data recording had finished. This procedure did not affect hatching success. The thermal conductance of plaster-of-paris is practically identical to that of natural eggs (see Ward 1990). T egg in unattended nests was recorded, for which a scrape was made ~40 cm from the focal nest, into which pebbles and debris were added, and a Kentish-plover egg filled with plaster-of-paris connected to a datalogger with a 36-gauge probe was put in this hand-made nest. These hand-made nests were always in exposed sites. T a5 and T egg were recorded every 40 s between 1000 and 1500 hours (GMT).
T e was recorded from unheated taxidermic Kentish plovers (Bakken 1992) in both standing and sitting postures in 1999, in both covered and exposed sites, using 24-gauge probes inserted into the central abdominal region of the taxidermic mounts, which were cast of copper. Although the use of this technique to characterise T e was criticised by Walsberg and Wolf (1996; but see Larochelle 1998), we used it as an indication of heat loads experienced by plovers nesting in different sites. Two male and two female taxidermic mounts were used, in standing (a male and a female) and sitting (a male and a female) postures, and connected to dataloggers that recorded temperatures every 5 min. The taxidermic mounts were placed facing southeast in sites used by nesting Kentish plovers. The sites were completely exposed, and some of them were artificially covered by twigs of Arthrocnemum bushes, so that these nests resembled concealed ones (nest cover category 3 of Amat et al. 1999a). A mount in sitting posture was placed on a nest scrape, and another in standing posture was placed ~15 cm from the nest scrape. About 70 cm from the nest scrape, a scrape similar in size and depth to those of Kentish plovers was made in an exposed site, into which we added pebbles and small twigs to resemble nest lining. Another taxidermic mount in sitting posture was placed on this hand-made scrape, and its corresponding pair in standing posture ~15 cm from the scrape. Each pair of mounts was in either the exposed or the covered site. We recorded T a5 with a 20-gauge probe placed between the covered and exposed site. Temperatures were recorded from sunrise to sunset (approximately 0530–1930 hours) at each pair of nest scrapes. Individual mounts were alternated between concealed and exposed sites for 19 recording days.
The instruments used to record temperatures were calibrated to the nearest 0.1°C against a standard thermometer.
Thermoregulatory behaviour
The thermoregulatory behaviour of incubating Kentish plovers was observed from blinds located 15–20 m from focal nests during 1997–1999. The observations lasted 2–5 h and were conducted between 1000 and 1500 hours. Two groups of nests were established, one of which had no cover and served as control. The other nests had no cover but they were covered as described above. Treatments were randomly assigned to nests. Although males incubate mainly during the night, they may also participate in diurnal incubation (Fraga and Amat 1996; see also below). To keep conditions constant, males were removed from the two groups of nests before 0530 hours, and kept in captivity for the duration of observations at their respective nests. Food and water were provided ad libitum to males maintained in captivity.
All observations started when the females returned to their nests after we had entered the blinds. The thermoregulatory behaviour was allocated to the following categories (see Grant 1982): standing, holding wings, erecting dorsal feathers (ptiloerection), gaping, panting and belly-soaking. The time of day in which each one of these behaviours was performed was recorded with a timer to the nearest minute, as well as the moments when females left the nests and returned to them. The incubating behaviour was matched to T a5, for which we synchronised the timer and the dataloggers to the nearest 20 s.
To calculate an index of thermoregulatory behaviour that integrates all thermal behaviours recorded, a procedure similar to that of Bartholomew and Dawson (1979) was followed. The time that each bird performed every thermal behaviour [standing (ST), gaping (GA), panting (PA), ptiloerection (PT), wing-dropping (WD), and belly-soaking (BS)] was calculated relative to the total time that the bird was on the nest (TN). The index of thermoregulatory behaviour (TB) was calculated as:
TB may be >1 because some of the thermal behaviours may be performed simultaneously.
Wind may have some effect on thermoregulation because of its effects on convective heat loss (Whittow 1986), but its effects on thermoregulatory behaviour were not measured because, as stated, the aim of the study was not to characterise the thermal environment of incubating plovers, but rather to study the responses of the birds to acute thermal stress. When ambient temperature is high, leaving a nest unattended even for a short period may compromise nesting success (Grant 1982; Jehl and Mahoney 1987).
Incubation bouts during daytime
Female Kentish plovers typically incubate during daytime and males at night. However, during the second half of the nesting season (after 15 May), males spent some time incubating during the day, mainly around midday (Fraga and Amat 1996). The participation of males in diurnal incubation during the hottest part of the day may be related to the inability of females to stay at nests for long periods when T a is high. We compared the frequencies with which males were captured at nests during midday (1000–1500 hours) in naturally covered and exposed sites. We also compared whether males were more frequently captured during midday at exposed nests during the second half of the nesting season, when T a was higher, than during the first half of the season. Furthermore, it was also tested whether there was a relationship between the probability of capturing males at nests during midday and maximum ambient temperatures at a nearby (<5 km) meteorological station the same days on which the captures were made.
Because observations of male and female incubation bouts as a function of time of day would provide more information to relate male participation in diurnal incubation to microclimate conditions than the capture frequencies of males, we also recorded whether incubation bouts during three parts of the day were dependent on T a5, for which we made observations in exposed nests in which neither adults had been removed. Neither of the nests on which these observations were conducted was included in the experiments described above. The observations were made from blinds as indicated above, and three periods were considered: early morning (0530–0730 hours), midday (1000–1500 hours) and late afternoon (1700–1930 hours). During each one of these periods, T a5 was recorded as indicated above. Observations started when adults returned to the nests and resumed incubation after we had entered the blinds, and lasted 1.6±0.22 (SD) h at morning nests (n=11), 4.6±0.40 h at midday nests (n=12) and 1.6±0.37 h at afternoon nests (n=10). The participation of males in incubation was calculated as the proportion of time attending the nest in relation to total time that the focal nest was attended by either adult during each one of the periods considered above.
Statistical analyses
Data were tested for normality before being analysed with parametric tests. If they were not normally distributed, they were transformed following Zar (1984). To compare operative temperatures among sites (covered, exposed) and bird postures (sitting, standing), we averaged the temperatures recorded at the same time during the 19 recording days. As temperatures were recorded every 5 min, this yielded 79 data points for each site and posture. Statistical tests were conducted using SYSTAT (Wilkinson 1990) or STATISTICA (StatSoft 1993). Unless otherwise indicated, mean values are presented with ±1 SD.
Results
Operative temperatures
When the taxidermic models were in exposed sites (Fig. 1), mean temperatures during diurnal time (n=79) were higher when they were in sitting (39.96±7.91°C) than in standing posture (39.39±6.62°C) (paired t=3.38, P=0.001). Mean temperatures in covered sites (n=79) were higher for models in standing (30.82±4.07°C) than in sitting posture (29.71±3.74°C) (paired t=15.85, P<0.001). Mean temperatures were much higher for models in exposed than in covered sites (Fig. 1), in both standing (paired t=22.81, P<0.001) and sitting postures (paired t=17.88, P<0.001).
Mean operative environmental temperatures for Kentish plovers in covered and exposed nesting sites, and mean environmental temperature 5 cm above ground surface, every 5 min during daytime (n=19 recording days). Operative environmental temperatures were measured using taxidermic mounts in sitting and standing postures. Standard errors of means are not presented for clarity
The equations that described the relationship between T e and T a5 are presented in Table 1. During the hottest part of the day (1000−1600 hours), there was a thermal difference of 10–15°C in the operative temperatures of plovers between covered and exposed sites (Fig. 1). Using equation 20 of Calder and King (1974), and assuming a body temperature (T b) of 41°C (J.A. Amat, unpublished data), the thermo-neutral zone for breeding Kentish plovers (mean body mass=42 g; Fraga and Amat 1996) may be estimated to extend down to about 28°C. Therefore, the thermal range encountered by birds in covered nest sites during most of daytime was closer to the thermal neutral zone than that encountered by plovers in exposed sites.
Thermal behaviour and egg temperatures
None of the Kentish plovers incubating in experimentally covered sites was observed exhibiting any thermoregulatory behaviour indicative of heat dissipation during observation periods lasting on average 230±29 min (n=12). Mean T a5 during such observation periods was 29.82±2.97°C. In contrast, in exposed sites, the plovers were frequently observed performing behavioural responses to dissipate heat (Table 2). On average, observations at exposed sites lasted 229±44 min (n=12), and mean T a5 at such sites (30.54±3.23°C) did not differ statistically from that recorded when observing experimentally covered nests (t 22=0.57, P=0.577)
The thermal behaviour in exposed sites can be arranged in a graded series, according to the lower T a at which each particular behaviour was first recorded, from standing and gaping to belly-soaking (Fig. 2). The frequencies of occurrence of these thermal behaviours (expressed as percentage of time that the plovers performed them relative to the time that the birds were in their nests) that were more closely related to T a5 were those of panting (r 10=0.89, P<0.001) and ptiloerection (r 10=0.85, P<0.001). The thermoregulatory index was also related to ambient temperature (Fig. 3, r 10=0.75, P=0.005). Part of the variation in this index may be explained by the action of wind on convective heat loss, and hence the differences in the value of the index for a same T a5.
Mean T a5 (±1 SE) at which female Kentish plovers (n=12) incubating in exposed nests performed thermoregulatory behaviours to dissipate body heat. Each data point is the average of the mean temperatures computed for all occurrences of a particular behaviour for individual birds. Observation of each individual lasted 3–5 h
Relationship between T a5 and an index of thermoregulatory behaviour of female Kentish plovers (n=12) incubating in exposed sites. The index was calculated as the sum of times expended in each thermoregulatory behaviour in relation to total time at nests, during observation periods lasting 3–5 h. A number denotes two overlapping points
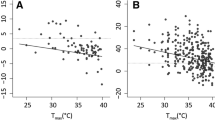
Mean T egg in covered attended nest (30.4±3.40°C, n=12) was lower than in unattended exposed sites (36.5±4.36°C, n=12) (t 22=4.78, P=0.001). There were no differences in T egg between exposed nests (36.0±1.62°C, n=12) and unattended exposed sites (37.1±5.61°C, n=12) (t 22=0.83, P=0.425). Nevertheless, maximum T egg in attended exposed nests was 38.8°C, whereas in unattended exposed sites it was 50.8°C, which may be a lethal temperature (Grant 1982).
We regressed T egg in unattended nests on T a5 (both in °C) to obtain the equation describing the relationship between both variables
Incubation bouts during daytime
In covered sites 14.7% of 75 individuals caught during midday were males, whereas in exposed sites 35.6 % of 135 individuals were males, a significant difference (G 1=11.2, P=0.001). As expected, during the first half of the season, the frequencies of captures of males during midday were lower (18.5% of 65) than during the second half of the season (32.4% of 145) (G 1=4.55, P=0.033). Interestingly, there was a significant relationship between the probability of capturing males at nests during midday and maximum ambient temperatures the same days in which the captures were made, but only for exposed nests (logistic regression: χ1 2=10.33, P=0.001, n=130), and not for covered ones (χ1 2=0.18, P=0.667, n=68).
Observations of male and female incubation bouts as a function of time of day also indicated that the participation of males in diurnal incubation was related to T a5. Indeed, the proportion of time that males spent in diurnal incubation was significantly related to T a5, but only during midday (r 10=0.63, P=0.027), and not during the early morning (r 9=−0.19, P=0.584) or late afternoon (r 8=0.13, P=0.714) when ambient temperatures were lower.
Nest desertion
During most breeding seasons in 1991–1999, there were some heat waves during which maximum daily T a5 was >45°C (J.A. Amat, unpublished data). These high temperatures may determine that the plovers are unable to incubate for long periods in exposed sites, and even if they resort to shortening incubation bouts, the incubating bird may be able to stay at the nest for only a short period, so that exposure to direct solar radiation would result in T egg≥57°C, according to Eq. 1, which may be fatal for developing embryos. Propensity of plovers to desert their nests was affected by proximity of the nest to water, with nests located close to water being deserted less frequently (39.0% of 346) than nests located far from water (57.6% of 118) (G 1=12.32, P<0.001). This was so in spite of the nests located close to water being more frequently placed in exposed sites (62.4% of 346) than were the nests in sites far from water (51.7% of 118) (G 1=4.17, P=0.041).
Discussion
Because incubating Kentish plovers incur greater risk of being depredated in covered than in exposed sites, the benefits of nesting in thermally favourable sites are constrained by predation risk (Amat and Masero 2004). The results of this study suggest that thermoregulatory costs during daytime are much higher in exposed than in covered sites. Consequently, the rate at which birds nesting in exposed sites exhibit behaviours related to heat dissipation should be higher than that of birds nesting in covered sites. Indeed, we have shown that Kentish plovers exhibit a variety of behavioural strategies to face high ambient temperatures when incubating in exposed nests. Lima (1993) indicated that avian species using exposed habitats should experience more thermally stressful conditions, and asked whether these species should exhibit enhanced abilities to withstand such physiological stress. Tieleman and Williams (1999) assessed the role of hyperthermia in the water economy of desert birds, and found no differences between desert and non-desert birds in their degree of hyperthermia.
We used only four taxidermic mounts to characterise the operative temperatures of plovers. To avoid pseudoreplication, we should have used a larger number of mounts if temperatures were affected by the mounts (see Walsberg and Wolf 1996). In any case, the differences in temperatures that we found between exposed and covered sites were so large that the biological effects should have been much greater than the statistical effects were likely to be.
When we conducted the behavioural observations, the temperatures experienced by plovers in covered sites were likely to be within their thermo-neutral zone, which may explain why birds incubating under shade did not exhibit any thermoregulatory behaviour. However, in exposed sites, the plovers experienced temperatures that stressed them, and they reacted to these heat loads with behavioural responses, ranging from standing instead of sitting on the eggs, to belly-soaking. The results of experiments with taxidermic models showed that T e was indeed lower for birds standing that for those sitting (see also Downs and Ward 1997; Brown and Downs 2003). When T a5 rose (up to ~35°C), Kentish plovers exhibited behavioural responses that probably increased water evaporation (gaping, panting, ptiloerection, wing dropping). Also, when ambient temperature increased, pair members shortened incubation bouts.
Birds can regulate their T b at 45°C T a, but few are capable of withstanding T a ≥50°C (Dawson and Hudson 1970; Marder and Ben-Asher 1983). In fact, Jehl and Mahoney (1987) found that T a >47°C caused mortality in chicks of the California gull (Larus californicus). Kentish plovers may experience T a >45°C during incubation (George Kainady and Al-Dabbagh 1976; Grant 1982). Although when we conducted the behavioural observations T a5 was usually <40°C, during some of the breeding seasons there were some heat waves, lasting 3–10 days, during which maximum T a5 was >45°C (J.A. Amat, unpublished data). Using equations in Table 1, at T a varying 45−50°C, T e of plovers in covered sites would vary 42−45°C, whereas in exposed sites would vary 63–68°C. These very high values of T e may determine that, although pair members shorten incubation bouts, the plovers in exposed sites would not be able to withstand such thermal conditions. Leaving a nest unattended would mean that T egg would reach lethal levels in only 2 min (Grant 1982), and if this happens, the nest is likely to be abandoned. However, susceptibility to thermal stress seemed to change in relation to proximity to water, as suggested by the higher frequency of nest desertion in sites far from water, where there was no possibility of belly-soaking, than in sites near water. Therefore, proximity to water would allow a more continuous nest attendance.
Although birds may have some physiological mechanisms to respond to heat stress (Coleman et al. 1995; Tieleman and Williams 1999; Zhou et al. 1999), few species are able to survive exposure to T a >48°C for periods of 270 min (Marder 1983). In fact, for homeotherms, a likely approximation of the upper lethal temperature of the environment may be about 3°C above body temperature (Peter 1993). Such temperatures may be experienced by Kentish plovers during heat waves, and to survive they probably have to desert their nests.
In summary, because of the likely lack of physiological adaptations to reduce heavy heat loads, Kentish plovers have to resort to behavioural solutions, but even so, nesting success is vulnerable to these behavioural solutions because heat stress for extended periods constrains parental nest attendance in exposed sites. This suggests that due to environmental factors a conflict may arise when incubating under very hot conditions, i.e. continuing incubation at the risk of being dehydrated or deserting the nest and incubation. This type of conflict may limit uniparental incubation by shorebirds in hot environments, and may restrict sexual conflicts over parental care.
References
Amat JA, Fraga RM, Arroyo GM (1999a) Replacement clutches by Kentish plovers. Condor 101:746–751
Amat JA, Fraga RM, Arroyo GM (1999b) Brood desertion and polygamous breeding in the Kentish plover Charadrius alexandrinus. Ibis 141:596–607
Amat JA, Masero JA (2004) Predation risk on incubating adults constrains the choice of thermally favourable nest sites in a plover. Anim Behav 67 (in press)
Bakken GS (1976) A heat transfer analysis of animals: unifying concepts and the application of field metabolism transfer to field ecology. J Theor Biol 60:337–384
Bakken GS (1992) Measurement and application of operative and standard operative temperatures in ecology. Am Zool 32:194–216
Bakken GS, Buttemer WA, Dawson WR, Gates DM (1981) Heated taxidermic mounts: a means of measuring the standard operative temperature affecting small animals. Ecology 62:311–318
Bartholomew GA, Dawson WR (1979) Thermoregulatory behavior during incubation in Heermann’s gulls. Physiol Zool 52:422–437
Bergstrom PW (1989) Incubation temperatures of Wilson’s plovers and killdeers. Condor 91:634–641
Brown M, Downs CL (2003) The role of shading behaviour in the thermoregulation of breeding crowned plovers (Vanellus coronatus). J Therm Biol 28:51–58
Calder WA III, King JR (1974) Thermal and caloric relations in birds. In: Farner DS, King JR (eds) Avian biology, vol IV. Academic, New York, pp 259–413
Coleman JS, Heckalthorn SA, Hallberg RL (1995) Heat-shock proteins and thermotolerance: linking molecular and ecological perspectives. Trends Ecol Evol 10:305–306
Davies SJJF (1982) Behavioural adaptations of birds to environments where evaporation is high and water in short supply. Comp Biochem Physiol 71A:557–566
Dawson WR, Hudson JW (1970) Birds. In: Wittow GC (ed) Comparative physiology of thermoregulation, vol 1. Academic, New York, pp 224–302
Downs CT, Ward D (1997) Does shading behavior of incubating shorebirds in hot environments cool the eggs or the adults? Auk 114:717–724
Fraga RM, Amat JA (1996) Breeding biology of a Kentish plover (Charadrius alexandrinus) population in an inland saline lake. Ardeola 43:69–85
George Kainady PV, Al-Dabbagh KY (1976) Some observations on the behaviour of incubating Charadrius alexandrinus on hot summer days. Bull Basrah Nat Hist Mus 3:121–137
Grant GS (1982) Avian incubation: egg temperature, nest humidity, and behavioral thermoregulation in a hot environment. Ornithol Monogr 30:1–75
Huey RB (1991) Physiological consequences of habitat selection. Am Nat 137:S91–S115
Jehl JR Jr, Mahoney SA (1987) The roles of thermal environment and predation in habitat choice in the California gull. Condor 89:850–862
Kalsi RS, Khera S (1986) Some observations on breeding and displacement behaviour of the redwatted lapwing, Vanellus indicus indicus (Aves: Charadriidae). Res Bull Sci Punjab Univ 37:131–141
Kosztolányi A, Székely T (2002) Using a transponder system to monitor incubation routines of snowy plovers. J Field Ornithol 73:199–205
Kosztolányi A, Székely T, Cuthill IC (2003) Why do both parents incubate in the Kentish plover? Ethology 109:645–658
Larochelle J (1998) Comments on a negative appraisal of taxidermic mounts as tools for studies of ecological energetics. Physiol Zool 71:596–598
Lessells CM (1984) The mating system of Kentish plovers Charadrius alexandrinus. Ibis 126:474–483
Lima SL (1993) Ecological and evolutionary perspectives on escape from predatory attack: a survey of North American birds. Wilson Bull 105:1–47
Maclean GL (1967) The breeding biology and behaviour of the double banded courser Rhinoptilus africanus (Temminck). Ibis 109:556–569
Maclean GL (1975) Belly-soaking in the Charadriiformes. J Bombay Nat Hist Soc 72:74–82
Marder J (1983) Cutaneous water evaporation. II. Survival of birds under extreme thermal stress. Comp Biochem Physiol 75A:433–439
Marder J, Ben-Asher J (1983) Cutaneous water evaporation. I. Its significance in heat-stressed birds. Comp Biochem Physiol 75A:425–431
Nakazawa R (1979) Incubation behaviour of the Kentish plover, Charadrius alexandrinus, with special reference to the share of the sexes and of effect ground surface temperature. Misc Rep Yamashina Inst Ornithol 11:54–63
Page GW, Stenzel LE, Ribic CA (1985) Nest site selection and clutch predation in the snowy plover. Auk 102:347–353
Peter RH (1993) The ecological implications of body size. Cambridge University Press, Cambridge
Pianka ER (1994) Evolutionary ecology, 5th edn. Harper Collins, New York
Purdue JR (1976) Thermal environment of the nest and related parental behavior in snowy plovers, Charadrius alexandrinus. Condor 78:180–185
Salzman AG (1982) The selective importance of heat stress in gull nest location. Ecology 63:742–751
Schreiber EA (2002) Climate and weather effects on seabirds. In: Schreiber EA, Burger J (eds) Biology of marine birds. CRC, Boca Raton, pp 179–215
StatSoft (1993) STATISTICA, Release 4.5. Statsoft, Tulsa
Székely T (1996) Brood desertion in Kentish plover Charadrius alexandrinus: an experimental test of parental quality and remating opportunities. Ibis 138:749–755
Székely T, Cuthill IC, Kis J (1999) Brood desertion in Kentish plover: sex differences in remating opportunities. Behav Ecol 10:185–190
Thomas DH, Maclean GL (1981) Comparison of physiological and behavioural thermoregulation and osmoregulation in two sympatric sandgrouse species (Aves: Pteroclididae). J Arid Environ 4:335–348
Thomson SC, Brooke AP, Speakman JR (1998) Diurnal activity in the Samoan flying fox, Pteropus samoensis. Philos Trans R Soc Lond B 353:1595–1606
Tieleman BI, Williams JB (1999) The role of hyperthermia in the water economy of desert birds. Physiol Biochem Zool 72:87–100
Walsberg GE (1985) Physiological consequences of microhabitat selection. In: Cody M (ed) Habitat selection in birds. Academic, Orlando, pp 389–413
Walsberg GE, Wolf BO (1996) An appraisal of operative temperature mounts as tools for studies of ecological energetics. Physiol Zool 69:658–681
Ward D (1990) Incubation temperatures of crowned, black-winged, and lesser black-winged plovers. Auk 107:10–17
Weathers WW (1981) Physiological thermoregulation in heat-stressed birds: consequences of body size. Physiol Zool 54:345–361
Whittow GC (1986) Regulation of body temperature.In: Sturkie PD (ed) Avian physiology, 4th edn. Springer, Berlin Heidelberg New York, pp 221–252
Whittow GC (2002) Seabird reproductive physiology and energetics. In: Schreiber EA, Burger J (eds) Biology of marine birds. CRC, Boca Raton, pp 409–437
Wiebe KL, Martin K (1998) Costs and benefits of nest cover for ptarmigan: changes within and between years. Anim Behav 56:1137–1144
Wilkinson L (1990) SYSTAT: the system for statistics. SYSTAT, Evanston
Wolf BO, Walsberg GE (1996) Thermal effects of radiation and wind on a small bird and implications for microsite selection. Ecology 77:2228–2236
Zar JH (1984) Biostatistical analysis, 2nd edn. Prentice-Hall, London
Zerba E, Morton ML (1983) The rhythm of incubation from egg laying to hatching in mountain white-crowned sparrows. Ornis Scand 14:188–197
Zhou WT, Fujita M, Yamamoto S (1999) Thermoregulatory responses and blood viscosity in dehydrated heat-exposed broilers (Gallus domesticus). J Therm Biol 24:185–192
Acknowledgements
Consejería de Medio Ambiente (Junta de Andalucía) gave permission to conduct this research at Reserva Natural Laguna de Fuente de Piedra, as well as providing lodging and many other facilities. Manuel Rendón-Martos, director of the Nature Reserve, facilitated our work during all phases of the project. In the field, we were assisted by A. Arroyo, J.L. Arroyo, R. Camarena, J.M. Ramírez, J. Rubio, M. Siquier and M. Vázquez. G. Nehls assembled the taxidermic mounts, and F. Chiara gave us technical assistance with these mounts. Our thanks also go to F. Álvarez, I. Quintero, T. Székely, J. Wright and a referee for many helpful comments on the manuscript. Financial support was provided by Plan Andaluz de Investigación (research group RNM 0105) and Dirección General de Investigación Científica y Técnica (grants PB92-0115 and PB95-0110).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J. Graves
Rights and permissions
About this article
Cite this article
Amat, J.A., Masero, J.A. How Kentish plovers, Charadrius alexandrinus, cope with heat stress during incubation. Behav Ecol Sociobiol 56, 26–33 (2004). https://doi.org/10.1007/s00265-004-0758-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-004-0758-9