Abstract
Purpose
For complex proximal humeral fractures, severe displacement and comminuted fragments lead to poor clinical outcomes. Despite considerable management, the locking plate fixation is still a preference and the proper selection of its strategy for each individual seems to be essential.
Method
The available classification system of the fracture, determination of surgical intervention, the common complications and related causes, and the latest critical surgical strategies with locking plate fixation were discussed in this review.
Results
The frequent complications with complex proximal humeral fractures after operative treatment present a great challenge to orthopedic surgeons. In order to maintain the stability of locking plate fixation, several options including calcar screw, bone graft, bone cement augmentation, dual plate fixation, and fracture impaction were available for restoration of medial support.
Conclusion
Restoration of medial support seems of importance to provide solid stability and reduced complication for the complex fractures with fixation of locking plates.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Proximal humeral fractures (PHF) are seen most commonly in aged population, accounting for 4 to 9% of all fractures in adults [1]. The overall incidence considerably increased in the elderly population, particularly in the elderly over 65 years old [2]. The incidence of complex fractures types is usually positively associated with increasing age. The mechanism of PHF is largely attributed to low-energy fall, especially in the elderly population [3]. That is partly due to the decreased bone quality and mobility of the elderly.
The majority of PHF are either nondisplaced or minimally displaced, which are usually treated with conservative management [4, 5]. Various surgical techniques of open or closed reduction and internal fixation were applied for displaced and unstable PHF. Compared with conservative management, surgical treatment of displaced unstable fractures yields relatively satisfactory clinical results, especially for functionally active patients. The surgical treatment of PHF is expected to up to 20%, due to potential complications with conservative treatment in an aging population [6].
Currently, the treatment strategy of displaced and unstable PHF remains demanding. Various operative implants and techniques, such as percutaneous pinning, intramedullary nailing, plating, or shoulder arthroplasty, were described [7,8,9]. Locking plate fixation is the standard option with better stability for displaced fractures of the proximal humerus clinically, although there is lack of clear superiority among those methods. There is a trend that surgeons prefer locking plate fixation in patients with complex fractures of proximal humerus. The locking techniques with plate fixation were developed with various advantages for complex fractures of the proximal humerus, although several technique-related complications, such as hardware failure, osteonecrosis, nonunion, malunion, rotator cuff impairment, and impingement, were still reported [10]. The selection of management strategy may yield different clinical outcomes in the treatment of complex PHF with locking plates. This article aims to review the current management strategy of complex PHF with locking plate fixation. In addition, we also review available classification system, determination of plate treatment, management strategy, approaches, related complications, and rehabilitation regime in detail.
Fracture classification
The fracture type classification is of importance for the determination of appropriate treatment. Currently, there is not well-received fracture classification with entirely satisfactory guidance for treatment and prediction of outcome. The available classification systems were always associated with poor intra-observer and inter-observer reliability [11, 12]. In 1934, Codman defined the proximal humerus into four parts for fracture classification: the humeral head; the lesser tuberosity; the greater tuberosity; and the humeral shaft. Since then, several fracture type classifications were described for PHF [13, 14].
The Neer classification [13] is most widely used in the classification of the PHF. The displacement but the fracture line was emphasized in terms of Neer classification. The fracture displacement is defined as over 45 of angulation between fragment and at least 1 cm of displacement of fragments. Siebenrock et al. [12] reported that the Neer classification system seems to be slightly more useful than others in guidance of clinical treatment. Base on the potential risk of ischemia of proximal humerus and the severity of the fractures, including fracture location, dislocation, and status of the surgical neck, the AO classification was developed. The AO classification includes a total of 27 subgroups, indicating its complexity. Recently, Herbert et al. [14] published a new fracture type classification named pathomorpologic classification, based on the pathomorphologic analysis of these fractures. The relative position of head and shaft fragment is the determinant for main classification criterion. A radiology study reported that the pathomorphologic classification achieved a higher reliability than the Neer classification [15]. Recently, Articular, Surgical neck, Tuberosities (AST) classification described by Christian et al. [16], fracture line morphology of complex PHF described by Afsana et al. [17], and HGLS system described by Atul et al. [18] were developed for classification of PHF; their efficacy and need further validation. Familiarity with anatomy and pathomechanics of the proximal humerus helps understand the PHF. In a short summary, the fracture type classifications are still an important factor for the determination of intervention and prediction of clinical outcomes.
The determination of surgical intervention
The indications for treatment of PHF remain controversial. A previous treatment algorithm is emphasized on fracture classification based on radiographs, but the poor intra-observer reliability and relationship with clinical outcomes make less emphasis on them [11, 12]. The trend of determination of surgical intervention is moving to patients’ characteristics and expectation, considering the requirement of mobility and quality of life. Prior to moving forward to treatment, it is critical to assess advantages and disadvantage of each surgical strategy and influence factors. The severity of the fractures, bone quality, the integrity of rotator cuff, damage to adjacent structure, blood supply, and patient’s comorbidities should be evaluated before surgery.
Most of PHF were minimally displaced; these fractures can be treated with conservative management. The severe displaced fractures still need surgical intervention. Operative interventions improve fragment alignment by achievement of a more anatomical reduction and allow early mobility by providing better stability during healing period. In general, for displaced two-part surgical neck fractures, percutaneous wiring, intramedullary nailing, plating are commonly used and achieve efficient functional outcomes. Screwing or wiring perpendicular to the fracture plane or suturing is recommending in the treatment of two-part tuberosity fractures [19]. Although intramedullary nail is reported in fixation of two-part, three-part, and four-part fractures, the most frequent condition for intramedullary nail is the two-part surgical neck fractures. The locking plate system is usually an option for complex fracture with considerable advantage of angle stability, rigid fixation of fragment, stable fixation, and early functional exercises [19, 20]. The locking plates were widely used for the treatment of PHF, especially in patient with poor bone quality. For severe fractures with potential avascular necrosis, difficulty in restoration of proper functional anatomy due to comminuted fractures, arthroplasty, is a viable option as a stabilizing treatment [21].
Critical factor for treatment
Bone mineral density
One critical reason of fixation failure may be attributed to bone quality, especially osteoporotic bone in the elderly. Some studies reported that there was a direct relationship between bone mineral density (BMD) and mechanical stability in the proximal humerus, and the distribution of local BMD individually varies within the humeral head [22, 23]. Consistently, the medial, dorsal, and proximal aspects of proximal humeral have a higher BMD and bone strength than the remaining regions [24]. The screws of standard implants may not usually reach the high BMD region to enable the best bone purchase in the humeral head. Meyer et al. claimed that it is the local BMD and aging not surgical technique or initial displacement or angulation that have a significant effect on the fixation failure [25]. A mechanical assessment investigated whether DensiProve predicted the failure of the PHF where increasing cyclic mechanical loading until fixation failure was performed [26]. These data showed BMD significantly correlated with cycles with failure. The measurement of local bone quality provides valuable information, which partially make it suitable for surgeon to take measures to improve fixation stability.
Fracture head ischaemia
The anterolateral branch of the anterior circumflex artery is the main blood supply of the proximal humerus while only the posterior portion of the greater tuberosity and part of the humeral head were perfused by the posterior circumflex artery. The blood supply of the proximal head after injury and whether revascularization occurred is a critical factor of avascular necrosis (AVN) of the head fragment [27]. The development of necrosis of the post-fracture humeral head is independent of initial ischaemia, which is partially due to quick revascularization or creeping substitution [27]. Indeed, Hertal et al. [28] developed a method of prediction of humeral head ischaemia after intracapsular fracture of the proximal humerus. The length of posteromedial metaphyseal extension (calcar) and the integrity the medial hinge are the most relevant predictors of ischaemia after intracapsular fracture of the proximal humerus. The length of posteromedial metaphyseal extension is relatively negative with the severity of ischemia of proximal humeral head [28].
Medial support
Maintaining fracture fixation is the major challenge in the treatment of the complex PHF [29]. Several studies confirmed that the lack of proper anatomic reduction is the strongest predictor of the implant failure, especially in osteoporotic bone [29,30,31]. Varus malreduction lead to increased varus torque and high stress at the tip of the locking screws or pins due to altered arm lever of the rotator cuff [31]. The lack of medial column support may exaggerate these forces. Generally, patients with type C fractures, four part fracture, and/or medial calcar disruption were vulnerable to the varus collapse [32]. Several strategies that may improve the mechanical stability are under the control of surgeons. A medial cortical buttress or achieving mechanical support of the inferomedial region of the proximal humerus counteracts these forces, which seems to be of importance for maintaining fracture reduction [30, 33].
Medial support strategies of PHF
Generally, locking plates alone are unable to provide effective stability of the humeral head from the lateral position [34]. Complex PHF usually challenged the reduction and stability of the osteosynthesis, especially complicated with metaphyseal extension or comminution [31, 33]. In complex PHF, a large metaphyseal defect of the proximal humerus compromised the medial cortical support. Restoration of medial cortical support is usually required to achieve better stability. Currently, there were commonly the four type methods of medial support restoration.
Locking plate with inferomedial calcar screw
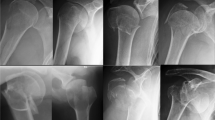
The locking plate combined with inferomedial calcar screw support maybe the most commonly used method of medial support for PHF (Fig. 1). The inferomedial calcar screw was introduced to the cancellous bone of the humeral head close to the calcar region. A better stability was achieved by the combination of locking plate fixation and additional inferomedial calcar screws when medial support is deficient, which is verified by lining biomechanical evidence [35, 36]. Nevertheless, the combination of locking plate and inferomedial calcar screw may be not a good option in severe comminuted PHF. A retrospective study illustrated that treatment with locking plate fixation and fibular allograft augmentation achieved significantly greater shoulder functional score than the combination of locking plate fixation and inferomedial screw in patients with four part PHF, partially because of better medial support [37]. For a better stability of medial support, an additional inferomedial calcar screw should be combined with other method of restoration of medial support, especially in severe comminuted PHF and in patients of low-quality bone.
Locking plate fixation with an inferomedial calcar screw for proximal humeral fracture. a The pre-operative antero-posterior X-ray of a 62-year-old healthy male patient shows a 3-part proximal humeral fracture. b The pre-operative antero-posterior X-ray of the proximal humeral fracture fixed with locking plate and an inferomedial calcar screw
Bone graft augmentation
Bone allograft is a well-received method of structural augmentation of unstable PHF. Gardner et al. [38] firstly introduced a segment of fibula allograft endosteally to aid reduction and restoration of medial support of the proximal humerus with comminuted medial column. Panchal et al. [39] reported that 26 patients (72.2%) patients with unstable PHF were evaluated as good or excellent of UCLA rating scale and achieved good humerus neck-shaft angle after treatment with a locking plate and fibular strut allograft at two years of follow-up. There were only two case of avascular necrosis of the humeral head and varus collapse in the case series. In addition, iliac crest bone autologous graft was adopted for additional medial support in unstable displaced PHF [40]. A randomized controlled trial compared the clinical and radiographic outcomes of locking plate and the combination of locking plate and iliac crest bone graft in the treatment of comminuted PHF. The combination application contributed to better fracture healing clinically and radiographically without severe complications in all 18 patients. Two patients underwent total shoulder replacement due to nonunion in the locking plate group. Notably, the allograft was suggested to be placed maximally near the medial calcar to have a direct reduction of the medial column and inserted into the subchondral bone of the humeral head to lift it superiorly [39]. The biomechanical analysis of cadavers [41] proved the superior advantages of additional bone graft to locking plate. Bone graft with locking plate increased stiffness and load of varus failure with a direct medial support but not an anti-rotation stability.
Bone cement augmentation
Augmentation of bone cement shares a similar principle with bone graft in restoration of medial support of PHF (Fig. 2). But bone cement provides additional screw augmentation to improve primary stability [42, 43]. Comminuted PHF usually occur in aged patients with osteoporosis. The low bone density usually fails to provide stable anchorage for plate and screws, because of shear force at the bone implant interface. There is usually a bone defect after reduction of PHF due to impaction-induced cancellous bone compression, then bone cement is an alternative for augmentation and fulfill of cavity. Therefore, it was critical to inject enough bone cement to fulfill the cavity and to the right position to ensure inferomedial screw augmentation, based on which medial support can be further augmented. The locking plate with cement contributed to significantly better biomechanical stability than locking plate alone, which was verified by lining biomechanical studies [44]. A retrospective case series analyzed the locking plate in conjunction with calcium sulfate augmentation in the treatment PHF and fracture dislocation; 18 out of 22 cases achieved good clinical outcomes [45]. The major concern about bone cement may be attributed to necrosis induced by exothermic reaction. Although the bone cement induced by temperature theoretically did not reach the threshold for osteonecrosis and apoptosis of cartilage and subchondral bone, surgeons still need to pay attention to that. Another concern is the leakage of bone cement, resulting in subsequent complication. The medial calcar should be well reduced before injection of bone cement to minimize the possibility of leakage. If satisfactory reduction of medial calcar cannot be reached, bone cement needs to be injected slowly and smoothly under the fluoroscopy, and the needle tip of the syringe should be placed far away from medial calcar.
Dual plate fixation
Regarding severe complex PHF, conventional locking plates with or without a calcar screw fail to achieve efficient mechanical stability from lateral tension band position, which usually induced higher risk of implant failure and subsequent varus collapse and bone nonunion. Therefore, hybrid double plate is described to be another alternative. The double plates that were intended to restore medial support of the proximal humerus were developed.
Lateral and anterior plates
The double plates were firstly report by Wanner et al. [46], displaced two-part, three-part, or four-part PHF were treated by two one-third tubular plates on the anterior and lateral aspects of the proximal humerus. In the series of 71 patients, 63% patients achieved a good or very good clinical outcome, based on their self-design protocol [46]. Combined application of a monoaxial locking plated and a third tubular plate were introduced in a pilot study of seven patients and yielded a constant score of 80 points one year after injury [47]. A biomechanical study compared the hybrid double-plate osteosynthesis with calcar screw to test whether there is a superior method [48]. The hybrid method consists of locking plate and an additive one-third tube plate in the bicipital groove region achieved similar rigid and robust effect to calcar screw. Although the hybrid double plate osteosynthesis tended to confer higher stiffness than the medial support screws at higher cycles, no statistical significance was detected between hybrid plate and calcar screw. The additional anterior plate may contribute to a higher mechanical stability when PHF come with comminution of medial column (Fig. 3a).
Lateral and posterior plates
Choi et al. [49] developed a dual plate fixation technique for the treatment of multi-fragmentary PHF (Fig. 3b). The combination of low-profile locking plate and polyaxially locking distal radial plate theoretically directly provides a rigid and strong two column stability. In this model, the distal radius plate was anticipated to not only prevent varus collapse of the humeral head but also maintain anterior-posterior angulation of the humeral head from posterior aspect.
Lateral and medial plates
Theoretically, the combination of lateral locking plate and medial locking plate provides direct medial support and anti-rotational stability relative to the combination of lateral and dorsal locking plates (Fig. 3c). A finite element analysis conducted by He et al. [50] compared the biomechanical stability among a locking plate, a locking plate with an intramedullary fibular graft, and a locking plate with medial distal radius plate in the treatment of PHF without medial support. The data demonstrated that the combined application of lateral locking plate and medial locking plate contributed to significantly greater and regional construct stiffness and decreased stress on lateral locking plate. Sam-Guk Park et al. [51] performed two cases of combination fixation of PHF with lateral plate with an additional medial plate in osteoporotic aging people. The medial locking plate was applied when the locking plate cannot provide sufficient stability of comminuted PHF and anterior circumflex humeral arteries were compromised after fracture. The clinical data showed that the fixation allows immediate exercise after surgery and patients achieved a satisfactory outcome without inclination of the humeral neck and osteonecrosis of the humeral head.
Fracture impaction
In addition to the above method, fracture impaction and shaft medialization with locking plate fixation was described to restore the integrity of the medial support (Fig. 4). In this method, the humeral shaft was medially translated and impacted to into the humeral head to achieve medial buttress [52]. A biomechanical cadaveric study compared the biomechanical stability of fracture impaction with nonaugmented plate fixation for PHF with medial communication [52]. None of augmented constructs with fracture impaction failed whereas five of six (83.3%) of nonaugmented constructs collapsed before the peak loading, revealing that the additional fracture impaction has a superior ability to resist repetitive varus loading to locking plate fixation alone. Another study used numerical methods to compare the biomechanical effect of PHF fixed by fracture impaction with locking plate or intramedullary nail [53]. The data demonstrated that the impaction with locking plate or intramedullary nail increased fixation stability and decreased peak stress by sharing loading with implant when all loading concentrated on nonimpacted model, despite the normal bone quality or poor bone quality. Compared with other methods of restoring medial support, the unique advantage of fraction impaction is free of an allograft or bone cement and subsequent reduction of expense. Additionally, treatment with fracture impaction shortens surgical time and avoids further surgical injury due to additional procedure. However, fracture impaction inevitably shortens the length of the humerus, subsequently altering the torque of the surrounding muscle, especially the deltoid, and the mobility of extremity of the injured side maybe affected. Therefore, in order to avoid severe affected outcome, the altered length of the humerus should not exceed 2 cm after fracture impaction [52]. Notably, a clinical study about PHF treated with fracture impaction remains unreported; thus, the fracture impaction with locking plate may need to be further explored.
Complications
Despite development of modern plates and techniques, the incidence of complications remains at a relative high level. The incidence of complications varied from 17.8 to 35.7% [20, 54], and the revision rate is reported to up to 25%. Generally, most complications occurred within 3 months postoperatively, but a minimum of two years of follow-up may be appropriate to monitor the vitality of the proximal head.
Varus collapse
Varus collapse was one of the most post-operative complications. Varus collapse was frequent and commonly followed by secondary screw penetration and secondary subacromial impingement. Varus collapse leads to a limited range of shoulder motion, especially in active forward elevation and abduction. Varus collapse of the head fragment with an incidence of 16.3% was reported as the most common complication. The insufficient restoration of medial support might be responsible for varus collapse. As a result, an increasing number of surgeons paid special attention to restore medial support by kinds of methods like above described, which contributed to significantly reduced varus collapse. Additionally, Voigt et al. [55] showed the decreased rate of varus collapse with polyaxial plate relative to monoaxial plate, because of more options for inferomedial screw polyaxial plates. Additionally, Schliemann et al. [56] believed that the plate with similar elastic modulus to bone may induce a lower rate of varus collapse.
Intra-articular perforation
Intra-articular perforation of the head screws maybe the most frequent implant-related complications with rates ranging from 0 to 23% [57]. The screw penetration can be divided into primary and secondary screw penetration. The primary screw penetration was referred as a screw into glenohumeral joint during surgery, which is iatrogenic injury. The routine application of fluoroscopy in three planes is recommended to minimize the incidence of primary screw penetrations. The second screw penetration was referred as screw violence into glenohumeral joint due to the collapse of the humeral head. Several studies recommended to place the screw away from subchondral bone, ranging from 2–3 mm to 5–10 mm, and a subsequent incidence of intra-articular screw penetration decreased significantly [58, 59]. The restoration of medial support, such as intramedullary graft and calcium phosphate cement augmentation, also effectively decreased the incidence of secondary screw penetration. Both primary and secondary screw penetrations were the main cause of glenoid destruction.
Subacromial impingement
Subacromial impingement was reported at a rate around 4.8%, which were mainly caused by a poor position of lateral plate, malunion of the greater tuberosity, or the sequelae of varus collapse of the humeral head [60]. Therefore, a special attention should be paid on satisfactory position of plate and prevention of varus deformity to reduce this complication. Konrad et al. [61] reported five cases in the setting of 270 patients; the superior placement of plate leads to impingement; removal of the plate might be required if the symptom of subacromial impingement was severe. Taking the shoulder through a full range of motion arc before closing the wound was critical to detect any symptoms of impingement. Ricchetti et al. [62] proposed that the plate placed 15–20 mm beneath the tip of the greater tuberosity may be helpful to avoid impingement.
AVN
AVN is a problematic complication with PHF historically, with a rate of 4.4–10.8% [63, 64]. The compromising blood supply to the humeral head is responsible for AVN, and the integrity of the medial hinge, length of the dorsomedial metaphyseal extension, and fracture types were essential to predict the AVN. The deltoid splitting or minimally invasive approaches have been attempted to preserve the blood supply via less disruption of adjacent soft tissue. Notably, Fjalestad et al. [65] illustrated a higher rate of AVN with non-operative treatment than operative treatment in patients with displaced 3- and 4-part PHF. However, a one year follow-up study compared the deltoid splitting and deltopectoral approaches and showed no significantly difference in the rate of AVN [66]. A long-term follow-up study with over a mean follow-up of 2.5 years reported a lower rate of AVN in patients treated with minimally invasive deltoid splitting approach compared with the conventional deltopectoral approach [67].
Nonunion
Nonunion, as an uncommon complication during PHF, often causes severe shoulder pain and dysfunction. The complications were mainly induced by over-aggressive shoulder rehabilitation, inadequate fixation of fracture, and poor patient compliance. In addition, the fracture nonunion is associated with patients’ comorbidities, like osteoporosis, mental illness, and alcoholism.
Salvage treatment
Fracture malunion, nonunion, infection, iatrogenic destruction, the occurrence of avascular necrosis of the humeral head, and loss of plate fixation are not rare complications during an operative treatment, which is mainly caused by severe communication of the fractures, poor surgical technique, or osteoporosis. All of the above complications lead to malfunction of the shoulder and inevitable shoulder pain. Few effective options are available for orthopaedic surgeons when plate fixation fails during surgery or post-operative. Removal and revision of the fixation, capsular release and arthrolysis, and conversion to shoulder arthroplasty can be an alternative as salvage treatment.
For patients with shoulder stiffness who achieved a satisfactory fracture union, capsular release under anaesthesia or arthroscopic arthrolysis could be a good strategy for restoration of shoulder function and satisfactory range of motion. When patients were complicated with implant-related infection, removal of implant, and debridement and irrigation of infection were essential before further intervention including re-fixation or shoulder arthroplasty. Primary revision is required when the occurrence of malreduction and subsequent malunion, mostly in the greater tuberosity and the humeral head, because of severely affected shoulder function and negative effect on patients’ routine activity. Another common fixation failure is secondary screw cut out due to the reduction of fracture fragment, which leads to the subsequent removal of screw. Comes with primary or secondary screw cut out, which commonly followed by glenoid destruction.
For complex PHF, in which function reduction and fixation cannot be achieved by locking plate, because of malunion and nonunion, more than 50% of humeral head was involved in impaction; a severe displacement of fracture due to loss of reduction and a resultant damage to blood supply to the humeral head were observed [68]; hemiarthroplasty (HA) is a viable and stabilizing treatment, especially in aged patients with low bone stock [21]. Reverse shoulder arthroplasty (RSA) is an ideal candidate when patients are with late symptomatic complication of existing impaired shoulder rotator cuff, clinically significant osteoarthritis, or fracture nonunion. Moreover, RSA demonstrated a promising result as secondary intervention following failure of open reduction and internal fixation [69]. Compared with HA, RSA is independent with the tuberosity healing, and RSA can be selected as the final solution when other revision strategies cannot be appropriately performed, such as patients with poor bone quality and without intact rotator cuff.
Rehabilitation strategy
Physiotherapeutic intervention and rehabilitation regimen of shoulder after plate fixation remained inconsistent currently. The Neer-described rehabilitation regime is the mostly accepted clinically [70]. The passive exercises are initiated with an injured arm immobilized by a shoulder bandage during the first three weeks after surgery. The active exercise of the shoulder can be started with a sling immobilization during the following three weeks. Before the union of the greater tuberosity, active elevation over the shoulder should be avoided and the exercise began nine weeks after surgery at least. A time point of 12 weeks after surgery is for the start of a passive stretching and weighted exercise. It should be noted that the post-operative rehabilitation following plate fixation of PHF should be established and modified by the responsible surgeon based on the previously reported rehabilitation regimes and the evaluation of patient’s individual condition. Regarding the rehabilitation regime establishment, all conditions including the bone quality, fracture type, patient’s comorbidities, patient’s requirement of shoulder function, age, and radiological results during follow-up should be taken into consideration.
In summary
Currently, restoration of medial support for prevention of complications is well received by surgeons clinically. The locking plate is still a preference of orthopaedic surgeons. For effectively maintaining mechanical stability of complex PHF, one or more methods of medial support with locking plate are expected, taking BMD, potential ischaemia of the head fragment, and other factors into consideration. The algorithm of the selection of medial support combined with locking plate in the treatment of PHF was summarized (Fig. 5), and the determination of treatment is also dependent on patients’ characteristics and expectation. Subsequently, patients with locking plate fixation of complex PHF should follow the rehabilitation protocol to achieve a satisfactory clinical outcome, and the surgeon should be responsible for the modification of the rehabilitation regime. For severe complication occurrence of complex PHF after plate fixation, such as avascular necrosis, malunion or nonunion compromising normal shoulder function, and other joint incongruity, shoulder arthroplasty may be an effective salvage method.
References
Badman BL, Mighell M (2008) Fixed-angle locked plating of two-, three-, and four-part proximal humerus fractures. J Am Acad Orthop Surg 16(5):294–302
Nguyen TV, Center JR, Sambrook PN, Eisman JA (2001) Risk factors for proximal humerus, forearm, and wrist fractures in elderly men and women: the Dubbo Osteoporosis Epidemiology Study. Am J Epidemiol 153(6):587–595. https://doi.org/10.1093/aje/153.6.587
Bergdahl C, Ekholm C, Wennergren D, Nilsson F, Moller M (2016) Epidemiology and patho-anatomical pattern of 2,011 humeral fractures: data from the Swedish Fracture Register. BMC Musculoskelet Disord 17:159. https://doi.org/10.1186/s12891-016-1009-8
Nho SJ, Brophy RH, Barker JU, Cornell CN, MacGillivray JD (2007) Management of proximal humeral fractures based on current literature. J Bone Joint Surg Am 89(Suppl 3):44–58. https://doi.org/10.2106/jbjs.G.00648
Tejwani NC, Liporace F, Walsh M, France MA, Zuckerman JD, Egol KA (2008) Functional outcome following one-part proximal humeral fractures: a prospective study. J Shoulder Elb Surg 17(2):216–219. https://doi.org/10.1016/j.jse.2007.07.016
Egol KA, Kubiak EN, Fulkerson E, Kummer FJ, Koval KJ (2004) Biomechanics of locked plates and screws. J Orthop Trauma 18(8):488–493. https://doi.org/10.1097/00005131-200409000-00003
Park MC, Murthi AM, Roth NS, Blaine TA, Levine WN, Bigliani LU (2003) Two-part and three-part fractures of the proximal humerus treated with suture fixation. J Orthop Trauma 17(5):319–325
Rowles DJ, McGrory JE (2001) Percutaneous pinning of the proximal part of the humerus. An anatomic study. J Bone Joint Surg Am 83(11):1695–1699. https://doi.org/10.2106/00004623-200111000-00012
Park YK, Kim SH, Oh JH (2017) Intermediate-term outcome of hemiarthroplasty for comminuted proximal humerus fractures. J Shoulder Elb Surg 26(1):85–91. https://doi.org/10.1016/j.jse.2016.05.008
Robinson CM, Page RS, Hill RM, Sanders DL, Court-Brown CM, Wakefield AE (2003) Primary hemiarthroplasty for treatment of proximal humeral fractures. J Bone Joint Surg Am 85(7):1215–1223. https://doi.org/10.2106/00004623-200307000-00006
Siebenrock KA, Gerber C (1993) The reproducibility of classification of fractures of the proximal end of the humerus. J Bone Joint Surg Am 75(12):1751–1755. https://doi.org/10.2106/00004623-199312000-00003
Papakonstantinou MK, Hart MJ, Farrugia R, Gabbe BJ, Kamali Moaveni A, van Bavel D, Page RS, Richardson MD (2016) Interobserver agreement of Neer and AO classifications for proximal humeral fractures. ANZ J Surg 86(4):280–284. https://doi.org/10.1111/ans.13451
Neer CS 2nd (1970) Displaced proximal humeral fractures. I. Classification and evaluation. J Bone Joint Surg Am 52(6):1077–1089
Resch H, Tauber M, Neviaser RJ, Neviaser AS, Majed A, Halsey T, Hirzinger C, Al-Yassari G, Zyto K, Moroder P (2016) Classification of proximal humeral fractures based on a pathomorphologic analysis. J Shoulder Elb Surg 25(3):455–462. https://doi.org/10.1016/j.jse.2015.08.006
Gracitelli MEC, Dotta TAG, Assuncao JH, Malavolta EA, Andrade-Silva FB, Kojima KE, Ferreira Neto AA (2017) Intraobserver and interobserver agreement in the classification and treatment of proximal humeral fractures. J Shoulder Elb Surg 26(6):1097–1102. https://doi.org/10.1016/j.jse.2016.11.047
Cuny C, Baumann C, Mayer J, Guignand D, Mb I, Berrichi A, Ionescu N, Guillemin FJ (2013) AST classification of proximal humeral fractures: introduction and interobserver reliability assessment. Eur J Orthop Surg Traumatol 23(1):35–40
Hasan AP, Phadnis J, Jaarsma RL, Bain GI (2017) Fracture line morphology of complex proximal humeral fractures. J Shoulder Elb Surg 26(10):e300–e308
Sukthankar A, Leonello D, Ding G, Sandow M (2011) A Comprehensive classification of proximal humeral fractures–“HGLS classification”. In: Orthopaedic Proceedings. vol SUPP_II. The British Editorial Society of Bone & Joint Surgery, pp 163–163
Schumaier A, Grawe B (2018) Proximal humerus fractures: evaluation and management in the elderly patient. Geriatr Orthop Surg Rehabil 9:2151458517750516. https://doi.org/10.1177/2151458517750516
Sun Q, Ge W, Li G, Wu J, Lu G, Cai M, Li S (2018) Locking plates versus intramedullary nails in the management of displaced proximal humeral fractures: a systematic review and meta-analysis. Int Orthop 42(3):641–650. https://doi.org/10.1007/s00264-017-3683-z
Greiner SH, Kääb MJ, Kröning I, Scheibel M, Perka C (2008) Reconstruction of humeral length and centering of the prosthetic head in hemiarthroplasty for proximal humeral fractures. J Shoulder Elb Surg 17(5):709–714
Hepp P, Lill H, Bail H, Korner J, Niederhagen M, Haas NP, Josten C, Duda GN (2003) Where should implants be anchored in the humeral head? Clin Orthop Relat Res 415:139–147. https://doi.org/10.1097/01.blo.0000092968.12414.a8
Tingart MJ, Lehtinen J, Zurakowski D, Warner JJ, Apreleva M (2006) Proximal humeral fractures: regional differences in bone mineral density of the humeral head affect the fixation strength of cancellous screws. J Shoulder Elb Surg 15(5):620–624. https://doi.org/10.1016/j.jse.2005.09.007
Lill H, Hepp P, Gowin W, Oestmann JW, Korner J, Haas NP, Josten C, Duda GN (2002) Age- and gender-related distribution of bone mineral density and mechanical properties of the proximal humerus. RöFo 174(12):1544–1550. https://doi.org/10.1055/s-2002-35944
Meyer C, Alt V, Hassanin H, Heiss C, Stahl JP, Giebel G, Koebke J, Schnettler R (2005) The arteries of the humeral head and their relevance in fracture treatment. Surg Radiol Anat 27(3):232–237. https://doi.org/10.1007/s00276-005-0318-7
Roderer G, Brianza S, Schiuma D, Schwyn R, Scola A, Gueorguiev B, Gebhard F, Tami A (2013) Mechanical assessment of local bone quality to predict failure of locked plating in a proximal humerus fracture model. Orthopedics 36(9):e1134–e1140. https://doi.org/10.3928/01477447-20130821-14
Lee CK, Hansen HR (1981) Post-traumatic avascular necrosis of the humeral head in displaced proximal humeral fractures. J Trauma 21(9):788–791. https://doi.org/10.1097/00005373-198109000-00006
Hertel R, Hempfing A, Stiehler M, Leunig M (2004) Predictors of humeral head ischemia after intracapsular fracture of the proximal humerus. J Shoulder Elb Surg 13(4):427–433. https://doi.org/10.1016/j.jse.2004.01.034
Hertel R (2005) Fractures of the proximal humerus in osteoporotic bone. Osteoporos Int 16(Suppl 2):S65–S72. https://doi.org/10.1007/s00198-004-1714-2
Moda SK, Chadha NS, Sangwan SS, Khurana DK, Dahiya AS, Siwach RC (1990) Open reduction and fixation of proximal humeral fractures and fracture-dislocations. J Bone Joint Surg (Br) 72(6):1050–1052
Gardner MJ, Weil Y, Barker JU, Kelly BT, Helfet DL, Lorich DG (2007) The importance of medial support in locked plating of proximal humerus fractures. J Orthop Trauma 21(3):185–191. https://doi.org/10.1097/BOT.0b013e3180333094
Wang Q, Zhu Y, Liu Y, Wang L, Chen Y (2017) Correlation between classification and secondary screw penetration in proximal humeral fractures. PLoS One 12(9):e0183164. https://doi.org/10.1371/journal.pone.0183164
Krappinger D, Bizzotto N, Riedmann S, Kammerlander C, Hengg C, Kralinger FS (2011) Predicting failure after surgical fixation of proximal humerus fractures. Injury 42(11):1283–1288. https://doi.org/10.1016/j.injury.2011.01.017
Leonard M, Mokotedi L, Alao U, Glynn A, Dolan M, Fleming P (2009) The use of locking plates in proximal humeral fractures: comparison of outcome by patient age and fracture pattern. Int J Should Surg 3(4):85–89. https://doi.org/10.4103/0973-6042.63214
Vachtsevanos L, Hayden L, Desai AS, Dramis A (2014) Management of proximal humerus fractures in adults. World J Orthop 5(5):685–693. https://doi.org/10.5312/wjo.v5.i5.685
Bai L, Fu Z, An S, Zhang P, Zhang D, Jiang B (2014) Effect of calcar screw use in surgical neck fractures of the proximal humerus with unstable medial support: a biomechanical study. J Orthop Trauma 28(8):452–457. https://doi.org/10.1097/bot.0000000000000057
Kim DS, Lee DH, Chun YM, Shin SJ (2018) Which additional augmented fixation procedure decreases surgical failure after proximal humeral fracture with medial comminution: fibular allograft or inferomedial screws? J Shoulder Elb Surg 27(10):1852–1858. https://doi.org/10.1016/j.jse.2018.03.020
Gardner MJ, Boraiah S, Helfet DL, Lorich DG (2008) Indirect medial reduction and strut support of proximal humerus fractures using an endosteal implant. J Orthop Trauma 22(3):195–200. https://doi.org/10.1097/BOT.0b013e31815b3922
Panchal K, Jeong JJ, Park SE, Kim WY, Min HK, Kim JY, Ji JH (2016) Clinical and radiological outcomes of unstable proximal humeral fractures treated with a locking plate and fibular strut allograft. Int Orthop 40(3):569–577. https://doi.org/10.1007/s00264-015-2950-0
Zhu L, Liu Y, Yang Z, Li H, Wang J, Zhao C, Chen X, Zhang Y (2014) Locking plate fixation combined with iliac crest bone autologous graft for proximal humerus comminuted fracture. Chin Med J 127(9):1672–1676
Mathison C, Chaudhary R, Beaupre L, Reynolds M, Adeeb S, Bouliane M (2010) Biomechanical analysis of proximal humeral fixation using locking plate fixation with an intramedullary fibular allograft. Clin Biomech (Bristol, Avon) 25(7):642–646. https://doi.org/10.1016/j.clinbiomech.2010.04.006
Roderer G, Scola A, Schmolz W, Gebhard F, Windolf M, Hofmann-Fliri L (2013) Biomechanical in vitro assessment of screw augmentation in locked plating of proximal humerus fractures. Injury 44(10):1327–1332. https://doi.org/10.1016/j.injury.2013.05.008
Kathrein S, Kralinger F, Blauth M, Schmoelz W (2013) Biomechanical comparison of an angular stable plate with augmented and non-augmented screws in a newly developed shoulder test bench. Clin Biomech (Bristol, Avon) 28(3):273–277. https://doi.org/10.1016/j.clinbiomech.2012.12.013
Laux CJ, Grubhofer F, Werner CML, Simmen HP, Osterhoff G (2017) Current concepts in locking plate fixation of proximal humerus fractures. J Orthop Surg Res 12(1):137. https://doi.org/10.1186/s13018-017-0639-3
Somasundaram K, Huber CP, Babu V, Zadeh H (2013) Proximal humeral fractures: the role of calcium sulphate augmentation and extended deltoid splitting approach in internal fixation using locking plates. Injury 44(4):481–487. https://doi.org/10.1016/j.injury.2012.10.030
Wanner GA, Wanner-Schmid E, Romero J, Hersche O, von Smekal A, Trentz O, Ertel W (2003) Internal fixation of displaced proximal humeral fractures with two one-third tubular plates. J Trauma 54(3):536–544. https://doi.org/10.1097/01.Ta.0000052365.96538.42
Theopold J, Marquass B, Fakler J, Steinke H, Josten C, Hepp P (2016) The bicipital groove as a landmark for reconstruction of complex proximal humeral fractures with hybrid double plate osteosynthesis. BMC Surg 16:10. https://doi.org/10.1186/s12893-016-0125-6
Theopold J, Schleifenbaum S, Muller M, Werner M, Hammer N, Josten C, Hepp P (2018) Biomechanical evaluation of hybrid double plate osteosynthesis using a locking plate and an inverted third tubular plate for the treatment of proximal humeral fractures. PLoS One 13(10):e0206349. https://doi.org/10.1371/journal.pone.0206349
Choi S, Kang H, Bang H (2014) Technical tips: dualplate fixation technique for comminuted proximal humerus fractures. Injury 45(8):1280–1282. https://doi.org/10.1016/j.injury.2014.04.029
He Y, Zhang Y, Wang Y, Zhou D, Wang F (2017) Biomechanical evaluation of a novel dualplate fixation method for proximal humeral fractures without medial support. J Orthop Surg Res 12(1):72. https://doi.org/10.1186/s13018-017-0573-4
Park S-G (2016) Medial and lateral dual plate fixation for osteoporotic proximal humerus comminuted fracture: 2 case reports. J Korean Fract Soc 29(1):61–67
Weeks CA, Begum F, Beaupre LA, Carey JP, Adeeb S, Bouliane MJ (2013) Locking plate fixation of proximal humeral fractures with impaction of the fracture site to restore medial column support: a biomechanical study. J Shoulder Elb Surg 22(11):1552–1557. https://doi.org/10.1016/j.jse.2013.02.003
Chen YN, Chang CW, Lin CW, Wang CW, Peng YT, Chang CH, Li CT (2017) Numerical investigation of fracture impaction in proximal humeral fracture fixation with locking plate and intramedullary nail. Int Orthop 41(7):1471–1480. https://doi.org/10.1007/s00264-017-3401-x
Sudkamp N, Bayer J, Hepp P, Voigt C, Oestern H, Kaab M, Luo C, Plecko M, Wendt K, Kostler W, Konrad G (2009) Open reduction and internal fixation of proximal humeral fractures with use of the locking proximal humerus plate. Results of a prospective, multicenter, observational study. J Bone Joint Surg Am 91(6):1320–1328. https://doi.org/10.2106/jbjs.H.00006
Voigt C, Geisler A, Hepp P, Schulz AP, Lill H (2011) Are polyaxially locked screws advantageous in the plate osteosynthesis of proximal humeral fractures in the elderly? A prospective randomized clinical observational study. J Orthop Trauma 25(10):596–602
Schliemann B, Hartensuer R, Koch T, Theisen C, Raschke MJ, Kösters C, Weimann A (2015) Treatment of proximal humerus fractures with a CFR-PEEK plate: 2-year results of a prospective study and comparison to fixation with a conventional locking plate. J Shoulder Elb Surg 24(8):1282–1288
Ricchetti ET, DeMola PM, Roman D, Abboud JA (2009) The use of precontoured humeral locking plates in the management of displaced proximal humerus fracture. J Am Acad Orthop Surg 17(9):582–590
Zhu Y, Lu Y, Shen J, Zhang J, Jiang CJJ (2011) Locking intramedullary nails and locking plates in the treatment of two-part proximal humeral surgical neck fractures: a prospective randomized trial with a minimum of three years of follow-up. J Bone Joint Surg Am 93(2):159–168
Olerud P, Ahrengart L, Ponzer S, Saving J, Tidermark J (2011) Internal fixation versus nonoperative treatment of displaced 3-part proximal humeral fractures in elderly patients: a randomized controlled trial. J Shoulder Elb Surg 20(5):747–755
Sproul RC, Iyengar JJ, Devcic Z, Feeley BT (2011) A systematic review of locking plate fixation of proximal humerus fractures. Injury 42(4):408–413
Konrad G, Hirschmüller A, Audige L, Lambert S, Hertel R, Südkamp NP (2012) Comparison of two different locking plates for two-, three-and four-part proximal humeral fractures—results of an international multicentre study. Int Orthop 36(5):1051–1058
Ricchetti ET, Warrender WJ, Abboud JA (2010) Use of locking plates in the treatment of proximal humerus fractures. J Shoulder Elb Surg 19(2):66–75
Jost B, Spross C, Grehn H, Gerber C (2013) Locking plate fixation of fractures of the proximal humerus: analysis of complications, revision strategies and outcome. J Shoulder Elb Surg 22(4):542–549. https://doi.org/10.1016/j.jse.2012.06.008
Kavuri V, Bowden B, Kumar N, Cerynik D (2018) Complications associated with locking plate of proximal humerus fractures. Indian J Orthop 52(2):108–116. https://doi.org/10.4103/ortho.IJOrtho_243_17
Fjalestad T, Hole MØ, Hovden IAH, Blücher J, Strømsøe K (2012) Surgical treatment with an angular stable plate for complex displaced proximal humeral fractures in elderly patients: a randomized controlled trial. J Orthop Trauma 26(2):98–106
Buecking B, Mohr J, Bockmann B, Zettl R, Ruchholtz S (2014) Deltoid-split or deltopectoral approaches for the treatment of displaced proximal humeral fractures? Clin Orthop Relat Res 472(5):1576–1585
Martetschläger F, Siebenlist S, Weier M, Sandmann G, Ahrens P, Braun K, Elser F, Stöckle U, Freude T (2012) Plating of proximal humeral fractures. 35 (11):e1606-e1612
Hoel S, Jensen T, Falster O, Ulstrup A (2016) Hemiarthroplasty for proximal humerus fracture and consequences of a comminuted greater tubercle fragment. Musculoskelet Surg 100(1):9–14
Kupfer N, Garbis N (2020) Shoulder arthroplasty in the treatment of proximal humeral fractures. In: Huri G, Familiari F, Moon YL, Doral MN, Marcheggiani Muccioli GM (eds) Shoulder Arthroplasty: the shoulder club guide. Springer International Publishing, Cham, pp 149–158. https://doi.org/10.1007/978-3-030-19285-3_16
Neer CS 2nd (1970) Displaced proximal humeral fractures. II. Treatment of three-part and four-part displacement. J Bone Joint Surg Am 52(6):1090–1103
Funding
This work was supported by Shanghai Science and Technology Commission (19411963100).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sun, Q., Wu, X., Wang, L. et al. The plate fixation strategy of complex proximal humeral fractures. International Orthopaedics (SICOT) 44, 1785–1795 (2020). https://doi.org/10.1007/s00264-020-04544-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-020-04544-7