Abstract
Background
Non-enhanced computed tomography (CT) is a valuable modality in the diagnosis of non-alcoholic fatty liver disease (NAFLD). However, it is not clear if low-dose CT attenuation correction (CTAC) scans have the same accuracy to diagnose NAFLD. Our aim is to evaluate the diagnostic accuracy of low-dose CTAC in the diagnosis of NAFLD using non-enhanced CT as a gold standard.
Methods
A total of 864 patients who underwent a clinically indicated hybrid nuclear imaging scanning between May 2011 and April 2014 were included in the study. Diagnosis of fatty liver was established if an absolute liver attenuation was <40 Hounsfield units and/or a liver-to-spleen ratio was <1.1. The diagnostic accuracy parameters were calculated to detect NAFLD by low-dose CTAC using unenhanced CT as a gold standard.
Results
The prevalence of fatty liver by diagnostic CT and low-dose attenuation correction were 9.9 and 12.9% (using liver attenuation <40HU and liver-to-spleen ratio <1.1), respectively, with 32.9 and 34.9% (using absolute liver attenuation or ratio-to-spleen criteria), correspondingly. Low-dose CTAC had sensitivity (81.3%), specificity (94.0%), positive predictive value (60.2%), and negative predictive value (97.8%) using both diagnostic criteria. Using either of the diagnostic criteria resulted in sensitivity (76.8%), specificity (83.5%), PPV (66.3%), and NPV (89.5%).
Conclusion
Low-dose CT could be used as a tool to rule out the presence of fatty liver if neither liver attenuation of less than 40 HU nor liver-to-spleen below 1.1 is present.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Background
The term Non-alcoholic fatty liver disease (NAFLD) refers to the abnormal accumulation of hepatic triglyceride in the absence of significant alcohol intake [1–4]. Multiple modalities are utilized in NAFLD diagnosis [5, 6]. In biopsy-based studies, the prevalence of NAFLD varies between 3 and 46% [7–10]. Although, liver ultrasound and biomarkers are the most commonly used non-invasive tools in the assessment of NAFLD [11–14], liver biopsy remains the gold standard for that diagnosis [15–17].
NAFLD is associated with multiple medical disorders including metabolic syndrome, coronary heart disease, and atherosclerosis [18–24]. Additionally, it is labeled as a precursor to non-alcoholic steatohepatitis which increased the risk of developing hepatic cirrhosis over time. Computed tomography (CT) is a widely available tool that may aid in diagnosing hepatosteatosis and its potential complications [25, 26]. Absolute liver measurement less than 40 Hounsfield units (HU) or liver-to-spleen HU ratio (LSR) less than <1.1 has been adopted as a CT diagnostic criterion to diagnose NAFLD [26–28].
Non-diagnostic low-dose CT attenuation correction (CTAC) is used as a tool of attenuation correction in cardiac positron emission tomography/CT (PET/CT) imaging. In our cardiac PET/CT practice, we routinely perform a diagnostic unenhanced coronary calcium CT and low-dose CTAC at the same setting. Such a scan includes parts of the liver and spleen in the imaging field [25, 26]. However, it is not clear if the same cutoffs can be used to non-diagnostic low-dose CTAC to diagnose NAFL. Thus, the aim of this study is to evaluate the diagnostic accuracy of low-dose CTAC in diagnosis of NAFLD using non-enhanced CT as a gold standard.
Materials and methods
Study population
A total of 864 consecutive patients (mean age 60.6 ± 11.45 years, 55% females) who underwent a clinically indicated cardiac PET/CT between May 2011 and April 2014 were included. Patients had a diagnostic unenhanced cardiac CT for the evaluation of coronary calcium and a low-dose CTAC in the same setting. We excluded patients younger than 18 years and those who are known to have coronary artery disease, since they did not undergo coronary calcium score CT scans as per our laboratory protocol. Patients with prior history of liver diseases, liver transplant, or alcohol intake were also excluded.
The patients’ demographics (age, gender, blood pressure, height, weight, and body mass index (BMI)), past medical history, traditional cardiovascular risk factors (including hypertension, dyslipidemia, diabetes, peripheral vascular disease, and smoking), recent medications, and indication for PET/CT scanning were collected at the time of scanning.
Image acquisition and analysis
All participants were scanned using a sixty-four-detector row CT scanner (PET CT D690 with a VCT CT from General Electric Healthcare, Milwaukee, WI, USA). Both scans started at the level of the carina until beyond the level of the apex of the heart including cuts from the liver and spleen. The diagnostic unenhanced coronary calcium CT was initially performed, followed by the non-diagnostic low-dose CTAC. The diagnostic calcium scoring CT was acquired with prospectively ECG triggering during respiratory breath holding. Utilizing 120 kVp, 200 mA filtered back projection reconstruction, and 2.5 mm slice thickness. The total dose length product of this scan was 47–60 mGy-cm. The CTAC was done as a free breathing spiral non-gated acquisition utilizing the following parameters: 140 kVp and 10–40 mA in the adaptive mode. Low-dose CTAC was reconstructed with a slice thickness of 3.75 mm. The total dose length product of this scan ranged between 10 and 21 mGy-cm (due to the variable mA). All images were reviewed on Advantage Windows workstation (General Electric Healthcare, Milwaukee, WI, USA), with window length of 40 and window width of 400.
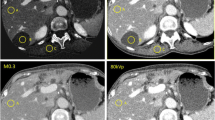
A single reader measured the liver and splenic attenuation of the two scans (i.e., low-dose CTAC and diagnostic unenhanced coronary calcium CT) blinded to the patients’ clinical parameters. Hepatic Hounsfield unit (HU) values were measured using circular ROI area greater than 165 mm2, placed in the anterolateral right hepatic lobe and avoiding the vessels. The splenic parenchymal attenuation was calculated by a single random ROI value which was usually greater than 100 mm2. The measurements of the hepatic and the splenic attenuation for the two views (between the diagnostic CT and CTAC) were taken at the same level of imaging as much as possible with the same measured area (Fig. 1).
Definition of non-alcoholic fatty liver disease
In the present study, NAFLD was defined as CT criteria for the diagnosis of fatty liver: liver attenuation of <40 HU and/or LSR of <1.1.
Other study definitions
Hypertension was defined as a systolic blood pressure ≥140 mm Hg, diastolic blood pressure ≥90 mm Hg, or use of antihypertensive medications. Dyslipidemia was defined as low-density lipoprotein >130 mg/dL, high-density lipoprotein <40 mg/dL in men and <50 mg/dL in women, or use of lipid-lowering medications. Diabetes mellitus was defined as fasting blood glucose ≥126 mg/dL or use of insulin or oral hypoglycemic medications.
Statistical analysis
Descriptive statistical analysis was utilized to describe the baseline characteristics. It was reported as mean, standard deviation, or percentage when applicable. The sensitivity, specificity, positive predictive value, negative predictive value, and their 95% confidence intervals for the low-dose CTAC diagnosis of NAFLD were calculated. We also calculated the diagnostic accuracy measures using both the liver attenuation and the LSR criteria versus that of using each criterion alone. Pearson’s correlation coefficients were calculated for the liver attenuation and the LSR on the two CT studies and a scatter plots were created. All analysis was performed using SPSS, version 21, (IBM Statistics, IL, USA).
Results
Table 1 summarizes the patients’ characteristics. Our cohort had high prevalence of cardiac risk factors, including hypertension (78%), diabetes (49%), and dyslipidemia (83%). The mean BMI was 31.71 ± 6.6 kg/m2. A total of 485 patients (56%) had a BMI of more than 30 kg/m2.
The liver and spleen attenuations were measured in 864 and 674 participants, respectively. The mean liver attenuation was 49.35 ± 12.53 HU on the unenhanced CT and 50.5 ± 12.24 HU on the low-dose CTAC. The mean spleen attenuation was 36.38 ± 11.8 HU on the unenhanced CT and 44.16 ± 7.49 HU on the low-dose CTAC.
A total of 187 patients (21.6%) and 151 patients (17.5%) had liver attenuation <40 HU on the diagnostic calcium scoring CT scan and low-dose CTAC, respectively. A total of 134 patients (19.9%) and 202 patients (30%) had LSR <1.1 on the diagnostic unenhanced CT scan and low-dose CTAC, respectively. A positive correlation was shown between the diagnostic calcium scoring CT and the low-dose CTAC measurements for both liver attenuation (r 2 = 0.456, p = 0.001; Fig. 2) and LSR data (r 2 = 0.159, p = 0.001; Fig. 3).
Scatter plot of liver-to-spleen attenuation ratio in terms of diagnostic CT versus low-dose attenuation CT shows non-constant positive relationship between the two studies. In our data, most of the liver-to-spleen ratios below 1.5 for all low-dose CT with wide variable ranges for diagnostic CT affect the slope of the linear correlation
When the diagnosis of NAFLD was based on the presence of both liver attenuation < 40 HU and LSR <1.1, the prevalence of fatty liver on the diagnostic calcium scoring CT and low-dose CTAC were 9.9 and 12.9%, respectively. Using this criterion, the sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) of low-dose CTAC in diagnosing fatty liver were 81.25, 94.01, 60.19, and 97.83%, respectively.
On the other hand, when the diagnosis of fatty liver was based on the presence of either liver attenuation <40 HU or liver-to-spleen ratio <1.1, the prevalence of fatty liver on diagnostic CT and CTAC were 32.9 and 34.9%, respectively. Using this criterion, the sensitivity, specificity, PPV, and NPV of non-diagnostic low-dose CT in diagnosing fatty liver were 76.81, 83.50, 66.25, and 89.52%, respectively (Table 2).
Given the high prevalence of obesity in our cohort, we stratified our analysis according to BMI. A high BMI of more than 30 kg/m2 reduces the accuracy of the low-dose CT in detecting fatty liver when the diagnosis is made using the liver attenuation and LSR measures combined (Fig. 4).
Discussion
NAFLD encompasses the entire spectrum of fatty liver disease in individuals without significant alcohol consumption. Establishing the diagnosis of NAFLD requires either imaging or histological evidence of hepatic steatosis, and exclusion of other secondary causes of hepatic fat accumulation. CT can be used to assess for hepatic steatosis by measuring the absolute attenuation of the liver parenchyma or calculating the LSR [11, 25].
Cardiac PET as a non-invasive nuclear imaging uses radioactive tracers to produce images of the heart. These scans are usually performed in patients with coronary risk factor, many of which are shared with non-alcoholic fatty liver. Thus, quantifying liver fat on the routinely performed low-dose attenuation correction CT would help in the risk stratification of these patients [29, 30]. Low-dose CTAC is becoming increasingly used in myocardial perfusion imaging [31–34]. Also, screening low-dose lung scans are increasingly utilized, which typically includes parts of the liver and spleen. Thus, it is conceivable that low-dose scans could be used to assess for NAFLD. To our knowledge, the ability of low-dose scans in diagnosing NAFLD was not assessed before. Our analysis shows that low-dose CTAC has good specificity and NPV compared to an unenhanced diagnostic calcium scoring CT in detecting NAFLD. However, the PPV of low-dose CTAC is comparably low. Thus, a low-dose CTAC from a cardiac PET examination can be reasonably used to exclude NAFLD, but it is limited in confirming the presence of the disease. We also noted that an increased BMI negatively affects the diagnostic accuracy of low-dose CTAC in the diagnosis of NAFLD.
Boyce et al. [35] demonstrated that low-dose colonography CT is a reasonable objective and non-invasive measure of asymptomatic patients, with more reliability than clinical risk assessment alone. In their study, a lower radiation dose was achieved using 120 kVp and 25–75 mAs tube output, but it did not affect hepatic attenuation. In another study, Cho et al. [36] evaluated the ability of different imaging modalities in diagnosing fatty liver, including the assessment of unenhanced CT studies. Assessment was performed for both the preoperative and postoperative CT scans in patients who underwent partial hepatectomy. Their findings were corroborated by hepatic histological analysis for the presence of fatty fat. The sensitivity and specificity for unenhanced CT were 33 and 100%. Similar to our study, CT was better in excluding fatty liver than in confirming its presence. Additionally, obesity had a detrimental effect on the diagnostic accuracy of CT in diagnosing fatty liver. Lee et al. [37] assessed the accuracy of unenhanced CT in diagnosing fatty liver in histologically proven fatty liver in living hepatic donors, concluding that unenhanced CT is reliable in diagnosing fatty liver, using both subjective and qualitative methods.
Although we included a large real-world sample of clinically indicated cardiac patients, our study has several limitations. First of all, not all our patients had their spleen imaged on their low-dose CTAC scan. Secondly, we did not evaluate our ability in diagnosing fatty liver on low-dose CTAC using subjective visual assessment, a method that was used in other studies [36, 37]. Thirdly, we used a single large ROI to measure hepatic and splenic attenuations, rather than obtaining different smaller ROIs. The use of the latter method would have reasonably accounted for potential cases of non-uniform fatty liver distribution. Fourthly, the fact that the low-dose CTAC was performed with a different x-ray tube output compared to that of the diagnostic calcium scoring scan may have an effect on the overall HU measured and on noise levels. Finally, since our study was retrospective in nature, we did not have the opportunity to correlate our results with histological liver analysis or any other cardiac imaging modalities, which was not clinically indicated in many patients.
In conclusion, compared to an unenhanced diagnostic CT, a low-dose CTAC scan can be reliably used to exclude NAFLD, if neither liver attenuation of <40 HU nor LSR of <1.1 is present. High BMI reduces the overall diagnostic accuracy of low-dose CTAC in excluding NAFLD.
References
Hamaguchi M, Takeda N, Kojima T, et al. (2012) Identification of individuals with non-alcoholic fatty liver disease by the diagnostic criteria for the metabolic syndrome. World J Gastroenterol 18(13):1508–1516
Chalasani N, Younossi Z, Lavine JE, et al. (2012) The diagnosis and management of non-alcoholic fatty liver disease: practice Guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology 55(6):2005–2023
Clain DJ, Lefkowitch JH (1987) Fatty liver disease in morbid obesity. Gastroenterol Clin N Am 16(2):239–252
Nasrallah SM, Wills CE Jr, Galambos JT (1981) Hepatic morphology in obesity. Dig Dis Sci 26(4):325–327
Fleischman MW, Budoff M, Ifran Zeb DL, Foster T (2014) NAFLD prevalence differs among hispanic subgroups: the multi-ethnic study of atherosclerosis. World J Gastroenterol 20(17):4987
Kan H, Kimura Y, Hyogo H, et al. (2014) Non-invasive assessment of liver steatosis in non-alcoholic fatty liver disease. Hepatol Res 44(14):E420–E427. doi:10.1111/hepr.12330
Adams LA, Lymp JF, St Sauver J, et al. (2005) The natural history of nonalcoholic fatty liver disease: a population-based cohort study. Gastroenterology 129(1):113–121
Dixon JB, Bhathal PS, O’Brien PE (2001) Nonalcoholic fatty liver disease: predictors of nonalcoholic steatohepatitis and liver fibrosis in the severely obese. Gastroenterology 121(1):91–100
Wanless IR, Lentz JS (1990) Fatty liver hepatitis (steatohepatitis) and obesity: an autopsy study with analysis of risk factors. Hepatology 12(5):1106–1110
Adler M, Schaffner F (1979) Fatty liver hepatitis and cirrhosis in obese patients. Am J Med 67(5):811–816
Machado MV, Cortez-Pinto H (2013) Non-invasive diagnosis of non-alcoholic fatty liver disease. A critical appraisal. J Hepatol 58(5):1007–1019. doi:10.1016/j.jhep.2012.11.021
Hashimoto E, Tokushige K, Ludwig J (2015) Diagnosis and classification of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis: current concepts and remaining challenges. Hepatol Res 45(1):20–28. doi:10.1111/hepr.12333
Koebnick C, Wagner I, Leitzmann P, Stern U, Zunft HF (2003) Probiotic beverage containing Lactobacillus casei Shirota improves gastrointestinal symptoms in patients with chronic constipation. Can J Gastroenterol 17(11):655–660
Saadeh S, Younossi ZM, Remer EM, et al. (2002) The utility of radiological imaging in nonalcoholic fatty liver disease. Gastroenterology 123(3):745–750
Sumida Y, Nakajima A, Itoh Y (2014) Limitations of liver biopsy and non-invasive diagnostic tests for the diagnosis of nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. World J Gastroenterol 20(2):475–485. doi:10.3748/wjg.v20.i2.475
Mofrad P, Contos MJ, Haque M, et al. (2003) Clinical and histologic spectrum of nonalcoholic fatty liver disease associated with normal ALT values. Hepatology 37(6):1286–1292. doi:10.1053/jhep.2003.50229
Joy D, Thava VR, Scott BB (2003) Diagnosis of fatty liver disease: is biopsy necessary? Eur J Gastroenterol Hepatol 15(5):539–543
Sanches PL, de Piano A, Campos RM, et al. (2014) Association of nonalcoholic fatty liver disease with cardiovascular risk factors in obese adolescents: the role of interdisciplinary therapy. J Clin Lipidol 8(3):265–272. doi:10.1016/j.jacl.2014.02.007
Vilar CP, Cotrim HP, Florentino GS, et al. (2013) Association between nonalcoholic fatty liver disease and coronary artery disease. Rev Assoc Med Bras 59(3):290–297
Oni ET, Agatston AS, Blaha MJ, et al. (2013) A systematic review: burden and severity of subclinical cardiovascular disease among those with nonalcoholic fatty liver; should we care? Atherosclerosis 230(2):258–267
Feitosa MF, Reiner AP, Wojczynski MK, et al. (2013) Sex-influenced association of nonalcoholic fatty liver disease with coronary heart disease. Atherosclerosis 227(2):420–424. doi:10.1016/j.atherosclerosis.2013.01.013
Targher G, Day CP, Bonora E (2010) Risk of cardiovascular disease in patients with nonalcoholic fatty liver disease. N Engl J Med 363(14):1341–1350. doi:10.1056/NEJMra0912063
Hamaguchi M, Kojima T, Takeda N, et al. (2007) Nonalcoholic fatty liver disease is a novel predictor of cardiovascular disease. World J Gastroenterol 13(10):1579–1584
Ekstedt M, Franzen LE, Mathiesen UL, et al. (2006) Long-term follow-up of patients with NAFLD and elevated liver enzymes. Hepatology 44(4):865–873. doi:10.1002/hep.21327
Shen F, Zheng RD, Mi YQ, et al. (2014) Controlled attenuation parameter for non-invasive assessment of hepatic steatosis in Chinese patients. World J Gastroenterol 20(16):4702–4711. doi:10.3748/wjg.v20.i16.4702
Zeb I, Li D, Nasir K, et al. (2012) Computed tomography scans in the evaluation of fatty liver disease in a population based study: the multi-ethnic study of atherosclerosis. Acad Radiol 19(7):811–818. doi:10.1016/j.acra.2012.02.022
Kodama Y, Ng CS, Wu TT, et al. (2007) Comparison of CT methods for determining the fat content of the liver. Am J Roentgenol 188(5):1307–1312. doi:10.2214/AJR.06.0992
Hamer OW, Aguirre DA, Casola G, et al. (2006) Fatty liver: imaging patterns and pitfalls. Radiographics 26(6):1637–1653. doi:10.1148/rg.266065004
Akabame S, Hamaguchi M, K-i Tomiyasu, et al. (2007) Evaluation of vulnerable coronary plaques and non-alcoholic fatty liver disease (NAFLD) by 64-detector multislice computed tomography (MSCT). Circ J 72(4):618–625
Lautamäki R, Borra R, Iozzo P, et al. (2006) Liver steatosis coexists with myocardial insulin resistance and coronary dysfunction in patients with type 2 diabetes. Am J Physiol Endocrinol Metab 291(2):E282–E290
Kalra MK, Maher MM, Blake MA, et al. (2004) Detection and characterization of lesions on low-radiation-dose abdominal CT images postprocessed with noise reduction filters. Radiology 232(3):791–797. doi:10.1148/radiol.2323031563
Zhu X, Yu J, Huang Z (2004) Low-dose chest CT: optimizing radiation protection for patients. Am J Roentgenol 183(3):809–816. doi:10.2214/ajr.183.3.1830809
Wormanns D, Ludwig K, Beyer F, Heindel W, Diederich S (2005) Detection of pulmonary nodules at multirow-detector CT: effectiveness of double reading to improve sensitivity at standard-dose and low-dose chest CT. Eur Radiol 15(1):14–22
Arcadi T, Maffei E, Sverzellati N, et al. (2014) Coronary artery calcium score on low-dose computed tomography for lung cancer screening. World J Radiol 6(6):381
Boyce CJ, Pickhardt PJ, Kim DH, et al. (2010) Hepatic steatosis (fatty liver disease) in asymptomatic adults identified by unenhanced low-dose CT. Am J Roentgenol 194(3):623–628. doi:10.2214/AJR.09.2590
Cho CS, Curran S, Schwartz LH, et al. (2008) Preoperative radiographic assessment of hepatic steatosis with histologic correlation. J Am Coll Surg 206(3):480–488. doi:10.1016/j.jamcollsurg.2007.08.020
Lee SS, Park SH, Kim HJ, et al. (2010) Non-invasive assessment of hepatic steatosis: prospective comparison of the accuracy of imaging examinations. J Hepatol 52(4):579–585. doi:10.1016/j.jhep.2010.01.008
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for this study.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study data was collected retrospectively after obtaining local ethical approval.
Informed consent
Informed consent was waved.
Rights and permissions
About this article
Cite this article
Ahmed, A.M., Ebid, M.E., Ajlan, A.M. et al. Low-dose attenuation correction in diagnosis of non-alcoholic fatty liver disease. Abdom Radiol 42, 2454–2459 (2017). https://doi.org/10.1007/s00261-017-1166-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-017-1166-8