Abstract
Purpose
To evaluate the influence of 18F-FDG PET/CT in comparison to CT alone on treatment decisions in patients with advanced melanoma and to analyse the 5-year survival data in comparison to literature data.
Methods
Therapy management in 64 consecutive patients (primary staging n = 52; surveillance n = 12) with stage III/IV melanoma who underwent 18F-FDG PET/CT between 2004 and 2005 in our department was retrospectively analysed. Treatment decisions were made by two dermatooncologists for each patient twice, first based on the CT results and then based on the PET/CT results. Therapy changes based on the PET/CT results were classified as “major” (e.g. change from metastasectomy to systemic therapy) or “minor” (e.g. change from first to second line chemotherapy). The 5-year survival data of different patient cohorts were calculated.
Results
In the 52 patients in the primary staging group, the results of 18F-FDG PET/CT led to therapy change in 59 % and a major therapy change in 52 %. 18F-FDG PET/CT led to the avoidance of futile operations in 13 patients with suspicious lesions on CT that were deemed nontumorous on PET/CT. In the 12 patients in the surveillance group, the results of 18F-FDG PET/CT led to therapy change in 33 % and a major change in 17 %. The 5-year survival rates were 30 % in the entire cohort, 34 % in the primary staging group, and 17 % in the surveillance group. A significant overall survival benefit was observed in patients in whom 18F-FDG PET/CT excluded metastases or in whom metastases could be completely removed compared with patients who were not eligible for surgery (41 % vs. 10 %).
Conclusion
Primary staging of patients with stage III/IV melanoma should be performed with 18F-FDG PET/CT, leading to higher diagnostic accuracy and enabling individualized therapeutic management, especially optimal patient selection for metastasectomy. This strategy may extend long-term survival even in patients with advanced disease.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The prognosis of advanced melanoma is unfavourable, with 5-year survival rates of 78 %, 59 % and 40 % for stage IIIA, IIIB and IIIC disease, respectively, and 1-year survival rates of 62 % for M1a, 53 % for M1b and only 33 % for M1c disease [1]. Early detection of metastatic spread is essential for the initiation of adequate individual therapy. In recent years, promising results concerning new targeted and immunotherapeutic drugs for therapy of disseminated melanoma have been reported [2, 3]. However, in patients with limited systemic disease, resection of all metastases is probably the most effective method for prolonging survival [4]. On the other hand, patients will only benefit from complete metastasectomy, whereas simple tumour debulking does not improve prognosis [5]. Therefore, accurate identification of all sites of metastatic spread is essential before surgery [6–8]. In 2012, Weide et al. reported the outcome in 220 of 855 melanoma patients with distant metastases treated by complete surgical metastasectomy as primary therapy [4]. The 2-year and 5-year survival rates were 50 % and 40 % after surgical excision vs. 20 % and 10 % after other treatment modalities. There is still no other therapy that improves survival rates to a comparable extent.
Up to now, whole-body CT is regularly used as the standard imaging modality for tumour staging in patients with advanced melanoma. However, a recently published meta-analysis by Xing et al. [9] that analysed 74 studies including a total of 10,528 melanoma patients showed that 18F-FDG PET/CT is clearly superior to CT alone for the detection of extracerebral metastases. The aim of this study was twofold: first, to evaluate the influence of 18F-FDG PET/CT results on treatment decisions in patients with stage III/IV melanoma in comparison to CT alone, and second to analyse long-term outcomes in relation to primary staging and the choice of the imaging modality used for staging.
Materials and methods
Patients
The present study analysed datasets from patients with advanced melanoma who underwent whole-body CT and 18F-FDG PET/CT imaging between 2004 and 2005 in our department. The study cohort of 64 patients (41 men, 23 women) included 25 patients with stage III and 39 patients with stage IV disease. Their mean age was 58 years (median 61 years) ranging between 23 and 79 years. Primary tumours included superficial spreading melanoma (42 %), nodular melanoma (20 %), occult melanoma (19 %), acral lentiginous malignant melanoma (6 %), lentigo maligna melanoma (3 %), vulvar melanoma (2 %) and others (8 %). The mean tumour-thickness was 2.7 mm (median 2.0 mm). Whole-body imaging was mainly performed for further characterization of abnormal radiological and clinical findings or because of increased tumour markers (S100, LDH). Whole-body imaging was performed in 52 patients for primary staging and in 12 patients referred for restaging (called the surveillance group).
18F-FDG PET/CT imaging
18F-FDG PET/CT was performed according to a standard protocol including oral and intravenous administration of contrast agent. Patients were given 370 MBq 18F-FDG intravenously 55 – 65 min before scanning with a Hi-Rez Biograph 16 (Siemens Medical Solutions, Knoxville, TN) with 3 min emission scan per bed position. CT was performed with 120 kV, 120 – 160 mAs, a rotation time of 0.5 s, collimation of 0.75 mm (thorax) and 1.5 mm (abdomen), table feeds of 12 mm and 24 mm, and reconstructed slice thicknesses of 5 mm (axial, with an increment 5 mm) and 3 mm (coronal, with an increment of 2 mm), respectively. In all patients a multiphase CT scan including intravenous administration of 120 ml iodinated contrast agent (Ultravist 370; Schering GmbH, Berlin, Germany) was performed to obtain full diagnostic CT data. The attenuation-corrected PET data were iteratively reconstructed and coregistered with the CT data using commercial software (eSoft; Siemens, Erlangen, Germany). Full details of the imaging protocol were as previously reported [10].
Analysis plan and endpoints
The CT and PET/CT examinations in all 64 patients were reevaluated by an interdisciplinary team consisting of two dermatooncologists from the Skin Cancer Program of our Department of Dermatology (C.G., T.E.) and two radiologists experienced in reading CT and PET/CT scans (C.P., S.-C.S.). All suspected lesions in each patient were reclassified either as malignant or nonmalignant on the basis of the whole-body CT scan alone and then on the basis of the PET/CT scan. In each patient the CT-based management plan was compared with that after PET/CT and whether the PET/CT scan led to a change in therapy was recorded. Changes in therapy were classified in “major” (e.g. surgical to systemic therapy and vice versa) or “minor” (e.g. change from first line to second line chemotherapy). Details are summarized in Table 1. Imaging results were also correlated with anatomopathological findings in patients who had resection. Furthermore, follow-up in all patients starting at the time of the first staging until the last observation or death was documented.
Statistical analysis
Descriptive analysis was used for the calculation of sensitivity and specificity in the lesion-based analysis. Survival curves and median survival with relative 95 % confidence intervals (95 % CI) were generated according to the Kaplan-Meier estimators. Differences in survival were determined using the log rank test. Follow-up time was defined as the time from the date of staging to the date of the last follow-up or death. All statistical computations were performed using R 3.0 by the R Foundation of Statistical Computing (Vienna, Austria) [11].
Results
Lesion-based analysis
The lesion-based re-evaluation of the CT and 18F-FDG PET/CT scans individually in each patient on the basis of the original datasets revealed results similar to those of a former study [10]. Of the 297 metastatic lesions according to the reference standard, 229 were detected on CT as true-positive lesions (sensitivity 77.1 %, specificity 69.9 %), and 269 were detected on FDG PET/CT (sensitivity 90.6 %, specificity 77.2 %).
Patient-based analysis
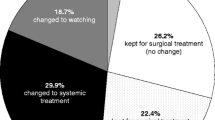
The patient-based reevaluation of the whole-body CT and 18F-FDG PET/CT scans in the 64 patients showed that the 18F-FDG PET/CT findings led to a change in the primary CT-based treatment decisions in 35 (55 %) of the patients. In 29 patients (45 %), this change was classified as “major” and in 6 patients as “minor”. The results of this patient-based reevaluation of the CT and 18F-FDG PET/CT scans are summarized in Figs. 1 and 2.
Details of therapy changes based on the results of 18F-FDG PET/CT compared to the decisions based on CT alone in 29 patients with major changes and 6 patients with minor changes from among the whole group of 64 patients with stage III/IV melanoma (CTX chemotherapy, FU follow-up, N.pts number of patients, RT radiotherapy)
In the 52 patients in the primary staging group, the results of 18F-FDG PET/CT led to therapy change in 59 % and a major therapy change in 52 %. In this group, 21 patients (40 %) underwent complete metastasectomy. In the 12 patients in the surveillance group, the results of 18F-FDG PET/CT led to therapy change in four patients (33 %) and a major therapy change in two patients. In this group, one patient was treated by metastasectomy. In 16 (25 %) of the 64 patients, the recommendation for complete metastasectomy based on conventional CT findings were revised on the basis of the PET/CT findings. In detail, PET/CT detected further metastases in 3 (5 %) of the 64 patients (Fig. 3) and excluded metastases in 13 patients (Fig. 4). PET/CT led to a change in the extent of metastasectomy in 4 (6 %) of the 64 patients with expansion of the surgical field in 3 patients and a reduction in the surgical field in 1 patient. PET/CT led to surgical treatment in 8 (12 %) of the 64 patients who had been falsely classified following CT alone: false-positive CT interpretation would have resulted in systemic therapy in 1 patient, and CT alone failed to detect metastatic lesions in 7 patients (Fig. 5).
a Contrast-enhanced CT image shows an enlarged suspicious lymph node in the left inguinal region. b 18F-FDG PET/CT image confirms the lymph node metastasis in the left inguinal region. c, d 18F-FDG PET/CT image (d) shows another soft-tissue metastasis in the left popliteal fossa not seen on the CT image (c) that resulted in extension of the surgical field
Overall survival
The 5-year survival rate for the entire cohort was 30.1 % (Fig. 6a). In the 52 patients in the primary staging group, the 5-year survival rate was 33.6 % (Fig. 6b). In the 12 patients in the surveillance group, the 5-year survival rate was 16.7 % (Fig. 6c). The overall survival rate in 43 patients in whom metastases were excluded by 18F-FDG PET/CT or in whom metastases were completely removed was significantly higher than in 21 patients not eligible for surgery (40.5 % vs. 10.0 %; p = 0.000185; Fig. 6d).
Kaplan-Meier plots of overall survival in a the entire study cohort (64 patients), b the primary staging group (52 patients), and c the surveillance (restaging) group (12 patients). d Comparison of overall survival in patients in whom metastases were excluded or surgically removed (upper curve, 43 patients) and in patients with metastatic spread not eligible for surgery (lower curve. 21 patients; p = 0.000185)
Discussion
Despite improvements in medical treatment of advanced melanoma (for example, BRAF/MEK inhibitors and CTLA-4- or PD-1 antibodies) [2, 3, 12–15], complete metastasectomy seems to improve overall survival the most [4, 5, 7, 8]. In contrast, tumour debulking without achieving complete metastasectomy does not improve overall survival and will stress the patient without achieving substantial benefit [5].
In this study, we compared the treatment decisions in 64 patients with advanced melanoma on the basis of findings of a conventional CT scan and the treatment decisions based on additional information provided by 18F-FDG PET/CT in the same patient cohort. Astonishingly, the majority (52 %) of treatment decisions had to be revised on the basis of the 18F-FDG PET/CT findings. This was especially true in patients undergoing primary staging procedures. In contrast, during the further clinical course, the likelihood of a major treatment change on the basis of the PET/CT results seemed to decrease. In a previous lesion-based analysis of 420 lesions suspicious for melanoma metastases, 18F-FDG PET/CT was significantly more accurate than other staging modalities (PET alone, CT alone, whole-body MRI) in the overall detection of malignant lesions [10]. Confirming results were published by Xing et al. in 2011 in a meta-analysis including over 10.000 melanoma patients of different tumour stages [9]. In that study, ultrasonography had the highest sensitivity for the detection of lymph node metastases, whereas CT and 18F-FDG PET/CT were more accurate for the detection of distant metastases. 18F-FDG PET/CT was superior to CT alone for primary staging (sensitivity 80 % vs. 51 %) and surveillance (sensitivity 86 % vs. 69 %).
In a systematic review of seven studies on the value of 18F-FDG PET scans in the follow up of patients with cutaneous melanoma, the mean sensitivity and specificity of PET was 96 % and 92 %, respectively [16]. Bronstein et al. [17] investigated the impact of an additional PET/CT scan on the management of patients with stage III/IV melanoma designated for metastasectomy. The surgical management changed in 12 % of the patients (4/32). In our cohort, the treatment decision was a surgical approach in 19 %, including those patients in whom extension of the surgical field was changed based on the PET/CT results. So far only a few studies have compared PET/CT and other whole-body imaging modalities in the staging of patients with advanced melanoma. Whole-body MRI including diffusion-weighted imaging is known to offer good diagnostic performance with sensitivities and specificities comparable to, or even higher than, those offered by PET/CT for staging of melanoma patients. However, the value of whole-body MRI is impaired not only by specific contraindications (e.g. metal implants) and long examination times but also by reduced diagnostic accuracy in the detection of lung nodules and high interreader variability [18–21].
Besides providing more accurate detail on metastatic spread, the question arises as to whether the knowledge provided by PET/CT leads to improved treatment recommendations and improved survival. Compared to the published data given by the AJCC of an overall 5-year survival rate of 5 – 10 % in patients with for stage IV melanoma and 22 – 65 % in patients with stage III melanoma [1], the 5-year survival rates in our patients of 30.1 % for the entire cohort and 33.6 % for the primary staging group seem to be more favourable. We analysed a mid-sized cohort only, and analysis of larger cohorts is required to confirm whether these survival rates are generally as favourable as in our population. However, Dalrymple-Hay et al. [22] found an increased 3-year and 5-year overall survival benefit in 121 melanoma patients who underwent a PET scan before thoracotomy for lung metastases. This improvement appears to derive mainly from improved selection of patients eligible for metastasectomy [5, 23, 24]. In a retrospective analysis [25], the prognosis in patients with stage IV melanoma who underwent metastasectomy (with or without additional medical treatment) was compared with that in patients with medical treatment only. The median survival was 15.8 months and the 4-year survival rate was 20.8 % in patients receiving surgery (with or without medical treatment), and 6.9 months and 7.0 %, respectively, in patients receiving medical treatment alone (p < 0.0001). In a recent study complete metastasectomy was also a major significant variable in a Cox proportional hazards model of overall survival in 855 patients with stage IV melanoma together with S100 and LDH [4].
In conclusion, 18F-FDG PET/CT is an accurate method for the staging and detection of distant metastases in patients with stage III/IV melanoma and was shown to be superior to CT alone. It offers improved therapy stratification in these patients, especially proper patient selection for metastasectomy. Thus 18F-FDG PET/CT leads to optimal individualized therapy management that may be able to extend long-term survival even in patients with advanced disease.
References
Balch CM, Gershenwald JE, Soong SJ, Thompson JF, Atkins MB, Byrd DR, et al. Final version of 2009 AJCC melanoma staging and classification. J Clin Oncol. 2009;27(36):6199–206.
Chapman PB, Hauschild A, Robert C, Haanen JB, Ascierto P, Larkin J, et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N Engl J Med. 2011;364(26):2507–16.
Hodi FS, O’Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363(8):711–23.
Weide B, Elsasser M, Buttner P, Pflugfelder A, Leiter U, Eigentler TK, et al. Serum markers lactate dehydrogenase and S100B predict independently disease outcome in melanoma patients with distant metastasis. Br J Cancer. 2012;107(3):422–8.
Caudle AS, Ross MI. Metastasectomy for stage IV melanoma: for whom and how much? Surg Oncol Clin N Am. 2011;20(1):133–44.
Pflugfelder A, Kochs C, Blum A, Capellaro M, Czeschik C, Dettenborn T, et al. Malignant melanoma S3-guideline “diagnosis, therapy and follow-up of melanoma”. J Dtsch Dermatol Ges. 2013;11 Suppl 6:1–116.
Kroon BB. Surgery for distant metastatic melanoma improves survival. Ann Surg Oncol. 2012;19(8):2426–7.
Tsao H, Atkins MB, Sober AJ. Management of cutaneous melanoma. N Engl J Med. 2004;351(10):998–1012.
Xing Y, Bronstein Y, Merrick I. Contemporary diagnostic imaging modalities for the staging and surveillance of melanoma patients: a meta-analysis. J Natl Cancer Inst. 2011;103:129–42.
Pfannenberg C, Aschoff P, Schanz S, Eschmann SM, Plathow C, Eigentler TK, et al. Prospective comparison of 18F-fluorodeoxyglucose positron emission tomography/computed tomography and whole-body magnetic resonance imaging in staging of advanced malignant melanoma. Eur J Cancer. 2007;43(3):557–64.
R Development Core Team. R: a language and environment for statistical computing. 31st ed. Vienna: R Foundation for Statistical Computing; 2014.
Larkin J, Ascierto PA, Dreno B, Atkinson V, Liszkay G, Maio M, et al. Combined vemurafenib and cobimetinib in BRAF-mutated melanoma. N Engl J Med. 2014;371(20):1867–76.
Long GV, Stroyakovskiy D, Gogas H, Levchenko E, de Braud F, Larkin J, et al. Combined BRAF and MEK inhibition versus BRAF inhibition alone in melanoma. N Engl J Med. 2014;371(20):1877–88.
Flaherty KT, Robert C, Hersey P, Nathan P, Garbe C, Milhem M, et al. Improved survival with MEK inhibition in BRAF-mutated melanoma. N Engl J Med. 2012;367(2):107–14.
Robert C, Long GV, Brady B, Dutriaux C, Maio M, Mortier L, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372(4):320–30.
Danielsen M, Hojgaard L, Kjaer A, Fischer BM. Positron emission tomography in the follow-up of cutaneous malignant melanoma patients: a systematic review. Am J Nucl Med Mol Imaging. 2013;4(1):17–28.
Bronstein Y, Ng CS, Rohren E, Ross MI, Lee JE, Cormier J, et al. PET/CT in the management of patients with stage IIIC and IV metastatic melanoma considered candidates for surgery: evaluation of the additive value after conventional imaging. AJR Am J Roentgenol. 2012;198(4):902–8.
Schwenzer N, Pfannenberg C. PET/CT, MR and PET/MR in lymphoma and melanoma. Semin Nucl Med. 2015;45(4):322–31.
Pfannenberg C, Schwenzer N. Whole-body staging of malignant melanoma: advantages, limitations and current importance of PET-CT, whole body MRI and PET-MRI. Radiologe. 2015;55(2):120–6.
Hausmann D, Jochum S, Utikal J, Hoffmann RC, Zechmann C, Neff KW, et al. Comparison of the diagnostic accuracy of whole-body MRI and whole-body CT in stage III/IV malignant melanoma. J Dtsch Dermatol Ges. 2011;9:212–22.
Laurent V, Trausch G, Bruot O, Olivier P, Felbinger J, Régent D. Comparative study of two whole-body imaging techniques in the case of melanoma metastases: advantages of multi-contrast MRI examination including a diffusion-weighted sequence in comparison with PET-CT. Eur J Radiol. 2010;75(3):376–83.
Dalrymple-Hay MJR, Rome PD, Kennedy C, Fullham M, Cuaghan BC. Pulmonary metastatic malanoma – the survival benefit of positron emission tomography scanning. Eur J Cardiothorac Surg. 2002;21(4):611–5.
Younes R, Abrao FC, Gross J. Pulmonary metastasectomy for malignant melanoma: prognostic factors for long-term survival. Melanoma Res. 2013;23(4):307–11.
Colman MW, Kirkwood JM, Schott T, Goodman MA, McGough 3rd RL. Does metastasectomy improve survival in skeletal melanoma? Melanoma Res. 2014;24(4):354–9.
Howard JH, Thompson JF, Mozzillo N, Nieweg OE, Hoekstra HJ, Roses DF, et al. Metastasectomy for distant metastatic melanoma: analysis of data from the first Multicenter Selective Lymphadenectomy Trial (MSLT-I). Ann Surg Oncol. 2012;19(8):2547–55.
Compliance with ethical standards
ᅟ
Conflicts of interest
None.
Informed consent
For this type of study formal consent is not required.
Author information
Authors and Affiliations
Corresponding author
Additional information
Susann-Cathrin Schüle and Thomas Kurt Eigentler contributed equally to this work.
Rights and permissions
About this article
Cite this article
Schüle, SC., Eigentler, T.K., Garbe, C. et al. Influence of 18F-FDG PET/CT on therapy management in patients with stage III/IV malignant melanoma. Eur J Nucl Med Mol Imaging 43, 482–488 (2016). https://doi.org/10.1007/s00259-015-3187-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-015-3187-2