Abstract
Objective
To prospectively compare the prevalence and frequency of subchondral bone marrow edema (BME) in the lumbar facet joints of low back pain patients and healthy subjects.
Materials and methods
Lumbar magnetic resonance imaging (MRI) examinations were performed on 55 asymptomatic participants (18 men; age range 21–63; mean 36 ± 12 years; body mass index (BMI) range 16–31; mean 22.6 ± 3.2 kg/m2) and 79 low back pain patients (36 men; age range 18–77; mean 47 ± 14 years; BMI range 18–40; mean 27.8 ± 4.4 kg/m2). In both groups, facet joint subchondral BME signal was evaluated using T2-weighted STIR imaging, and facet joint osteoarthritis was characterized as mild, moderate, and severe.
Results
The BME signal was found in seven asymptomatic participants (12.7%) and 28 low back pain patients (35.4%) (P = 0.003). A significant portion of the patients (15.2%) presented more than one BME signal (P = 0.011). By pooling the ten facet joints of all subjects in each group, a significant difference in osteoarthritis grade distribution was observed between the two groups (P < 0.001). When adjusted for low back pain status, age, BMI, Modic type 1, disk herniation, and facet joint osteoarthritis maximal grade, only the latter was significantly associated with the facet joint BME signal (P < 0.001).
Conclusion
Despite the higher prevalence and frequency of the BME signal in facet joints of low back pain patients compared to that in healthy subjects, the signal was found to be associated with the severity of the patients’ osteoarthritis and not with their low back pain status.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Low back pain is the leading cause of disability, and it is most commonly related to disk-related disease, including disk herniation, and/or facet joint osteoarthritis [1, 2]. The symptoms of facet joint osteoarthritis can mimic those associated with disk herniation through the so-called “pseudo-radicular” referral pattern, which often makes it difficult to distinguish between the two conditions [2,3,4]. Magnetic resonance imaging (MRI) is a useful technique to assess disk and facet joint abnormalities, though it lacks specificity regarding the determination of the causes of low back pain because these abnormalities are also common in asymptomatic subjects [4,5,6,7].
On MR images, bone marrow edema (BME) patterns manifest with moderately low signal intensity on T1-weighted images and high signal intensity on T2-weighted images without sharp abnormal to normal marrow interfaces [8]. In the subchondral vertebral end plates, this signal, known as Modic type 1, and its resolution are both associated with low back pain and its relief [8,9,10,11,12,13,14]. By contrast, these abnormalities are not as common in healthy subjects [10]. In their retrospective study, Friedrich et al. [15] reported this type of BME signal within the facet joint subchondral bone in 14% of patients with low back pain, though their results were not compared to those of healthy subjects. If this signal is indeed more frequent in low back pain patients than in asymptomatic subjects, its detection would be useful in the development and implementation of effective treatment plans (e.g., selecting a particular facet joint level for facet block therapy).
This study aimed to compare the prevalence and frequency of the facet joints BME signal between symptomatic patients and healthy subjects.
Materials and methods
Subjects
This study was conducted at two hospitals (Centre Hospitalier Universitaire Ambroise Paré, Mons; Hôpital Universitaire des Enfants Reine Fabiola, H.U.D.E.R.F., Brussels, Belgium), and the study methodology and procedures were approved by the local ethics committee of each institution, and written informed consent was obtained from all participants.
Two subject groups were considered in this study. The first group of 55 healthy volunteer participants (i.e., healthy group) included 18 men and 37 women, aged from 21 to 63 years old (mean ± standard deviation (SD), 36 years ±12) with a body mass index (BMI) ranging from 16 to 31 kg/m2 (mean ± standard deviation (SD), 22.6 kg/m2 ± 3.2). These subjects were recruited from the medical and paramedical staffs of both institutions subject to the following inclusion criteria: more than 18 years old; never having consulted a physician, physiotherapist, or osteopath for low back pain; never been absent from work for low back pain; no history of spine trauma, spine infection, spine surgery, lumbar spine infiltration, neoplasia, or rheumatic disease; and no MRI contraindications. The second group of 79 symptomatic patients (i.e., low back pain group) included 36 men and 43 women, aged from 18 to 77 years old (mean ± SD, 47 years ± 14) with a BMI ranging from 18 to 40 kg/m2 (mean ± SD, 27.8 kg/m2 ± 4.4). These participants were consecutively recruited among patients who consulted with their physician and underwent a lumbar MRI examination subject to the following inclusion criteria: more than 18 years old with persistent low back pain for more than 3 months; no history of spine trauma, spine infection, spine surgery, neoplasia, or rheumatic disease; and no MRI contraindications. All participants completed the Nordic Musculoskeletal Questionnaire (NMQ) that consisted of questions regarding the participants’ gender, age, weight, height, physical activities, possible symptoms, and/or treatments [16].
MRI imaging
The MRI examinations were performed with a 1.5 T MRI scanner (Intera®, Philips Healthcare, Best, the Netherlands; Signal HDxt, GE Healthcare, Fairfield, CT). The standardized protocol was applied to both groups, which included sagittal spin-echo T1-weighted sequence (repetition time msec/echo time msec, 550–600/10–11; section thickness, 4 mm; 512 × 512 matrix), sagittal turbo spin T2-weighted sequence (repetition time msec/echo time msec, 4400–4600/80–120; slice thickness, 4 mm; matrix 512 × 512 mm), and axial short tau inversion recovery (STIR) T2-weighted sequence (repetition time msec/echo time msec, 4400–9600/45–80; inversion recovery msec, 130–150; slice thickness, 4 mm; 512 × 512 matrix).
Image analysis
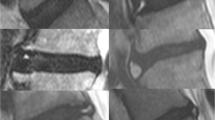
Two radiologists (AM, CN) with 15 and 16 years of experience in neuroradiology and musculoskeletal MRI scanning and interpretation were blinded to the subject data that were based on the parameters listed below and then asked to independently grade facet joint osteoarthritis according to the Weishaupt’s grading system [10] on both sides of each lumbar level. This grading system organizes joint conditions in the following manner: grade 0 corresponds to participants with a normal facet joint space; grade 1 corresponds to participants exhibiting a narrowing of the facet joint space thinner than 2 mm, small osteophytes, and/or mild hypertrophy of the articular process; grade 2 corresponds to participants with narrowing of the facet joint space, moderate osteophyte, moderate hypertrophy of the articular process, and/or mild subarticular bone erosion; and grade 3 corresponds to participants with narrowing of the facet joint space, large osteophyte, severe hypertrophy of the articular process, severe subarticular bone erosion, and/or subchondral cysts. For this study, the BME signal of the facet joint subchondral bone was defined as a region of high signal intensity with ill-defined margins on the T2-weighted STIR images (Figs. 1 and 2). If present, facet joint effusion was also coded for both sides of each lumbar level. To resolve discrepancies between the radiologists’ interpretations, they reviewed the images together to reach a consensus.
Axial short tau inversion recovery (STIR) T2-weighted sequence (a) at the L5-S1 level showing left grade I facet joint osteoarthritis with bone marrow edema (BME) signal (white arrow); (b) at the L5-S1 level, showing right grade II facet joint osteoarthritis with BME signal (white arrow) and left grade III facet joint osteoarthritis without significant BME signal; (c) at the L4-L5 level, showing right grade II and left grade III facet joint osteoarthritis with BME signal (white arrows); (d) at the L4-L5 level, showing bilateral grade III facet joint osteoarthritis with subchondral bone cysts (black arrow). No BME signal was observed
The radiologists were also asked to classify disk abnormalities, such as bulging, protruding, or extruding at each lumbar level according to the North American Spine Society’s (NASS) Clinical Guidelines [17]. Finally, they were asked to code end plate and adjacent bone marrow abnormalities on sagittal MR images according to the definitions provided in Modic et al. [10] for each category: no abnormality, type I (low signal intensity on T1-weighted images and high signal intensity on T2-weighted images compared to fatty bone marrow), type II (high signal intensity with both images), and type III (low signal intensity on both images). When two types were simultaneously present on both sides of the intervertebral space, only one diagnosis was applied in the following manner: first priority, type I; second priority, type II; and last priority, type III.
Statistical analysis
Proportion comparisons were performed by exact Fisher tests. The factors that influence the probability of observing BME signal were assessed with multivariate logistic regression using R (version 3.3.2) software [18]. The level of significance was 0.05.
Results
BME signals were present in 13 facet joints in seven healthy participants (12.7%) and 50 facet joints in 28 patients with low back pain (35.4%) (P = 0.003). The frequency of facet joint BME in each subject is presented in Table 1, and notably, the frequency of subjects with 2 or more areas of BME signals in the whole spine was significantly higher in the low back pain group than in the healthy group (P = 0.011).
The frequency and grades of lumbar facet joint osteoarthritis are presented in Table 2.
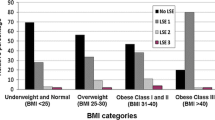
By pooling the facet joint levels of all subjects, the osteoarthritis grade distributions for each were found to be statistically significantly different (P < 0.001), and notably, there was a higher proportion of grade 3 in the low back pain group. The prevalences of BME signals according to the maximal grade of facet joint osteoarthritis in all subjects are presented in Fig. 3. The distribution of disk abnormalities and vertebral end plate abnormalities according to Modic are shown in Table 3. The groups were statistically different in terms of age and BMI (P < 0.001). The factors that influence the probability of observing more than one BME signal are provided in Table 4. Based on the results of logistic regression analysis adjusted for confounding factors, which included low back pain status, age, BMI, Modic type 1 vertebral end plate, disk herniation, and facet joint osteoarthritis maximal grade, only the latter was found to be significantly associated with the facet joint BME signal.
Discussion
This study results revealed the following: (1) the facet joint BME signals were more prevalent and frequent in the low back pain patients than they were in healthy subjects and (2) the BME signal was associated with the severity of facet joint osteoarthritis regardless of the pain status.
Although the prevalence and frequency of the facet joint BME signals were higher in the low back pain group than in the healthy group, 12% of the facet joints of healthy participants presented a BME signal, which indicated that relationship to low back pain was inconsistent, which may have been due to BME signals’ association with a wide variety of diseases, including trauma-induced lesions and degenerative and inflammatory osteoarthropathies with various histopathological substrates [9, 19, 20]. BME signals in vertebral end plates, known as Modic type 1 signals, are commonly related to a particular chain of events. Specifically, an initial mechanical phenomenon is followed by microfractures of the cancellous bone and end plate. An inflammatory response characterized by edema in the end plates and/or adjacent bone marrow is followed by the onset of fibrovascular marrow replacement and the diffusion of low-virulence anaerobic bacteria, which can further propagate peridiscal marrow inflammation [21, 22]. In the knee, such signal corresponds histologically to medullary necrosis, fibrosis, or trabecular abnormalities, whereas edema is a minor consideration in this study [20, 23]. Histopathological analyses in rheumatoid arthritis patients have shown that the BME signal is related to lymphocyte and osteoclast infiltrates and is predictive of erosive lesions [24].
Without histopathological analysis, the significance of the facet joint BME signal in low back pain patients is therefore unclear. Our results revealed that the facet joint BME signal was related to the severity of the facet joint osteoarthritis in both low back pain patients and healthy subjects, with BME signal and osteoarthritis predominating in the overloaded L4/L5 and L5/S1 lumbar levels. Similar signal intensity was reported by Morrison et al. in lumbar pedicles associated with adjacent facet degenerative joint disease [25]. Nevertheless, the BME signal does not appear to be by itself the causal process of subchondral bone marrow disease. On the contrary, the causal process is typically associated with biomechanical stress, joint instability, or a degenerative disease, with the extent and severity of the BME signal being linked to cartilage loss [9, 19, 23, 26, 27].
Moreover, our multivariate logistic regression analyses revealed that pain status does not influence the probability of observing a facet joint BME signal. This observation stands in contrast to the retrospective study by Friedrich et al., who reported that the prevalence of facet joint BME signal in low back patients is only 14%, which is much lower than that found in the low back pain group and close to that observed in our healthy group, although these authors did suggest that this signal may be relevant to the planning of facet joint treatment [15]. The prevalence of facet joints osteoarthritis has differed from one study group to another [7, 10, 28]. Jensen et al. and Weishaupt et al. reported that this prevalence varied from 8 to 12% in asymptomatic subjects between the ages of 35 and 42 [10, 28], while it reached 90% in young asymptomatic tennis players [7]. Although our healthy group are very different in terms of sport activities, 83% of our asymptomatic subjects showed mild facet joint osteoarthritis.
Disk abnormalities, particularly extrusion, also commonly cause low back pain. In our healthy participants, no one presented with disk protrusion or extrusion. These results differed somewhat from those in Weishaupt et al. who reported prevalences of protrusion and extrusion in 38% and 18% of their asymptomatic subjects, respectively [10]. The incidentally low prevalence of disk abnormalities in our healthy participants allowed us to focus on a uniform study group of patients with facet joint osteoarthritis.
Our study had several limitations. First, our healthy participants were younger than our low back pain patients, and the prevalence of low back pain increased with age, making it difficult to recruit older healthy participants. However, the logistic regression analysis took this difference into account, and it did not influence the probability of observing a facet joint BME signal. Secondly, a vast majority of our patients complained of chronic low back pain, so it was not possible to evaluate the impact of low back pain duration on the BME signal. Investigating its prevalence in patients suffering from acute low back pain and its evolution over time would provide important insight regarding this relationship. Thirdly, the MRI scans could have underestimated the severity of facet joint osteoarthritis compared to CT scans [29]. However, in their study, comparing MRI and CT scanning results obtained in the assessment of lumbar facet joint osteoarthritis, Weishaupt et al. observed moderate to good agreement between both techniques, and excellent agreement was found when differences of one grade were omitted [30]. Fourthly, selective facet joint block testing was not performed on our low back pain group to distinguish facet joint syndrome from other confounding factors that can cause low back pain. However, even when properly conducted, false-positive rates of 25–44% have been reported for lumbar facet joint nerve blocks [31]. Moreover, multivariate logistic regression analyses have taken certain other factors into account, including age, BMI, Modic type 1 vertebral end plate, and disk herniation, which have also been found to contribute to low back pain. Finally, maybe our MR protocol including axial T2-weighted STIR sequences was not optimal to assess small facet joint osteoarthritis especially in vertebral alignment deformities such as scoliosis or spondylolisthesis, and we should probably have used additional sagittal T2-weighted STIR combined with axial T2-weighted sequences. Nevertheless, the aim of our study was to evaluate the presence of even small areas of bone marrow edema and to differentiate them from partial volume effect of the joint fluid or subchondral bone cysts. Moreover, we took into account all MR sequences to grade osteoarthritis.
In conclusion, the prevalence and frequency of the BME signal of the facet joint in low back pain patients were higher compared to the signal in healthy subjects, and this signal appears to be related to the severity of the osteoarthritis, but not to the low back pain status.
References
GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;39:21789–858. https://doi.org/10.1016/S0140-6736(18)32279-7.
Kalichman L, Hunter DJ. Lumbar facet joint osteoarthritis: a review. Semin Arthritis Rheum. 2007;37:69–80. https://doi.org/10.1016/j.semarthrit.2007.01.007.
Varlotta GP, Lefkowitz TR, Schweitzer M, Errico TJ, Spivak J, Bendo JA, et al. The lumbar facet joint: a review of current knowledge: part II: diagnosis and management. Skelet Radiol. 2011;40:149–57. https://doi.org/10.1007/s00256-010-0984-3.
Eubanks JD, Lee MJ, Cassinelli E, Ahn NU. Prevalence of lumbar facet arthrosis and its relationship to age, sex, and race: an anatomic study of cadaveric specimens. Spine. 2007;32:2058–62. https://doi.org/10.1097/BRS.0b013e318145a3a9.
Weishaupt D, Zanetti M, Hodler J, Min K, Fuchs B, Pfirrmann CW, et al. Painful lumbar disk derangement: relevance of endplate abnormalities at MR Imaging. Radiology. 2001;218:420–7. https://doi.org/10.1148/radiology.218.2.r01fe15420.
Alyas F, Turner M, Connell D. MRI findings in the lumbar spines of asymptomatic, adolescent, elite tennis players. Br J Sports Med. 2007;41:836–41. https://doi.org/10.1136/bjsm.2007.037747.
Rajeswaran G, Turner M, Gissane C, Healy JC. MRI findings in the lumbar spines of asymptomatic elite junior tennis players. Skelet Radiol. 2014;43:925–32. https://doi.org/10.1007/s00256-014-1862-1.
Moulopoulos LA, Koutoulidis V In: Bone Marrow MRI-A pattern-based Approach-Springer –Verlag Italia 2015;115.
Rahme R, Moussa R. The modic vertebral endplate and marrow changes: pathologic significance and relation to low back pain and segmental instability of the lumbar spine. AJNR Am J Neuroradiol. 2008;29:838–42. https://doi.org/10.3174/ajnr.A0925.
Weishaupt D, Zanetti M, Hodler J, Boos N. MR imaging of the lumbar spine: prevalence of intervertebral disk extrusion and sequestration, nerve root compression, end plate abnormalities, and osteoarthritis of the facet joints in asymptomatic volunteers. Radiology. 1998;209:661–6. https://doi.org/10.1148/radiology.209.3.9844656.
Kuisma M, Karppinen J, Niinimäki J, Ojala R, Haapea M, Heliövaara M, et al. Modic changes in endplates of lumbar vertebral bodies: prevalence and association with low back and sciatic pain among middle-aged male workers. Spine. 2007;32:1116–22. https://doi.org/10.1097/01.brs.0000261561.12944.ff.
Thompson KJ, Dagher AP, Eckel TS, Clark M, Reinig JW. Modic changes on MR images as studied with provocative diskography: clinical relevance--a retrospective study of 2457 disks. Radiology. 2009;250:849–55. https://doi.org/10.1148/radiol.2503080474.
Mok FP, Samartzis D, Karppinen J, Fong DY, Luk KD, Cheung KM. Modic changes of the lumbar spine: prevalence, risk factors, and association with disc degeneration and low back pain in a large-scale population-based cohort. Spine J. 2016;16:32–41. https://doi.org/10.1016/j.spinee.2015.09.060.
Borg B, Modic MT, Obuchowski N, Cheah G. Pedicle marrow signal hyperintensity on short tau inversion recovery- and T2-weighted images: prevalence and relationship to clinical symptoms. AJNR Am J Neuroradiol. 2011;32:1624–31. https://doi.org/10.3174/ajnr.A2588.
Friedrich KM, Nemec S, Peloschek P, Pinker K, Weber M, Trattnig S. The prevalence of lumbar facet joint edema in patients with low back pain. Skelet Radiol. 2007;36:755–60. https://doi.org/10.1007/s00256-007-0293-7.
Kuorinka I, Jonsson B, Kilbom A, Vinterberg H, Biering-Sørensen F, Andersson G, et al. Standardised Nordic questionnaires for the analysis of musculoskeletal symptoms. Appl Ergon. 1987;18:233–7.
Fardon DF, Williams AL, Dohring EJ, Murtagh FR, Gabriel Rothman SL, Sze GK. Lumbar disc nomenclature: version 2.0: recommendations of the combined task forces of the North American Spine Society, the American Society of Spine Radiology and the American Society of Neuroradiology. Spine J. 2014;14:2525–45. https://doi.org/10.1016/j.spinee.2014.04.022.
R Development Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.r-project.org/. Published 2013. Accessed November 20, 2014.
Starr AM, Wessely MA, Albastaki U, Pierre-Jerome C, Kettner NW. Bone marrow edema: pathophysiology, differential diagnosis, and imaging. Acta Radiol. 2008;49:771–86. https://doi.org/10.1080/02841850802161023.
Zanetti M, Bruder E, Romero J, Hodler J. Bone marrow edema pattern in osteoarthritic knees: correlation between MR imaging and histologic findings. Radiology. 2000;215:835–40.
Crockett MT, Kelly BS, van Baarsel S, Kavanagh EC. Modic type 1 vertebral endplate changes: injury, inflammation, or infection? AJR Am J Roentgenol. 2017;209:167–70. https://doi.org/10.2214/AJR.16.17.
Dudli S, Liebenberg E, Magnitsky S, Lu B, Lauricella M, Lotz JC. Modic type 1 change is an autoimmune response that requires a pro-inflammatory milieu provided by the 'modic disc'. Spine J. 2018;18:831–44. https://doi.org/10.1016/j.spinee.2017.12.004.
Kijowski R, Stanton P, Fine J, De Smet A. Subchondral bone marrow edema in patients with degeneration of the articular cartilage of the knee joint. Radiology. 2006;238:943–9. https://doi.org/10.1148/radiol.2382050122.
McQueen FM, Ostendorf B. What is MRI bone oedema in rheumatoid arthritis and why does it matter? Arthritis Res Ther. 2006;8:222. https://doi.org/10.1186/ar2075.
Morrison JL, Kaplan PA, Dussault RG, Anderson MW. Pedicle marrow signal intensity changes in the lumbar spine: a manifestation of facet degenerative joint disease. Skelet Radiol. 2000;29:703–7.
Karchevsky M, Schweitzer ME, Carrino JA, Zoga A, Montgomery D, Parker L. Reactive endplate marrow changes: a systematic morphologic and epidemiologic evaluation. Skeletal Radiol. 2005;34:125–9. https://doi.org/10.1007/s00256-004-0886-3.
Modic MT. Modic type 1 and type 2 changes. J Neurosurg Spine. 2007;6:150–1.
Jensen MC, Brant-Zawadzki MN, Obuchowski N, Modic MT, Malkasian D, Ross JS. Magnetic resonance imaging of the lumbar spine in people without back pain. N Engl J Med. 1994;331:69–73. https://doi.org/10.1056/NEJM199407143310201.
Fujiwara A, Tamai K, Yamato M, An HS, Yoshida H, Saotome K, et al. The relationship between facet joint osteoarthritis and disc degeneration of the lumbar spine: an MRI study. Eur Spine J. 1999;8:396–401.
Weishaupt D, Zanetti M, Boos N, Hodler J. MR imaging and CT in osteoarthritis of the lumbar facet joints. Skelet Radiol. 1999;28:215–9.
Boswell MV, Manchikanti L, Kaye AD, Bakshi S, Gharibo CG, Gupta S, et al. A best-evidence systematic appraisal of the diagnostic accuracy and utility of facet (Zygapophysial) joint injections in chronic spinal pain. Pain Physician. 2015;18:E497–533.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were done so in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Madani, A., Katz, R., Muylem, A.V. et al. Prevalence and frequency of subchondral bone marrow edema in the lumbar facet joints of asymptomatic and symptomatic individuals. Skeletal Radiol 49, 1141–1147 (2020). https://doi.org/10.1007/s00256-020-03400-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-020-03400-4