Abstract
We present two cases of peripheral nerve schwannoma which showed an increased accumulation of 2-deoxy-[18F] fluoro-D-glucose (FDG) in the tumors on positron emission tomography (PET) imaging acquired at both 1 h (early phase) and 2 h (delayed phase) after FDG injection. FDG-PET scans were performed with a dedicated PET scanner (HeadtomeV/ SET2400 W, Shimadzu, Kyoto, Japan) and the PET data analyzed the most metabolically active region of interest (ROI). We set the maximum standardized uptake value (SUV max) with a cut-off point of 3.0 to distinguish benign and malignant lesions. Although the mechanism responsible for the increased FDG uptake in benign schwannomas remains unknown, we discuss our findings in the context of tumor cellularity and briefly review other studies on the subject.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Schwannoma is a usually benign soft tissue tumor originating from the peripheral nervous system [1, 2, 3, 4], which may be difficult to differentiate from malignant soft tissue tumors [2, 3, 5, 6]. Both CT and MRI are helpful in determining the size, location, invasion, and involvement of surrounding organs [2, 4, 5]. Its morphology and signal characteristics may mimic a malignant soft tissue tumor [2].
Two-deoxy-[18F] fluoro-D-glucose (FDG) positron emission tomography (PET) is an evolving diagnostic modality used for tumor detection, staging, therapeutic monitoring, and follow-up evaluations for various malignant tumors [6, 7, 8]. Recently, to improve the accuracy of diagnosis, the use of delayed PET imaging has been proposed for various malignant tumors [6, 9].
We have performed FDG-PET scans for preoperative staging in patients with clinically suspected malignant musculoskeletal tumors and follow-up evaluations.
In this article we present and discuss two benign schwannomas which showed increased activity on PET scanning at both 1 and 2 h after FDG injection mimicking a finding seen with malignant soft tissue tumors.
PET imaging
Each patient fasted for at least 4 h before intravenous administration of approximately 370 MBq of FDG. Serum glucose levels were determined just before FDG injection. Both patients were normoglycemic (<100 mg/dl). Simultaneous emission–transmission PET scans were acquired 1 h after FDG injection (transmission source: 68 Ga line source). The PET scans after 1 and 2 h following FDG injection were performed using a dedicated scanner (Headtome/ SET2400 W, Shimadzu, Kyoto, Japan), which has 32 rings of Bismuth Germanate (BGO) detectors and simultaneously produces 63 slices 3.125-mm-thick along a 20-cm longitudinal field. The intrinsic resolution was 3.7 mm full width at half-maximum (FWHM), and the sensitivity of the device was 7.3 cps/Bq cm−3. Whole-body scans required four bed positions, each with an acquisition time of 7 min, resulting in a total scanning time of 30 min in each scan. Images were reconstructed with an iterative median root prior reconstruction algorithm (mask size 3×3, β 0.3, subsets 24, iteration1).
Data analysis
High-resolution transaxial, coronal, and sagittal image sets were displayed on a monitor in a linear gray scale and scaled from the count 0 to 7500 per voxel. Regions of interest (ROIs) were semi-automatically placed over the highest accumulation area corresponding to the tumors on the PET images. The transaxial slice with the highest radioactivity concentration within the tumor was identified, and the standardized uptake value of the highest point within the ROI (SUVmax) was calculated by dividing decay-corrected maximal count of tumor area by injected dose of FDG per unit of body weight. We set SUVmax with a cutoff of 3.0 to distinguish benign from malignant lesions.
Case reports
Case 1
A 28-year-old woman presented with a history of swelling and slight tenderness in the right neck that had been present for a few years. She had no clinical symptoms such as pain at rest or neurological deficit. A cervical radiograph was normal. On MR imaging, T1-weghted images (TR/TE: 500/13.5 ms) demonstrated a soft tissue mass (5×3.5×3 cm in size) in paravertebral muscle that exhibited iso-signal intensity with that of skeletal muscle. T2-weighted images (TR/TE: 3800/82.3 ms) showed an inhomogeneous high-signal-intensity mass. The tumor showed relatively homogenous contrast enhancement on T1-weighted images obtained after an intravenous gadolinium infusion (meglumine gadopentetate, Magnevist10 ml; Fig. 1).
Magnetic resonance imaging coronal T1-weighted image (TR/TE: 500/13.5 ms) demonstrates a soft mass lesion in paravertebral muscle with iso-signal intensity to muscle. Coronal T2-weighted image (TR/TE: 3800/82.3 ms) shows inhomogeneous high signal intensity within the mass. Coronal T1-weighted image after intravenous gadolinium shows homogenous enhancement of the mass.
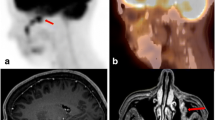
FDG-PET imaging was performed at 60 (early phase) and 120 min (delayed phase) after an intravenous injection of 366.3 MBq of FDG. The resulting image showed a mass with an increased accumulation of FDG. The SUVmax of this lesion was 5.95 in early phase and 7.04 in delayed phase, suggesting a malignant tumor (Fig. 2).
An open biopsy was performed and the tissue obtained was histologically diagnosed as a schwannoma. No evidence of malignancy was present in the surgically resected specimens (Fig. 3). Histologically, the tumor was composed of spindle cells with palisading in some areas (Fig. 3B, arrow), showing two distinct histological features: one with packed spindle cells and collagenous background consistent with Antoni A elements (Fig. 3C), the other with sparsely located cells in the myxoid background, identical to Antoni B elements (Fig. 3D). No mitotic figures or bizarre tumor cells were seen. These findings were consistent with a benign schwannoma.
Case 2
A 40-year-old woman presented with a 7-year history of swelling in the iliac crest. She was completely asymptomatic with no significant past medical or surgical history. A pelvic radiograph was normal. On MR imaging, T1-weghted images (TR/TE: 608/12 ms) before and after contrast enhancement (meglumine gadopentetate, Magnevist 10 ml) and T2-weighted images (TR/TE: 4000/96 ms) were obtained. An axial MRI revealed a soft tissue mass in the right retroperitoneum with a homogeneous, low-signal intensity on T1-weighted images and high-signal intensity on T2-weighted images. The tumor exhibited an inhomogeneous contrast enhancement on T1-weighted images obtained after an intravenous gadolinium infusion (Fig. 4).
Axial MR imaging reveals a soft tissue mass in the right retroperitoneum with a homogeneous, low signal intensity on T1-weighted images (TR/TE: 608/12 ms) and high signal intensity on T2-weighted images (TR/TE: 4000/96 ms). The tumor shows inhomogeneous contrast enhancement on T1-weighted images obtained after an intravenous gadolinium infusion.
FDG-PET imaging was performed at 1 h (early phase) and 2 h (delayed phase) after the intravenous injection of 374.8 MBq of FDG. The resulting image showed a mass with an increased accumulation of FDG. The SUVmax of this lesion was 3.91 in early phase and 4.31 in delayed phase, suggesting a malignant tumor (Fig. 5).
A histological diagnosis of schwannoma was obtained from the tissue of an open biopsy, and a subsequent marginal excision was performed. Histologically, the tumor showed similar feature to case 1, with distinct Antoni A (Fig. 6) and Antoni B areas (Fig. 6). The tumor had no mitosis or bizarre cells and was diagnosed as schwannoma.
Discussion
Schwannoma is generally a benign, slow-growing, encapsulated tumor originating from the nerve sheath that usually occurs in the extremities, and head and neck region [1, 2, 3]. A few cases of benign schwannomas in the retroperitoneum have also been reported [1, 2, 5]. Benign soft tissue masses and soft tissue sarcomas may have overlapping clinical and imaging features [2, 3, 5, 6]. Biopsy of an indeterminate mass is usually the most specific method for obtaining an accurate diagnosis.
Clinical symptoms may be of diagnostic value in differentiating benign and malignant tumors. Pain at rest is rare with benign tumors but almost always present if malignant [3]. Neurogenic deficits are also rare in benign tumors [3]. Our two patients were relatively asymptomatic.
The MR imaging features of benign nerve sheath tumors usually show a smooth margin, well-defined, fusiform mass that enters and exits the nerve [5]. Schwannomas generally are iso intense with muscle on T1-weighted images and hyperintense on T2-weighted images [1, 5]. The target sign (a peripheral hyperintense rim and central low intensity on T2 weighted images) is also useful in diagnosing a benign nerve sheath tumors [11]. Contrast-enhancement T1-weighted image is considered to be a defining feature of schwannoma. The enhancement pattern is typically inhomogeneous, with stronger enhancement peripherally, but may be homogeneous [9, 10, 11, 12]. Due to heterogeneity and degeneration, it may mimic malignancy.
Microscopically, schwannomas display a biphasic pattern with areas of highly ordered cellularity (Antoni A) and other, less cellular areas where a copious myxoid matrix predominates (Antoni B). The global degree of cellularity varied widely among lesions [13]. Based on Beaulieu et al.’s [13] observations, our cases were hypercellular tumors which consisted of lesions with more than 10% Antoni A areas. Beaulieu et al. [13] reported that the wide range of SUVs for FDG appears to be explained by the variable degree of cellularity for each lesion.
FDG-PET imaging is increasingly used in clinical oncology because it allows functional imaging of viable tumor tissue [14]. The use of FDG-PET for the detection and monitoring of patients with musculoskeletal lesions has also been reported [15]. Several previous studies have reported the usefulness of FDG-PET in distinguishing high-grade soft tissue sarcomas from low-grade or benign soft tissue tumors [6, 7, 16, 17]; however, considerable overlap in the SUV has been observed between benign and malignant lesions [16, 18, 19]. The large accumulation of FDG in benign soft tissue tumors has been reported in giant cell tumors of tendon sheath, sarcoidosis, and desmoid [16, 18, 19]. The SUV of schwannomas has been reported to show a wide variation of 0.33–3.7 [4], 1.75±0.84 [16], and 0.7–2.84 [18], but the SUV of the present cases are some of the highest figures reported to date.
To improve the accuracy of FDG-PET for differential diagnoses, delayed PET imaging has been recently introduced as a supplementary method to conventional SUV analysis. Lodge et al. [6] reported that high-grade soft tissue sarcomas reached a peak activity concentration approximately 4 h after injection, whereas benign lesions reached a maximum activity concentration within 30 min. Kubota et al. [8] also reported that most malignant tumors showed a higher FDG uptake at 2 h rather than at 1 h, whereas most normal tissue showed a lower FDG uptake at 2 h in comparison with uptake at 1 h.
We obtained FDG-PET images 1 and 2 h after the injection of FDG. Both our cases of schwannomas showed greater accumulations at 2 h than at 1 h. The results based on the accumulation of FDG in both phases resembled those of malignant soft tissue tumors.
The large accumulation of FDG in schwannomas might result from the potential ability of schwann cells to transport glucose for axonal repolarization in the peripheral nerve system [18, 20, 21]; however, a consensus has not been reached on the precise mechanism of FDG accumulation in schwannomas.
The biological significance of the difference in FDG uptake among various tumors is not clear [21]. Although Lodge et al. [6] reported that benign lesions reach a peak activity concentration within 30 min, FDG uptake in these schwannomas did not reach a plateau within 30 min after injection. Soft tissue lesions exhibiting large accumulations of FDG might not always be malignant [16], care must be taken in interpreting FDG-PET imaging for preoperative staging, even when delayed PET imaging is performed.
Further studies on the intratumoral distribution of FDG in schwannomas with a heterogeneous cellular composition may help to clarify the role of FDG-PET in distinguishing these benign tumors from a malignancy.
References
Hide IG, Baudouin CJ, Murray SA, Malcolm AJ. Giant ancient schwannoma of the pelvis. Skeletal Radiol 2000; 29:538–542.
Schindler OS, Dixon JH, Case P. Retroperitoneal giant schwannomas: report on two cases and review of the literature. J Orthop Surg (Hong Kong) 2002; 10:77–84.
Ogose A, Hotta T, Morita T, Yamamura S, Hosaka N, Kobayashi H, et al. Tumors of peripheral nerves: correlation of symptoms, clinical signs, imaging features, and histologic diagnosis. Skeletal Radiol 1999; 28:183–188.
Ahmed AR, Watanabe H, Aoki J, Shinozaki T, Takagishi K. Schwannoma of the extremities: the role of PET in preoperative planning. Eur J Nucl Med 2001; 28:1541–1551.
Daneshmand S, Youssefzadeh D, Chamie K, Boswell W, Wu N, Stein JP, et al. Benign retroperitoneal schwannoma: a case series and review of the literature. Urology 2003; 62:993–997.
Lodge MA, Lucus JD, Marsden PK, Cronin BF, O’Doherty MJ, Smith MA. A PET study of18FDG uptake in soft tissue masses. Eur J Nucl Med 1999; 26:22–30
Schulte M, Brecht-Krauss D, Heymer B, Guhlmann A, Hartwig E, Sarkar MR, et al. Fluorodeoxyglucose positron emission tomography of soft tissue tumours: is a non-invasive determination of biological activity possible? Eur J Nucl Med 1999; 26: 599–605.
Kubota K, Itoh M, Ozaki K, Ono S, Tashiro M, Yamaguchi K, et al. Advantage of delayed whole-body FDG-PET imaging of tumor detection. Eur J Nucl Med 2001; 28:696–703.
Soderlund V, Goranson H, Bauer HC. MR imaging of benign peripheral nerve sheath tumors. Acta Radiol 1994; 35:282–286.
Lee JA, Boles CA. Peripheral schwannoma lacking enhancement on MRI. Am J Roentgenol 2004; 182:534–535.
Varma DG, Moulopoulos A, Sara AS, Leeds N, Kumar R, Kim EE, et al. MR imaging of extracranial nerve sheath tumors. J Comput Assist Tomogr 1992; 16:448–453.
Beggs I. Pictorial review: imaging of peripheral nerve tumours. Clin Radiol 1997; 52:8–17
Beaulieu S, Rubin B, Djang D, Conrad E, Turcotte E, Eary JF. Positron emission tomography of schwannomas: emphasizing its potential in preoperative planning. Am J Roentgenol 2004; 182:971–974.
Strauss LG, Conti PS. The applications of PET in clinical oncology. J Nucl Med 1991; 32:623–648.
Kern KA, Brunetti A, Norton JA, Chang AE, Malawer M, Lack E, et al. Metabolic imaging of human extremity musculoskeletal tumors by PET. J Nucl Med 1988; 29:181–186.
Aoki J, Watanabe H, Shinozaki T, Takagishi K, Tokunaga M, Koyama Y, et al. FDG-PET for preoperative differential diagnosis between benign and malignant soft tissue masses. Skeletal Radiol 2003; 32:133–138.
Adler LP, Blair HF, Makley JT, Williams RP, Joyce MJ, Leisure G, et al. Noninvasive grading of musculoskeletal tumors using PET. J Nucl Med 1991; 32:1508–1512.
Aoki J, Endo K, Watanabe H, Shinozaki T, Yanagawa T, Ahmed AR, et al. FDG-PET for evaluating musculoskeletal tumors: a review. J Orthop Sci 2003; 8:435–441.
Watanabe H, Inoue T, Shinozaki T, Yanagawa T, Ahmed AR, Tomiyoshi K, et al. PET imaging of musculoskeletal tumours with fluorine-18α- methyltyrosine: comparison with fluorine-18 fluoro-deoxyglucose PET. Eur J Nucl Med 2000; 27:1509–1517.
Magnani P, Thomas TP, Tennekoon G, DeVries GH, Greene DA, Brosiu FC III. Regulation of glucose transport in cultured Schwann cells. J Peripher Nerv Syst 1998; 3:28–36.
Waki A, Fujibayashi Y, Yokoyama A. Recent advances in the analyses of the characteristics of tumor on FDG uptake: mini review. Nucl Med Biol 1998; 25:589–592.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hamada, K., Ueda, T., Higuchi, I. et al. Peripheral nerve schwannoma: two cases exhibiting increased FDG uptake in early and delayed PET imaging. Skeletal Radiol 34, 52–57 (2005). https://doi.org/10.1007/s00256-004-0845-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-004-0845-z