Abstract
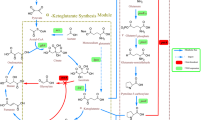
Monascus sp. is an important food microbial resource with the production of cholesterol-lowering agent lovastatin and other healthy metabolites. However, the mycotoxin citrinin naturally produced by Monascus sp. and the insufficient productivity of lovastatin limit its large-scale use in food industry. The aim of this paper is to modify a lovastatin-producing strain Monascus pilosus GN-01 through metabolic engineering to obtain a citrinin-free M. pilosus strain with higher yield of lovastatin. The citrinin synthesis regulator gene ctnR was firstly disrupted to obtain GN-02 without citrinin production. Based on that, the lovastatin biosynthesis genes (mokC, mokD, mokE, mokF, mokH, mokI, and LaeA) were, respectively, overexpressed, and pigment-regulatory gene (pigR) was knocked out to improve lovastatin production. The results indicated ctnR inactivation effectively disrupted the citrinin release by M. pilosus GN-01. The overexpression of lovastatin biosynthesis genes and pigR knockout could lead higher contents of lovastatin, of which pigR knockout strain achieved 76.60% increase in the yield of lovastatin compared to GN-02. These studies suggest that such multiplex metabolic pathway engineering in M. pilosus GN-01 is promising for high lovastatin production by a safe strain for application in Monascus-related food.
Key points
• Disruption of the regulator gene ctnR inhibited citrinin production of M. pilosus.
• Synchronous overexpression of biosynthesis gene enhanced lovastatin production.
• pigR knockout enhanced lovastatin of ΔctnR strain of M. pilosus.







Similar content being viewed by others
Data availability
On reasonable request, the corresponding authors will provide the datasets created and/or analyzed during the current work.
References
Bryksin AV, Matsumura I (2010) Overlap extension PCR cloning: a simple and reliable way to create recombinant plasmids. Biotechniques 48(6):463–465. https://doi.org/10.2144/000113418
Cheng MJ, Wu MD, Chen IS, Chen CY, Lo WL, Yuan GF (2010) Secondary metabolites from the red mould rice of Monascus purpureus BCRC 38113. Nat Prod Res 24(18):1719–1725. https://doi.org/10.1080/14786410902941477
de Oliveira Filho JWG, Islam MT, Ali ES, Uddin SJ, Santos JVO, de Alencar M, Junior ALG, Paz M, de Brito M, JMC ES, Shaw S, de Medeiros M, Dantas S, Rolim HML, Ferreira PMP, Kamal MA, Pieczynska MD, Das N, Gupta VK, Mocan A, Dos Santos Andrade TJA, Singh BN, Mishra SK, Atanasov AG, Melo-Cavalcante AAC (2017) A comprehensive review on biological properties of citrinin. Food Chem Toxicol 110:130–141. https://doi.org/10.1016/j.fct.2017.10.002
DeBose-Boyd RA (2008) Feedback regulation of cholesterol synthesis: sterol-accelerated ubiquitination and degradation of HMG CoA reductase. Cell Res 18(6):609–621. https://doi.org/10.1038/cr.2008.61
Duan Y, Du Y, Yi Z, Wang Z, Pei X, Wei X, Li M (2022) Systematic metabolic engineering for the production of azaphilones in Monascus purpureus HJ11. J Agric Food Chem 70(5):1589–1600. https://doi.org/10.1021/acs.jafc.1c07588
Duan Y, Tan Y, Wei X, Pei X, Li M (2023) Versatile strategy for the construction of a transcription factor-based orthogonal gene expression toolbox in Monascus spp. ACS Synth Biol 12(1):213–223. https://doi.org/10.1021/acssynbio.2c00500
Embaby AM, Hussein MN, Hussein A (2018) Monascus orange and red pigments production by Monascus purpureus ATCC16436 through co-solid state fermentation of corn cob and glycerol: an eco-friendly environmental low cost approach. PLOS One 13(12):e0207755. https://doi.org/10.1371/journal.pone.0207755
Farawahida AH, Palmer J, Flint S (2022) Monascus spp. and citrinin: identification, selection of Monascus spp. isolates, occurrence, detection and reduction of citrinin during the fermentation of red fermented rice. Int J Food Microbiol 379:109829. https://doi.org/10.1016/j.ijfoodmicro.2022.109829
Heckman KL, Pease LR (2007) Gene splicing and mutagenesis by PCR-driven overlap extension. Nat Protoc 2(4):924–932. https://doi.org/10.1038/nprot.2007.132
Kamle M, Mahato DK, Gupta A, Pandhi S, Sharma N, Sharma B, Mishra S, Arora S, Selvakumar R, Saurabh V, Dhakane-Lad J, Kumar M, Barua S, Kumar A, Gamlath S, Kumar P (2022) Citrinin mycotoxin contamination in food and feed: impact on agriculture, human health, and detection and management strategies. Toxins (basel) 14(2):85. https://doi.org/10.3390/toxins14020085
Khang CH, Park SY, Lee YH, Kang S (2005) A dual selection based, targeted gene replacement tool for Magnaporthe grisea and Fusarium oxysporum. Fungal Genet Biol 42(6):483–492. https://doi.org/10.1016/j.fgb.2005.03.004
Lee CL, Hung HK, Wang JJ, Pan TM (2007) Improving the ratio of monacolin K to citrinin production of Monascus purpureus NTU 568 under dioscorea medium through the mediation of pH value and ethanol addition. J Agr Food Chem 55(16):6493–6502. https://doi.org/10.1021/jf0711946
Lee SS, Lee JH, Lee I (2013) Strain improvement by overexpression of the laeA gene in Monascus pilosus for the production of Monascus-fermented rice. J Microbiol Biotechn 23(7):959–965. https://doi.org/10.4014/jmb.1303.03026
Li YP, Pan YF, Zou LH, Xu Y, Huang ZB, He QH (2013) Lower citrinin production by gene disruption of ctnB involved in citrinin biosynthesis in Monascus aurantiacus Li AS3.4384. J Agr Food Chem 61(30):7397–7402. https://doi.org/10.1021/jf400879s
Li YP, Tang X, Wu W, Xu Y, Huang ZB, He QH (2015) The ctnG gene encodes carbonic anhydrase involved in mycotoxin citrinin biosynthesis from Monascus aurantiacus. Food Addit Contam A 32(4):577–583. https://doi.org/10.1080/19440049.2014.990993
Li Z, Liu Y, Li Y, Lin F, Wu L (2020) Screening and identification of Monascus strains with high-yield monacolin K and undetectable citrinin by integration of HPLC analysis and pksCT and ctnA genes amplification. J Appl Microbiol 129(5):1410–1418. https://doi.org/10.1111/jam.14689
Liang B, Du XJ, Li P, Sun CC, Wang S (2018) Investigation of citrinin and pigment biosynthesis mechanisms in Monascus purpureus by transcriptomic analysis. Front Microbiol 9:1374. https://doi.org/10.3389/fmicb.2018.01374
Lin TS, Chiu SH, Chen CC, Lin CH (2023) Investigation of monacolin K, yellow pigments, and citrinin production capabilities of Monascus purpureus and Monascus ruber (Monascus pilosus). J Food Drug Anal 31(1):85–94. https://doi.org/10.38212/2224-6614.3438
Liu BH, Wu TS, Su MC, Chung CP, Yu FY (2005) Evaluation of citrinin occurrence and cytotoxicity in Monascus fermentation products. J Agric Food Chem 53(1):170–175. https://doi.org/10.1021/jf048878n
Liu A, Juan Chen A, Liu B, Wei Q, Bai J, Hu Y (2022) Investigation of citrinin and monacolin K gene clusters variation among pigment producer Monascus species. Fungal Genet Biol 160:103687. https://doi.org/10.1016/j.fgb.2022.103687
Long CN, Zeng X, Xie J, Liang YM, Tao JJ, Tao QQ, Liu MM, Cui JJ, Huang ZW, Zeng B (2019) High-level production of Monascus pigments in Monascus ruber CICC41233 through ATP-citrate lyase overexpression. Biochem Eng J 146:160–169. https://doi.org/10.1016/j.bej.2019.03.007
Lv J, Zhang BB, Liu XD, Zhang C, Chen L, Xu GR, Cheung PCK (2017) Enhanced production of natural yellow pigments from Monascus purpureus by liquid culture: the relationship between fermentation conditions and mycelial morphology. J Biosci Bioeng 124(4):452–458. https://doi.org/10.1016/j.jbiosc.2017.05.010
McKenney JM (1988) Lovastatin: a new cholesterol-lowering agent. Clin Pharm 7(1):21–36
Mozsik L, Pohl C, Meyer V, Bovenberg RAL, Nygard Y, Driessen AJM (2021) Modular synthetic biology toolkit for filamentous fungi. ACS Synth Biol 10(11):2850–2861. https://doi.org/10.1021/acssynbio.1c00260
Siedenberg D, Mestric S, Ganzlin M, Schmidt M, Punt PJ, van den Hondel C, Rinas U (1999) GlaA promoter controlled production of a mutant green fluorescent protein (S65T) by recombinant aspergillus niger during growth on defined medium in batch and fed-batch cultures. Biotechnol Prog 15(1):43–50. https://doi.org/10.1021/bp980105u
Suwannarach N, Kumla J, Nishizaki Y, Sugimoto N, Meerak J, Matsui K, Lumyong S (2019) Optimization and characterization of red pigment production from an endophytic fungus, Nigrospora aurantiaca CMU-ZY2045, and its potential source of natural dye for use in textile dyeing. Appl Microbiol Biotechnol 103(17):6973–6987. https://doi.org/10.1007/s00253-019-09926-5
Tobert JA (2003) Lovastatin and beyond: the history of the HMG-CoA reductase inhibitors. Nat Rev Drug Discov 2(7):517–526. https://doi.org/10.1038/nrd1112
Xie NN, Liu QP, Chen FS (2013) Deletion of pigR gene in Monascus ruber leads to loss of pigment production. Biotechnol Lett 35(9):1425–1432. https://doi.org/10.1007/s10529-013-1219-1
Zhang C, Liang J, Yang L, Sun B, Wang C (2017) De novo RNA sequencing and transcriptome analysis of Monascus purpureus and analysis of key genes involved in monacolin K biosynthesis. PLoS One 12(1):e0170149. https://doi.org/10.1371/journal.pone.0170149
Zhang C, Liang J, Zhang A, Hao S, Zhang H, Zhu Q, Sun B, Wang C (2019) Overexpression of monacolin K biosynthesis genes in the Monascus purpureus azaphilone polyketide pathway. J Agric Food Chem 67(9):2563–2569. https://doi.org/10.1021/acs.jafc.8b05524
Zhang C, Zhang H, Zhu Q, Hao S, Chai S, Li Y, Jiao Z, Shi J, Sun B, Wang C (2020) Overexpression of global regulator LaeA increases secondary metabolite production in Monascus purpureus. Appl Microbiol Biotechnol 104(7):3049–3060. https://doi.org/10.1007/s00253-020-10379-4
Zhang S, Zeng X, Lin Q, Liu J (2022) Analysis of secondary metabolite gene clusters and chitin biosynthesis pathways of Monascus purpureus with high production of pigment and citrinin based on whole-genome sequencing. PLoS One 17(6):e0263905. https://doi.org/10.1371/journal.pone.0263905
Zhen Z, Xiong X, Liu Y, Zhang J, Wang S, Li L, Gao M (2019) NaCl inhibits citrinin and stimulates Monascus pigments and monacolin K production. Toxins (Basel) 11(2). https://doi.org/10.3390/toxins11020118
Funding
This work is financially supported by the grant from National Natural Science Foundation of China (32001634) and Natural Science Foundation of Fujian Province, China (2020J01488).
Author information
Authors and Affiliations
Contributions
HXK and GTL contributed equally to this work. HXK and GTL conducted experiments and wrote the manuscript. XXQ and LJ conceived and designed research. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Xiaokun Hong and Tianlong Guo contributed equally to this work.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hong, X., Guo, T., Xu, X. et al. Multiplex metabolic pathway engineering of Monascus pilosus enhances lovastatin production. Appl Microbiol Biotechnol 107, 6541–6552 (2023). https://doi.org/10.1007/s00253-023-12747-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-023-12747-2