Abstract
The plasma membrane (PM) is a key structure for the survival of cells during dehydration. In this study, we focused on the concomitant changes in survival and in the lateral organization of the PM in yeast strains during desiccation, a natural or technological environmental perturbation that involves transition from a liquid to a solid medium. To evaluate the role of the PM in survival during air-drying, a wild-type yeast strain and an osmotically fragile mutant (erg6Δ) were used. The lateral organization of the PM (microdomain distribution) was observed using a fluorescent marker related to a specific green fluorescent protein-labeled membrane protein (Sur7-GFP) after progressive or rapid desiccation. We also evaluated yeast behavior during a model dehydration experiment performed in liquid medium (osmotic stress). For both strains, we observed similar behavior after osmotic and desiccation stresses. In particular, the same lethal magnitude of dehydration and the same lethal kinetic effect were found for both dehydration methods. Thus, yeast survival after progressive air-drying was related to PM reorganization, suggesting the positive contribution of passive lateral rearrangements of the membrane components. This study also showed that the use of glycerol solutions is an efficient means to simulate air-drying desiccation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Microorganism dispersal allows yeasts to colonize new habitats and reach sites where resources favor growth, and enables their survival (Gorbushina and Broughton 2009; Gunde Cimerman et al. 2000; Quispel 1998). The yeast Saccharomyces cerevisiae occupies and flourishes in numerous habitats such as soil, plants, and rocks (Landry et al. 2006). Therefore, it can be found on surfaces constantly subjected to changes in atmospheric conditions with irregular fluctuations of temperature, humidity, and radiation (Gorbushina 2007). Among these stresses, the cell’s capacity to resist dehydration and rehydration has been essential for the efficient colonization of terrestrial environments (Tunnacliffe and Lapinski 2003). Moreover, understanding of cellular mechanisms occurring during air-drying desiccation is essential to improve industrial preservation techniques (e.g., ferment, probiotics) (Beker and Rapoport 1987; Meng et al. 2008).
During moderate hydric perturbations (final a w value >0.90), yeast cells actively maintain turgor pressure (Klipp et al. 2005). To adjust to such perturbations of the environment and to prevent the resulting loss of cytoplasmic water, microorganisms accumulate intracellular ions, such as K+, or organic solutes to reestablish the cell volume and, at the same time, preserve metabolic activities (Brown 1990; Santos and Da Costa 2002). For higher perturbations, no measurable metabolic activity can be detected anymore and only osmotic rules apply. The study of cell survival mechanisms that occur during desiccation, for instance on plants or soil, is scientifically complex to study and observe because of the change from a liquid to a solid state. Thus, many data connecting the structural and functional changes in cells during dehydration stress are available in liquid media with high concentrations of osmolytes (Dupont et al. 2010; Mille et al. 2002; Simonin et al. 2007; Turk et al. 2007). The main results obtained from these approaches show that cell death caused by extreme dehydration and subsequent rehydration is the result of a sequence of plasma membrane (PM) events (Crowe et al. 1984; Ragoonanan et al. 2008; Scherber et al. 2009; Simonin et al. 2007).
The extensive loss of water causes PM condensation and restructuration, with a lipid phase transition (Crowe et al. 1992) and also an increase of the cell surface-to-volume ratio (s/v) (Ragoonanan et al. 2008; Wolfe et al. 1986). After rapid osmotic perturbations, invagination and structural instability promote internalization of the PM into the cell cytoplasm and result in a decrease of the cell’s surface area. During rehydration and cell reexpansion, this lack of available surface area leads to PM rupture and further lysis of almost the whole cell population. By contrast, cell survival can be maintained after rehydration following more gentle and progressive osmotic perturbations. Recently, the mechanisms behind such cell survival have been identified as entailing lateral PM reorganization, which does not occur during abrupt perturbations (Dupont et al. 2010).
The occurrence of such mechanisms during yeast desiccation in air, which involves a transition from liquid to solid state, has not been demonstrated and experimental data are still lacking. During desiccation, the early stages correspond to the maintenance of cells in a concentrated liquid medium, as in a condition of osmotic stress. However, the subsequent events are more complex because of matrix solidification and the presence of air and particularly oxygen in direct contact with the yeast. Therefore, understanding how reductions in water availability influence the PM’s behavior during air-drying desiccation is of great interest in the field of microbial ecology and exobiology.
This work was intended to determine whether the yeast survival mechanisms extensively studied in liquid medium are similar to those that occur during air-drying desiccation. For this, we examined how water limitation and high osmolarity influenced S. cerevisiae survival, respectively. Thus, three amplitudes of dehydration, all below the water activity value of 0.90, and two rates of change (kinetics), one rapid and one slow, were used to mimic the various disturbances of the yeast natural environments. To measure the PM damage induced by both dehydration methods, a wild-type and a mutant strain with an osmotically fragile PM (erg6Δ) were used. Instead of ergosterol, the mutant accumulates molecular precursors and it has already been shown that this strain exhibits lower PM plasticity with a low resistance to deformation and environmental stress (Abe and Hiraki 2009; Rodriguez-Vargas et al. 2007). Moreover, the yeast PM contains two non-overlapping subcompartments called MCC (an ergosterol-rich membrane compartment of Can1, arginine/H+-symporter) and MCP (a membrane compartment occupied by Pma1, H+-ATPase protein) (Grossmann et al. 2007). Several proteins, such as Sur7 proteins, are integrated within the MCC microdomains. Under physiological conditions, when fused to green fluorescent protein (GFP), these proteins form microscopically visible patches in the plasma membrane (Malinska et al. 2004). A recent study showed that yeast survival to osmotic dehydration was accompanied by a lateral rearrangement of membrane components through modification of the distribution of specific proteins (Sur7-GFP) (Dupont et al. 2010). Therefore, to understand the role of the PM in the yeast’s air-drying response, we focused on the behavior of these proteins, which constitute permanent fluorescent PM markers during desiccation.
Materials and methods
Yeast strains and growth conditions
The S. cerevisiae strain BY4742 Wild Type (WT) (MATα his3Δ1 leu2Δ0 lys2Δ0 ura3Δ0) and erg6Δ mutant (MATα his3Δ1 leu2Δ0 lys2Δ0 YMR008c::kanMX4) (Euroscarf, Frankfurt, Germany) were used in this study. Cells were grown aerobically at 25 °C in 250-mL conical flasks containing 100 mL of Malt Wickerham (MW) modified medium. The MW medium contained 10 g glucose, 3 g pancreatic peptone, 3 g yeast extract, and 1.5 g NaH2PO4 (Sigma-Aldrich, Saint Quentin Fallavier, France) in 1 L of water–glycerol, and had an osmotic pressure of 1.4 MPa. This osmotic pressure has been recommended for the optimal growth of S. cerevisiae (Anand and Brown 1968). For growth, a subculture (1 mL) was transferred into a conical flask containing MW medium. The cultures were placed on a rotary shaker (New Brunswick Scientific, Edison, NY, USA) at 250 rpm for 24 h and allowed to grow to the early stationary phase. The final population was nearly 108 cells mL−1.
Yeast transformation
Plasmid amplification was carried out in the Escherichia coli DH5α host, grown at 37 °C in Luria–Bertani medium supplemented with 100 μg/mL ampicillin. The S. cerevisiae transformants were selected on Yeast Nitrogen Base casa plates (0.67 % Difco yeast nitrogen base without amino acids, 0.5 % NH4Cl, 1 % glucose, 0.1 % Difco Bacto Casamino acids, and 1.8 % agar). The strain BY4742 was transformed with the plasmid YIp211SUR7GFP after linearization using Eco52I (SUR7 gene was inserted as a HindIII–BamHI fragment) (Grossmann et al. 2007; Dupont et al. 2010). Strain transformation of S. cerevisiae with the linearized YIp211SUR7GFP plasmid was performed using the LiAc method (Ito et al. 1983).
Preparation of binary water–glycerol solutions of different osmotic pressures
Glycerol (Sigma-Aldrich) was the solute used for the hyperosmotic treatments in all experiments. The mass of solute to be added to 1,000 g of distilled water to obtain the desired water activity (a w) was calculated using the Norrish equation (Norrish 1966):
where X s is the molar fraction of the solute and K is the Norrish coefficient of the solute used to increase the osmotic pressure. For glycerol, K = 1.16 (Chirife and Fontan 1980). Osmotic pressure π (Pa) and water activity are related by the following equation:
where R is the universal gas constant (J. mol−1. K−1), T the temperature (K), and \( \overline {{V_{\text{w}}}} \) the partial molar volume of water (m3. mol−1). The quantities of glycerol were 51 g, 1,063 g, 3,986 g, and 6,654 g for 1,000 g of water to obtain final a w values of 0.99, 0.80, 0.45, and 0.30. respectively. Cell suspensions and rehydration solutions were kept in an air-conditioned room at 25 °C. The a w values of the solutions and the samples were checked using a dew-point osmometer (Decagon Devices Inc., Pullman, WA, USA).
Osmotic treatments
Three osmotic treatments were used: moderate (a w = 0.80), which is slightly higher than that allowing osmoregulation, and two levels of severe (a w = 0.45 and a w = 0.30). Culture samples (20 mL) were centrifuged (5 min, 2,200 × g), washed twice in the binary water–glycerol mixture (a w = 0.99), and the pellets were resuspended in 10 mL of water–glycerol mixture. Growth of cells in the presence of glycerol leads to its uptake into the cytoplasm. Therefore, during osmotic treatments, rapid cell water outflow allows the equilibration of internal and external osmotic pressures and changes in cell volume (Dupont et al. 2011). The level of volume contraction reached after this one-step response remains constant as long as cells are not permeabilized (for at least 1 h after hyperosmotic shock). Thus, by preventing glycerol uptake during glycerol perturbations, the amount of water transfer and the volume contraction reached after dehydration in liquid and gaseous environments were similar. For rapid perturbations (osmotic shock, 166 MPa. s−1), 1-mL aliquots of this suspension were placed in microsample tubes, which were then centrifuged (10 min, 5,100 × g) and the supernatant was removed. Hyperosmotic shock was induced by quickly introducing 1 mL of a binary water–glycerol solution (final a w values of 0.80, 0.45, and 0.30) to the pellets. For progressive perturbations (osmotic slope, 0.086 MPa. s−1), the external osmotic pressure was increased linearly by slowly adding pure glycerol to an agitated conical flask containing the initial cell suspension at a w = 0.99; the glycerol solution was injected using a syringe pump (KD Scientific, Boston, MA, USA). The cells were maintained under hyperosmotic conditions for 1 h before rehydration. For rapid rehydration, the hyperosmotic solution was removed from the microsample tube after centrifugation (10 min, 5,100 × g) and 1 mL of the binary water–glycerol solution (a w = 0.99) was added abruptly to the cell pellet. Progressive rehydration was performed using successive dilutions in solutions with increasing osmotic pressure to reach a w = 0.99.
Desiccation
The desiccation chamber consisted of an airtight plastic box with controlled relative humidity (RH). Saturated salt solutions were used to control the average RH inside the box (Greenspan 1977). Samples were placed on a rack above the salt solutions and the area of exposure was kept as large as possible in order to allow free diffusion of water. The chambers were maintained at a temperature of 25 °C and ventilated to increase mass transfer. Three saturated salt solutions (CH3COOK, K2CO3, or NaCl) were used to control the water activities to dry samples to obtain a w values of 0.30, 0.45, and 0.75. Saturation was assured by mixing a large excess (50 %) of each salt with deionized water. Culture samples (40 mL) were centrifuged for 5 min at 2,200 × g, washed twice with a NaCl solution (9 g/L), and the pellets were then resuspended in 10 mL of the binary NaCl solution (a w = 0.99). For the fast treatment (shock), the cultures were harvested by filtration using a Buchner funnel. A thin layer was then deposited on a glass microscope slide and placed in the drying chamber for 1 h. For the progressive treatment (slope), the cultures were also harvested by filtration but they were extruded using a syringe (ø200 μm) forming small filaments, as in industrial processes, and placed in the drying chambers for 1 h. For rapid rehydration, yeast cells were plunged directly into water. Progressive rehydration was performed by placing the glass microscope slides in specific chambers (RH = 100 %).
Measurement of yeast viability
Yeast viability was estimated in triplicate using the colony-forming unit (CFU) method. After osmotic dehydration or air-drying, fully rehydrated cells were serially diluted and the appropriate dilutions were plated on MW medium with 15 g. L−1 of agar. Colonies were counted after incubation for 36 h at 25 °C and recorded as CFU/mL (liquid medium) or CFU/mL per gram of hydrated paste (gaseous environment).
Microscopy
WT yeast strains expressing Sur7-GFP were observed using a Nikon Eclipse TE 2000 U microscope with a multispectral confocal head (D Eclipse C1). Excitation was performed at 488 nm, and the emission signal was measured between 500 and 545 nm. Images were acquired with a 100× (NA = 1.4) Plan Apo oil-immersion objective (Nikon) and collected using EZ-C1 software 3.50 (Nikon). Cells dehydrated in liquid medium were observed as described (Dupont et al. 2010). Dried yeast cells were harvested by filtration, and a thin layer was then deposited on a glass microscope slide and dried progressively (slope) or within a few milliseconds (shock). After dehydration, a drop of mineral oil was placed on the dried yeast, which was covered with a glass slide and subjected to microscopy.
Results
Impact of osmotic and desiccation stress on yeast viability
This study compared cell survival of two yeast strains dehydrated using two different techniques (osmotic dehydration and air-drying desiccation). The kinetics of the dehydration/rehydration phases and the dehydration amplitude were compared between the WT strain, which accumulates ergosterol in its PM, and the erg6Δ mutant, which is a membrane-sensitive strain.
Dehydration and rehydration kinetics
The kinetics of dehydration and rehydration in liquid and gaseous environments are presented in Fig. 1. Very similar values of cell water activity were observed between the two environments, during the dehydration and rehydration steps. The time delay during rehydration in a gaseous environment is due to the gain in moisture of the sample which depends on the transfer of water from the liquid to the air and from the air to the yeast. The results presented in Fig. 2a compare the viability of the WT strain after dehydration and rehydration cycles in liquid and gaseous environments. Progressive or rapid dehydration and progressive or rapid rehydration were tested, generating four possible combinations.
Dehydration and rehydration kinetics of Saccharomyces cerevisiae BY4742 from 1.4 to 166 MPa during air-drying and osmotic dehydration. Shock (broken lines) or slope (gray squares) air-dried cells and shock (broken lines) or slope (gray diamonds) osmotic dehydration in liquid medium. Temperature and relative humidity were checked with a thermocouple and a humidity data logger
Impact of different dehydration and rehydration kinetics on yeast viability. Survival of wild type strain (a) and erg6Δ mutant (b) were measured after osmotic treatments in water/glycerol solutions (dark gray bars) or air-drying (light gray bars). In both cases, viability was evaluated using the CFU method. Error bars correspond to the SD calculated from three repeated experiments. Asterisks denote statistical significance (*P < 0.05)
First, for osmotic dehydration and air-drying desiccation, the lowest survival rates were observed when the dehydration and rehydration steps were performed rapidly (0.35 % and 2.80 % survival, respectively, after osmotic and air-drying treatments), whereas the highest survival rates were observed with both techniques after progressive dehydration followed by progressive rehydration (78.7 % and 42.3 %, respectively). The two other treatments that combined a rapid and a progressive step showed intermediate cell survival rates: 19.2 % and 21.6 %, respectively, during osmotic and air-drying treatments after shock–slope cycles, and 42.6 % and 27.5 %, respectively, after slope–shock cycles.
The erg6Δ mutant yeast cells were subjected to the same four combinations (Fig. 2b). The viability measures of this strain after treatments in liquid and gaseous environments were, respectively, 3.65 % and 13.2 % after slope–slope, 0.26 % and 6.92 % after slope–shock, 0.16 % and 7.58 % after shock–slope, and 0.30 % and 4.27 % after shock–shock cycles. These results confirm that this strain has a fragile PM that makes it particularly sensitive to osmotic dehydration and air-drying desiccation.
Magnitude of dehydration
For both strains, cells were treated with a shock–shock cycle (water activity of 0.80, 0.45, or 0.30) for 60 min and then rehydrated. Figure 3a presents cell viability of the WT strain after dehydration in liquid medium or in drying chambers. With these treatments, higher viability ratios were observed at a w values of 0.80 with 82.5 % and 65.3 % in liquid and gaseous environments, respectively. The viability was, respectively, 12.5 % and 0.35 % after dehydration in liquid medium and 30.1 % and 2.80 % after air-drying treatments at a w values of 0.45 and 0.30. For the WT strain, dehydration in liquid medium or by air-drying led to similar behavior. Moreover, for both techniques, cell viability clearly decreased with decreasing water activity.
Impact of different dehydration and rehydration amplitudes (shock–shock cycle) on yeast viability. Survival of wild-type strain (a) and erg6Δ mutant (b) were measured after osmotic treatments in water/glycerol (dark gray bars) or air-drying (light gray bars) using the CFU method. Error bars correspond to the SD calculated from three repeated experiments. Asterisks denote statistical significance (*P < 0.05)
Figure 3b shows the cell viability of the erg6Δ mutant strain after the same dehydration–rehydration cycles. In this case, the threshold was lower, i.e., there was a stronger impact of the osmotic level on erg6Δ destruction than on the WT strain. For the three osmotic pressures tested, viability was between 3.29 % and 0.01 % after shock–shock cycles in liquid medium and between 3.50 % and 4.57 % in a gaseous environment. These results showed that dehydration, in liquid medium or by air-drying, led to low and very low survival rates, respectively. Thus, for erg6Δ, all cellular damage preventing cultivability and colonization occurred between a w values of 0.99 and 0.80. As previously observed with the kinetic effect, the level of dehydration appeared to have no impact on survival rates of this strain.
Impact of osmotic and desiccation stress on PM organization
In addition to yeast survival, the PM organization of the WT strain was studied in order to observe whether the mechanisms of cell death/cell survival were the same in liquid and gaseous environments. Thus, the kinetic effect on the lateral organization of the PM through Sur7-GFP localization was studied for each technique, after dehydration and rehydration. The impacts of drying kinetics and the magnitude of dehydration in liquid and gaseous environments were also tested with the transformed WT strain and the results obtained were similar to those obtained with the non-transformed strain (data not shown).
Lateral PM organization after dehydration
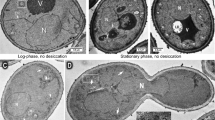
The results presented in Fig. 4a show the lateral organization of the PM after dehydration in liquid and gaseous environments. In the control cells, which were totally hydrated (a w = 0.99), Sur7-GFP proteins were concentrated in discrete patches (Fig. 4a, A). According to the literature, it is possible to observe around 15 GFP areas evenly distributed throughout the PM in yeasts (Dupont et al. 2010; Grossmann et al. 2007). After shock osmotic dehydration (Fig. 4a, C), the distribution of Sur7-GFP microdomains remained in distinct patches as in the control cells, whereas progressive dehydration in liquid medium (Fig. 4a, B) led to lateral redistribution of Sur7-GFP proteins. In the latter case, there were fewer but larger patches than in the control, suggesting PM reorganization.
Impact of dehydration (a) and rehydration (b) kinetics on Sur7-GFP microdomain distribution in the plasma membrane of transformed WT. Images (a) show control cells (A), cells in liquid medium after progressive (B) or rapid dehydration (C), and cells after progressive (D) or rapid air-drying (E). Images (b) show cells dehydrated in liquid medium after progressive (A) or rapid (B) rehydration and cells dehydrated in gaseous environment after progressive (C) or rapid rehydration (D). Median optical cross-sections of the cells are shown. Scale bar = 5 μm
After shock air-drying (Fig. 4a, E), the microdomains were still evenly distributed. These results were similar for the control and the yeast shock-dehydrated in liquid medium. After progressive dehydration in a gaseous environment (Fig. 4a, D), several distribution profiles were observed. Few cells showed evenly distributed microdomains, as seen in the controls. Nevertheless, most cells showed more diffuse Sur7 areas, fewer but larger than in the controls. However, these domains were slightly different, being less extensive from those obtained in liquid medium (Dupont et al. 2010). This might have arisen from the observation technique under oil. Thus, microdomains stayed concentrated in discrete patches after rapid dehydration with both dehydration techniques, whereas the patches were larger and fewer after progressive dehydration suggesting kinetics-dependent PM reorganization.
Lateral PM organization after rehydration
Figure 4b shows lateral organization of the PM after rehydration visualized by Sur7-GFP localization. The effect of rehydration was studied by observation of the cells after dehydration (final a w of 0.30) followed by rehydration (a w of 0.99). While Sur7-GFP was still mainly confined in the PM after rapid dehydration in liquid or gaseous environments, after a shock–shock cycle, it was localized in the cell cytoplasm and was not observed in the PM either in liquid medium (Fig. 4b, B) or through air-drying (Fig. 4b, D). For both techniques, rapid rehydration resulted in the presence of the protein throughout the cell. This observation could be related to membrane rupture and disorganization of the cytoplasm during rehydration. After slope–slope cycles (Fig. 4b, C), numerous patches of Sur7-GFP were distributed in the PM. After rehydration, the reorganization of the PM in cells dehydrated in liquid medium or by air-drying was similar to the profiles of the control cells.
Discussion
This study was conducted to help understand the role of PM in the yeast survival mechanisms in response to hydric perturbations. For this, the resistance of two yeasts (WT and a membrane sensitive strain) to dehydration in a liquid medium (osmotic stress) or a gaseous environment (osmotic and matric stress) was measured. The effect of the dehydration technique on the lateral organization of the PM through Sur7-GFP localization was then studied. Our main conclusion is that there was similar behavior in liquid and gaseous environments for both yeast strains. Similarities were also found with PM modifications after observation using confocal microscopy. Finally, our results emphasize the major role of kinetics for optimal preservation of yeast cells during air-drying desiccation.
Similar yeast survival during osmotic dehydration and air-drying desiccation
For the WT strain, viability depended on the lowering rate of water activity with both methods of dehydration (Fig. 2a). A comparison of yeast viability after shock–slope and slope–shock cycles showed the importance of controlling dehydration and rehydration kinetics to enhance yeast survival (Rodriguez-Porrata et al. 2008). As in liquid medium, the choice of optimal drying kinetics (progressive dehydration and rehydration) improved cell preservation. The largest difference between osmotic dehydration and air-drying desiccation (78.7 % and 42.3 %, respectively) was observed after the slope–slope cycle and can be explained by the more complex phenomena occurring during air-drying desiccation (Fig. 2a). Indeed, authors have reported in S. cerevisiae that drying leads to an increase in the intracellular level of malondialdehyde (MDA), a product of lipid peroxidation (Pereira et al. 2003). It has also recently been shown that cell oxidation levels increase significantly during air-drying desiccation (Garre et al. 2010). Therefore, the difference in yeast resistance between air-drying and osmotic dehydration might be associated with cell oxidation during the transition to an aerobic phase (França et al. 2007; Teixeira et al. 1996). Nevertheless, although the survival rates are slightly different, the general behavior of the strains is the same after osmotic dehydration and air-drying desiccation. Indeed, we can observe the same kinetic behavior (Fig. 2a) and the same decrease in viability with increasing osmotic magnitude (Fig. 3a), showing the importance of the mechanical component. Consequently, both effects (mechanical constraint and oxidative stress) exist but with different impacts on the yeast survival rate. Thus, oxidative stress has a minor effect compared to mechanical stress, certainly because the yeast S. cerevisiae can activate a broad response involving several defense mechanisms (enzymes, low molecular mass antioxidants) (Estruch 2000; Herdeiro et al. 2006; Landolfo et al. 2010).
After dehydration with moderate shock, cell survival remained relatively high in liquid and gaseous environments (82.5 % and 65.3 %, respectively), showing a limited kinetic effect at high water activity values (Fig. 3a). At an a w of 0.80, cell growth and metabolism stopped but the viability was mainly preserved, whereas dehydration attaining water activities of 0.45 or 0.30 resulted in important cell destruction in both liquid and gaseous environments. This may be because of the membrane lipid phase transition from liquid crystalline to gel during dehydration and from gel to liquid during rehydration, which occurs between a w values of 0.80 and 0.45 (Beker and Rapoport 1987). Authors showed in dry pollen that the transition from gel to liquid crystalline in membrane phospholipids that accompanies rehydration is the main cause of permeabilization and cell leakage (Crowe et al. 1989). A more recent study, conducted in liquid medium, showed that inappropriate kinetics or high levels of stress can cause cell death (Dupont et al. 2010). Indeed, rapid dehydration causes contraction of the yeast PM with progressive internalization leading to a reduction of the PM surface area during dehydration and lysis of the cells because of a lack of surface area during shock rehydration. Conversely, if the PM has time to reorganize, the yeast’s survival is better. This might be associated with the slowness of lyotropic chain-disordering phenomena (Milhaud 2004). Indeed, the redistribution of water molecules between the different parts of the sample could be a question of time. Moreover, the use of slow kinetics could allow the synthesis of protectants in the early stages of air-drying (Ratnakumar et al. 2011). However, the results obtained with the mutant strain suggest that the maintenance of PM integrity is the main parameter determining survival during air-drying desiccation. Indeed, the dehydration and rehydration kinetics had no impact on erg6Δ cell viability after osmotic and desiccation stress (Fig. 2b). The magnitude of dehydration also had no impact (Fig. 3b). It has already been shown that this strain, with a deleted ERG6 gene, is very sensitive to osmotic disturbances due to changes of PM mechanical properties (Dupont et al. 2011). Indeed, the nature of the membrane sterols governs the mechanical behavior of the PM during osmotic perturbation. These results also underline the importance of the mechanical constraint during air-drying desiccation. Thus, volume contraction resulting from the release of water—similar in liquid and gaseous environments—completely destabilizes the cells and determines their death at all the magnitudes and rates of dehydration tested (Simonin et al. 2007). Finally, the use of erg6Δ underlines how the yeast’s PM composition, in particular the sterol composition, is essential for the survival of S. cerevisiae at the solid–liquid interface, where hydration is often subjected to instability.
Implication of the PM in yeast drying tolerance
To understand desiccation survival mechanisms at the atmosphere–lithosphere interface, the lateral organization of the PM through Sur7-GFP localization was studied for both dehydration methods on the WT strain. A microscopy technique under oil was used to maintain the effects of air-drying desiccation during observation. A redistribution of Sur7-GFP was observed after progressive disturbance in liquid and gaseous environments (Fig. 4a, B, D). These observations were accompanied by significant yeast survival (Fig. 2a) and might be linked with PM rearrangement during the lipid phase transition. The mechanisms responsible for cell resistance were perhaps not exactly identical but in both cases, they resulted in yeast survival (Figs. 2a and 4b, A, C). Observation of the Sur7-GFP distribution after shock–shock cycles revealed similar behavior after dehydration (Fig. 4a, C, E) and after rehydration (Fig. 4b, B, D) in both liquid and gaseous environments. Patches were distributed as in the controls on quickly dried cells, revealing that these proteins were not internalized during the first phase of drying. After rehydration, cells showed a single cluster of Sur7-GFP, indicative of a cell bursting during rehydration, as obtained in liquid medium.
Whether in the case of cell death or survival, these results suggest similar mechanisms during desiccation to those already observed during dehydration in liquid medium, with similar PM behavior. During air-drying, the PM is a key structure in cell survival. Moreover, the cells had the same volume before dehydration and the yeast response was similar when the osmotic gradient was obtained in liquid or gaseous environments. Therefore, both dehydration strategies induced an accumulation of intracellular glycerol. Results from the WT and erg6Δ mutant strains showed that the mechanical component was the main stress experienced by the yeasts during hydric perturbations of their environment. For adapted microorganisms, such as the WT strain, slow kinetics seem to be essential in permitting partial disorganization of the PM, then survival and colonization.
In conclusion, for each of the two tested strains, survival rates were closed for air-drying desiccation and osmotic dehydration. There were similar patterns of PM disorganization and reorganization during progressive kinetics and preservation of the Sur7-GFP distribution and cell death after rapid kinetics. Therefore, the PM appears to be the main target of dehydration treatments and its slow lateral reorganization allows cell survival. Moreover, observation of dried cells under oil immersion optics is an interesting approach that could be used to stain other cellular structures and allow them to be studied in poorly hydrated conditions. The dehydration techniques used in this study, by addition of a solute (osmotic stress) and by elimination of water (osmotic and matric stress), were different in terms of molecular diffusion, matrix solidification, and cellular environment. Indeed, in the first case, cells were constantly surrounded by liquid and protected from the harmful effects of activated oxygen molecules. In the second case, as water flowed out, the different cellular structures were gradually exposed to air. Thus, if free radical production caused by hydric perturbation exists in liquid and gaseous environments (Petrovic 2006), yeasts are more exposed to oxidative stress associated with gaseous oxygen (singlet oxygen) during air-drying desiccation (Piette 1991). Although we observed a greater destructive effect in the case of air-drying desiccation, yeast survival, depending on the kinetics or amplitude, changed in parallel for both types of dehydration (glycerol and air-drying). Therefore, these results could be explained by the predominance of the mechanical dehydration component and modification of the PM surface area for both strains of yeast. This work allows proposing a hypothesis on the similar cellular behavior during dehydration in liquid medium and during air-drying desiccation. Thus, dehydration in liquid medium represents a convenient method for studying yeast survival mechanisms during air-drying caused by natural fluctuations in the level of hydration. We plan to test other yeast mutants more sensitive to oxidative stress.
References
Abe F, Hiraki T (2009) Mechanistic role of ergosterol in membrane rigidity and cycloheximide resistance in Saccharomyces cerevisiae. Biochim Biophys Acta 1788:743–752
Anand JC, Brown AD (1968) Growth rate patterns of the so-called osmophilic and non-osmophilic yeasts in solutions of polyethylene glycol. Microbiology 52:205
Beker M, Rapoport A (1987) Conservation of yeasts by dehydration. Adv Biochem Eng Biotechnol 35:127–171
Brown A (1990) Microbial water stress physiology. Principles and perspectives. Wiley, New York
Chirife J, Fontan CF (1980) A study of the water activity lowering behavior of polyethylene glycols in the intermediate moisture range. J Food Sci 45:1717–1719
Crowe JH, Crowe LM, Chapman D (1984) Preservation of membranes in anhydrobiotic organisms: the role of trehalose. Science 223:701–703
Crowe J, Hoekstra F, Crowe L (1989) Membrane phase transitions are responsible for imbibitional damage in dry pollen. Proc Natl Acad Sci U S A 86:520
Crowe J, Hoekstra F, Crowe L (1992) Anhydrobiosis. Annu Rev Physiol 54:579–599
Dupont S, Beney L, Ritt JF, Lherminier J, Gervais P (2010) Lateral reorganization of plasma membrane is involved in the yeast resistance to severe dehydration. Biochim Biophys Acta 1798:975–985
Dupont S, Beney L, Ferreira T, Gervais P (2011) Nature of sterols affects plasma membrane behavior and yeast survival during dehydration. Biochim Biophys Acta 1808:1520–1528
Estruch F (2000) Stress controlled transcription factors, stress induced genes and stress tolerance in budding yeast. FEMS Microbiol Rev 24:469–486
França MB, Panek AD, Eleutherio ECA (2007) Oxidative stress and its effects during dehydration. Comp Biochem Physiol A 146:621–631
Garre E, Raginel F, Palacios A, Julien A, Matallana E (2010) Oxidative stress responses and lipid peroxidation damage are induced during dehydration in the production of dry active wine yeasts. Int J Food Microbiol 136:295–303
Gorbushina AA (2007) Life on the rocks. Environ Microbiol 9:1613–1631
Gorbushina AA, Broughton WJ (2009) Microbiology of the atmosphere–rock interface: how biological interactions and physical stresses modulate a sophisticated microbial ecosystem. Annu Rev Microbiol 63:431–450
Greenspan L (1977) Humidity fixed points of binary saturated aqueous solutions. J Res Natl Bur Stand 81a:89–96
Grossmann G, Opekarová M, Malinsky J, Weig-Meckl I, Tanner W (2007) Membrane potential governs lateral segregation of plasma membrane proteins and lipids in yeast. EMBO J 26:1–8
Gunde Cimerman N, Zalar P, Hoog S, Plemenitaš A (2000) Hypersaline waters in salterns–natural ecological niches for halophilic black yeasts. FEMS Microbiol Ecol 32:235–240
Herdeiro R, Pereira M, Panek A, Eleutherio E (2006) Trehalose protects Saccharomyces cerevisiae from lipid peroxidation during oxidative stress. Biochim Biophys Acta 1760:340–346
Ito H, Fukuda Y, Murata K, Kimura A (1983) Transformation of intact yeast cells treated with alkali cations. J Bacteriol 153:163–168
Klipp E, Nordlander B, Krüger R, Gennemark P, Hohmann S (2005) Integrative model of the response of yeast to osmotic shock. Nat Biotechnol 23:975–982
Landolfo S, Zara G, Zara S, Budroni M, Ciani M, Mannazzu I (2010) Oleic acid and ergosterol supplementation mitigates oxidative stress in wine strains of Saccharomyces cerevisiae. Int J Food Microbiol 141:229–235
Landry CR, Townsend JP, Hartl DL, Cavalieri D (2006) Ecological and evolutionary genomics of Saccharomyces cerevisiae. Mol Ecol 15:575–591
Malinska K, Malinsky J, Opekarova M, Tanner W (2004) Distribution of Can1p into stable domains reflects lateral protein segregation within the plasma membrane of living S. cerevisiae cells. J Cell Sci 117:6031–6041
Meng XC, Stanton C, Fitzgerald GF, Daly C, Ross RP (2008) Anhydrobiotics: the challenges of drying probiotic cultures. Food Chem 106:1406–1416
Milhaud J (2004) New insights into water–phospholipid model membrane interactions. Biochim Biophys Acta 1663:19–51
Mille Y, Beney L, Gervais P (2002) Viability of Escherichia coli after combined osmotic and thermal treatment: a plasma membrane implication. Biochim Biophys Acta 1567:41–48
Norrish RS (1966) An equation for the activity coefficients and equilibrium relative humidities of water in confectionery syrups. Int J Food Sci Technol 1:25–39
Pereira E, Panek AD, Eleutherio ECA (2003) Protection against oxidation during dehydration of yeast. Cell Stress Chaperones 8:120–124
Petrovic U (2006) Role of oxidative stress in the extremely salt-tolerant yeast Hortaea werneckii. FEMS Yeast Res 6:816–822
Piette J (1991) New trends in photobiology: biological consequences associated with DNA oxidation mediated by singlet oxygen. J Photochem Photobiol B Biol 11:241–260
Quispel A (1998) Lourens GM Baas Becking (1895–1963). Inspirator for many (micro) biologists. Int Microbiol 1:69–72
Ragoonanan V, Malsam J, Bond DR, Aksan A (2008) Roles of membrane structure and phase transition on the hyperosmotic stress survival of Geobacter sulfurreducens. Biochim Biophys Acta 1778:2283–2290
Ratnakumar S, Hesketh A, Gkargkas K, Wilson M, Rash BM, Hayes A, Tunnacliffe A, Oliver SG (2011) Phenomic and transcriptomic analyses reveal that autophagy plays a major role in desiccation tolerance in Saccharomyces cerevisiae. Mol Biosyst 7:139–149
Rodriguez-Porrata B, Novo M, Guillamón J, Rozes N, Mas A, Otero RC (2008) Vitality enhancement of the rehydrated active dry wine yeast. Int J Food Microbiol 126:116–122
Rodriguez-Vargas S, Sanchez-Garcia A, Martinez-Rivas J, Prieto J, Randez-Gil F (2007) Fluidization of membrane lipids enhances the tolerance of Saccharomyces cerevisiae to freezing and salt stress. Appl Environ Microbiol 73:110–116
Santos H, Da Costa MS (2002) Compatible solutes of organisms that live in hot saline environments. Environ Microbiol 4:501–509
Scherber C, Schottel J, Aksan A (2009) Membrane phase behavior of Escherichia coli during desiccation, rehydration, and growth recovery. Biochim Biophys Acta 1788:2427–2435
Simonin H, Beney L, Gervais P (2007) Sequence of occurring damages in yeast plasma membrane during dehydration and rehydration: mechanisms of cell death. Biochim Biophys Acta 1768:1600–1610
Teixeira P, Castro H, Kirby R (1996) Evidence of membrane lipid oxidation of spray-dried Lactobacillus bulgaricus during storage. Lett Appl Microbiol 22:34–38
Tunnacliffe A, Lapinski J (2003) Resurrecting Van Leeuwenhoek’s rotifers: a reappraisal of the role of disaccharides in anhydrobiosis. Phil Trans R Soc Lond 358:1755–1771
Turk M, Abramovi Z, Plemenitaš A, Gunde Cimerman N (2007) Salt stress and plasma membrane fluidity in selected extremophilic yeasts and yeast like fungi. FEMS Yeast Res 7:550–557
Wolfe J, Dowgert MF, Steponkus PL (1986) Mechanical study of the deformation and rupture of the plasma membranes of protoplasts during osmotic expansions. J Membr Biol 93:63–74
Acknowledgments
We are grateful to W. Tanner and G. Grossmann (University of Regensburg, Cell Biology and Plant Physiology, Regensburg, Germany) for providing the plasmid YIp211SUR7GFP. We thank the personnel of the Plateau Technique “Imagerie Spectroscopique” IFR 92 (Université de Bourgogne, Dijon, France). This work was supported by French Ministry of Research, the Regional Council of Burgundy, Merck MF (Dijon, France), and the “FUI Probiotique” program supported by Vitagora (Dijon, France).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lemetais, G., Dupont, S., Beney, L. et al. Air-drying kinetics affect yeast membrane organization and survival. Appl Microbiol Biotechnol 96, 471–480 (2012). https://doi.org/10.1007/s00253-012-4014-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-4014-3