Abstract
Capsaicin is a versatile plant product which has been ascribed several health benefits and anti-inflammatory and analgesic properties. We have investigated the effect of capsaicin on the molecular stability, self-assembly, and fibril stability of type-I collagen. It was found that capsaicin suppresses collagen fibril formation, increases the stability of collagen fibers in tendons, and has no effect on the molecular stability of collagen. Turbidity assay data show that capsaicin does not promote disassembly of collagen fibrils. However, capsaicin moderately protects collagen fibrils from enzymatic degradation. Computational studies revealed the functions of the aromatic group and amide region of capsaicin in the collagen–capsaicin interaction. The results may have significant implications for capsaicin-based therapeutics that target excess collagen accumulation-linked pathology, for example thrombosis, fibrosis, and sclerosis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Capsaicin (8-methyl-N-vanillyl-6-nonenamide; Fig. 1) is a plant product widely used in foods and medicines. Topical application of capsaicin is known to have benefits against a variety of pain complications associated with such diseases as diabetic neuropathy and osteoarthritis (Tandan et al. 1992; Hempenstall et al. 2005). Other known health benefits of capsaicin involve its antihypertension (Hayes et al. 1981; Wang et al. 1984; Clozel et al. 1985), antitumorigenic (Huang et al. 2009; Wutka et al. 2014; Mori et al. 2006), and anti-inflammatory (Frydas et al. 2013) properties. Studies have proved that inactivation of capsaicin-sensitive nerve fibers reduces the pulmonary remodeling which occurs in collagen and elastic fibers and capsaicin treatment reduces the presence of collagen fibers in the vessels and lung tissues (Prado et al. 2011). It has also been reported that capsaicin is capable of penetrating collagen matrixes (Ko et al. 1998). Because capsaicin can penetrate the skin (Fang et al. 2001) by crossing the extra cellular matrix barrier, there is a possibility of interaction between collagen assemblies and capsaicin. To the best of our knowledge, direct studies of the capsaicin–collagen relationship are scarce in the literature. Hence, it is important to understand the effects of capsaicin on the biophysical properties of both the molecular and fibrillar forms of collagen.
Collagens are structural proteins in the extracellular matrix which are crucial in providing the mechanical framework of many tissues, for example skin, tendons, and blood vessels (Kielty and Grant 2002). In tissues the triple-helical collagen molecules occur as self-assembled supramolecular structures which are essential for proper functioning of tissues. Collagens, directly or indirectly, are linked to many pathological complications. Excess platelet aggregation mediated by fibrillar collagens can cause thrombosis, which leads to myocardial infarction and stroke (Farndale et al. 2004). Type VIII collagen is involved in atherosclerosis lesions and plaque formation (Plenz et al. 2003), and rupture of the plaque leads to formation of a platelet thrombus which causes sudden heart attack, if the coronary artery is the affected vessel, or a stroke, if the cerebral artery is involved. Blockage of cardiac arteries, after angioplasty, by accumulation of collagen in restenosis has been found to be a major challenge (Lafont et al. 1999). Recent research has verified the possibility of a link between accumulation of fibrillar collagen and tissue stiffness in hypertensive heart disease. Because accumulation of excess collagen is directly linked to severe disease, it is important to find inhibitors of collagen fibril formation. Any compound which is capable of inhibiting collagen fibril formation could, we believe, be useful in treating collagen-activated diseases.
In this study we attempted to determine whether capsaicin has any effect on the biophysical properties of collagen. We used a combination of biophysical and computational studies to aid understanding of the collagen–capsaicin interaction. Strong suppression of type-I collagen fibril formation occurs in the presence of capsaicin. Studies were also conducted to analyze the protective effect of capsaicin against the enzymatic degradation of collagen fibers in tendons. We also investigated the effect of capsaicin on the thermal stability of molecular collagen and on the shrinkage temperature of the tendons.
Materials and methods
Reagents
Type-I collagen was isolated by use of the method described below. Capsaicin, dihydrocapsaicin, and type-IA collagenase were obtained from Sigma–Aldrich Chemicals, USA. Other chemicals and buffers used for the study were of analytical grade from SRL (India) and Himedia (India).
Isolation of type-I collagen
Tendons were teased from the tails of 6-month-old male albino Wistar rats. The isolated tendons were washed thoroughly with saline then repeatedly with distilled water. The temperature of both saline and distilled water was maintained at 4 °C during washing. Acid-soluble type-I collagen was isolated from the tendons by use of the method of Chandrakasan et al. (1976). The purified type-I collagen was then dialyzed extensively against 50 mM acetic acid, to remove all traces of salts, then the pure collagen was lyophilized. An appropriate amount of lyophilized collagen powder was dissolved in 50 mM acetic acid solution for preparation of the collagen sample.
Collagen fibril formation and fibril disassembly
Collagen fibril formation, in the absence and presence of capsaicin (1:0 to 1:100 molar ratios of collagen to capsaicin), was assessed by monitoring absorbance at 313 nm by UV–visible spectrophotometry (Cary 100 and Cary 4000; Agilent Technologies). The concentration of collagen was maintained at 0.3 mg ml−1 (in PBS, pH 7.4), and measurements were recorded at 37 °C. The collagen sample (100 µl from 3 mg ml−1 stock) was incubated with capsaicin (10–100 µl from 2,000 µM stock) for ~5 min. PBS was added to the sample just before kinetic measurements. The reference solution for every experiment was prepared from water (0–90 µl), capsaicin, and buffer, without collagen, to normalize the background. For disassembly experiments, a suspension of mature collagen fibrils was prepared and its optical density at 313 nm was monitored in the presence and absence of capsaicin. The same amount of capsaicin was added to the reference cell. All the experiments were repeated at least twice. Capsaicin stock solution was prepared in 50 % ethanol. Control collagen fibril formation was conducted under similar conditions without capsaicin.
Circular dichroism
A Jasco CD spectropolarimeter (model J815-150L) was used to assess the effect of capsaicin on the conformational properties of type-I collagen. CD scans of collagen samples were obtained in the presence of different concentrations of capsaicin (1:10, 1:25, 1:50 and 1:100 molar ratios of collagen to capsaicin). The concentration of the collagen solution was maintained at 0.5 mg ml−1. Thermal unfolding experiments were performed by monitoring the CD signal at 222 nm as the temperature of the sample was increased from 20 to 60 °C at 2 °C min−1.
Study of the enzymatic degradation of collagen tendons
Enzymatic degradation, by collagenase, of native and capsaicin-treated rat tail tendons (RTT) was studied by estimating the amount of hydroxyproline released from the sample by hydrolysis. RTT fibers were treated with capsaicin (1–100 μM) for 24 h at 27 °C before addition of collagenase. Native RTT and capsaicin-treated RTT were treated with type-IA collagenase in 0.04 M CaCl2 solution (0.05 M tris–HCl buffer at pH 7.2). The ratio of collagen to enzyme was maintained at 50:1 and enzyme-treated samples were incubated at 37 °C for ~96 h. Degradation of RTT was monitored by measuring soluble hydroxyproline released from insoluble collagen (Ryan and Woessner 1971). Samples of the supernatant (750 μl) were withdrawn after centrifugation of the sample at 10,000 rpm for 10 min. The collagenase hydrolysate was hydrolyzed with 6 M HCl at 120 °C for 16 h in sealed hydrolysis tubes. To remove excess acid, hydrolysates were evaporated in a porcelain dish by use of a water bath. The acid-free residue was diluted to known volume and the amount (%) of hydroxyproline was determined by use of Woessner’s method (1961).
Hydroxyproline is an amino acid unique to collagen and acts as a useful marker for identification of collagen in the presence of non-collagenous proteins. Woessner’s method for determination of hydroxyproline involves oxidation of hydroxyproline to pyrrole-2-carboxylic acid. This forms a complex with p-dimethylaminobenzaldehyde for which maximum absorbance is at 557 nm.
Measurement of hydrothermal shrinkage of tendons
A micro-shrinkage tester technique was used to measure the shrinkage temperature of native RTT fibers in the presence and absence of capsaicin. After incubation, a small strip of RTT fiber was placed on a saline-filled grooved microscope slide. The slide was mounted on a heating stage and the temperature of the collagen fibers in the saline was increased at a constant rate of 2 °C min−1 until the onset of shrinkage was observed. The temperature at which the fibers had shrunk to one-third of their original size was taken as the shrinkage temperature in this study.
Studies of docking of capsaicin with peptides in triple-helical collagen molecules
X-ray crystal structures of selected collagen model peptides were obtained from the Protein Data Bank (PDB; PDB IDs: 1CAG, 1QSU, 2DRT, 1Q7D) (Bella et al. 1994). The structures were prepared by use of the protein-preparation wizard of the software Discovery Studio 4.0. Pre-processing and protonation were performed, by use of CHARMm force fields, to balance free valences of molecules (Brooks et al. 2009). The docking sites were chosen around specific triplets for each of these collagen peptides: “GPO” for 1CAG, “EKG” for 1QSU, “GLO” for 2DRT, and “GFOGER” for 1Q7D. The 3D coordinate profile of capsaicin was downloaded in SDF format from PubChem (CID 1548943) (Bolton et al. 2008) and prepared by use of the general purpose wizard with the default settings. Preparation also nullified formation of any isomers or tautomers by the molecule. Docking between collagen and capsaicin was performed by use of the Cdocker procedure, which is based on CHARMm.
Molecules structurally similar to capsaicin were identified from the ZINC database (Irwin and Shoichet 2005) by using a similarity score of 50 %. Of the best matches, 10 molecules (ZINC7911905, ZINC7911909, ZINC35317807, ZINC38032190, ZINC44869451, ZINC44869455, ZINC48325216, ZINC50950504, ZINC60968773, and ZINC78400413) were selected as decoys such that they, while having high structural similarity to capsaicin, were dissimilar with regard to the substructures reported to be involved in interactions with the molecule. These decoys were subjected to docking with the same procedure that was used for capsaicin. All docking studies were performed with Discovery Studio 4.0 on a 10 core Dell Precision 5610 Workstation.
Results and discussion
Effect of capsaicin on formation of fibrils of type-I collagen
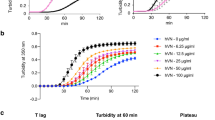
The process of collagen fibril formation was monitored by recording the optical density (absorbance at 313 nm) of collagen (0.3 mg ml−1) in PBS buffer at 37 °C (Mehta et al. 2014). In the absence of capsaicin a typical turbidity curve consisting of a lag phase, a growth phase, and a plateau phase was observed (Fig. 2a, black line); such a curve confirms self-assembly of soluble triple-helical collagen molecules into higher-order fibrils. Gradual suppression of collagen fibril formation was observed when increasing concentrations of capsaicin were added to the sample (Fig. 2a). The nature of the turbidity data, shown in Fig. 2a, suggests that the lag time of the self-assembly process is not much delayed in the presence of capsaicin; rather, the extent fibril formation if substantially reduced. At 1:100 molar ratio of collagen to capsaicin, we observed almost 90 % reduction in the amount of fibrils formed with a slight delay in the lag time. Because the lag time is not much affected, it is possible the suppression effect is achieved during the growth phase in which inter-fibrillar association occurs. It is also possible that interaction of capsaicin with the fibrillar form of collagen is more favorable than capsaicin–molecular collagen interaction. Another reason for this inhibition effect could be promotion of fibril disassembly by capsaicin. To clarify the effect of capsaicin on fibril disassembly, we added capsaicin to a suspension of mature collagen fibrils and monitored the change in optical density of the sample as a function of time (Fig. 2b). The results, shown in Fig. 2b clearly indicate that capsaicin does not promote disassembly of collagen fibrils (Fig. 2b, filled circles).
Effect of capsaicin on type-I collagen fibril formation and fibril disassembly. a The kinetics of formation of collagen fibrils in the presence and absence of capsaicin were studied by monitoring optical density at 313 nm. Molar ratios of collagen to capsaicin in different samples were: 1 1:0, 2 1:10, 3 1:25, 4 1:50, and 5 1:100. The concentration of collagen was constant at ~0.3 mg ml−1 in all the samples and the measurements were recorded in PBS buffer (pH 7.4) at 37 °C. b Effect of capsaicin on disassembly of collagen fibrils. The molar ratio of the collagen to capsaicin was 1:100 and measurements were recorded at 25 °C
Effect of capsaicin on enzymatic degradation of fibrillar collagen
To determine the binding properties of capsaicin with collagen fibers, we conducted collagenolytic degradation of capsaicin-treated rat tail tendons. The amount of hydroxyproline released from collagenase-treated RTT was measured to quantify the degradation of collagen fibrils in the presence of capsaicin. Approximately 99 % of the native collagen fibers were degraded in the absence of capsaicin (Fig. 3). Capsaicin-treated RTT were, however, resistant to collagenolytic hydrolysis (Fig. 3a). As shown in the same figure, approximately 20 % less hydrolysis was observed in the presence of 100 μM capsaicin. Because capsaicin-treated collagen fibers were thoroughly washed before exposure to collagenase, interaction between collagenase and capsaicin seems unlikely. Hence, this result suggests that capsaicin may have the ability to bind to collagen fibers, and this binding may interfere with the catalytic action of the collagenase.
a Protective effect of capsaicin against enzymatic degradation of rat tail tendons (RTT). Percentage degradation of collagen tendons by collagenase enzyme, by use of the procedure discussed in the “Materials and methods” section, was determined in the presence and absence of capsaicin (10, 25, 50, and 100 μM). The collagen-to-collagenase ratio was maintained at 50:1 and the samples were incubated at 37 °C for 96 h. All measurements were repeated at least twice. b Correlation between the increase in shrinkage temperature and percentage protection against collagenase. R 2 for the linear regression fit is 0.76
Hydrothermal shrinkage of tendons in the presence of capsaicin
For further clarification of the capsaicin–tendon interaction we measured the shrinkage temperature of RTT in the presence of capsaicin. Measurement of the hydrothermal shrinkage temperature of RTT fibers, i.e. the temperature at which the collagen tendon shrinks to one-third of its original length when heated in an aqueous medium, furnishes direct evidence of the thermal stability of ordered collagen fibers. A variety of factors can affect the denaturation temperature of collagen fibers (Usha and Ramasami 2000; Sionkowska 2000). We measured the shrinkage temperature of native tendons and capsaicin-treated tendons (as discussed in the “Materials and methods” section); the results are summarized in Table 1. For native collagen fibers the shrinkage temperature was ~56 °C, which is higher than the denaturation temperature of collagen triple-helical molecules (~41 °C). We observed an increase in the shrinkage temperature for collagen fibers pretreated with capsaicin (Table 1). The shrinkage temperature of RTT collagen fibers treated with 100 μM capsaicin was 60.5 °C. This increase suggests a possible interaction between capsaicin and collagen fibers in the tendon, an assumption consistent with the results obtained from enzymatic degradation experiments of collagen fibers. A correlation between the increase in shrinkage temperature and protection against collagenase is clearly evident from the plot shown in Fig. 3b.
Effect of capsaicin on the conformational stability of the collagen triple-helix
Because the shrinkage temperature of collagen tendons was found to increase in the presence of capsaicin (Table 1), as the next step we examined the effect of capsaicin on the thermal stability of molecular collagen. The molecular structure of collagen is a triple helix and has a characteristic CD signal at 222 nm. We acquired the CD spectra of collagen in the presence and absence of capsaicin (Fig. 4a). In the presence of capsaicin, collagen molecules retained their inherent triple-helical conformation (a positive peak at ~222 nm), and we noticed an increase in the intensity of the peak at 222 nm in the presence of capsaicin. We then performed thermal unfolding of collagen triple-helical molecules and monitored the change in the CD signal at 222 nm. The thermal unfolding curves suggested a slight increase of T m for collagen in the presence of capsaicin (Fig. 4b). This result suggests that in the presence of capsaicin the conformational stability of the triple-helical collagen molecule remains almost unaltered whereas the thermal stability of collagen fibers is substantially increased (Table 1).
Circular dichroism studies of the collagen–capsaicin interaction. a CD profiles of the collagen sample in the presence and absence of capsaicin. b Thermal unfolding of type-I collagen in the presence and absence of capsaicin. The change in the CD signal at 222 nm for the triple-helix was monitored as the temperature of the sample was increased from 20 to 60 °C at a rate of 1 °C min−1. All the CD scans were averages from three accumulations
Computational studies of docking of capsaicin with peptides in triple-helical collagen molecules
Because these experimental results indicated possible interaction of capsaicin and collagen, we conducted computational docking studies to gain further insight. Capsaicin was docked around specific triplets in each of the collagen peptides investigated (Supplementary Figures S2–S5). Results from 1CAG and capsaicin binding studies indicate that capsaicin binds with Gly–Pro–Hyp triplets (Fig. 5). Capsaicin formed three hydrogen bonds (between HA of Pro37 and O2 of capsaicin, =O of Gly36 and H45 of capsaicin, and =O of Gly36 and H46 of capsaicin) and participated in a mixed pi/alkyl hydrophobic interaction (between the electron clouds of the benzene subunit of capsaicin and the cyclopentane subunit of Pro37). The Cdocker energy score was −4.977 and interaction energy score was −17.404, values indicative of a stable interaction between capsaicin and collagen. To confirm the effect of functional groups of capsaicin identified as being involved in interactions with Gly–Pro–Hyp triplets, 10 capsaicin decoys (listed in the “Materials and methods” section) were identified and subjected to docking studies by use of the same procedure. Except for one molecule (ZINC35317807; Fig. S1b), none interacted with Gly–Pro–Hyp regions.
Docking of capsaicin with a Gly–Pro–Hyp triplet of the triple-helical collagen like peptide (PDB ID: 1CAG). a Capsaicin docked with the collagen triple helix, the triple-helix surface of which is rendered as its electron-density map. b Interaction of capsaicin with the triple helix. Hydrogen bonds are shown with gray dotted lines, whereas the electrostatic interaction is shown with a light purple dashed line. The docking studies were performed by use of Discovery Studio 4.0 (as discussed in the “Materials and methods” section)
We also conducted studies of docking of capsaicin with selected triple-helical model peptides containing sequences that are found in type-I collagen. The results have been summarized in supplementary Figs. S2–S5. As observed from the interactions, capsaicin could potentially bind to the “GFOGER”, “EKG”, and “GLO” sequences within the triple-helical conformation.
A possible mechanism of the capsaicin–collagen interaction
Capsaicin can be regarded as a molecule consisting of three segments: an aromatic component, an amide bond, and a hydrophobic side chain (Fig. 1). The aromatic ring of capsaicin is known to be important to its binding properties, because of its ability to act as both hydrogen bond donor and acceptor (Katritzky et al. 2000). It is also possible that amide hydrogen and carbonyl oxygen participate in H-bonding interactions. A hydrophobic effect of the hydrocarbon chain of capsaicin is also possible. With such properties, the capsaicin molecule seems capable of interfering with both hydrophobic and hydrophilic interactions. Aromatic residues are of major importance in the process of self-association of collagen triple-helical peptides into higher-order structures (Kar et al. 2009; Cejas et al. 2008). Inhibition of self-assembly of collagen model peptides has also been observed in the presence of aromatic residues (Kar et al. 2009). Synthetic peptides with aromatic-rich sequences in the telopeptide regions of the collagen sequence have been found to inhibit self-assembly of type-I collagen in an in vitro system (Prockop and Fertala 1998). Our computational studies indicated that the aromatic moiety and amide region of capsaicin are involved in critical interactions that stabilize the complex. Hence the aromatic part of the capsaicin and its amide region seem crucial for the collagen–capsaicin interaction. To understand the importance of the hydrophobic region of capsaicin, we also investigated the effect of dihydrocapsaicin on collagen fibril formation at 37 °C in PBS. We observed no effect of dihydrocapsaicin on the onset of collagen fibril formation (Supplementary Fig. 6). It seems that the rigidity of the hydrophobic chain of capsaicin facilitates collagen–capsaicin interactions.
In this study we observed strong inhibition of collagen fibril formation when the concentration of capsaicin was increased to ~100 fold higher than the concentration of collagen. The type-I collagen triple helix is a hetero trimer which contains two α1 chains and one α2 chain. Each α chain contains approximately ~1,000 amino acids. It is possible that the whole sequence of both the α1 and α2 chains may have multiple sites facilitating binding of capsaicin. Computational docking studies suggest that Gly and Pro participate in collagen–capsaicin interactions. Further studies of docking of capsaicin with other collagen peptides (supplementary data, Figs. S2–S5) also indicated its interaction with the “GFOGER”, “EKG”, and “GLO” regions. Sequence analysis of type-I rat collagen showed that the α1 chain contains 44 Gly–Pro–Hyp regions and 127 Gly–Pro sites. Similarly, the α2 chain contains 29 Gly–Pro–Hyp regions and 116 Gly–Pro sites (supplementary Fig. S7). This could be one reason for the requirement of such a high molar ratio of capsaicin to collagen (100:1) to prevent collagen fibril formation.
The molecular structure of collagen has a characteristic triple-helical conformation consisting of three polyproline II-like chains supercoiled around a common axis as a result of interchain hydrogen bonding (Ramachandran and Kartha 1955; Rich and Crick 1961; Brodsky and Persikov 2005). Each polypeptide chain has a unique (Gly–Xaa–Yaa) n sequence pattern in which almost every third residue is glycine. It is believed that non-specific interactions which involve hydration and intermolecular H-bonding are the force driving initiation of self-assembly of triple-helical collagen molecules (Kar et al. 2006; Leikin et al. 1995, 1997). Sugars and polyols are known to inhibit fibrillogenesis of type-I collagen by disrupting hydrogen-bonded water bridges between the helices (Kuznetsova et al. 1998). Sugars are also known to be able to inhibit self-association of collagen triple-helical peptides (Kar et al. 2006). Considering all these factors, we believe that both hydrophobic and hydrophilic interactions mediated by capsaicin are directly linked to the mechanism of suppression of collagen fibril formation.
Inhibition of fibril formation by capsaicin suggests that capsaicin may interfere with aggregation of collagen molecules as a result of its interaction with monomolecular collagen. The interactions that occur between capsaicin and collagen in the monomolecular and fibrillar forms might not have the same effect. Although capsaicin does not alter the thermal stability of triple-helical collagen molecules, it increases the shrinkage temperature of tendons and protects them from enzymatic degradation. Collagen forms a highly ordered, quarter-staggered arrangement in which collagen triple helical molecules are fixed at defined dimensions in 3D space. It is, therefore, possible that capsaicin can participate in multipoint interactions between collagen molecules (in the fibrillar form), which may enhance the stability of collagen fibers.
Conclusions
Accumulation of collagen is one of the main causes for the onset of lethal diseases such as fibrosis and atherosclerosis. A straightforward approach targeting collagen fibril formation could therefore be critical for treatment of such diseases. This study revealed the potential of capsaicin to suppress collagen fibril formation. This unique property of capsaicin may have potential in the development of drugs for treatment of diseases linked with excess collagen fibrillogenesis.
Abbreviations
- CD:
-
Circular dichroism
- RTT:
-
Rat tail tendon
- T m :
-
Transition temperature
References
Bella J, Eaton M, Brodsky B, Berman HM (1994) Crystal and molecular structure of a collagen-like peptide at 1.9 A resolution. Science 266:75–81
Bolton EE, Wang Y, Thiessen PA, Bryant SH (2008) Chapter 12 PubChem: integrated platform of small molecules and biological activities. Annu Rep Comput Chem 4:217–241
Brodsky B, Persikov AV (2005) Molecular structure of the collagen triple helix. Adv Protein Chem 70:301–339
Brooks BR, Brooks CL, Mackerell AD, Nilsson L, Petrella RJ, Roux B et al (2009) CHARMM: the biomolecular simulation program. J Comput Chem 30:1545–1614
Cejas MA, Kinney WA, Chen C, Vinter JG, Almond HR Jr, Balss KM, Maryanoff CA, Schmidt U, Breslav M, Mahan A, Lacy E, Maryanoff BE (2008) Thrombogenic collagenmimetic peptides: self-assembly of triple helix-based fibrils driven by hydrophobic interactions. Proc Natl Acad Sci USA 105:8513–8518
Chandrakasan G, Torchia DA, Piez KA (1976) Preparation of intact monomeric collagen from rat tail tendon and skin and the structure of the non-helical ends in solution. J Biol Chem 251(19):6062–6067
Clozel JP, Roberts AM, Hoffman JI, Coleridge HM, Coleridge JC (1985) Vagal chemoreflex coronary vasodilation evoked by stimulating pulmonary C-fibers in dogs. Circ Res 57(3):450–460
Fang JY, Fang CL, Hong CT, Chen HY, Lin TY, Wei HM (2001) Capsaicin and nonivamide as novel skin permeation enhancers for indomethacin. Eur J Pharm Sci 12(3):195–203
Farndale RW, Sixma JJ, Barnes MJ, de Groot PG (2004) The role of collagen in thrombosis and hemostasis. J Thromb Haemost 2(4):561–573
Frydas S, Varvara G, Murmura G, Saggini A, Caraffa A, Antinolfi P, Tete’ S, Tripodi D, Conti F, Cianchetti E, Toniato E, Rosati M, Speranza L, Pantalone A, Saggini R, Di Tommaso LM, Theoharides TC, Conti P, Pandolfi F (2013) Impact of capsaicin on mast cell inflammation. Int J Immunopathol Pharmacol 26(3):597–600
Hayes AG, Skingle M, Tyers MB (1981) Effects of single doses of capsaicin on nociceptive thresholds in the rodent. Neuropharmacology 20(5):505–511
Hempenstall K, Nurmikko TJ, Johnson RW, A’Hern RP, Rice AS (2005) Analgesic therapy in postherpetic neuralgia: a quantitative systematic review. PLoS Med 2(7):e164
Huang SP, Chen JC, Wu CC, Chen CT, Tang NY, Ho YT et al (2009) Capsaicin-induced apoptosis in human hepatoma HepG2 cells. Anticancer Res 29(1):165–174
Irwin JJ, Shoichet BK (2005) ZINC––a free database of commercially available compounds for virtual screening. J Chem Inf Model 45:177–182
Kar K, Amin P, Bryan MA, Persikov AV, Mohs A, Wang Y, Brodsky B (2006) Self-association of collagen triple helix peptides into higher order structures. J Biol Chem 281:33283–33290
Kar K, Ibrar S, Nanda V, Getz TM, Kunapuli SP, Brodsky B (2009) Aromatic interactions promote self-association of collagen triple-helical peptides to higher-order structures. Biochemistry 48:7959–7968
Katritzky AR, Xu YJ, Vakulenko AV, Wilcox AL, Bley KR (2000) Model compounds of caged capsaicin: design, synthesis, and photoreactivity. J Org Chem 68:9100–9104
Kielty CM, Grant ME (2002) The collagen family: structure assembly and organization in the extracellular matrix. Wiley, New York, pp 159–221
Ko F, Diaz M, Smith P, Emerson E, Kim YJ, Krizek TJ, Robson MC (1998) Toxic effects of capsaicin on keratinocytes and fibroblasts. J Burn Care Rehabil 19(5):409–413
Kuznetsova N, Chi SL, Leikin S (1998) Sugars and polyols inhibit fibrillogenesis of type-I collagen by disrupting hydrogen-bonded water bridges between the helices. Biochemistry 37(34):11888–11895
Lafont A, Durand E, Samuel JL, Besse B, Addad F, Lévy BI, Desnos M, Guérot C, Boulanger CM (1999) Endothelial dysfunction and collagen accumulation: two independent factors for restenosis and constrictive remodeling after experimental angioplasty. Circulation 100(10):1109–1115
Leikin S, Rau DC, Parsegian VA (1995) Temperature-favoured assembly of collagen is driven by hydrophilic not hydrophobic interactions. Nat Struct Biol 2(3):205–210
Leikin S, Parsegian VA, Yang W, Walrafen GE (1997) Raman spectral evidence for hydration forces between collagen triple helices. Proc Natl Acad Sci 94(21):11312–11317
Mehta A, Rao JR, Fathima NN (2014) Effect of ionic liquids on different hierarchical order of type-I collagen. Colloids Surf B 117:376–382
Mori A, Lehmann S, O’Kelly J, Kumagai T, Desmond JC, Pervan M, McBride WH, Kizaki M, Koeffler HP (2006) Capsaicin, a component of red peppers, inhibits the growth of androgen-independent, p53 mutant prostate cancer cells. Cancer Res 66(6):3222–3229
Plenz GA, Deng MC, Robenek H, Völker W (2003) Vascular collagens: spotlight on the role of type VIII collagen in atherogenesis. Atherosclerosis 1–11
Prado CM, da Rocha GZ, Leick-Maldonado EA, Starling CM, Capelozzi VL, Martins MA, Tibério IF (2011) Inactivation of capsaicin-sensitive nerves reduces pulmonary remodeling in guinea pigs with chronic allergic pulmonary inflammation. Braz J Med Biol Res 44(2):130–139
Prockop DJ, Fertala A (1998) Inhibition of the self-assembly of collagen I into fibrils with synthetic peptides. Demonstration that assembly is driven by specific binding sites on the monomers. J Biol Chem 273:15598–15604
Ramachandran GN, Kartha G (1955) Structure of collagen. Nature 176:593–595
Rich A, Crick FH (1961) The molecular structure of collagen. J Mol Biol 3:483–506
Ryan JN, Woessner JF (1971) Mammalian collagenase: direct demonstration in homogenates of involuting rat uterus. Biochem Biophys Res Commun 44(1):144–149
Sionkowska A (2000) Modification of collagen films by ultraviolet irradiation. Polym Degrad Stab 68(2):147–151
Tandan R, Lewis GA, Krusinski PB, Badger GB, Fries TJ (1992) Topical capsaicin in painful diabetic neuropathy. Controlled study with long-term follow-up. Diabetes Care 15:8–14
Usha R, Ramasami T (2009) Effect of crosslinking agents (basic chromium sulfate and formaldehyde) on the thermal and thermomechanical stability of rat tail tendon collagen fibre. Thermochim Acta 59–66
Wang JP, Hsu MF, Teng CM (1984) Antiplatelet activity of capsaicin. Thromb Res 36:497–507
Woessner JF (1961) The determination of hydroxyproline in tissue and protein samples containing small proportions of this imino acid. Arch Biochem Biophys 93:440–447
Wutka A, Palagani V, Barat S, Chen X, El Khatib M, Götze J, Belahmer H, Zender S, Bozko P, Malek NP, Plentz RR (2014) Capsaicin treatment attenuates cholangiocarcinoma carcinogenesis. PLoS One 9(4):e9560
Acknowledgments
We thank the Indian Institute of Technology, Jodhpur, for research facilities. We are grateful to Central Leather Research Institute, Chennai, for laboratory support with some of the biophysical studies. Authors SP and BM are grateful for CSIR-YSA grants from CSIR (CSIR-CLRI Communication number 1122). Authors KK and GB thank the Indian Institute of Technology, Jodhpur, for the Seed Grant, and author KK is grateful for BRNS [No.37(1)/14/38/2014-BRNS/1357] funding support.
Author information
Authors and Affiliations
Corresponding authors
Additional information
S. Perumal and K. Dubey contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Perumal, S., Dubey, K., Badhwar, R. et al. Capsaicin inhibits collagen fibril formation and increases the stability of collagen fibers. Eur Biophys J 44, 69–76 (2015). https://doi.org/10.1007/s00249-014-1002-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-014-1002-9