Abstract
Background
Reversible lesions of the corpus callosum with initial restricted diffusion on diffusion-weighted imaging (DWI) are rare and mainly described in the south Asiatic population.
Objective
The purpose of this study was to describe the clinical presentation, imaging findings, prognosis and etiology of transient restricted diffusion lesions of the corpus callosum in a series of Caucasian children.
Materials and methods
Seven children presenting with a transient restricted DWI lesion of the corpus callosum were included. Their clinical presentations and paraclinical examinations were investigated in addition to their MRI findings during the acute phase and at follow-up.
Results
Five patients initially presenting with prodromal flu-like symptoms were diagnosed with mild encephalopathy with reversible corpus callosum lesions, three of which were due to the influenza virus. For two patients (twins) with a stroke-like presentation and without febrile illness, a central nervous system manifestation of X-linked Charcot-Marie-Tooth disease with connexin 32 mutation was diagnosed. All patients had a good clinical prognosis without clinical sequelae or residual MRI lesion for all patients at follow-up.
Conclusion
A transient lesion of the corpus callosum with restricted diffusion should prompt the radiologist to suggest an infectious trigger in children. The prognosis of these patients was good with normalization of clinical symptoms and MRI without any specific treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Several disorders can affect the corpus callosum in children with a wide prognostic spectrum according to the etiology and associated encephalic lesions [1,2,3]. Among the different etiologies, some have an initial diffusion restriction at the acute phase but a reversible course with complete and rapid fading as well as normalization of clinical symptoms in the following weeks or months. If most cases reported have been related to potential infectious triggers, the etiologies and physiopathologies have not been completely elucidated. Reversible splenial lesion syndrome (RESLES) [4, 5] is a recent radiologic syndrome that encompasses all etiologies associated with transient lesional diffusion restriction. In children, most of the cases have been reported in East Asia during febrile illness. This entity was previously known as a clinical-radiologic syndrome mild encephalopathy/encephalitis with reversible splenial lesion (MERS) [5,6,7] including encephalopathy signs lasting more than 12 h. According to the extension of the lesion on magnetic resonance imaging (MRI), two types of MERS have been described: MERS type 1 [8, 9] corresponds to an isolated focal lesion of the splenium of the corpus callosum whereas MERS type 2 [10, 11] includes an anterior extension up to the entire corpus callosum and/or a symmetrical bilateral extension to the contiguous deep white matter of the centrum semiovale. However, some authors have reported many cases with the same radiologic features and prognosis as MERS, except for the encephalopathy signs. Thus, a larger entity termed reversible splenium lesion with febrile illness (RESLEF) was recently suggested [12].
The purpose of this study was to report and describe the clinical, biological and MRI findings, prognosis and different etiologies in a series of Caucasian children presenting with a transient diffusion-restricted lesion of the corpus callosum.
Materials and methods
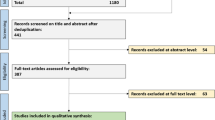
We retrospectively included all children with a transient lesion of the corpus callosum with relative diffusion restriction who underwent an MRI between 2007 and 2014 in our pediatric radiology department. All patients were initially admitted to the pediatric emergency department of our institution. A retrospective analysis of clinical, biological and imaging features was conducted. Formal research ethics approval was waived for this retrospective study.
The following clinical data were collected from medical files: initial clinical presentation and follow-up with particular attention to neurological signs such as seizure, neurological deficits or encephalopathy symptoms including consciousness or behavioral disturbance. A pediatric neurologist had assessed all patients. The following biological data were collected from the medical files: standard initial blood test, serology, nasopharyngeal swabs, stool sample examinations, cerebrospinal fluid examination (including polymerase chain reaction for the genome detection of parainfluenza viruses, herpes virus, cytomegalovirus, varicella zona virus, Epstein-Barr virus, human herpes virus 6, enteroviruses and adenoviruses), electroencephalography and electroneuromyography.
All children were imaged using a Siemens 1.5-T Symphony or Aera scanner with a 16-channel head coil (Erlangen, Germany). The brain MRI protocol included: diffusion-weighted imaging (DWI; TR 5,700 or 5,000 ms, TE 93 or 105 ms, one excitation, field of view 310 cm × 310 cm, slice thickness 5 mm, inter-slice gap 5 mm), fluid-attenuated inversion recovery (FLAIR), T2-w spin echo and T1-w spin echo before and after intravenous contrast administration. A three-dimensional time of flight MR angiography sequence was also performed when a vascular disease was clinically suspected. The MRI examination was performed at the acute phase and a follow-up MRI was performed between 1 month and 6 months after the initial clinical episode and included at least a DWI and FLAIR sequence.
Two pediatric radiologists from our department (B.B. and C.T., with 15 and 20 years of experience, respectively) analyzed the MR images retrospectively. The checklist included distribution and extent of white matter lesions on DWI and FLAIR images as well as the presence of contrast enhancement.
Results
Seven children were included, ages 6.4 to 16 years. There were five boys and two girls. The main clinical, laboratory and imaging findings are summarized in Table 1.
Clinical and laboratory findings
All patients were healthy without any relevant medical history except for one girl who had a history of epilepsy treated with valproate. Febrile illness with prodromal flu-like symptoms was reported 2 to 10 days before the initial MRI examination for 5 patients (patients 1–5). Among these patients, three presented disorders of consciousness (patients 2, 3 and 4), two cases were associated with behavioral disturbance (aggressiveness) (patients 2 and 3) and two cases were associated with seizures (patients 2 and 4). Patient 5 had a behavioral disturbance (visual hallucinations with fleeting episodes of sudden blindness and myodesopsia) without disorders of consciousness. One patient (patient 1) had no clinical symptoms. The lesion of the corpus callosum was incidentally discovered during a follow-up MRI scan performed for growth delay. Anamnesis revealed prodromal flu-like symptoms with fever and headache the day before.
The diagnosis of acute influenza virus A or B infection was established for three of these five patients (patients 1, 3 and 4); this was based on nasopharyngeal swab analysis in two patients and on serological analysis in one patient. Patient 5 presented with concomitant acute parainfluenza virus and adenovirus infections associated with a meningitis profile on analysis of cerebrospinal fluid. No pathogen was found in patient 2 despite a meningitis profile established on cerebrospinal fluid analysis (microbiological cerebrospinal fluid, nasopharyngeal swab, and blood and stool sample analysis remained negative).
The C-reactive protein was moderately higher in all patients except patient 1, who had no significant neuropsychological symptoms.
The two remaining male patients (patients 6 and 7) were identical twins. They presented to the emergency department at 3-year intervals with multiple regressive stroke-like episodes including hemiplegia and dysarthria but no neuropsychological or systemic symptoms. They had neither febrile illness nor prodromal symptoms. The viral and bacterial investigations of the blood, cerebrospinal fluid, nasopharyngeal swabs and stool samples were negative. On clinical and radiologic follow-up 3 months later, the outcome was good without any clinical symptoms or residual MRI lesions. The acute clinical and imaging presentation of the second twin was similar, but hollow feet were found during the clinical follow-up examination. An electroneuromyogram, subsequently performed, revealed a peripheral neuropathy. A review of the family medical history revealed that the maternal grandfather had suffered from a peripheral neuropathy. For these two patients, a final diagnosis of X-linked Charcot-Marie-Tooth disease type 1 with connexin 32 mutation was made thanks to the electroneuromyogram examination and investigation of their mother’s genetic history.
Apart from the residual peripheral neuropathy in the twins, a good final clinical outcome and total fading of the MRI lesions were observed for all patients.
MRI patterns
All patients presented with a restricted lesion of the corpus callosum on DWI (Fig. 1). In three children, an extension to the deep white matter was observed. Four of the children with febrile illness presented with an isolated ovoid lesion of the splenium of the corpus callosum with restricted diffusion during the acute phase. The remaining patient with febrile illness (patient 2) presented with a lesion of the genu with symmetrical bilateral deep white matter extension to the centrum semiovale (Fig. 2). For the two patients with stroke-like episodes, who were finally diagnosed with X-linked Charcot-Marie-Tooth disease type 1, 3-D time of flight MR angiography of the circle of Willis showed no vascular abnormalities. MRI showed diffuse areas of restricted diffusion involving the splenium of the corpus callosum and with additional extensive bilateral symmetrical confluent areas predominantly in the posterior deep white matter (Fig. 3). These lesions extended superiorly toward the centrum semiovale but spared the subcortical U-fibers. There was no posterior fossa, cortical gray matter or basal ganglia involvement in any patient nor any abnormal enhancement after intravenous contrast administration in the acute phase or during follow-up imaging.
A 16-year-old boy (patient 1) with fever, headache and flu-like symptoms 2 days before an MRI examination scheduled for investigation of a growth delay. a-d There is a lesion in the splenium of corpus callosum (arrows) on axial diffusion-weighted image (a, repetition time/echo time 5,700 ms/93 ms), axial apparent diffusion coefficient map b-1000 (b), axial fluid-attenuated inversion recovery spin echo (c, repetition time/echo time/inversion time 9,000 ms/114 ms/2.5 ms) and reformatted axial 3-D T1-weighted gradient echo (repetition time/echo time/flip angle/inversion time 2,000 ms/4 ms/15°/1,100 ms) after gadolinium administration
A 9-year-old girl (patient 2) with acute disorder of consciousness and behavioral disturbance following flu-like symptoms. No infectious agent was found on investigations. The interval between symptoms onset and MRI was 2 days. a Axial diffusion-weighted image (repetition time/echo time 5,700 ms/93 ms) demonstrates high signal intensity in the genu of the corpus callosum (arrows) The interval between symptoms onset and MRI was 2 days b-1000. b Corresponding apparent diffusion coefficient map shows low values in the same area (arrows). c Axial diffusion-weighted MR image (repetition time/echo time 5,700 ms/93 ms) demonstrates high signal intensity in the centrum semiovale (arrowheads) b-1000. d Corresponding apparent diffusion coefficient map shows reduced values in the same area (arrowheads)
A 6-year-old boy (patient 6) initially hospitalized for stroke-like episodes and finally diagnosed with an acute central nervous system manifestation of X-linked Charcot-Marie-Tooth disease with connexin 32 mutation. The interval between symptoms onset and MRI was 1 day. a Axial diffusion-weighted image (repetition time/echo time 5700 ms/93 ms) shows high signal intensity lesion of the whole splenium of corpus callosum (arrows) b-1000. b Axial diffusion-weighted image (repetition time/echo time 5700 ms/93 ms) shows symmetrical bilateral extension to the deep white matter of the centrum semiovale (arrowheads) b-1000. c Axial apparent diffusion map corresponding to (a) shows reduced values in the splenium (arrows). d Axial apparent diffusion map corresponding to (b) shows reduced values in the centrum semiovale (arrowheads)
In all patients, complete fading of the lesion in the corpus callosum was observed on the follow-up MRI performed between 1 and 6 months later (Fig. 4).
A 7-year-old boy with visual disorders (patient 5). Magnetic resonance imaging 2 days days after symptom onset demonstrates an isolated ovoid high signal intensity lesion of the corpus callosum (arrow) at axial diffusion-weighted image (a, repetition time/echo time: 5,000 ms/105 ms) and axial T2-weighted spin echo image b-1000 (b, repetition time/echo time: 5,900 ms/99 ms) shows the same lesion with high signal intensity (arrow). c-d The lesion has disappeared at MRI 1 days days after the study in (a-b) as seen on axial diffusion-weighted MR image (c, repetition time/echo time: 5,000 ms/105 ms) and axial T2-weighted spin echo image b-1000 (d, repetition time/echo time 5,900 ms/99 ms)
Discussion
This study concerned seven children with a transient restricted diffusion-weighted lesion of the corpus callosum at MRI. A few pediatric cases have been reported in the Caucasian population [12,13,14], but most previous studies describing this type of corpus callosum lesion have been reported by centers in East Asia [8, 12]. As reported in the literature, we found that transient restricted diffusion-weighted MRI lesions of corpus callosum associated with febrile illness due to an infectious pathogen seem to be the main etiology in our series.
We found the same clinical symptoms for patients with febrile illness as in other studies [8, 12] (mainly disorders of consciousness and behavior) but with a greater prevalence of seizures (2/5 patients). Conversely, patient 1 had a lesion that was incidentally discovered on a MRI examination performed for another indication. No symptoms were observed except for headache and prodromal flu-like symptoms 2 days earlier. He was finally diagnosed with an acute influenza A infection. This observation may suggest that transient lesions of the corpus callosum are probably underestimated in influenza virus infections and may not always be associated with neurological symptoms. As in the other studies [8, 12], influenza virus type A or B appears to be the most common causative infectious agent in our series. Evidence of acute infection with parainfluenza virus and adenovirus was reported in one of our patients. A variety of other pathogens have previously been described in literature, including viruses (Epstein-Barr virus, mumps, rotavirus, etc.), bacteria (Mycoplasma pneumoniae, Escherichia coli, Salmonella Enteritiditis, etc.) and exceptionally parasites (Plasmodium falciparum [15]). In the largest cohort, up to 41% of negative infection tests were reported [8]. In our series, only one patient with febrile illness tested negative for infectious investigations, despite a cerebrospinal fluid meningitis profile and a C-reactive protein increase.
The clinical and radiologic outcome of children presenting with transient lesions of the corpus callosum associated with febrile illness is good with a rapid decline in clinical symptoms as well as MRI lesions within the following weeks or months. In the two main studies [8, 12], clinical outcome was good and without neurological sequelae, regardless of whether the patient received steroid-specific treatment, and irrespective of the lesion extension to the deep white matter and the type of infectious pathogen. Two patients with neurological sequelae have been reported [10], but with atypical MRI features. In these cases, frontal cortical and corpus striatum lesions were described in addition to the standard transient corpus callosum and deep white matter lesion. This probably implies that these patients should not have been given a simple MERS diagnosis. Finally, only one patient was reported with a persistent corpus callosum lesion following a MERS type 1 lesion [16], but no information was available on his clinical outcome or neuropsychological development.
Some differential diagnoses on MRI require a mention. The etiologies of reversible lesions of corpus callosum are various, including metabolic disorders such as hypoglycemia [17,18,19] and hypernatremia [20], high-altitude cerebral edema [21] and epileptic contexts following a seizure or associated with antiepileptic drugs [22, 23], including their withdrawal [24, 25]. Most pediatric cases reported in the literature were of transient corpus callosum lesions in the setting of a febrile illness. When corpus callosum lesions extend contiguously to the deep white matter, a metabolic leukoencephalopathy spectrum disorder such as adrenoleukodystrophy could be suspected [26]. However, in these disorders, MRI deep white matter lesions are barely reversible [27] and contrast enhancement is often found. Moreover, the clinical presentation is different with prior cognitive or behavioral regression.
X-linked Charcot-Marie-Tooth disease type 1 is another differential diagnosis found in our series. The corpus callosum lesions in the twins had a wide extension to the splenium and symmetrical bilateral extension within the deep white matter of the centrum semiovale. Neither twin had encephalopathy signs, febrile illness or prodromal flu-like symptoms. Central nervous system dysfunction caused by connexin 32 mutations has been reported in patients as young as 5 years of age [28]. Although this form of Charcot-Marie-Tooth is X-linked, at least one manifesting female carrier has been reported with deep white matter changes [29], although not during her childhood. In our study, the diagnosis of X-linked Charcot-Marie-Tooth disease type 1 was unknown for the two boys and the stroke-like episodes were the first manifestation of the disease. Other cases of stroke-like episodes as the first clinical manifestation of X-linked Charcot-Marie-Tooth disease type 1 with connexin 32 mutation have been reported previously [30, 31]. An increasing number of studies have described transient central nervous system dysfunction in males with X-linked Charcot-Marie-Tooth disease, manifesting as hemiplegia, sensory loss, dysarthria, ataxia, aphasia and even confusion, and accompanied by transient corpus callosum and deep white matter MRI abnormalities. These symptoms last hours to weeks and in rare cases can precede peripheral neuropathy onset of the disease, whereas MRI changes often resolve completely within a few months [31,32,33,34]. Two slight differences in MRI signs can help distinguish X-linked Charcot-Marie-Tooth disease. First, an extension of the MRI abnormalities within the corticospinal tract [34]. Second, a persistent mild hyperintense signal on T2-weighted images within the centrum semiovale at 1 month and even at 6 months [31, 33], which is never the case in MERS 2 patients. These abnormalities were not found in our twin patients.
The pathogenesis of corpus callosum lesions in RESLES remains unknown. According to the clinical and underlying pathogenesis condition, several theories have been put forward to explain the transiently restricted diffusion-weighted image lesions on MRI.
One of the most relevant theories is based on the preponderance of the reported pediatric RESLEF or MERS cases in East Asia and more particularly in Japan, as well as some familial cases [35], which suggests the important role played by unidentified genetic factors. Secondly, some theories mention the histological structure of the corpus callosum containing tightly packed myelin sheaths and a relatively high water component probably rendering the corpus callosum more susceptible to cytotoxic edema than other brain areas. However, this theory appears incomplete, as it does not explain the deep white matter lesion that may be associated.
Third, an inflammatory theory has been suggested to explain lesions found in RESLEF and MERS associated with infectious pathogens. Indeed, some authors found a high production of cytokines in the cerebral spinal fluid [36]. However, the symmetry and constant absence of RESLEF or MERS lesion enhancement do not support an inflammatory etiology.
The main pathogenesis theory, even if not fully understood, is a transient dysfunction of intercellular metabolic or ionic exchange between oligodendrocyte, astrocyte and axon fibers implying the formation of cytotoxic edema [3]. This theory appears relevant for the main etiologies of transient restricted DWI corpus callosum lesions including MERS/RESLEF, Charcot Marie-Tooth type 1X disease and anti-epileptic drug withdrawal.
The main limitation of our study is the small number of subjects. Despite many attempts to cluster reversible splenial lesions into distinct entities, at the end, with the exception that most described cases in children have been attributed to infection, these continue to be highly nonspecific and we still do not understand completely their pathophysiology.
Conclusion
A transient lesion of the corpus callosum with restricted diffusion during a febrile illness episode should prompt the radiologist to suggest an infectious origin in children, particularly the influenza virus. Associated mild encephalopathy sign may be present or absent. The prognosis of these patients is good with rapid normalization of clinical symptoms and fading of the MRI lesion within one week to a month without any specific treatment.
References
Uchino A, Takase Y, Nomiyama K et al (2006) Acquired lesions of the corpus callosum: MR imaging. Eur Radiol 16:905–914
Doherty MJ, Jayadev S, Watson NF et al (2005) Clinical implications of splenium magnetic resonance imaging signal changes. Arch Neurol 62:433–437
Starkey J, Kobayashi N, Numaguchi Y, Moritani T (2017) Cytotoxic lesions of the corpus callosum that show restricted diffusion: mechanisms, causes, and manifestations. Radiographics 37:562–576
Garcia-Monco JC, Cortina IE, Ferreira E et al (2011) Reversible splenial lesion syndrome (RESLES): what’s in a name? J Neuroimaging 21:e1–e14
Zhang S, Ma Y, Feng J (2015) Clinicoradiological spectrum of reversible splenial lesion syndrome (RESLES) in adults. Medicine (Baltimore) 94:e512
Tada H, Takanashi J, Barkovich AJ et al (2004) Clinically mild encephalitis/encephalopathy with a reversible splenial lesion. Neurology 63:1854–1858
Takanashi J, Barkovich AJ, Yamaguchi K, Kohno Y (2004) Influenza-associated encephalitis/encephalopathy with a reversible lesion in the splenium of the corpus callosum: a case report and literature review. AJNR Am J Neuroradiol 25:798–802
Takanashi J (2009) Two newly proposed infectious encephalitis/encephalopathy syndromes. Brain Dev 31:521-528
Takanashi J, Imamura A, Hayakawa F, Terada H (2010) Differences in the time course of splenial and white matter lesions in clinically mild encephalitis/encephalopathy with a reversible splenial lesion (MERS). J Neurol Sci 292:24–27
Takanashi J, Barkovich AJ, Shiihara T et al (2006) Widening spectrum of a reversible splenial lesion with transiently reduced diffusion. AJNR Am J Neuroradiol 27:836–838
Notebaert A, Willems J, Coucke L et al (2013) Expanding the spectrum of MERS type 2 lesions, a particular form of encephalitis. Pediatr Neurol 48:135–138
Kashiwagi M, Tanabe T, Shimakawa S et al (2014) Clinico-radiological spectrum of reversible splenial lesions in children. Brain Dev 36:330–336
Ka A, Britton P, Troedson C et al (2015) Mild encephalopathy with reversible splenial lesion: an important differential of encephalitis. Eur J Paediatr Neurol 19:377–382
Harini C, Das RR, Prabhu SP et al (2015) Clinical and neuroimaging profile of children with lesions in the corpus callosum. J Neuroimaging 25:824–831
Laothamatas J, Sammet CL, Golay X et al (2014) Transient lesion in the splenium of the corpus callosum in acute uncomplicated falciparum malaria. Am J Trop Med Hyg 90:1117–1123
Hashimoto Y, Takanashi J, Kaiho K et al (2009) A splenial lesion with transiently reduced diffusion in clinically mild encephalitis is not always reversible: A case report. Brain Dev 31:710-712
Kagawa K, Okada H (2009) Reversible splenial lesion of the corpus callosum on diffusion-weighted magnetic resonance imaging in hypoglycemic hemiparesis: report of two cases. No Shinkei Geka 37:473–478
Malik AM (2013) The reversible corpus callosum splenium lesion associated with hypoglycemic encephalopathy. Neurohospitalist 3:169
Kim JH, Choi JY, Koh S-B, Lee Y (2007) Reversible splenial abnormality in hypoglycemic encephalopathy. Neuroradiology 49:217–222
Maeda M, Tsukahara H, Terada H et al (2006) Reversible splenial lesion with restricted diffusion in a wide spectrum of diseases and conditions. J Neuroradiol 33:229–236
Hackett PH, Yarnell PR, Hill R et al (1998) High-altitude cerebral edema evaluated with magnetic resonance imaging: clinical correlation and pathophysiology. JAMA 280:1920–1925
Kim SS, Chang KH, Kim ST et al (1999) Focal lesion in the splenium of the corpus callosum in epileptic patients: antiepileptic drug toxicity? AJNR Am J Neuroradiol 20:125–129
Güven H, Delibaş S, Comoğlu SS (2008) Transient lesion in the splenium of the corpus callosum due to carbamazepine. Turk Neurosurg 18:264–270
Gürtler S, Ebner A, Tuxhorn I et al (2005) Transient lesion in the splenium of the corpus callosum and antiepileptic drug withdrawal. Neurology 65:1032–1036
Mori H, Maeda M, Takanashi J et al (2012) Reversible splenial lesion in the corpus callosum following rapid withdrawal of carbamazepine after neurosurgical decompression for trigeminal neuralgia. J Clin Neurosci 19:1182–1184
Loes DJ, Fatemi A, Melhem ER et al (2003) Analysis of MRI patterns aids prediction of progression in X-linked adrenoleukodystrophy. Neurology 61:369–374
Kim JH, Kim HJ (2005) Childhood X-linked adrenoleukodystrophy: clinical-pathologic overview and MR imaging manifestations at initial evaluation and follow-up. Radiographics 25:619–631
Siskind C, Feely SME, Bernes S et al (2009) Persistent CNS dysfunction in a boy with CMT1X. J Neurol Sci 279:109–113
Basri R, Yabe I, Soma H et al (2007) X-linked Charcot-Marie-Tooth disease (CMTX) in a severely affected female patient with scattered lesions in cerebral white matter. Intern Med 46:1023–1027
Al-Mateen M, Craig AK, Chance PF (2014) The central nervous system phenotype of X-linked Charcot-Marie-Tooth disease: a transient disorder of children and young adults. J Child Neurol 29:342–348
Anand G, Maheshwari N, Roberts D et al (2010) X-linked hereditary motor sensory neuropathy (type 1) presenting with a stroke-like episode. Dev Med Child Neurol 52:677–679
Paulson HL, Garbern JY, Hoban TF et al (2002) Transient central nervous system white matter abnormality in X-linked Charcot-Marie-Tooth disease. Ann Neurol 52:429–434
Wang Y, Yin F (2015) A review of X-linked Charcot-Marie-Tooth disease. J Child Neurol 31:761–772
Kassubek J, Bretschneider V, Sperfeld A-D (2005) Corticospinal tract MRI hyperintensity in X-linked Charcot-Marie-Tooth disease. J Clin Neurosci 12:588–589
Imamura T, Takanashi J, Yasugi J et al (2010) Sisters with clinically mild encephalopathy with a reversible splenial lesion (MERS)-like features; Familial MERS? J Neurol Sci 290:153–156
Morichi S, Kawashima H, Ioi H et al (2012) High production of interleukin-10 and interferon-γ in influenza-associated MERS in the early phase. Pediatr Int 54:536–538
Bulakbasi N, KocaogluM, Tayfun C, Ucoz T (2006) Transient splenial lesion of the corpus callosum in clinicallymild influenza-associated encephalitis/encephalopathy. AJNR Am J Neuroradiol 27:1983–1986
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Rights and permissions
About this article
Cite this article
Le Bras, A., Proisy, M., Kuchenbuch, M. et al. Reversible lesions of the corpus callosum with initially restricted diffusion in a series of Caucasian children. Pediatr Radiol 48, 999–1007 (2018). https://doi.org/10.1007/s00247-018-4124-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-018-4124-x