Abstract
Purpose
The aim of this study was to investigate the clinical-radiological determinants of diffusion-weighted image (DWI) abnormalities in patients with suspected acute ischemic stroke (AIS) seen at the emergency room (ER).
Methods
During the study period, 882 consecutive patients were screened at Clínica Alemana de Santiago, Chile; 786 had AIS and 711 (90.4%) were included.
Results
DWI demonstrated 87.3% sensitivity and 99.0% specificity, with a positive likelihood ratio of 79 and a negative likelihood ratio of 0.13 for the detection of AIS. In the univariate analysis, a positive DWI in AIS was associated with admission National Institute of Health Stroke Scale (NIHSS) score (OR 1.09, 95% CI 1.04–1.1%), time from symptom onset to DWI (OR 1.03, 95% CI 1.01–1.05), presence of a relevant intracranial artery occlusion (OR 3.18, 95% CI 1.75–5.76), posterior circulation ischemia (OR 0.44, 95% CI 0.28–0.7), brainstem location of the AIS (OR 0.16, 95% CI 0.093–0.27), infratentorial location of AIS (OR 0.44, 95% CI 0.28–0.70), and lacunar (OR 0.27, 95% CI 0.11–0.68) or undetermined stroke etiology (OR 0.12, 95% CI 0.3–0.31). In multivariate analysis, only admission NIHSS score (OR 1.07, 95% CI 1.01–1.13), time from symptom onset to DWI (OR 1.04, 95% CI 1.01–1.13), brainstem location (OR 0.13, 95% CI 0.051–0.37), and lacunar (OR: 0.4, 95% CI 0.21–0.78) or undetermined etiology (OR: 0.4, 95% CI 0.22–0.78) remained independently associated.
Conclusion
DWI detects AIS accurately; the positivity of these evaluations in the ER is associated only with NIHSS on admission, time to DWI, brainstem location, and AIS etiology.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
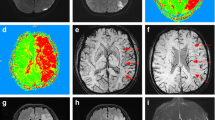
Diffusion-weighted image (DWI) is highly sensitive for the diagnosis of acute ischemic stroke (AIS) [1,2,3,4], but may yield false-negative results [1,2,3,4,5,6].
The variables associated with these false results have not been evaluated in detail; in some previous experiences, patients were already known to have an AIS [5], used normal subjects as control [6], or did retrospective analysis [7], leading to an increased DWI sensitivity. Other studies included patients with transient ischemic attacks [8], or minor strokes [6], increasing the chance of negative results.
We aimed to investigate determinants of positive DWI in patients with AIS seen in the emergency room (ER) at Clínica Alemana in Santiago, Chile, in a large prospective cohort of consecutive unselected patients with focal symptoms suggestive of AIS.
Methods
In this prospective study, patients with suspected AIS admitted to the ER between December 2012 and April 2017 were evaluated by the neurologist on call. Age, stroke risk factors, and time from stroke onset to arrival at the ER, defined as the last time at which the patient was known to be free of any neurological deficits, were recorded. Stroke severity was assessed with the National Institute of Health Stroke Scale (NIHSS). Patients were then studied with our local neuroimaging protocol, which has been previously described [9] consisting of a non-contrast brain computed tomography (NCCT) and, in those patients without contraindication, an immediate spiral computed tomographic angiography of the cervical and intracranial arteries (CTA), and then DWI was performed. If the CTA could not be obtained, acute magnetic resonance angiography (MRA) was performed.
The neuroradiologist on call was informed about the admission of a possible stroke patient and the suspected location of the lesion.
Relevant intracranial large vessel disease was defined as symptomatic disease involving segments M1 or M2 of the middle cerebral artery, the terminal internal carotid artery, the A1 segment of the anterior W cerebral artery, the P1 segment of the posterior cerebral artery, or the vertebral and basilar arteries.
We recorded the time from symptom onset to the time when DWI was performed and the MRI equipment in which this evaluation was done.
DWI evaluations were obtained either in one of the two GE HdxT 1.5-T MRIs or in a Simmens Skyra 3 T, and the machine used depended only on current availability at patient presentation. A standardized protocol was applied, including axial acquisitions and, in those patients with negative results thin coronal sections. DWI parameters are shown in Table 1.
Patients eligible for intravenous thrombolytic therapy were treated in a window of 4.5 h; the recombinant tissue plasminogen activator (rtPA) bolus was administered after the NCCT scan, CTA and DWI were performed usually immediately after the NCCT evaluation. Patients that arrived in the first 24 hours to the ER frequently were evaluated with transcranial Doppler as part of the head position in stroke trial (HEADPOST)—pilot and HEADPOST main studies. After the initial evaluation patients were hospitalized in the Stroke Unit for at least 48. Telemetry monitoring was performed in all cases and, as soon as possible, cardiologists carried out a transthoracic echocardiogram. A transesophageal echocardiogram was performed according to the treating vascular neurologist’s opinion. Additional evaluations could be done to detect infrequent causes of acute ischemic stroke.
Patients with negative DWI results in the ER were re-evaluated after 24 h with either brain NCCT or MRI.
AIS was diagnosed in patients with a history, clinical examination, and typical vascular brain damage clinical outcome, with signs of brain ischemia on NCCT/DWI according to our stroke neuroimaging protocol, on follow-up imaging, or if an occluded vessel was observed in the symptomatic territory. Patients with repeated negative imaging but with an evident neurovascular syndrome and no other alternative diagnosis after an extensive workup were finally diagnosed as stroke by a stroke specialist neurologist [1]. Transient ischemic attacks were excluded.
On discharge, patients were stratified according to the Trial of Org 10172 in Acute Stroke Treatment (TOAST) [10], and the infarct location was defined based on the initial DWI imaging, presence of an occluded vessel in the symptomatic territory, or in the follow-up imaging by the treating stroke neurologist that was not aware of this study. For those cases, were all images were normal, but the stroke neurologist considered that the patient diagnosis was an AIS; clinical criteria were used to locate the stroke (for instance, Wallenberg syndrome was located in the brain stem).
The diagnosis of stroke mimics (SMs) was based on the presence of acute or subacute focal symptoms with no demonstrable ischemia or arterial occlusion on neuroimaging and normal brain imaging on follow-up (24 to 48 h later); a definitive, alternative diagnosis that explains the patient’s initial symptoms must have been made before discharge.
The Ethics Committee of Universidad del Desarrollo, Clínica Alemana de Santiago approved the protocol, and patients or their relatives provided written informed consent.
Statistical analysis
Continuous variables were described using mean and standard deviation, while categorical variables are described using frequencies and proportions. Univariate analysis based on the presence of a positive DWI in the ER was carried out applying Student’s t test for continuous variables while categorical variables were compared using the Fisher exact test of independence. Variables in the multivariate model were selected using the criteria of Hosmer-Lemeshow with a retention P value of 0.25. With these selected variables, a logistic regression model using a stepwise process was built, using backward selection. Sensitivity, specificity, positive (PLR) and negative (NLR) likelihood ratios, and their respective 95% CIs were calculated to estimate DWI validity in detecting AIS in the unselected patients arriving at the ER with symptoms suggestive of AIS. All confidence intervals were set at 95%, with 5% for significance. Data were processed with the Stata v 14.0 software.
Results
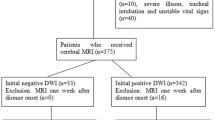
During the study period, a total of 882 consecutive patients were screened; of these, 786 had suffered an AIS and 711 were included (90.4%) in this analysis. A group of 75 patients were excluded because of MRI contraindication (peacemaker, agitation, or critical medical condition). Ninety-six patients were classified as SMs and nine of these did not undergo DWI. Except for one false positive (a small subcortical tumor interpreted as an AIS), all DWIs performed in SMs were negative.
In this non-selected group of patients with suspected AIS, DWI demonstrated a sensitivity of 87.3% (CI 95%, 84.7–89.7) and a specificity of 99.0% (95 CI%, 94.3–100), with a positive predictive value of 99.8% (95 CI%, 98.8–99.9) and a negative predictive value of 51.3% (95 CI%, 46.5–56.1). The PLR was 79 (CI 95%, 11.7–550.1), and the NLR was 0.13 (CI 95%, 0.11–0.16).
All stroke patients had blood tests, EKG, and brain CT scan; 615 of these (86.4%) had a CTA of the cervical and intracranial arteries performed, and a MRA of the cervical and intracranial arteries was carried out in 22 cases (3%). A total of 526 patients (73.9%) had transcranial Doppler as part of their evaluation; 79 patients (11. 1%) had ultrasound ECO-Duplex examinations of the cervical vessels. Of the 711 patients, 665 (93.5%) had transthoracic echocardiography and 33 (4.6%) a transesophageal exploration. Finally, 71 cases (9.9%) were evaluated with DSA angiography. Only 14 patients (1.9%) had incomplete evaluations.
The characteristics of the AIS patients and univariate analysis correlating the presence of a positive DWI in the ER are shown in Table 2. The mean age was 69.5 ± 16.8 years; 390 (54.9%) were females, and 296 (41.6%) were evaluated with DWI within 4.5 h of symptom onset and 570 (80.1%) within the first 24 h; 75 (10.5%) cases were evaluated between 24 and 48 h of evolution and only 66 (9.4%) after 48 h. A 3-T MRI was used in 133 (18.7%) of the studies, and a total of 191 patients (26.8%) were treated with rtPA.
In the univariate analysis correlating the presence of a positive DWI in the ER with the clinical and radiological variables, the variables that reached significance were admission NIHSS score (OR 1.09, 95% CI 1.04–1.1%), time to DWI (OR 1.03, 95% CI 1.01–1.05), the presence of a relevant intracranial occlusion (OR 3.18, 95% CI 1.75–5.76), posterior circulation ischemia (OR 0.44 95% CI 0.28–0.7), brainstem (OR 0.16, 95% CI 0.093–0.27) and infratentorial localizations (OR 0.44, 95% CI 0.28–0.70), and lacunar (OR 0.27, 95% CI 0.11–0.68) or undetermined stroke etiology (OR 0.12, 95% CI 0.3–0.31).
In the multivariate analysis (Table 3), only admission NIHSS (OR 1.07, 95% CI 1.01–1.13; P = 0.01), time from symptom onset to DWI (OR 1.04, 95% CI 1.01–1.13; P < 0.001), brainstem location (OR 0.13, 95% CI 0.051–0.37; P < 0.001), and lacunar (OR 0.4, 95% CI 0.21–0.78; P = 0.007) or undetermined etiology (OR 0.4, 95% CI 0.22–0.78; P = 0.007) remained statistically significant.
In the group of 90 (13.7%) patients with AIS and initially negative DWI, 14 (15.5%) cases had an intracranial vessel occlusion in the symptomatic territory in the acute imaging protocol. In three cases, the family rejected a new brain imaging on the basis of an unfavorable clinical outcome due to severe AIS. In the remaining 73 (81.1%) patients, the follow-up brain imaging performed between 24 and 48 h demonstrated an AIS in 42 (57.5%) cases (40 evaluated with MRI and 2 with NCCT scans). Most patients with negative follow-up imaging had only a brain NCCT scan as imaging control and AIS anatomical location was classified by the attending neurologist on the basis on their clinical symptoms. Brainstem syndromes (Wallenberg or Weber) were evident in ten patients; nine cases presented with a classical lacunar syndrome defined as follows: pure motor hemiparesis, pure sensory stroke, sensorimotor syndrome, ataxic hemiparesis, and dysarthria—clumsy hand with the absence of agnosia, aphasia, apraxia, homonymous hemianopsia, monoplegia, seizures, and stupor [11,12,13,14,15,16].
Five cases had isolated cortical symptoms (acute aphasia or constructive apraxia) and seven had other symptoms, including one case of acute chorea in a patient with an anti-phospholipid syndrome.
SM patients had a final diagnosis of metabolic encephalopathies in 19 cases, migraine disorders in 16 patients, seizures in 15 cases, conversion disorders in 12, and other diseases in the other 25 cases.
Discussion
In this study of consecutive unselected patients arriving to an ER with symptoms suggestive of AIS, a positive DWI is almost synonymous with AIS on the basis of a PLR of 79. On the contrary, a result of 0.13 in the NLR suggests that a negative DWI has only moderate significance for a decrease probability of AIS. Indeed, in our experience, almost one out of eight patients with AIS had a negative DWI: this could be as high as one third in patients with nondisabling AIS [15].
Several variables were associated with the positivity of DWI in our patients; for every 1 point of increase in admission NIHSS, the risk of having a positive DWI increased by 7%. Higher NIHSS values represent more extensive areas of ischemic tissue, as postulated by Chalela et al. [3]. Most of their negative DWIs were seen in AIS whose NIHSS were less than 4, which is comparable to our findings and previous studies [1]. Time from symptom onset to DWI was another important factor associated with positive results in the ER; for every 60 min that the DWI is delayed, the chances of a positive result increases by 4%. During the early stages of AIS, the magnitude of the hypo-perfusion may cause symptoms, but could be not be severe enough to cause water restriction evident by DWI. With cerebral blood flow levels of 15 to 20 ml/100 g/min, infarction may not have developed after more than 120 min although evident neurological symptoms are present [16]; this could explain the presence of important cerebral focal symptoms but with negative DWI. Previous studies demonstrated that most false-negative DWI occurs among patients evaluated within the first 3 h after the onset of symptoms, in our experience, false-negative DWI, could be found even 8 h after symptom onset [3].
Brainstem AIS was associated with 87% less probabilities of positive results in DWI as demonstrated by other investigators [5, 17], this reflecting the fact that the brainstem is frequently affected by small infarcts that are difficult to detect. As stated by Oppenheimer and a recent meta-analysis, the posterior brain circulation is associated with these negative DWI results [2, 5]. However, in our experience, the brainstem location of AIS is strongly associated with these false-negative DWIs; in our multivariate analysis that included the presence of posterior circulation, infratentorial AIS, and brainstem location as variables, it was shown that this location was the only one of these variables that was associated with false-negative DWI results.
Finally, stroke etiology is associated with positivity of the DWI in the ER. Lacunar and undetermined AIS etiologies were found to have 60% less chance of an abnormal DWI evaluation, probably because of their small sizes. In this experience, both stroke subtypes demonstrated lower NIHSS values than other TOAST subtypes (P = 0.0001). In a recent meta-analysis by Hart et al. [18], AIS of indeterminate etiology demonstrated a mean NIHSS of 4, a value that correlates with small stroke sizes [19], similar to the values described by Chalela et al. [3] and found in this study to be associated with negative DWI evaluations. In our study, undetermined strokes arrive earlier to the ER (P = 0.0001), an element that, as we explain earlier, correlates with more negative DWI results.
We found no association between the MRI machine used and a positive result, although a small group of patients was studied in the 3-T MRI.
Our study has several strengths; it includes a large number of consecutive patients with a suspected AIS at the moment DWI was done, which exclude selection bias, the evaluation of multiple variables that could influence the results of DWI, some of which had not been investigated previously. These include the presence of an intracranial vessel occlusion and a univariate and multivariate analysis, including supra- versus infratentorial lesion location, anterior versus posterior circulation, and brainstem lesion versus other localizations, allowing us to conclude that those cases described previously in which the posterior brain circulation was associated with negative DWI are caused mainly by brainstem locations of the stroke. In addition, we added thin coronal sections for those with negative DWI that increases the possibility of positive results [20].
Our study has also several limitations; mainly, it is a single-center experience whose patients were not randomized to the different MRI equipment. The neuroradiologist on call informed the results of DWI and his results were not compared to those of other specialists or stroke neurologist in order to calculate interobserver and intraobserver k values. Components of the itemized NIHSS that could possibly correlate with the positivity of DWI were not evaluated [21]. Of those patients with an initial negative DWI, less than half had an MRI as the follow-up brain imaging. Also, we cannot rule out that some patients with negative imaging evaluations could be SMs and, finally, some patients received a rtPA bolus before DWI, although this treatment has been shown to avert a positive infarction imaging in only 0.9% of cases [22].
In conclusion, our findings demonstrate that the positivity of DWI is independently associated with admission NIHSS, time to DWI, brainstem location, and the etiology of the stroke; AIS should be considered a clinical diagnosis, and as many DWI could be negative in an acute setting, medical acute therapies should not be based on the results of this imaging that should be used just to confirm this disease [23].
References
Brunser AM, Hoppe A, Illanes S, Díaz V, Muñoz P, Cárcamo D et al (2013) Accuracy of diffusion-weighted imaging in the diagnosis of stroke in patients with suspected cerebral infarct. Stroke 44:1169–1171
Edlow BL, Hurwitz S, Edlow JA (2017) Diagnosis of DWI-negative acute ischemic stroke: a meta-analysis. Neurology 89(3):256–262
Chalela JA, Kidwell CS, Nentwich LM, Luby M, Butman JA, Demchuk AM, Hill MD, Patronas N, Latour L, Warach S (2007) Magnetic resonance imaging and computed tomography in emergency assessment of patients with suspected acute stroke: a prospective comparison. Lancet 369:293–298
Lovblad KO, Laubach HJ, Baird AE, Curtin F, Schlaug G, Edelman RR et al (1998) Clinical experience with diffusion-weighted MR in patients with acute stroke. AJNR Am J Neuroradiol 19:1061–1066
Oppenheim C, Stanescu R, Dormont D, Crozier S, Marro B, Samson Y, Rancurel G, Marsault C (2000) False-negative diffusion-weighted MR findings in acute ischemic stroke. AJNR Am J Neuroradiol 21:1434–1440
Singer MB, Chong J, Lu D, Schonewille WJ, Tuhrim S, Atlas SW (1998) Diffusion-weighted MRI in acute subcortical infarction. Stroke 29(1):133–136 Erratum in: Stroke 1998 Mar;29:731
Zuo L, Zhang Y, Xu X, Li Y, Bao H, Hao J, Wang X, Li G (2015) A retrospective analysis of negative diffusion-weighted image results in patients with acute cerebral infarction. Sci Rep 5:8910
Sylaja PN, Coutts SB, Krol A, Hill MD, Demchuk AM, VISION Study Group (2008) When to expect negative diffusion-weighted images in stroke and transient ischemic attack. Stroke 39:1898–1900
Brunser AM, Lavados PM, Cárcamo DA, Hoppe A, Olavarría V, Diaz V, Rivas R (2010) Additional information given to a multimodal imaging stroke protocol by transcranial Doppler ultrasound in the emergency room: a prospective observational study. Cerebrovasc Dis 30:260–266
Adams HP Jr et al (1993) Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in acute stroke treatment. Stroke 24(1):35–41
Fisher CM, Curry HB (1965) Pure motor hemiplegia of vascular origin. Arch Neurol 13:30–44
Fisher CM (1967) A lacunar stroke: the dysarthria-clumsy hand syndrome. Neurology 17:614–617
Fisher CM (1965) Pure sensory stroke involving face, arm, and leg. Neurology 15:76–80
Fisher CM, Cole M (1965) Homolateral ataxia and crural paresis: a vascular syndrome. J Neurol Neurosurg Psychiatry 28:48–55
Makin SD, Doubal FN, Dennis MS, Wardlaw JM (2015) Clinically confirmed stroke with negative diffusion-weighted imaging magnetic resonance imaging: longitudinal study of clinical outcomes, stroke recurrence, and systematic review. Stroke 46:3142–3148
Gonzalez RG (2012) Clinical MRI of acute ischemic stroke. J Magn Reson Imaging 36:259–271
Saber Tehrani AS, Kattah JC, Mantokoudis G, Pula JH, Nair D, Blitz A, Ying S, Hanley DF, Zee DS, Newman-Toker DE (2014) Small strokes causing severe vertigo: frequency of false-negative MRIs and nonlacunar mechanisms. Neurology 83:169–173
Hart RG, Catanese L, Perera KS, Ntaios G, Connolly SJ (2017) Embolic stroke of undetermined source: a systematic review and clinical update. Stroke 48:867–872
Yaghi S, Herber C, Boehme AK, Andrews H, Willey JZ, Rostanski SK, Siket M, Jayaraman MV, McTaggart RA, Furie KL, Marshall RS, Lazar RM, Boden-Albala B (2017) The association between diffusion MRI-defined infarct volume and NIHSS score in patients with minor acute stroke. J Neuroimaging 27:388–391
Felfeli P, Wenz H, Al-Zghloul M, Groden C, Förster A (2017) Combination of standard axial and thin-section coronal diffusion-weighted imaging facilitates the diagnosis of brainstem infarction. Brain Behav 7:e00666
Yaghi S, Herber C, Willey JZ, Andrews HF, Boehme AK, Marshall RS, Lazar RM, Boden-Albala B (2015) Itemized NIHSS subsets predict positive MRI strokes in patients with mild deficits. J Neurol Sci 358:221–225
Freeman JW, Luby M, Merino JG, Latour LL, Auh S, Song SS, Magadan A, Lynch JK, Warach S, Hsia AW (2013) Negative diffusion-weighted imaging after intravenous tissue-type plasminogen activator is rare and unlikely to indicate averted infarction. Stroke 44:1629–1634
Abbott AL, Silvestrini M, Topakian R, Golledge J, Brunser AM, de Borst GJ, Harbaugh RE, Doubal FN, Rundek T, Thapar A, Davies AH, Kam A, Wardlaw JM (2017) Optimizing the definitions of stroke, transient ischemic attack, and infarction for research and application in clinical practice. Front Neurol 8:537
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for this study.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in the studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Brunser, A.M., Cavada, G., Venturelli, P.M. et al. Diffusion-weighted imaging determinants for acute ischemic stroke diagnosis in the emergency room. Neuroradiology 60, 687–692 (2018). https://doi.org/10.1007/s00234-018-2029-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-018-2029-x