Abstract
Purpose
The results of analyses of patients’ health problems related to medication use have been highly variable due to various factors, such as different study methodology, diverse variables determined, fields of study. The aim of our study was to determine the prevalence and preventability of negative clinical outcomes of medication (NCOMs).
Methods
This was a cross-sectional study performed in the emergency departments (EDs) of nine Spanish hospitals during a 3-month period. A two-stage probabilistic sampling method was used , and a systematic appraisal tool was used to identify the NCOMs based on information gathered through patient interview and review of the medical records. Case evaluations were conducted in two phases by pharmacists and physicians. The prevalence and preventability of NCOM were calculated. A homogeneity test was performed to assess potential differences in the prevalence for each hospital.
Results
A total of 4,611 patients were included in the study. The overall prevalence of NCOMs was 35.7 % [95 % confidence interval (CI) 33.3–38.1]. These NCOMs could be divided into three categories: ineffectiveness (18.2 %; 95 % CI 16.2–20.1), necessity (14.9 %; 95 % CI 13.4–16.6), and lack of safety (2.4 %; 95 % CI 1.9–2.8). About 81 % (95 % CI 80.1–82.3) of the NCOMs could have been prevented.
Conclusions
NCOMs provoked approximately one-third of visits to the EDs, and a high percentage of these were preventable. Implementation of strategies for patient safety and pharmaceutical care could help to prevent these problems and optimize the use of medications.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Medication-related health problems in patients are a relevant issue in public health systems worldwide. Studies published over the last 20 years reveal that between 0.2 and 38 % of all health problems are related to the use of medication [1–11] and that a high percentage of these problems are preventable [1, 9, 12–24]. The high variability in the published results makes it difficult to compare data from different studies and to develop solutions to the problem.
Multiple factors contribute to generate this variability between the results of such studies. One of these is the heterogeneity of the terminology used in the studies for measuring health problems associated to the use of medication. Most of these terms are typically related to medication safety [25] (e.g., adverse drug reactions, adverse drug events, or medication errors). However, health problems arising from therapeutic ineffectiveness and/or from the lack of use can also affect medication-related morbidity and should be considered to determine the actual magnitude of negative health outcomes related to the drug therapy in the healthcare process [1, 26, 27]. The negative clinical outcomes of medication are hereby considered as those health problems related to the use (or lack of use) of medication independently of the cause that generated them (i.e., medication errors, self-medication, non-compliance, drug–drug interaction, etc.) [26, 27].
An additional factor which contributes to generate high variability between the results of different analyses of health problems associated with medication use is the different methods of analysis applied in the studies [1, 6, 9, 12, 13, 28]. For example, the sources of information can influence the results, and the magnitude of health problems attributed to medications has been shown to be lower when medical records are used as the exclusive source of information compared to the incorporation of information from patient interviews [9, 13, 28, 29]. Studies utilizing both sources of information report higher association rates and are more consistent [1, 12, 14]. Similarly, it has been shown that the evaluation reliability influences the variability of the results [30].
Yet another factor that contributes to the variability of results is the setting of the study due to the differences in the characteristics of the populations being examined. Hospital emergency departments (EDs) are settings of interest for measuring the magnitude of health problems related to medications due to the frequent attendance of individuals with a variety of health problems. Spanish EDs are especially good settings for these studies because of their easy accessibility—patients can access EDs with no restriction and without having to first visit a family doctor or general practitioner.
Baena et al. [1] used a validated, protocolized detection method [31, 32] to investigate the prevalence of the negative clinical outcomes of medication (NCOMs) in a hospital. The results of this study showed a 33.1 % rate of NCOMs with a preventability of 73 %. However, the extrapolation of these results to other hospitals remains to be demonstrated.
We have conducted a study in nine hospitals to evaluate the prevalence and preventability of the NCOMs and have analyzed several risk factors that could be associated to these negative clinical outcomes.
Methods
A cross-sectional and multicenter study was performed in the EDs of nine Spanish hospitals from April 1 to June 30, 2003.
Study population and inclusion process
The study population was drawn from patients visiting the EDs of the nine Spanish hospitals listed in Table 1.
A two-stage probabilistic sampling method was performed at each hospital. In the first stage, a simple random sampling was used to select the sample collection days. In the second stage, a systematic sampling was performed to select patients. Sample size was estimated for a prevalence of NCOM of 33 % (based on data obtained by Baena et al. [1]), for the average number of patients visiting EDs in each hospital in 2001, with a confidence interval (CI) of 95 % and a maximum admissible error of 0.01 per hospital. The estimated sample size (n) was 3,760 (Table 1).
We considered three exclusion criteria: (1) acute voluntary drug poisoning; (2) the patient did not wait for the physician’s diagnosis and was therefore not diagnosed; (3) the patient was referred to another hospital without being diagnosed. Patients who visited the ED multiple times for the same health problem during the period of the study were included only once in the analysis.
Measurements and procedure
The principal variable measured was the presence of NCOMs that resulted in a visit to the ED [1]. NCOMs were defined as “patient health outcomes that are not consistent with the objectives of pharmacotherapy and are associated with the use or errors in the use of medicines” [32]. NCOM refers to a negative clinical outcome (i.e., a negative change in the state of the patient health) related to the use (or lack of use) of medication independently of the cause that generated the change (i.e., medication errors, self-medication, non-compliance, etc.).
Sources of information
Each patient was interviewed using a validated questionnaire [33] to collect information on the medications he/she was taking and his/her health problems. Information on the specific health problem that caused the ED visit was obtained from the medical record (i.e., the medical diagnosis).
A total of 98 pharmacists conducted interviews with the study patients. To minimize interviewer bias, all pharmacists received the same training for patient interviews and evaluations. Two pharmacists (MIB and PCF) and one physician (RM) formed the training team. Each case was evaluated using the Dáder method [31] for NCOM identification.
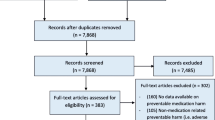
Case evaluations were conducted in two phases (Fig. 1). First, information on the patient was subjected to an initial evaluation by pharmacists to establish a suspected case of a NCOM was conducted by pharmacists. Secondly, a subsequent assessment to confirm this diagnosis was conducted with the assistance of a physician.
The case evaluation process of negative clinical outcomes of medication (NCOM) consisted of two phases and was conducted at each hospital. Phase 1: Preliminary evaluations by pairs of pharmacists (P). Participating pharmacists were grouped in pairs (PP1) at each hospital, with each pair receiving a number of cases for evaluation. To determine the number of patients each PP1 received, the total number of patients interviewed at each hospital was divided by the number of PP at the hospital. In this phase the pairs of pharmacists evaluated the information to establish a suspected case of a NCOM. Phase 2: Final evaluation. Two pairs of assessor groups (PA), each consisting of a physician (A) and a pharmacist (P) were formed in each hospital. One of the pharmacists from the preliminary evaluation teams (PP1, -2, -3) joined each of the two assessor groups (PA) for the final evaluation to form a team of three reviewers. All cases that were evaluated in phase 1 were re-evaluated by each of the final evaluation teams (PA1+P and PA2+P). This second evaluation confirmed or rejected each suspected case of NCOM that was identified in the first phase. To confirm or reject a potential NCOM the PA1 and PA2 assessments had to match; otherwise, an independent third party consisting of a physician and two pharmacists (the team that coordinated the study) evaluated the case and made a final decision
Phase 1. Preliminary evaluations by pairs of pharmacists
Participant pharmacists were grouped in pairs at each hospital. Every pharmacist pair (PP1) received a number of cases for evaluation. To determine the number of patients each PP1 received, the total number of patients interviewed at each hospital was divided by the number of PP1s at the hospital. Pharmacists used basic and specialized bibliography (Appendix 1) in addition to medical record and patient interviews to study the effects of medications. Pharmacists used all of the information available to determine whether the health problem during the ED consultation was related to the medications the patient was taking (i.e., the suspected case of NCOM).
Phase 2. Final evaluation
Two pairs of assessors (PA1 and PA2), a physician and a pharmacist, were formed in each hospital. One of the pharmacists from the preliminary evaluation teams (PP) joined each of the two groups for the final evaluation to form a team of three reviewers. All cases that were evaluated in phase 1 were re-evaluated by each of the final evaluation teams (PA1+P and PA2+P). This second evaluation confirmed or rejected each suspected case of NCOM that was identified in the first phase. To confirm or reject a potential NCOM the PA1 and PA2 assessments had to match; otherwise, an independent third party comprising a physician and two pharmacists (the team that coordinated the study) evaluated the case and made a final decision.
Criteria were established in this study to classify a case as a NCOM of necessity, effectiveness, or safety (Table 2), as agreed upon in the Third Consensus of Granada [32]. Take, for example, the case of a patient visiting the ED who was suffering from a new health problem and who was not on medication. This case would be classified as NCOM of necessity only when the patient meeting these two criteria would have had the health problem for at least 7 days. This time frame was considered to be long enough in the Spanish National Health System for a patient to visit a family doctor or a general practitioner and therefore to obtain the medication needed.
The preventability of each NCOM was measured according to the criteria of Baena et al. [34]
The characteristics of the study population were described using the variables in Table 3. The severity of each diagnosis (mild, moderate, severe, or death) was measured according to the classification criteria of the Spanish pharmacovigilance system for adverse drug reactions [35].
The research ethics committees of the respective participating hospitals approved this study.
Statistical analysis
The prevalence and preventability of NCOMs were calculated. The chi-square (χ2) test and Student’s t test were used to compare the characteristics (for qualitative and quantitative variables, respectively) between patients who were rejected from the statistical analysis (because insufficient information was obtained with the interviews and medical records) and patients who were included in the study. The Kappa index [36] measured the level of agreement between the assessments produced by the PA1 and PA2 assessor teams for the absence/presence of a NCOM. A chi-square homogeneity test was used to assess potential differences in the prevalence of NCOMs obtained by each hospital. A multivariate analysis was performed to characterize possible risk factors for the appearance of a NCOM. The 95 % confidence interval was calculated, and a P < 0.05 value was considered for statistical significance. The SPSS statistical package for Windows ver. 15.0 (SPSS, Chicago, Il) was used to store and analyze the data.
Results
A total of 5,380 patients were established as the sample framework once we subjected the numbers of EDs visits to each hospital during the period of the study to the two-stage probabilistic sampling method. Of these, 124 patients were excluded for one of the following two reasons: (1) the patient did not wait for the physician to diagnose the condition and therefore the patient was not diagnosed (n = 103); (2) the patient exhibited voluntary acute drug poisoning (n = 21). Another 167 patients (3.1 % of the sample framework) refused to participate in the study. This group of non-collaborative patients was not analyzed further due to its low percentage. In addition, 478 participants were rejected from the statistical analysis because insufficient information was obtained in the interview or present in the medical record. Therefore, 4,611 patients formed the sample population for the study. The group of rejected participants was 8.89 % of the sample and was analyzed further to determine if rejection caused a bias in the study. The variables chosen to define the characteristics of the patients in the sample are listed in Table 3.
The patients who were rejected from the statistical analysis due to insufficient information (n = 478) differed from those patients who were included in the study in that patients in the rejected group were older (P < 0.001) and had more severe diagnoses (P < 0.001), their visits resulted in higher rates of hospital admissions (P <0.001), and a higher percentage of interviews were answered by a caregiver (P < 0.001).
The prevalence of NCOMs in the nine hospitals ranged from 17.9 (95 % CI 14.7–21.1) to 41.2 % (95 % CI 36.5–45.9); the overall prevalence was 30.7 % (95 % CI 29.5–32.1) (Table 4).
The chi-square homogeneity test used to assess potential differences in the prevalence of NCOM obtained by each hospital showed a chi-square value of 134.9 (P < 0.001). The prevalence of NCOMs was homogenous in hospitals with high involvement (>85 %) and a Kappa of >80 % (almost perfect) (Table 4) as these were the only two features that varied significantly among the hospitals. The weighted overall prevalence of NCOMs was 35.7 % (95 % CI 33.3–38.1). The weighted overall prevalence of NCOMs of necessity, ineffectiveness, and lack of safety was 14.9 (95 % CI 13.4–16.6), 18.2 (95 % CI 16.2–20.1) and 2.4 % (95 % CI 1.9–2.8), respectively (Table 4).
Of the 1,416 detected NCOMs, 10.5 % (95 % CI 8.9–12.1) led to the patient being hospitalized, 68.2 % (95 % CI 65.7–70.6) were minor health problems, 23.2 % (95 % CI 21–25.4) were moderate events, 8.5 % (95 % CI 7–9.9) were severe events, and 0.1 % (95 % CI 0.0–0.2) caused death.
In addition, of these 1,416 detected NCOMs, 81.2 % (95 % CI 80.1–82.3) could have been prevented, with the preventability of NCOMs detected at each hospital being >70 %. NCOMs related to treatment necessity could have been prevented in 98.4 % (95 % CI 97.3–99.4) of cases, while those related to ineffectiveness were preventable in 71.1 % of cases (95 % CI 67.1–75.0) and those related to a lack of safety were preventable in 50.5 % (95 % CI 37.0–63.9) of cases. The preventability of any detected NCOM varied depending on the number of medications that the patient was taking. Specifically, 100 % (χ2 = 59.5; P < 0.001) of the NCOMs that developed in patients not taking medications were preventable. In comparison, 77.8 and 77.2 % of NCOMs that appeared in patients taking between one and four medications and those taking more than five drugs, respectively, were preventable. The preventability of NCOMs was greater with less serious diagnoses, with 82.5 (χ2 = 11.63; P < 0.001) 77.6, and 70.6 % of mild, moderate, and severe NCOMs, respectively, being preventable. Two deaths caused by preventable NCOM were recorded.
The multivariate analysis revealed associations between the appearance of a NCOM and sex, self-medication, and a high-frequency smoking habit. The risk of appearance of NCOMs was higher in women than in men [adjusted odds ratio (OR) 0.8; 95 % CI 0.68–0.99]. The risk of appearance of NCOMs increased to 70 % (adjusted OR 1.70; 95 % CI 1.49–1.95) for each different physician prescribing a medication per patient. In addition, self-medicating patients had a higher risk of suffering a NCOM than those whose medication was prescribed by a family doctor or by an emergency or a specialist doctor, with the latter associated with the lowest risk (adjusted OR 0.51; 95 % CI 0.36–0.72). A high-frequency smoking habit was associated with the NCOM variable. Patients smoking more than 20 cigarettes per day showed a higher risk of suffering a NCOM (adjusted OR 1.66; 95 % CI 1.2–2.3) (Table 5).
The other variables of the multivariate analysis, namely, the number of drugs, comorbidity, knowledge of the medication, and compliance, showed no association (P > 0.05) with the main variable of the study (NCOM).
Discussion
The results of this study provide a valuable contribution to the knowledge of patients’ health problems because the data demonstrate that more than one-third of visits to EDs at Spanish hospitals are related to the use (or lack of use) of medications, a finding that has significant implications for the country’s healthcare system.
The NCOMs resulting in the highest number of ED visits were due to ineffective treatments, followed by NCOMs that resulted from the lack of required treatments. Both problems are largely due to modifiable patient behavior, such as the partial or complete compliance of treatment recommendations, and may be prevented by professional healthcare services, such as improved pharmaceutical care or health education programs [37–40].
The reliability of our results lies in the following methodologies: (1) the selection of the sample; (2) the combination of different information sources; (3) the uniform training of all researchers, interviewers, and reviewers; (4) the use of a common protocol for detecting NCOMs and the incorporation of medical assessment [31]; (5) the use of a dual assessment system for all cases and the resolution of discrepancies by a referee evaluation team. Because this study included a large number of patients from Spanish National Health System hospitals throughout Spain, the sample likely provides a reasonable approximation of the general population that visits Spanish EDs. No significant organizational differences in the various hospitals or patients from those hospitals were observed.
The data presented here are consistent with the results of a the study by Baena et al. [1] who reported a similar prevalence and distribution of NCOMs in the ED at a Spanish hospital using a similar methodology. The distribution of the different categories of NCOM (i.e., necessity, effectiveness, and safety) was similar. Other studies using comparable methods have demonstrated similar results in different healthcare scenarios [12, 14]. The problems that we found to be associated with a lack of safety are also similar to those identified in previous studies that measured these problems exclusively in EDs [8, 13, 24, 29, 41]. Importantly, the variability in the prevalence of health problems associated with medication use was less in our study than in prospective studies that used a combination of patient interviews and medical record as their source of information [1, 7–9, 12].
The analysis of preventability showed that approximately eight of ten NCOMs resulting in ED visit could be prevented, a result that is consistent with previous studies [1, 9, 12–14, 24]. These results highlight the necessity for measures to be introduced in Health Services that aim to prevent these problems. The integration of pharmacists (i.e., professionals of Health specialized in medication) into multidisciplinary healthcare teams could minimize the occurrence of NCOMs. Indeed, among our patient cohort, some of the pharmaceutical care activities that are typically performed by pharmacists could have prevented the visit to the ED. Therefore, many of these problems could have been solved in a primary care setting had pharmacists been involved in the decision-making process [37]. Importantly, the preventability of NCOMs increases in patients who did not take the medication. All cases involving patients with NCOMs but who did not take medications could have been prevented because these visits were primarily the result of the abandonment of medication (non-compliance) by the patient. This result emphasizes the importance of improving patient adherence to the treatment regimen and patient knowledge regarding their medications.
The cases that were rejected from the our study included patients who were older, exhibited more severe diagnoses and whose interview was answered by someone else. Some of these features have been previously shown to be associated with an increased risk of NCOMs [1, 8, 13]. Therefore, the inclusion of these cases in the dataset would have produced an even greater prevalence of NCOM. However, the multivariate analysis performed in this study showed no association of those factors with NCOMs.
In our study, the case of patients visiting the ED with a new health problem and not having medication would be considered to be a NCOM of necessity only if the patient suffered the health problem for at least 7 days and had not received the necessary medication prior to time of the ED visit. This time frame was considered to be long enough in the Spanish National Health System for a patient to visit a family doctor or a general practitioner and therefore to obtain the medication needed. This criterion avoided the overestimation of NCOM prevalence because the accessibility of Spanish EDs makes them the primary gateway for treatment and, consequently, we could have classified many more cases as NCOM of necessity.
The multivariate analysis showed that self-medicated and smoking women belong to the patient group with the highest risk of NCOMs.
Several studies have shown an association between sex and NCOMs, while several others have found no association between these two variables [41–43], thereby illustrating the variability in the obtained results mentioned in the Introduction. Interestingly, in those studies (with the exception of the Courtman and Stallings study [44]) that showed an association between gender and NCOM, females were found to have the higher risk of NCOMs [41–43].
The association between self-medication and NCOM variables could be explained by the implications of the self-medication—i.e., it is when the patient selects a drug without the assessment of a healthcare professional, leading to a NCOM. Self-medication is a modifiable behavior, and it is therefore important that this problem receive the appropriate attention.
The association of tobacco and NCOM could be explained based on the implied effect of substances in cigarette smoke on the metabolism of drugs, which would directly affect their effectiveness and/or their safety [45, 46]. However, a more complete study that would consider the singularities of smoking patients (ex-smoker’s diseases, time being a ex-smoker, chronic smoker vs. sporadic one, etc.) should be performed to determine the reliable association of tobacco and NCOMs.
The results of this study focused on the Spanish population, but the data may be of general interest because of the relevance to clinical practice. The prevalence of NCOMs that result in ED visits may vary between countries due to differences in healthcare systems, but this problem likely affects the healthcare systems and patients of all countries. Therefore, our study serves as a starting point for other countries to evaluate the magnitude of NCOMs within their own hospitals. An improved knowledge of the magnitude and effects of these problems on the health of the general population may encourage healthcare administrations to undertake action on these issues.
Conclusion
Approximately 35 % of ED visits are produced by NCOMs, and approximately 15 % of these visits are associated with the need for treatment, while 20 % are associated with drug ineffectiveness. Our results indicate that > 80 % of these NCOM are preventable. The magnitude of medication-related health problems and the high degree of preventability demand the establishment and implementation of patient safety strategies aimed at optimizing the use of medications.
The relevant prevalence of NCOMs determined in this study reveals the necessity of evaluation methods of public health systems. The high preventability of NCOMs that we found in our study demonstrates that it is worthwhile to implement evaluation methods that produce comparable results among European countries. This task should be taken by various health and economic European authorities, such as the European Medicines Agency and the Organization for Economic Co-operation and Development. Actions taken in this direction would benefit millions of European patients.
References
Baena MI, Faus MJ, Fajardo PC, Luque FM, Sierra F, Martinez-Olmos J et al (2006) Medicine-related problems resulting in emergency department visits. Eur J Clin Pharmacol 62:387–393
Kaushal R, Bates DW, Landrigan C, McKenna KJ, Clapp MD, Federico F et al (2001) Medication errors and adverse drug events in pediatric inpatients. JAMA 285:2114–2120
Franceschi M, Scarcelli C, Niro V, Seripa D, Pazienza AM, Pepe G et al (2008) Prevalence, clinical features and avoidability of adverse drug reactions as cause of admission to a geriatric unit: a prospective study of 1756 patients. Drug Saf 31:545–556
Lazarou J, Pomeranz BH, Corey PN (1998) Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. JAMA 279:1200–1205
De Vries EN, Ramrattan MA, Smorenburg SM, Gouma DJ, Boermeester MA (2008) The incidence and nature of in-hospital adverse events: a systematic review. Qual Saf Health Care 17:216–223
Budnitz DS, Pollock DA, Mendelsohn AB, Weidenbach KN, McDonald AK, Annest JL (2005) Emergency department visits for outpatient adverse drug events: demonstration for a national surveillance system. Ann Emerg Med 45:197–206
Hohl CM, Robitaille C, Lord V, Dankoff J, Colacone A, Pham L et al (2005) Emergency physician recognition of adverse drug-related events in elder patients presenting to an emergency department. Acad Emerg Med 12:197–205
Trifirò G, Calogero G, Ippolito FM, Cosentino M, Giuliani R, Conforti A et al (2005) Adverse drug events in emergency department population: a prospective Italian study. Pharmacoepidemiol Drug Saf 14:333–340
Zed PJ, Abu-Laban RB, Balen RM, Loewen PS, Hohl CM, Brubacher JR et al (2008) Incidence, severity and preventability of medication-related visits to the emergency department: a prospective study. CMAJ 178:1563–1569
Cohen AL, Budnitz DS, Weidenbach KN, Jernigan DB, Schroeder TJ, Shehab N et al (2008) National surveillance of emergency department visits for outpatient adverse drug events in children and adolescents. J Pediatr 152:416–421
Leendertse AJ, Egberts ACG, Stoker LJ, Van den Bemt PMLA (2008) Frequency of and risk factors for preventable medication-related hospital admissions in the Netherlands. Arch Intern Med 168:1890–1896
García V, Marquina I, Olabarri A, Miranda G, Rubiera G, Baena MI (2008) Negative results associated with medication in the emergency department of a hospital. Farm Hosp 32:157–162
Queneau P, Bannwarth B, Carpentier F, Guliana J-M, Bouget J, Trombert B et al (2007) Emergency department visits caused by adverse drug events: results of a French survey. Drug Saf 30:81–88
Ramos Linares S, Díaz Ruiz P, Mesa Fumero J, Núñez Díaz S, Suárez González M, Callejón Callejón G et al (2010) Incidence rate of adverse drug effects in a hospital emergency unit and its associated factors. Farm Hosp 34:271–278
Leape LL, Brennan TA, Laird N, Lawthers AG, Localio AR, Barnes BA et al (1991) The nature of adverse events in hospitalized patients. Results of the Harvard Medical Practice Study II. N Engl J Med 324:377–384
Wilson RM, Runciman WB, Gibberd RW, Harrison BT, Newby L, Hamilton JD (1995) The Quality in Australian Health Care Study. Med J Aust 163:458–471
Thomas EJ, Studdert DM, Newhouse JP, Zbar BI, Howard KM, Williams EJ et al (1999) Costs of medical injuries in Utah and Colorado. Inquiry 36:255–264
Davis P, Lay-Yee R, Briant R, Ali W, Scott A, Schug S (2002) Adverse events in New Zealand public hospitals I: occurrence and impact. N Z Med J 115:U271
Schiøler T, Lipczak H, Pedersen BL, Mogensen TS, Bech KB, Stockmarr A et al (2001) Incidence of adverse events in hospitals. A retrospective study of medical records. Ugeskr Laeger 163:5370–5378
Vincent C, Neale G, Woloshynowych M (2001) Adverse events in British hospitals: preliminary retrospective record review. BMJ 322:517–519
Baker GR, Norton PG, Flintoft V, Blais R, Brown A, Cox J et al (2004) The Canadian Adverse Events Study: the incidence of adverse events among hospital patients in Canada. CMAJ 170:1678–1686
Michel P, Quenon JL, Djihoud A, Tricaud-Vialle S, De Sarasqueta AM (2007) French national survey of inpatient adverse events prospectively assessed with ward staff. Qual Saf Health Care 16:369–377
Aranaz-Andrés JM, Aibar-Remón C, Vitaller-Murillo J, Ruiz-López P, Limón-Ramírez R, Terol-García E (2008) Incidence of adverse events related to health care in Spain: results of the Spanish National Study of Adverse Events. J Epidemiol Community Health 62:1022–1029
Patel KJ, Kedia MS, Bajpai D, Mehta SS, Kshirsagar NA, Gogtay NJ (2007) Evaluation of the prevalence and economic burden of adverse drug reactions presenting to the medical emergency department of a tertiary referral centre: a prospective study. BMC Clin Pharmacol 7:8
Pintor-Mármol A, Baena MI, Fajardo PC, Sabater-Hernández D, Sáez-Benito L, García-Cárdenas MV et al (2012) Terms used in patient safety related to medication: a literature review. Pharmacoepidemiol Drug Saf 21:799–809
Fernández-Llimós F, Tuneu L, Baena MI, Garcia-Delgado A, Faus MJ (2004) Morbidity and mortality associated with pharmacotherapy. Evolution and current concept of drug-related problems. Curr Pharm Des 10:3947–3967
Fernandez-Llimos F, Faus MJ (2005) From “drug-related problems” to “negative clinical outcomes”. Am J Health Syst Pharm 62(2348):2350
Tipping B, Kalula S, Badri M (2006) The burden and risk factors for adverse drug events in older patients—a prospective cross-sectional study. S Afr Med J 96:1255–1259
Juntti-Patinen L, Kuitunen T, Pere P, Neuvonen PJ (2006) Drug-related visits to a district hospital emergency room. Basic Clin Pharmacol Toxicol 98:212–217
Van Doormaal JE, Mol PGM, Van den Bemt PMLA, Zaal RJ, Egberts ACG, Kosterink JGW et al (2008) Reliability of the assessment of preventable adverse drug events in daily clinical practice. Pharmacoepidemiol Drug Saf 17:645–654
Pharmaceutical Care Research Group, University of Granada (2006) Pharmacotherapy follow-up: the Dáder method (3rd rev.; 2005). Pharm Pract 4:44–53
Committee of Consensus (2007) Third Consensus of Granada on Drug Related Problems (DRP) and Negative Outcomes associated with Medication (NOM). Ars Pharm 48:5–17
Baena MI, Fajardo P, Luque F, Marín R, Arcos A (2001) Problemas relacionados con los medicamentos en usuarios de un servicio de urgencias hospitalario: resultados de la validación de un cuestionario. Pharm Care Esp 3:345–357
Baena MI, Marín R, Martínez J, Fajardo P, Vargas J, Faus M (2002) Nuevos criterios para determinar la evitabilidad de los problemas relacionados con los medicamentos. Pharm Care Esp 4:393–396
Madurga M, De Abajo F, Martín-Serrano G, Montero D (1998) El sistema Español de farmacovigilancia. In: Nuevas perpectivas de la farmacovigilancia en España y en la Unión Europea. Grupo ISFAS, Madrid, pp 37–62
Landis JR, Koch GG (1997) The measurement of observer agreement for categorical data. Biometrics 33:159–174
Amariles P, Sabater-Hernández D, García-Jiménez E, Rodríguez-Chamorro MÁ, Prats-Más R, Marín-Magán F et al (2012) Effectiveness of Dader Method for pharmaceutical care on control of blood pressure and total cholesterol in outpatients with cardiovascular disease or cardiovascular risk: EMDADER-CV randomized controlled trial. J Manag Care Pharm 18:311–323
Gastelurrutia P, Benrimoj SI, Espejo J, Tuneu L, Mangues MA, Bayes-Genis A (2011) Negative clinical outcomes associated with drug-related problems in heart failure (HF) outpatients: impact of a pharmacist in a multidisciplinary HF clinic. J Card Fail 17:217–223
Murray MD, Young J, Hoke S, Tu W, Weiner M, Morrow D et al (2007) Pharmacist intervention to improve medication adherence in heart failure: a randomized trial. Ann Intern Med 146:714–725
Lee JK, Grace KA, Taylor AJ (2006) Effect of a pharmacy care program on medication adherence and persistence, blood pressure, and low-density lipoprotein cholesterol: a randomized controlled trial. JAMA 296:2563–2571
Budnitz DS, Pollock DA, Weidenbach KN, Mendelsohn AB, Schroeder TJ, Annest JL (2006) National surveillance of emergency department visits for outpatient adverse drug events. JAMA 296:1858–1866
Raschetti R, Morgutti M, Menniti-Ippolito F, Belisari A, Rossignoli A, Longhini P et al (1999) Suspected adverse drug events requiring emergency department visits or hospital admissions. Eur J Clin Pharmacol 54:959–963
Zhang M, Holman CDJ, Price SD, Sanfilippo FM, Preen DB, Bulsara MK (2009) Comorbidity and repeat admission to hospital for adverse drug reactions in older adults: retrospective cohort study. BMJ 338:a2752
Courtman BJ, Stallings SB (1995) Characterization of drug-related problems in elderly patients on admission to a medical ward. Can J Hosp Pharm 48:161–166
Krämer SD, Testa B (2009) The biochemistry of drug metabolism--an introduction: part 7. Intra-individual factors affecting drug metabolism. Chem Biodivers 6:1477–1660
Kroon LA (2007) Drug interactions with smoking. Am J Health Syst Pharm 64:1917–1921
Acknowledgments
The authors would like to thank Dr. Ricardo Ocaña Riola, Andalusian School of Public Health, Granada, Spain, for his statistical support. The authors would also like to thank all contributing pharmacists and especially researchers of the participating hospitals: Concepción Alvarez del Vayo Benito (University Hospital of Virgen del Rocio, Sevilla), Jose Miguel Sotoca (University Hospital Clínic of Barcelona, Barcelona), Miguel Ángel Calleja Hernández (University Hospital of Reina Sofia, Córdoba), Isabel María Muñoz Castillo (University Hospital of Carlos Haya), Rosa María Simó Martínez (University Hospital Central of Asturias, Oviedo), María Antonia Mangues Baffalluy (Hospital Santa Creu I Sant Pau, Barcelona), Montserrat Alonso Díez (Cruces University Hospital, Barakaldo), Esther Durán-García (Gregorio Marañón University Hospital, Madrid) and Beatriz Fuentes Caparrós (Infanta Margarita Hospital, Cabra, Córdoba).
This study was supported by a Research Grant from Spain’s Health Research Fund (Fondo de Investigación Sanitaria—FIS)
Author information
Authors and Affiliations
Corresponding author
Appendix 1. Basic and specialized bibliography used in the evaluation process
Appendix 1. Basic and specialized bibliography used in the evaluation process
-
1)
La Base de datos de Medicamentos y Parafarmacia (BOT) Bases de datos del medicamento y parafarmacia. (CD-room). Consejo General de Colegios Oficiales de Farmacéuticos, Rome
-
2)
Database of medicine of European Medicines Agency. Available at: http://www.ema.europa.eu/ema/
-
3)
Centro de Información online de Medicamentos de la Agencia Española de Medicamentos y Productos Sanitarios (AEMPS – CIMA). Available at: http://www.aemps.gob.es/cima/fichasTecnicas.do?metodo=detalleForm
-
4)
Sean C. Sweetman (ed) (2002) Martindale: the complete drug reference, 2nd edn. Pharmaceutical Press, London
-
5)
J. Flórez y col. (eds) (1997) Farmacología humana. 3rd edn. Masson SA, Barcelona
-
6)
Sociedad Española de Medicina de Familia y Comunitaria (2002) Guía Terapéutica en Atención Primaria, 1st edn. Semfyc ed, Barcelona
-
7)
Merck Sharp & Dohme (1999) The Merck manual of diagnosis and therapy, 17th edn. Merck Sharp & Dohme, Whitehouse Station
Rights and permissions
About this article
Cite this article
Baena, M.I., Fajardo, P.C., Pintor-Mármol, A. et al. Negative clinical outcomes of medication resulting in emergency department visits. Eur J Clin Pharmacol 70, 79–87 (2014). https://doi.org/10.1007/s00228-013-1562-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-013-1562-0