Abstract
The grazing impacts of two Acartia species (Acartia omorii and A. steueri) on size-fractionated phytoplankton biomass were measured in Jangmok Bay, Korea (34°59′37.8" N, 128°40′28.2'' E) from January to May 2015. Total chlorophyll (Chl-a) concentrations ranged from 0.66 to 5.18 µg L−1, and micro-phytoplankton (> 20 µm) comprised up to 66% (range, 10.5–65.6%) of the total pigment. The total abundance of Acartia species ranged from 267 to 5931 ind. m−3, and these copepods accounted for 20.8 to 88.0% of the total copepod abundance. The ingestion rates of A. steueri (r2 = 0.904, P = 0.013) and A. omorii (r2 = 0.239, P = 0.046) showed a high correlation with micro-phytoplankton. The average grazing impact of Acartia species on phytoplankton biomass was approximately 6.8 ± 11.8% (range, 0.1–69.0%). Temperature–salinity (T–S) diagram analysis revealed distinct environmental preferences for each species; A. omorii preferred a broader temperature range of 6.2 to 17.1 °C and a salinity range of 31.8 to 33.5, whereas A. steueri was more restricted, preferring temperatures between 6.5 and 12.8 °C and a salinity range of 32.2 to 33.5. These findings not only contribute to our understanding of the ecological roles of these copepod species in marine ecosystems but also highlight the importance of continuous research regarding the mechanisms driving their coexistence and interaction with the coastal food web.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The role of food selectivity in controlling zooplankton communities in pelagic ecosystems has been well documented (Frost 1972; Dahms and Qian 2005). Selectivity in feeding plays a crucial role in interspecies competition for food resources and influences adaptive strategies, making it an important trophic pathway for the transfer of energy and material from lower to higher trophic levels (Mauchline 1998; Bamstedt et al. 2000).
The impact of herbivorous zooplankton grazing on phytoplankton abundance and structure on has been a focus of research (Sterner 1989). Copepoda, in general, feed on phytoplankton of varying sizes and show selectivity in their feeding habits (Wilson 1973; Cowles 1979). These organisms differentiate prey based on factors, such as size, quality, and chemical cues of the prey (Frost 1972; Huntley et al. 1986; Cowles et al. 1988) on their own life cycle stages. Phytoplankton biomass and species composition are altered by their selective feeding (Bautista and Harris 1992; Edgar and Green 1994), which is a key mechanism related to the interspecies competition for food resources in pelagic food webs (Katechakis et al. 2004). The relationship between phytoplankton size and copepod feeding has been reported, with some studies suggesting a direct correlation between copepod body size and grazing effects (Harris 1982; Morales et al. 1991). However, the degree of selectivity that copepods display in their natural environment with respect to the types, sizes, and qualities of available food is not yet fully understood (Huntley 1981; Jang et al. 2010).
High abundances of the planktonic Acartiidae are commonly found in coastal and estuarine areas in Korea, China, and Japan (Kang and Kang 1998; Park et al. 2015). Acartia omorii and A. steueri are particularly abundant in the Jangmok Bay of Korea during the spring and winter seasons (Hwang et al. 2011). A. omorii adults prefer water temperatures below 23 °C, and predominated from January to early July. However, A. steueri adults are mainly found during the relatively cooler months of January to May and November to December, with the population rising notably in December when water temperatures are below 10 °C. Acartia species are omnivores and also feed on phytoplankton of various sizes and species, with adults generally associated with phytoplankton of a specific size (Rollwagen-Bollens and Penry 2003; Liu et al. 2010). Their seasonal abundance in coastal and estuarine areas can be attributed to niche separation for food (Ueda 1987; Choi et al. 2021).
The objective of this study was to investigate the feeding selectivity of two species of the genus Acartia, which would elucidate the mechanisms that promote coexistence and diversity in marine ecosystems. Hence, the role of these species in relation to changes in phytoplankton biomass and size composition was examined, and the grazing effect was calculated by combining the estimated biomass of phytoplankton in different size fractions (micro, nano, and pico).
Materials and methods
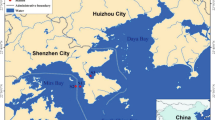
The study was conducted between January and May 2015 in Jangmok Bay (34°59′37.8" N, 128°40′28.2'' E) located in the northern part of the Geoje Island, South Sea of Korea. Sampling was conducted every 5 to 12 days at the research wharf (average depth, 8.5 m; maximum tidal range, 2.2 m) of the South Sea Research Institute of the Korea Institute of Ocean Science and Technology (KIOST) (Fig. 1).
The physical and biological features of the surface water were investigated. The water temperature (℃) and salinity were measured in situ using a YSI multimeter (model 63; Xylem Inc., Yellow Springs, OH, USA). Acartia species were collected by vertical hauling using a conical net (mesh size, 200 μm; mouth diameter, 45 cm). Specimens (Acartia omorii and A. steueri) were transferred to 20-L containers filled with surface water in a cooler and brought to the laboratory promptly. Except for March 30 and April 7, when we found a few other Acartia species, A. omorii and A. steueri made up almost all of the Acartia population. The other Acartia species we found on those two days were less than 3% of the total Acartia population. Healthy copepods were immediately isolated using a pipette with a wide opening by observing under a dissecting microscope (Zeiss Stemi SV11, Jena, Germany). Adult male and female copepods were sorted under a dissecting microscope and transferred to a 500-mL bottle containing seawater filtered through Whatman GF/F filters. Seven polycarbonate bottles (2.75 L) were filled with seawater filtered through a 200-μm mesh to remove other copepods and large grazers (Fig. 2). Each experiment was conducted using three experimental bottles, each populated with a set number of copepods (Table 1), alongside three control bottles containing only seawater filtered through a 200-μm mesh to exclude copepods. An additional bottle was set aside to establish initial conditions for measurement of chlorophyll (Chl-a) concentration. When specimens of both A. omorii and A. steueri were present simultaneously, 10 polycarbonate bottles were used. Details regarding variations in the specific Acartia species and number of individuals during the study period are provided in Table 1. In addition, the mean prosome length (PL) of adult females was measured for 10 individuals from each species. This incubation setup was housed in a custom-designed rotating incubation chamber, which ensured uniform conditions by simulating natural water movement, thereby maintaining the ecological validity of the study.
Seven polycarbonate bottles were filled to eliminate air bubbles. The incubation process was conducted over a 24-h period, during which surface water was continuously pumped from sizable acrylic tanks, and the bottles containing the specimens were rotated to ensure consistent conditions throughout the incubation period. After 24 h, water samples were collected from the polycarbonate bottles to measure Chl-a concentration and divided into three size categories (micro, nano, and pico) by size fractionation using 20-μm nylon mesh and 2-μm polycarbonate filter. To measure Chl-a concentration, 500-mL seawater from each bottle was filtered through a 47-mm Whatman GF/F filter. The filters were extracted with 90% acetone for 24 h at − 20 ℃ and subsequently analyzed by fluorometry (10 AU Fluorometer, Turner Designs, CA, USA) (Parsons et al. 1984).
The individual ingestion rates for the three size categories of Chl-a in Acartia species were calculated as follows using Frost’s equation (Frost 1972):
where F is the clearance rate (mL copepod−1 day−1), and C is the mean concentration of prey (mL−1) during the 24-h incubation period.
The clearance rate was calculated as follows:
where V is the volume (mL) of the incubation bottle, Z is the number of Acartia individuals added to the incubation bottle, t is the incubation time, C0 is the initial concentration of Chl-a, and Cc and Ce are the concentrations of prey in the experimental bottles at the end of the incubation period.
The mean concentration of prey (L−1) was calculated using the following formula:
Grazing impact (%) was calculated as the percentage of the initial concentration of Chl-a consumed by Acartia species.
Linear regression was calculated to determine the relationship between ingestion rates in copepods and phytoplankton concentrations. All the statistical analyses were performed using the IBM SPSS Statistics for Windows, version 20 (IBM Corp., Armonk, NY, USA). The significance level was set at P < 0.05.
Results
Environmental conditions
The water temperature in Jangmok Bay was 6.2–17.1 °C, with higher values in May (Table 2). The salinity of the water was 31.8–33.5, with the lowest values observed in May (Table 2). The total Chl-a concentration varied from 0.66 to 5.18 µg L−1, with the highest concentration in May and the lowest in March (Table 2). Micro-phytoplankton (> 20 μm) made up an average of 38.5 ± 16.5% of total Chl-a (range, 10.5–65.6%). Nano-phytoplankton (2–20 μm) accounted for an average of 34.1 ± 8.7% of total Chl-a (range, 15.6%–45.8%), with the highest percentage observed during March–April. The average contribution of pico-phytoplankton (< 2 μm) was 27.5 ± 14.8% of total Chl-a (range, 6.8% to 57.3%), with the highest concentration observed in April.
Occurrence of A. omorii and A. steueri
Acartiidae accounted for 20.8–88.0% of the total copepod population (Fig. 3A) with adults belonging to the species Acartia omorii and A. steueri. The abundance of Acartiidae was relatively high from January to February, contributing to more than an average of 60% of the total zooplankton, and it was low during mid-March to mid-April, accounting for an average of 26.6 ± 4.6% of the total zooplankton. The abundance of Acartiidae exhibited significant variability, ranging from 267 to 5931 ind. m−3. High abundance of more than 5000 ind. m−3 was recorded on January 21 (5931 ind. m−3), February 23 (5931 ind. m−3), and March 2 (5033 ind. m−3). The abundance of A. omorii adults ranged from 0 to 5931 ind. m−3 (mean: 2374 ± 1667 ind. m−3), with maximum abundance recorded in January and February (Fig. 3B). Except for a single occurrence at the end of March, A. omorii adults was consistently present throughout the study period. Conversely, the abundance of A. steueri adults ranged from 0 to 3733 ind. m−3 (mean: 463 ± 1027 ind. m−3), with a maximum abundance recorded for A. omorii adults, which appeared at its highest at the end of March. The temperature-salinity (T-S) diagram analysis indicated that A. omorii adults exhibit a broader tolerance range for both temperature and salinity (Fig. 4). In contrast, A. steueri adults demonstrated a more restricted pattern of occurrence, with higher abundances occurring particularly at lower temperatures (below 10 °C). Furthermore, A. steueri adults showed a preference for salinity levels between 32.2 and 33.5.
Ingestion and clearance rates
The maximum ingestion rate for total Chl-a was higher in A. steueri (396 ng Chl-a ind.−1 day−1) than that in A. omorii (170 ng Chl-a ind.−1 day−1) (Fig. 5). During the experiment, negative ingestion rates were observed once in A. steueri and three times in A. omorii during the measurement process. A. steueri showed positive ingestion rates of micro-phytoplankton (> 20 μm) throughout the experimental period, with the highest rate recorded in January 2015 (231 ng Chl-a ind.−1 day−1). The daily ingestion rate of A. omorii for micro-phytoplankton was greater than − 40 ng Chl-a ind.−1 day−1 in April, which was higher than the negative ingestion rate in other periods. The ingestion rate of nano-phytoplankton (2–20 μm) and pico-phytoplankton (< 2 μm) biomass was negative for A. steueri in all the experiments, except for that of those in January and April. This was lower than that of the total and micro-size phytoplankton. The ingestion rate for nano-phytoplankton (2–20 μm) and pico-phytoplankton (< 2 μm) biomass was more negative for A. omorii than for A. steueri.
A. omorii and A. steueri displayed higher clearance rates for total and micro-phytoplankton (> 20 μm) than those for nano-phytoplankton (2–20 μm) and pico-phytoplankton (< 2 μm) (Fig. 6). The clearance rate of total Chl-a concentration biomass ranged between − 0.64 and 3.21 mL ind.−1 day−1 for A. omorii and between − 0.06 and 8.42 mL ind.−1 day−1 for A. steueri (Fig. 6). A. steueri had positive micro-phytoplankton clearance rates throughout the experiment, with the highest rate (10.88 mL ind.−1 day−1) observed in April 2015. A. omorii had its highest micro-phytoplankton clearance rate (> 10 mL ind.−1 day−1) in March. A. steueri showed the highest nano- and pico-phytoplankton clearance rates in January, i.e., 10.93 mL ind.−1 day−1 and 6.46 mL ind.−1 day−1, respectively. However, overall, these rates were lower than the total and micro-phytoplankton clearance rates. The nano- and pico-phytoplankton clearance rates of A. omorii showed a similar pattern to the ingestion rate and were more negative than those of A. steueri.
Grazing impacts of Acartia species
The grazing impacts of Acartia species on phytoplankton varied seasonally. In some months, the grazing impact was low, while in others it was high, with consumption of < 0.1–50.5% of the initial biomass of total phytoplankton (Fig. 7). The grazing impact of A. omorii on micro-phytoplankton was 0.1–57.3%, with the highest impact in March. The grazing impact of A. steueri on micro-phytoplankton was 19.5–53.5%, with the highest impact in April. The grazing impact of both species on nano-phytoplankton (2–20 μm) and pico-phytoplankton (< 2 μm) was mostly negative, except in January and April. A. steueri had the highest impact on nano-phytoplankton (79.5%) in January.
Prosome length of Acartia species
The prosome length (PL) of Acartia omorii females ranged from 806.6 to 963.2 μm, with a mean of 873.2 ± 45.6 μm, while A. steueri females exhibited PL ranged from 865.1 to 1132.1 μm, with a mean of 1031.4 ± 72.7 μm (Fig. 8).
Discussion
The presence of congener species in overlapping ecological niches can lead to competition for resources, such as habitat, life cycle, and food. This study analyzed the feeding selectivity of adult females of two Acartia species based on the size of their phytoplankton prey. Our results revealed that micro-phytoplankton (> 20 µm) was a significant food source for both A. omorii and A. steueri, yet their ingestion rates and grazing impacts differed markedly.
Furthermore, the distinct feeding preferences of A. omorii and A. steueri observed in our study highlight the broader dietary patterns within the Acartia genus. In Ilkwang Bay, A. omorii adopts a herbivorous diet, mainly feeding on phytoplankton, as evidenced by the correlation between its production rate and Chl-a (Kang et al. 2007). However, this predominantly herbivorous tendency may not be a fixed trait for A. omorii across different environments. Acartia species, including A. steueri found in offshore waters, exhibit varied dietary behaviors in different ecological settings, ranging from omnivorous to carnivorous feeding patterns and also due to their developmental stages (Ara 2001; Kang and Kang 2005). This adaptability is crucial, particularly during periods of food scarcity, when Acartia species are known to alter their feeding strategies. Studies have indicated that Acartia species show preferences for a variety of prey, often driven by prey availability and environmental conditions. For instance, the diet of A. tonsa is diverse and comprises diatoms, dinoflagellates, heterotrophic protists, and nanoplankton, none of which is dominant prey (Kleppel and Hazzard 2000; Rollwagen-Bollens and Penry 2003). Not only when phytoplankton are scarce, Acartia species have been known to shift their diet to alternative prey, such as nauplii, ciliates, other calanoid stages, and heterotrophic dinoflagellates (Tackx and Polk 1982; Atkinson 1996; Levinsen et al. 2000; Calbet et al. 2007), or even to detritus derived from the macrophyte (Roman 1984). This flexibility in diet suggests that Acartia species, including A. omorii and A. steueri, might adjust their feeding preferences based on environmental conditions and prey availability, which is crucial for their survival, especially in areas where food sources are variable.
In our experimental design, the use of in situ temperatures was important in reflecting the natural conditions experienced by A. omorii and A. steueri in Jangmok Bay, thus ensuring the ecological validity of our findings. The difference in temperature preferences between Acartia species could significantly influence their feeding rates and behaviors (Durbin and Durbin 1992; Wlodarczyk et al. 1992). For instance, the metabolic and feeding efficiencies of A. omorii, which is dominant in cooler months, may differ from those of A. steueri, which thrives in relatively warmer conditions (Hwang et al. 2011). Previous studies showed that A. steueri appears throughout the year and reaches peak of abundance when water temperatures are above 20 °C in Ilkwang Bay, South Korea, and Sagami Bay, Japan (Jung et al. 2004; Kang and Kang 2005; Onoue et al. 2006). However, Hwang et al. (2011) found that in Jangmok Bay, A. steueri exhibits maximum densities at water temperatures below 10 °C and is absent when water temperatures exceed 20 °C. This divergence could affect their prey selection, grazing pressure, and ultimately, their roles in the marine food web, suggesting a complex interplay between local environmental conditions and species-specific adaptive strategies. Recognizing the crucial role of temperature in influencing the biological processes of these species, we also acknowledge that a comprehensive ecological assessment necessitates considering a wider array of environmental variables. Beyond water temperature, factors such as salinity, light availability, and nutrient levels are essential in fully capturing the ecological dynamics experienced by these copepods.
The accurate measurement of zooplankton feeding and grazing rates is essential. Numerous methods have been developed to measure these rates, but none has been fully effective, and their uncritical use can lead to misinterpretation of results (Peters 1984; Mauchline 1998). The incubation method used in this study has several limitations (crowding of grazers, algae growth due to excrement, and differences in turbulence) because of bottle effects (Roman and Rublee 1981; Sautour 1994). Nevertheless, it is a direct, convenient method to measure the impact of zooplankton feeding on various phytoplankton size categories (Paffenhöfer 1988; Liu et al. 2005; Olson et al. 2006). Kiørboe and Møhlenberg (1985) showed that culture methods can reliably help estimate natural feeding rates despite potential bottle effects, which can impact the in situ feeding rates of planktonic copepods. Thus, these methods are valuable for measuring the feeding impact of zooplankton on natural prey.
Ingestion and clearance rates of Acartia species fall within the range showed in Table 3. These rates were calculated as ng Chl-a ind.−1 day−1 and mL ind.−1 day−1, respectively. Although direct comparison with our study's methodology is not possible, the original units used in each study are included in the legend of the table because different units were used depending on the study. It is important to note that ingestion rates are influenced by various factors, including temperature, food size and density, body size and activity, and experimental methods (Meyer-Harms et al. 1999; Levinsen et al. 2000; Henriksen et al. 2007).
The present study showed that adults of A. omorii and A. steueri prefer phytoplankton larger than 20 μm, which is in line with other studies that have found similar food-size preferences in copepod species (Batten et al. 2001; Rollwagen-Bollens and Penry 2003; Gifford et al. 2007; Campbell et al. 2009; Fileman et al. 2010). Copepods generally feed selectively based on food size when food is abundant (Cowles 1979). Particles sized lesser than 5 μm are too small to serve as a food source and thus not preferred by some copepod species (Landry 1981; Pagano et al. 2003; Campbell et al. 2009). The present study demonstrated the limited utilization of nano- and pico-phytoplankton by Acartia adults as food sources, which was indicated by consistently negative ingestion rates for these sizes, except in January. The prey selectivity of A. steueri was decreased for nano-phytoplankton and that of both species was particularly poor for pico-phytoplankton. In addition, the negative ingestion rate of A. omorii for micro-phytoplankton (> 20 μm) suggests that Acartia species may require different diets to maintain populations in Jangmok Bay. This aligns with the concept that copepod feeding preferences and strategies are not solely influenced by prey size but also a combination of factors, including prey density, nutritional quality, and behavior. Studies have shown that copepods, including Acartia species, can adapt their diets based on environmental conditions, often switching to alternative prey, such as ciliates and other microzooplankton when phytoplankton is scarce (Calbet and Saiz 2005; Castellani et al. 2008). Yang et al. (2010) reported that A. hongi favors motile ciliates over similar-sized diatoms, indicating a preference for prey with active motility. This suggests that factors beyond prey size, such as motility and nutritional content, influence copepod feeding choices. In addition, experiments, wherein mixed diets of similar-sized Tintinnopsis angustior and Thalassiosira sp. were provided, the high filtration rate and selective feeding on certain ciliates by A. hongi could not be solely explained by prey size but rather by a selective feeding mechanism favoring quality or motile prey. Therefore, particle size alone may not fully explain selective feeding by copepods.
It is important to note that the grazing effect of copepods, including Acartia species, can vary based on region, year, and season and plays a role in controlling the composition and dynamics of phytoplankton communities in marine ecosystems (Gifford and Dagg 1988; Yang et al. 2010). The grazing pressure of the Acartia adults on total phytoplankton biomass in the present study was found to be 6.8 ± 11.8%, which is similar to that calculated in previous studies. This grazing pressure can equal or even exceed daily primary productivity, highlighting the significant role of Acartia species in marine food webs. Their selective feeding on larger phytoplankton could have important implications for the structure and dynamics of plankton communities, potentially influencing energy transfer and nutrient cycling in coastal ecosystems.
The present study demonstrated the preference of A. omorii and A. steueri for phytoplankton larger than 20 μm, which is consistent with other studies that showed similar food-size preferences in copepod species (Liu and Dagg 2003; Jang et al. 2010; Yoshida et al. 2012). A. steueri had significantly higher ingestion rates than A. omorii, and its ingestion rate was strongly correlated with food density in phytoplankton sized > 20 μm (r2 = 0.904, P = 0.013; Fig. 9). The results indicated that the high abundance of A. steueri in winter and spring can affect the biomass and composition of phytoplankton sized > 20 μm. Copepods feed more as their body size increases (Hansen et al. 1997). Paffenhöfer (1970) found that the total body length of an adult female copepod is directly related to the amount of food available during rearing. Larger copepods have greater gut volume because they require greater amount of energy for the metabolism associated with increased body sizes (Tseng et al. 2008, 2009). The variation in ingestion rates between A. steueri and A. omorii is because of the difference in their body lengths. In this study, the mean prosome length of adult females A. omorii was 873.2 ± 45.6 μm (n = 10), while that of adult females A. steueri was 1031.4 ± 72.7 μm (n = 10). By integrating observations on sex ratios, particularly on April 14 when the ratio approached 1:1, we further enriched our understanding of these dynamics. This balanced sex ratio among adults offered a pivotal moment to dissect the impact of physiological differences between the sexes on feeding behavior, especially as A. steueri demonstrated peak clearance rates and grazing pressures on micro-phytoplankton during this period. The additional energy requirements for oogenesis in adult females, necessitating a higher intake than their male counterparts (Roncalli et al. 2020), likely influenced these observed feeding behaviors and grazing pressures. This finding highlights the importance of considering both the species and size of copepods when studying their role in the food chain and energy transfer in coastal ecosystems. Further research is needed to fully understand the mechanisms associated with the differences in feeding behavior and ingestion rates in the two species, taking in consideration the different life cycle stages, different energetic needs of the two sexes, and in the same female sex in different ovogenesis periods (when producing diapause and subitaneous eggs).
Conclusions
In Jangmok Bay, the occurrence and abundance of A. omorii and A. steueri are closely linked to water temperature. T-S analysis indicated that A. omorii has a broader environmental tolerance, with a temperature range of 6.2 to 17.1 °C and a salinity spectrum of 31.8 to 33.5. However, A. steueri prefers a narrower temperature window of 6.5 to 12.8 °C and a salinity range of 32.2 to 33.5. This variation in temperature preference is critical to understanding their patterns of coexistence and competition, especially for micro-phytoplankton (> 20 µm), which is a key food resource. Our observations demonstrate the challenges that both A. omorii and A. steueri might face regarding food availability, and adaptability to temperature variations when their habitats overlap. Furthermore, this study sheds light on the adaptive feeding strategies of these species, suggesting a complex interplay between dietary preferences and environmental conditions. The findings particularly highlight A. omorii’s potential for broader dietary flexibility of adults, which may be vital for its survival in environments with limited food sources. Additional research is required to explore alternate food sources for these copepods and understand the impact of changing phytoplankton communities on their feeding behaviors and interspecies competition. Such investigations are crucial for a deeper comprehension of their ecological roles and the broader implications for the health of coastal marine ecosystems.
Data availability
Data will be made available from the corresponding authors upon reasonable request.
References
Ara K (2001) Daily egg production rate of the planktonic calanoid copepod Acartia lilljeborgi Giesbrecht in the Cananéia Lagoon estuarine system, São Paulo, Brazil. Hydrobiologia 445:205–215. https://doi.org/10.1023/A:1017573917281
Atkinson A (1996) Subantarctic copepods in an oceanic low chlorophyll environment: ciliate predation food selectivity and impact on prey population. Mar Ecol Prog Ser 130:85–86. https://doi.org/10.3354/meps130085
Ayukai T (1987) Discriminate feeding of the calanoid copepod Acartia clausi in mixtures of phytoplankton and inert particles. Mar Biol 94:579–587. https://doi.org/10.1007/BF00431404
Bamstedt U, Gifford D, Atkinson A, Roman MR, Gifford DJ (2000) Feeding. In: Harris R, Wiebe P, Lenz J, Skjoldal HR (eds) ICES Zooplankton methodology manual. Academic Press, San Diego CA
Batten SD, Fileman ES, Halvorsen E (2001) The contribution of microzooplankton to the diet of mesozooplankton in an upwelling filament off the north west coast of Spain. Prog Oceanogr 51:385–398. https://doi.org/10.1016/S0079-6611(01)00076-3
Bautista B, Harris RP (1992) Copepod gut contents, ingestion rates and grazing impact on phytoplankton in relation to size structure of zooplankton and phytoplankton during a spring bloom. Mar Ecol Prog Ser 82:41–50. https://doi.org/10.3354/meps082041
Broglio E, Johansson M, Jonsson PR (2001) Trophic interaction between copepods and ciliates: effects of prey swimming behavior on predation risk. Mar Ecol Prog Ser 220:179–186. https://doi.org/10.3354/meps220179
Calbet A, Saiz E (2005) The ciliate-copepod link in marine ecosystem. Aquat Microb Ecol 38:157–167. https://doi.org/10.3354/ame038157
Calbet A, Carlotti F, Gaudy R (2007) The feeding ecology of the copepod Centropages typicus (Kröyer). Prog Oceanogr 72(137):150. https://doi.org/10.1016/j.pocean.2007.01.003
Campbell RG, Sherr EB, Ashjian CJ, Plourde S, Sherr BF, Hill V, Stockwell DA (2009) Mesozooplankton prey preference and grazing impact in the western Arctic Ocean. Deep Sea Res Part II Top Stud Oceanogr 56:1274–1289. https://doi.org/10.1016/j.dsr2.2008.10.027
Castellani C, Irigoien X, Mayor DJ, Harris RP, Wilson D (2008) Feeding of Calanus finmarchicus and Oithona similis on the microplankton assemblage in the Irminger Sea, North Atlantic. J Plankton Res 30:1095–1116. https://doi.org/10.1093/plankt/fbn074
Choi SY, Seo MH, Soh HY (2021) Short-term variation in abundance of four Acartia species (Copepoda, Calanoida) in a eutrophic bay. Peer J 9:e10835. https://doi.org/10.7717/peerj.10835
Cowles TJ (1979) The feeding response of copepods from the Peru upwelling system: food size selection. J Mar Res 37:601–622
Cowles TJ, Olson RJ, Chisholm SW (1988) Food selection by copepods: discrimination on the basis of food quality. Mar Biol 100:41–49. https://doi.org/10.1007/BF00392953
Dahms HU, Qian PY (2005) Exposure of biofilms to meiofaunal copepods affects the larval settlement of Hydroides elegans (Polychaeta). Mar Ecol Prog Ser 297:203–214. https://doi.org/10.3354/meps297203
Durbin EG, Durbin AG (1992) Effects of temperature and food abundance on grazing and short-term weight change in the marine copepod Acartia hudsonica. Limnol Oceanogr 37(2):361–378. https://doi.org/10.4319/lo.1992.37.2.0361
Edgar NB, Green JD (1994) Calanoid copepod grazing on phytoplankton: seasonal experiments on natural communities. Hydrobiologia 273:147–161. https://doi.org/10.1007/BF00005638
Fileman E, Petropavlovsky A, Harris R (2010) Grazing by the copepods Calanus helgolandicus and Acartia clausi on the protozooplankton community at station L4 in the Western English Channel. J Plankton Res 32:709–724. https://doi.org/10.1093/plankt/fbp142
Frost BW (1972) Effects of size and concentration of food particles on feeding behavior of the marine planktonic copepod Calanus pacificus. Limnol Oceanogr 17:805–815. https://doi.org/10.4319/lo.1972.17.6.0805
Gifford DJ, Dagg MJ (1988) Feeding of the estuarine copepod Acartia tonsa Dana: carnivory vs. herbivory in natural microplankton assemblages. Bull Mar Sci 43:458–468
Gifford SM, Rollwagen-Bollens G, Bollens SM (2007) Mesozooplankton omnivory in the upper San Francisco Estuary. Mar Ecol Prog Ser 348:33–46. https://doi.org/10.3354/meps07003
Hansen PJ, Bjørnsen PK, Hansen BW (1997) Zooplankton grazing and growth: scaling within the 2–2, -μm body size range. Limnol Oceanogr 42:687–704. https://doi.org/10.4319/lo.1997.42.4.0687
Harris RP (1982) Comparison of the feeding behaviour of Calanus and Pseudocalanus in two experimentally manipulated enclosed ecosystems. J Mar Biol Assoc UK 62:71–91. https://doi.org/10.1017/S0025315400020117
Henriksen CI, Saiz E, Calbet A, Hansen BW (2007) Feeding activity and swimming patterns of Acartia grani and Oithona davisae nauplii in the presence of motile and non-motile prey. Mar Ecol Prog Ser 331:119–129. https://doi.org/10.3354/meps331119
Huntley M (1981) Nonselective, nonsaturated feeding by three calanid copepod species in the Labrador Sea1. Limnol Oceanogr 26:831–842. https://doi.org/10.4319/lo.1981.26.5.0831
Huntley M, Sykes P, Rohan S, Marin V (1986) Chemically-mediated rejection of dinoflagellate prey by the copepods Calanus pacificus and Paracalanus parvus: mechanisms, occurrence and significance. Mar Ecol Prog Ser 28:105–120
Hwang OM, Shin KS, Baek SH, Lee WJ, Kim SA, Jang MC (2011) Annual variations in community structure of mesozooplankton by short-term sampling in Jangmok Harbor of Jinhae Bay. Ocean Polar Res 33:235–253. https://doi.org/10.4217/OPR.2011.33.3.235
Isari S, Zervoudaki S, Saiz E, Pelejero C, Peters J (2015) Copepod vital rates under CO2-induced acidification: a calanoid species and a cyclopoid species under short-term exposures. J Plankton Res 37:912–922. https://doi.org/10.1093/plankt/fbv057
Jang MC, Shin K, Lee T, Noh I (2010) Feeding selectivity of calanoid copepods on phytoplankton in Jangmok Bay, south coast of Korea. Ocean Sci J 45:101–111. https://doi.org/10.1007/s12601-010-0009-0
Jung Y, Kang HK, Kang YJ (2004) In situ egg production rate of the planktonic copepod Acartia steueri in Ilkwang Bay, southeastern coast of Korea. J Plankton Res 26:1547–1553. https://doi.org/10.1093/plankt/fbh126
Kang HK, Kang YJ (1998) Egg production of the copepod Acartia steueri in Ilkwang Bay, southeastern coast of Korea. Korean J Fish Aquat Sci 31:288–295
Kang HK, Kang YJ (2005) Production of Acartia steueri (Copepoda: Calanoida) in Ilkwang Bay, southeastern coast of Korea. J Oceanogr 61:327–334. https://doi.org/10.1007/s10872-005-0043-1
Kang HK, Kang YJ, Park C (2007) Production of Acartia omorii (Copepoda: Calanoida) in Ilkwang Bay, southeastern coast of Korea. J Mar Syst 67:236–244. https://doi.org/10.1016/j.jmarsys.2006.05.014
Katechakis A, Stibor H, Sommer U, Hansen T (2004) Feeding selectivities and food niche separation of Acartia clausi, Penilia avirostris (Crustacea) and Doliolum denticulatum (Thaliacea) in Blanes Bay (Catalan Sea, NW Mediterranean). J Plankton Res 26:589–603. https://doi.org/10.1093/plankt/fbh062
Kibirige I, Perisinotto R (2003) In situ feeding rates and grazing impact of zooplankton in a South African temporarily open estuary. Mar Biol 142:357–367. https://doi.org/10.1007/s00227-002-0963-x
Kiørboe T, Møhlenberg F (1985) In situ feeding rates of plantonic copepods: a comparison of four methods. J Exp Mar Biol Ecol 88:67–81. https://doi.org/10.1016/0022-0981(85)90202-3
Kiørboe T, Tiselius PT (1987) Gut clearance and pigment destruction in a herbivorous copepod, Acartia tonsa, and determination of in situ grazing rates. J Plankton Res 9:525–534. https://doi.org/10.1093/plankt/9.3.525
Kleppel GS, Hazzard SE (2000) Diet and egg production of the copepod Acartia tonsa in Florida Bay. II. Role of the nutritional environment. Mar Biol 137:111–121. https://doi.org/10.1007/s002270000319
Landry MR (1981) Switching between herbivory and carnivory by the planktonic marine copepod Calanus pacificus. Mar Biol 65:77–82. https://doi.org/10.1007/BF00397070
Lee DB, Song HY, Park C, Choi KH (2012) Copepod feeding in a coastal area of active tidal mixing: diel and monthly variations of grazing impacts on phytoplankton biomass. Mar Ecol 33:88–105. https://doi.org/10.1111/j.1439-0485.2011.00453.x
Levinsen H, Turner JT, Nielsen TG, Hansen BW (2000) On the trophic coupling between protists and copepods in arctic marine ecosystems. Mar Ecol Prog Ser 204:65–77. https://doi.org/10.3354/meps204065
Liu H, Dagg M (2003) Interactions between nutrients, phytoplankton growth, and micro-and mesozooplankton grazing in the plume of the Mississippi River. Mar Ecol Prog Ser 258:31–42. https://doi.org/10.3354/meps258031
Liu H, Dagg MJ, Strom S (2005) Grazing by the calanoid copepod Neocalanus cristatus on the microbial foodweb in the coastal Gulf of Alaska. J Plankton Res 27:647–662. https://doi.org/10.1093/plankt/fbi039
Liu S, Li T, Huang H, Guo ZL, Huang LM, Wang WX (2010) Feeding efficiency of a marine copepod Acartia erythraea on eight different algal diets. Acta Ecol Sin 30:22–26. https://doi.org/10.1016/j.chnaes.2009.12.004
Mauchline J (1998) The biology of Calanoid copepods. In: Blaxter JHS, Southward AJ, Tyler PA (eds) Advances in marine biology. Academic Press, London, p 710
Meyer-Harms B, Irigoien X, Head R, Harris R (1999) Selective feeding on natural phytoplankton by Calanus finmarchicus before, during, and after the 1997 spring bloom in the Norwegian Sea. Limnol Oceanogr 44:154–165. https://doi.org/10.4319/lo.1999.44.1.0154
Morales CE, Bedo A, Harris RP, Tranter PRG (1991) Grazing of copepod assemblages in the north-east Atlantic: the importance of the small size fraction. J Plankton Res 13:455–472. https://doi.org/10.1093/plankt/13.2.455
Natori N, Toda T (2018) A multi-factor empirical model for calculation of naupliar ingestion rate of the embayment copepod Acartia steueri Smirnov (Copepoda: Calanoida). Mar Biol 165:1–11. https://doi.org/10.1007/s00227-018-3378-z
Olson MB, Lessard EJ, Wong CHJ, Bernhardt MJ (2006) Copepod feeding selectivity on microplankton, including the toxigenic diatoms Pseudo-nitzschia spp., in the coastal Pacific Northwest. Mar Ecol Prog Ser 326:207–220. https://doi.org/10.3354/meps326207
Onoue Y, Shimode S, Toda T, Kikuchi T (2006) Reproductive strategy of Acartia steueri in Sagami Bay, Japan. Coast Mar Sci 30:353–359. https://doi.org/10.15083/00040798
Paffenhöfer GA (1970) Cultivation of Calanus helgolandicus under controlled conditions. Helgol Meeresunters 20:346–359. https://doi.org/10.1007/BF01609912
Paffenhöfer GA (1988) Feeding rates and behavior of zooplankton. Bull Mar Sci 43:430–445
Pagano M, Kouassi E, Saint-Jean L, Arfi R, Bouvy M (2003) Feeding of Acartia clausi and Pseudodiaptomus hessei (Copepoda: Calanoida) on natural particles in a tropical lagoon (Ebrié, Côte d’Ivoire). Estuar Coast Shelf Sci 56:433–445. https://doi.org/10.1016/S0272-7714(02)00193-2
Park EO, Suh HL, Soh HY (2015) Spatio-temporal distribution of Acartia (Copepoda: Calanoida) species along a salinity gradient in the Seomjin River estuary, South Korea. J Nat Hist 49:2799–2812. https://doi.org/10.1080/00222933.2015.1022619
Parsons TR, Maita Y, Lalli CM (1984) A manual of chemical and biological methods for seawater analysis. Pergamon press, Oxford, p 173
Peters RH (1984) Methods for the study of feeding, grazing and assimilation by zooplankton. In: Downing JA, Rigler FH (eds) A manual of methods for the assessment of secondary production in freshwaters, 2nd edn. Blackwell, Oxford, pp 336–412
Rollwagen-Bollens GC, Penry DL (2003) Feeding dynamics of Acartia spp. copepods in a large, temperate estuary (San Francisco Bay, CA). Mar Ecol Prog Ser 257:139–158. https://doi.org/10.3354/meps257139
Roman MR (1984) Utilization of detritus by the copepod Acartia tonsa. Limnol Oceanogr 29:949–959. https://doi.org/10.4319/lo.1984.29.5.0949
Roman MR, Rublee PA (1981) A method to determine in situ zooplankton grazing rates on natural particle assemblages. Mar Biol 65:303–309. https://doi.org/10.1007/BF00397126
Roncalli V, Cieslak MC, Hopcroft RR, Lenz PH (2020) Capital breeding in a diapausing copepod: a transcriptomics analysis. Front Mar Sci 7:56. https://doi.org/10.3389/fmars.2020.00056
Sautour B (1994) Feeding habits of copepod plankton in the laboratory-incubation effects. Cah Biol Mar 35:113–129
Sterner RW (1989) The role of grazers in phytoplankton succession. In: Sommer U (ed) Plankton ecology: succession in plankton communities. Springer Verlag, Berlin, pp 107–170
Stoecker DK, Egloff DA (1987) Predation by Acartia tonsa Dana on planktonic ciliates and rotifers. J Exp Mar Biol Ecol 110:53–68. https://doi.org/10.1016/0022-0981(87)90066-9
Tackx M, Polk P (1982) Feeding of Acartia tonsa Dana (Copepoda, Calanoida): predation on nauplii of Canuella perplexa T. et A. Scott (Copepoda, Harpacticoida) in the sluice-dock at Ostend. Hydrobiologia 94(2):131
Tiselius P (1989) Contribution of aloricate ciliates to the diet of Acartia clausi and Centropages hamatus in coastal waters. Mar Ecol Prog Ser 56:49–56
Tseng LC, Kumar R, Dahms HU, Chen QC, Hwang JS (2008) Copepod gut contents, ingestion rates and feeding impact in relation to their size structure in the southeastern Taiwan Strait. Zool Stud 47:402–416
Tseng LC, Dahms HU, Chen QC, Hwang JS (2009) Copepod feeding study in the upper layer of the tropical South China Sea. Helgol Mar Res 63:327–337. https://doi.org/10.1007/s10152-009-0162-y
Ueda H (1987) Temporal and spatial distribution of the two closely related Acartia species A. omorii and A. hudsonica (Copepoda, Calanoida) in a small inlet water of Japan. Estuar Coast Shelf Sci 24:691–700. https://doi.org/10.1016/0272-7714(87)90107-7
Wilson DS (1973) Food size selection among copepods. Ecology 57:909–914. https://doi.org/10.2307/1935688
Wlodarczyk E, Durbin AG, Durbin EG (1992) Effect of temperature on lower feeding thresholds, gut evacuation rate, and diel feeding behavior in the copepod Acartia hudsonica. Mar Ecol Prog Ser 85:93–106. https://doi.org/10.3354/meps085093
Yamada Y, Sato W, Kobiyama A, Ogata T (2020) Feeding and reproduction including diapausing egg production as cold-water adaptations for overwintering of Acartia steueri (Copepoda, Calanoida) in Okkirai Bay, Sanriku, northern Japan. Plankton Benthos Res 15:269–278. https://doi.org/10.3800/pbr.15.269
Yang EJ, Ju SJ, Choi JK (2010) Feeding activity of the copepod Acartia hongi on phytoplankton and micro-zooplankton in Gyeonggi Bay, Yellow Sea. Estuar Coast Shelf Sci 88:292–301. https://doi.org/10.1016/j.ecss.2010.04.005
Yoshida T, Eio EJ, Toda T, Othman BHR (2012) Food size dependent feeding and egg production of Acartia pacifica from a tropical strait. Bull Mar Sci 88:251–266. https://doi.org/10.5343/bms.2011.1026
Acknowledgements
We would like to extend our sincere thanks to Professor Yen-Ju Pan from the National Taiwan Ocean University for his valuable feedback and constructive comments regarding our manuscript. His insightful suggestions have significantly contributed to the quality of this work.
Funding
This research was supported by Korea Institute of Marine Science & Technology Promotion (KIMST) funded by the Ministry of Oceans and Fisheries, Korea (RS-2018-KS181192) and the National Institute of Fisheries Sciences (NIFS) grant (Investigation of structure variations and development of assessment technology in the marine ecosystem; R2024059).
Author information
Authors and Affiliations
Contributions
SYC and HYS conceived the study and conducted the literature search. SYC, EHL, MHS, and M-C Jang collated data from the literature. SYC analyzed and visualized the data. SYC and HYS wrote the original draft of the manuscript, which was revised by all the authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
The authors declare that all applicable guidelines for sampling, care and experimental use of animals in the study have been followed.
Additional information
Responsible Editor: N. Aberle-Malzahn.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Choi, S.Y., Lee, E.H., Jang, MC. et al. Ecological implications and seasonal variability of grazing by marine copepods on phytoplankton: comparison between Acartia omorii and A. steueri in Jangmok Bay, Korea. Mar Biol 171, 125 (2024). https://doi.org/10.1007/s00227-024-04431-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-024-04431-4