Abstract
Elucidating the relative importance of niche and neutral processes in structuring ecological communities is a major goal of ecology. We considered multiple components of community structure (beta diversity, species abundance distributions, and niche specialisation) and spatial scales to identify the processes structuring marine fish communities along a tropical to temperate gradient. Beta diversity was explained by local scale environmental and spatial variables, with geographic distance and environmental gradients being stronger drivers of species turnover in tropical and temperate study areas, respectively. Species abundance distributions found a significant component of community structure in all regions to not differ from that expected by chance. Niche specialisation was more prevalent among the endemic, temperate fish species than in tropical fishes. Our results support a strong neutral component of marine fish community assembly, regardless of bioregion. A change in importance from dispersal limitation to niche filtering processes was found from tropical to temperate communities, superimposed over neutrally assembled marine fish communities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Understanding the processes by which communities are assembled and how these processes differ over spatial, environmental, or temporal gradients is one of the fundamental pursuits of community ecology (Hutchinson 1959). The contemporary belief is that niche and neutral processes exist as opposing ends of a continuum that determine community assembly (Chase and Myers 2011; Vellend et al. 2014). Both processes are involved and interact in the assembly of ecological communities, the relative importance of each is the feature often differing among ecosystems. Understanding the balance between these processes has been the motivation of many recent studies (e.g. Chase and Myers 2011; Myers et al. 2013; van der Plas et al. 2015; Viana et al. 2016). Niche processes are those such as environmental filtering and inter-specific competition (Stegen et al. 2013), where species without suitable phenotypes or traits are not able to establish or persist in a particular environment (Keddy 1992). Neutral processes are features such as ecological drift (Vellend et al. 2014), or priority effects, where early colonising species influence the success of subsequent colonisers (Fukami 2010).
Inspired by the theory of island biogeography (MacArthur and Wilson 1967), Hubbell’s (2001) unified neutral theory of biodiversity and biogeography (UNTB), in which species are deemed ecologically equivalent, is based in the neutral assembly of communities and provides a null hypothesis to test observed patterns against (Hubbell 2006). In the UNTB, differences in species composition among communities are a result of demographic stochasticity, dispersal, and speciation, as opposed to species’ ecological competitive abilities, and thus represents dispersal-based (or recruitment limited) structuring of communities (Etienne 2005; Matthews and Whittaker 2014). Hubbell’s neutral model is composed of two parameters, θ and m, the former being the ‘fundamental biodiversity number’ or richness of the species pool, and the latter a measure of dispersal limitation or immigration probability (Hubbell 2001). A further prediction of the UNTB is that species coevolving in species rich communities structured through dispersal limitation are likely to be functionally equivalent ecological generalists. This can occur as a result of conspecific individuals often being associated with different assemblages of species, and thus no consistent selection for niche differentiation, resulting in generalist species adapted to long term average environmental conditions (Hubbell 2006).
Complementary to Hubbell’s (2001) neutral theory, the lottery hypothesis of Sale (1977, 1978) proposed that community assembly of tropical reef fishes was the result of stochastic removal from and colonisation into the community by an individual. As with the UNTB, the lottery hypothesis has similarities to MacArthur and Wilson’s (1967) theory of island biogeography. Although the competitive abilities of species may differ, the existence of strong priority effects allows the persistence of an individual in a particular habitat once successfully settled from the larval phase (Ebeling and Hixon 1991). While this hypothesis implies a strong effect of stochastic processes in structuring communities, due to the random nature of vacant habitat becoming available and successful settlement from the larval pool (Hixon 2011; Sale 1978), the effect of dispersal limitation is likely to be enhanced in communities adhering to this assembly rule (Louette and De Meester 2007). Conversely, in temperate marine fish communities, niche filtering (Levin 1993) has been found to structure the communities, through either abiotic (Pérez-Matus and Shima 2010) or biotic effects (Johnson 2006).
Beta (β) diversity, the change in species composition along an environmental or spatial gradient, is the link between local [alpha (α)] and regional [gamma (γ)] diversities (Anderson et al. 2011). β diversity can be partitioned into turnover and nestedness components, the former being changes in species composition, and the latter when species in one sample are a subset of those observed in another (Baselga 2010). When combined with a null model approach, which provides the extent to which observed results deviate from that expected by chance (Chase et al. 2011), and variation partitioning methods (Legendre et al. 2009), β diversity has been advocated as a method to estimate the relative importance of neutral and niche processes in the assembly of communities (Myers et al. 2013; Tucker et al. 2016). A null model approach is necessary to compare patterns of β diversity among regions with varying γ diversity (size of species pools) (Chase and Myers 2011). However, constraining γ and α diversities in null model approaches has the potential to influence the β diversity results obtained from the null model (Ulrich et al. 2017).
Changes in β diversity associated with changes along environmental gradients have been attributed to deterministic processes (often referred to as environmental filtering), caused by differences in the relative fitness of species in different environments (Stegen et al. 2013; Vellend 2010). Changes in β diversity over geographical distances are thought to be the result of limited dispersal or spatially structured environmental variables (Hurtt and Pacala 1995; Myers et al. 2013); the former is Hubbell’s (2001) “dispersal-assembly” model. Variability in β diversity, unexplained through variance partitioning, can be attributed to neutral processes such as ecological drift, sampling effects due to different numbers of species with the potential to colonise sites within a region (species pools), and unmeasured environmental variables (Kraft et al. 2011; Myers et al. 2013; Vellend 2010; Vellend et al. 2014). Patterns in β diversity, however, cannot be directly used to infer process and should be used in conjunction with complementary approaches (Myers and LaManna 2016; Tucker et al. 2016).
Scale often has a profound effect on the processes being observed in ecological studies (Levin 1992; Trisos et al. 2014). As the spatial scale of the study increases, so does the time frame of operation of the processes responsible for the patterns observed, with broad-scale patterns resulting from speciation and extinction events, and finer-scale patterns the result of more contemporary processes (Jenkins and Ricklefs 2011). Environmental variables can appear more important than spatial variables at coarse scales, as the heterogeneity of the environment being considered will often increase as the spatial extent considered increases (Gilbert and Lechowicz 2004; Jones et al. 2006). This is observed when changes in communities corresponds more to changes in environmental variables, than geographic distance. The effects of dispersal limitation often become more apparent at finer scales due to a decrease in environmental heterogeneity relative to coarser scales, increasing the apparent importance of spatial variables (Arellano et al. 2016).
In a study of the marine fish communities along a temperate–tropical gradient associated with the Western Australian coastline, Ford et al. (2017) found distinct assemblages among the tropical locations sampled, and the temperate assemblages were defined by environmental variables. The broad-scale patterns were thought to be a result of tropical species colonising suitable environments in a region of high environmental heterogeneity, but the temperate species, many of which are endemic, have co-evolved in a region of relative geological stability and habitat homogeneity (Ford et al. 2017).
Recent studies of community assembly have typically been (i) performed on terrestrial plants and (ii) spatially restricted within a particular ‘climatic region’. Therefore, this study extends knowledge by (i) being in the marine realm, (ii) having replicate ‘locations’ within three bioregions across major climate regions—temperate to tropical, and (iii) by using an hierarchical analysis to identify the spatial scale at which geographical distance and/or environmental processes are relevant to the community assembly of marine fishes. This study was performed to reveal the scale at which assembly processes operate most strongly, and the relative importance of neutral, niche, and dispersal processes in determining the assembly of communities of marine fishes along a latitudinal gradient encompassing three bioregions. We expected that
-
(i)
based on the UNTB hypothesis of Hubbell (2001) and the lottery hypothesis of Sale (1977, 1978), neutral processes would be a strong component in the assembly of fish communities, and
-
(ii)
considering the results of Ford et al. (2017), we expected that dispersal limitation would display greater dominance in tropical locations, while niche processes would be of greater importance in temperate locations.
Materials and methods
Sampling
Fish abundances
Fish abundances were estimated through multiple recordings made with baited remote underwater video stereo systems (BRUVS) at 11 nearshore locations along the Western Australian continental shelf. Study locations were allocated to one of three bioregions: “North”, “Central”, or “South”, based on Spalding et al. (2007) and Last et al. (2011) (Fig. 1). BRUVS provide a non-extractive and consistent sampling method for demersal and benthopelagic fishes (Murphy and Jenkins 2010). The study region and data used in this study have previously been described (Ford et al. 2017), but the current study only incorporated samples from hard substrate habitats and fishes which had been identified to the species level. The analyses thus incorporated 746 samples comprising 42,637 individuals of 379 species of fishes from 71 families (Table 1).
Sample locations along Western Australian coastline. Regions defined according to Last et al. (2011)
Environmental variables
Fine-scale environmental variables were recorded for each sample site when BRUVs were deployed (depth), or derived from footage analysis (topography and benthos). Topography was classified as either “high-profile reef” (HPR), “medium-profile reef” (MPR), or “low-profile reef” (LPR). Benthos was determined as either “canopy-forming algae” (CA), “coral” (CO), “foliose algae” (FO), “seagrass” (SG), or “sessile invertebrates” (SI). Further details on topography and benthos classification are described in Ford et al. (2017).
Eighteen coarse-scale environmental variables were estimated from the BioOracle database (Tyberghein et al. 2012): sea surface temperature range (°C), mean sea surface temperature (°C), maximum sea surface temperature (°C), minimum sea surface temperature (°C), chlorophyll range (mg m−3), mean chlorophyll (mg m−3), maximum chlorophyll (mg m−3), minimum chlorophyll (mg m−3), mean photosynthetically active radiation (Einstein m−2 day−1), maximum photosynthetically active radiation (Einstein m−2 day−1), dissolved oxygen (ml L−1), mean diffuse attenuation (m−1), calcite (mol m−3), phosphate (µ mol L−1), silicate (µ mol L−1), salinity (PSS), nitrate (µ mol L−1), and pH.
Wave attributes were incorporated to represent typical magnitude of disturbances. We included: wave energy maximum period (max TE), wave energy mean period (mean TE), wave max height (max HS), and wave mean height (mean HS) created by CSIRO as part of their ‘National Marine Bioregionalisation’ project and sourced from http://www.marlin.csiro.au/. As an additional source of disturbance, cyclone frequency was included with data sourced from the International Best Track Archive for Climate Stewardship (IBTrACS) (Knapp et al. 2010). Kernel density estimation was used to convert the IBTrACS cyclone point data into grid format in QGIS 2.14.0 (Quantum GIS Development Team 2016). To place all coarse-scale environmental variables on an even spatial resolution, the layers were interpolated to ≈ 1 km2 resolution grids and values were extracted from these at the location of each sample.
Statistical analyses
We used several complementary methods to assess the relative importance of assembly processes, and the scale at which these processes acted the strongest. Variance partitioning of beta diversity was performed to assess the importance of environmental and spatial variables at regional and local scales. Species abundance distributions (SADs) and species’ ecological niche specialisation were then analysed to validate and complement the results of the variance partitioning.
Beta diversity
Following Myers et al. (2013), observed beta diversity (βobs) was calculated as the Bray–Curtis pairwise dissimilarity among samples, as dissimilarity-based metrics of β diversity are robust to changes in γ diversity and sampling intensity among regions (Anderson et al. 2011; Legendre and Cáceres 2013). Greater Bray–Curtis dissimilarity represents a greater difference in assemblage composition between samples. βobs was calculated between all sample pairs, Null models were created using 1000 randomisations of the data with the ‘permatfull’ method in the vegan package (Oksanen et al. 2017) in R V 3.2.0 (R Core Team 2017). To maintain ecological realism of the null model, species relative abundances and sample richness were fixed (both margins of the sample x species matrix were fixed), and randomisations were restricted to within study regions. Bray–Curtis dissimilarity matrices were calculated for each of the null models, the mean of the 1000 randomisations of each sample representing the expected beta diversity (βexp) of random communities. Beta diversity deviations (βdev), the standardised deviation of observed beta diversity (βobs) from that expected by chance (βexp), was calculated as
with positive βdev indicating greater beta diversity than expected by chance and negative βdev representing lower beta diversity than expected by chance. Comparisons of multivariate homogeneity of group dispersions were used to test for differences in βobs and βdev at coarse (among regions) and fine (among locations) scales. This test is analogous to Levene’s test for homogeneity of variances, and compares the average distance of samples to their group centroid among groups and can be applied to any distance/dissimilarity matrix (Anderson et al. 2006). For this test, we used the ‘betadisper’ function from the vegan package, with distances being calculated to the group centroid, and permutation tests using 9999 permutations to test for global and pairwise differences of dispersion of samples in multivariate space. Homogeneity of dispersion tests were performed first comparing regions, followed by location level comparisons.
Variation in βdev was partitioned among space, environmental variables, and the combination of space and environment (i.e. spatially structured environmental variables) using partial distance-based redundancy analysis (dbRDA) (Legendre and Anderson 1999). To incorporate the hierarchical nature of the data, the hierarchical partitioning of variance approach suggested by Cushman and McGarigal (2002) was adopted. The ‘capscale’ function in vegan was used to perform partial dbRDA with a Cailliez correction to account for negative eigenvalues (Borcard et al. 2011).
To incorporate the effect of spatial variables on βdev, Moran’s eigenvector maps (MEMs—previously called PCNM) (Dray et al. 2006) were created from the geographic co-ordinates of samples. MEMs were produced using the ‘PCNM’ algorithm in the PCNM package (Legendre et al. 2013), at both the coarse (region) and fine (locations) scales. Prior to analyses, a principal component analysis (PCA) was performed on the coarse-scale environmental variables to account for collinearity, while categorical fine-scale variables were converted to dummy binary variables.
Forward selection of explanatory variables was performed as proposed by Blanchet et al. (2008), using the ‘ordiR2step’ function of the vegan package. Initially, for each region, dbRDA was performed upon the full set of forward selected explanatory variables. Once significance of the global model was determined through the ‘anova.cca’ function in the vegan package, subsequent first-tier partial dbRDA analyses were performed on the data with fine-scale variables being partialled out to identify the variance explained by coarse-scale variables. Further partial dbRDA analysis was then performed with the coarse-scale variables being partialled out to ascertain the variance explained by fine-scale variables. For both analyses, the fine-scale variables were nested within locations. The proportion of the variance explained by the global model not explained by the sum of variance explained by coarse- and fine-scale variables was attributed to fine-scale variables being structured at the coarse scale (Borcard et al. 2011).
Second-tier partial dbRDA were then performed to partition the variance into that explained by coarse and fine-scale spatial and environmental variables. The second-tier partial dbRDA were performed again by partialling out the variation due to subsets of the explanatory variables. For example, to identify the variation explained by fine-scale environmental variables, the variability explained by fine-scale spatial, coarse-scale spatial, and coarse-scale environmental variables was partialled out (Cushman and McGarigal 2002). Again, fine-scale spatial and environmental variables were nested within locations and significance was tested using the ‘anova.cca’ function.
To ensure that the βdev created through the null model with restrained γ and α diversities was not an artefact of these diversities, we compared the βdev variation partition results to those of the turnover component of β diversity (βturn). βturn was calculated using the ‘bray.part’ function in the betapart package (Baselga et al. 2013) in R. This procedure calculates the Bray–Curtis dissimilarity among samples and then identifies the proportion of dissimilarity created by species replacement (or turnover), with the remaining dissimilarity being attributed to nestedness (Baselga 2010). Homogeneity of multivariate dispersion tests and variation partitioning of βturn were performed as for βdev.
Species abundance distributions
Species abundance distributions (SADs) were investigated to complement the variance partitioning analyses. Jabot and Chave (2011) proposed a non-neutral method which uses the unified neutral theory of Hubbell (2001) as a framework to test for non-neutrality in SADs. This method uses maximum-likelihood estimation of θ and m for each SAD, and then creates neutral SADs with values of species richness, θ, and m matching that of the observed SAD. Shannon’s evenness is then calculated for the observed SAD and that of the created neutral SADs, and the evenness of the observed SAD is compared to the distribution of evenness values of the neutral SADs. Deviation from neutrality is reported as δ, with values close to zero being neutral SADs. Positive values of δ indicate positive density dependence (abundant species have lower mortality rates), and negative values indicate negative density dependence (rare species have lower mortality rates) (Jabot and Chave 2011). We used 100,000 simulations for the approximate Bayesian computation in the Parthy software (Jabot and Chave 2011), with uniform prior distributions of ln(θ), ln(I), and δ being [0,25], [0,10], and [− 1,1], respectively, where I represents the estimated number of immigrants (Etienne 2005). Prior distributions were selected based on Jabot and Chave (2011), although with broader values of ln(θ). The natural log of priors was used, as opposed to log10, as this is the scale of θ and I at which richness and evenness respond (Jabot and Chave 2011). Densities of the distributions of δ were compared among regions using the ‘sm.density.compare’ function from the sm package (Bowman and Azzalini 2014), with holm adjustment for multiple comparisons. Jabot and Chave (2011) reported δ to not be influenced by number of species in communities of high richness; however, our samples contained fewer species than those of Jabot and Chave (2011). Within each region (log10) sample richness was regressed against values of δ to identify any influence of richness on values of δ.
With the approach of Jabot and Chave (2011), it is possible to estimate SAD departure from that expected under neutrality (δ), but θ and m cannot be estimated with this method (Jabot and Chave 2011). We used the method described by Munoz et al. (2007) to estimate θ and I, where θ is first calculated for a metacommunity (region in our case) through maximum-likelihood estimation, and then I is then calculated for each sample using the sampling formula of Etienne (2007). m can then be calculated as \( m = \frac{I}{I + J - 1} \). I and m were calculated using the untb package (Hankin 2007).
Niche specialisation
Species were placed on a continuum of niche specialisation to generalisation using the method proposed by (Fridley et al. 2007), termed θ (to avoid confusion with the SAD θ, we will refer to the generalist–specialist θ as θgs). Species niche specialisation is scored as the average value of beta diversity values of sites a species is observed in. Generalist species possess higher values of θgs as they are consistently associated or observed with different species. Specialist species score low values of θgs as they are found in communities which do not differ greatly among samples (Fridley et al. 2007). We used the multiple-site Simpson index as a measure of beta diversity. This measure is robust to changes in γ diversity (Zeleny 2008), species being found only in samples with low richness (skewed richness distributions) (Manthey and Fridley 2009), and changes in sample richness (Baselga et al. 2007). As the degree of a species’ ecological specialisation can change across environments (Fajmonová et al. 2013), analyses were performed on each region separately, using only species observed in at least ten samples. For each species, eight samples were randomly selected from the samples in which the species was observed, and the Simpson index calculated from these. This procedure was repeated 100 times to provide a measure of θgs unbiased by number of samples. Analyses were performed using the R code provided in Appendix 1 of Manthey and Fridley (2009). Differences in species’ m and θgs values were compared among regions with Kruskal–Wallis tests with subsequent pairwise tests performed using Dunn tests with Holm correction using the FSA package (Ogle 2017).
Results
Beta diversity
βobs differed among regions (Fig. 2a; homogeneity of multivariate dispersion test on average distance to medians, F = 43.514, p < 0.001) and locations (Fig. 2a; F = 21.207, p < 0.001). Pair wise comparisons revealed the North region to typically possess significantly higher βobs than the Central or South regions (Fig. 2a). This pattern is also reflected at the location level, with the greatest βobs typically observed in the northern locations and the lowest βobs typically in central locations (Fig. 2a). Differences in βdev among locations were not great, although significant (F = 9.152, p < 0.001), and significant differences were found among regions (Fig. 2b; F = 19.383, p < 0.001). βdev values were mostly positive at all locations, indicating some degree of non-random structure in the communities. Large, positive βdev outlier values at Dampier and Ningaloo are due to large, anomalous abundances of a species in a single sample, i.e. > 1200 individuals of the doubleline fusilier (Pterocaesio digramma) were recorded in one sample from Dampier. βturn demonstrated similar patterns to that of βobs and to a lesser extent βdev, indicating that species turnover is the dominant feature creating differences among samples (Fig. S1). βturn differed between regions (Fig. 2c, F = 36.988, p < 0.001), and locations (F = 14.093, p < 0.001). A common pattern observed with βobs, βdev, and βturn, with respect to among location patterns is that of greater β diversity in the North region locations, followed by a slight decrease at Shark Bay and Abrolhos Islands, a further decrease at Jurien, Rottnest Island, and Cape Naturaliste, then an increase in β diversity for the remaining South region locations (Broke Inlet, Albany, Bremer Bay, and Esperance) (Fig. 2). In addition, at the regional scale, the North region displays significantly greater βobs, βdev, and βturn, than the Central and South regions, with the Central region having significantly greater βdev than the South region. This implies greater β diversity among the northern locations, than among the southern locations, regardless of the within location β diversity (Fig. 2).
Sample pairwise dissimilarities of (a, b) Observed β diversity, (c, d) β deviations, and (e, f) β turnover. Plots to the left (a, c, e) are values within locations, plots to the right (b, d, f) are values within regions. Lowercase letters represent significance of comparisons among locations, with identical letters indicating no difference. Boxes represent first to third quartiles, and solid line in box represents median. Abbreviations are explained in Table 1 and Fig. 1. The y axis has been reduced on plot (b) for ease of interpretation, β deviation outliers at Dampier and Ningaloo extend to 73.88 and 43.42, respectively
Variance partitioning
Coarse-scale environmental attributes were characterised by PCA axes with the first two axes explaining 59.27 and 13.56% of variability, respectively, in the North region, with 99.04% explained by the first ten axes. The first two Central region PCA axes explained 56.75 and 19.04%, variability in coarse-scale environmental features, with 99.8% variability explained by the first ten axes. For the South region, the first two PCA axes explained 35.96 and 28.80% variability in coarse-scale environmental variables, respectively, with the first ten axes explaining 99.70% of variability in the coarse-scale environmental attributes (Fig. S2). The βdev variability explained by the spatial and environmental variables was 16, 21, and 22% for the North, Central, and South regions, respectively (Fig. 3). The large unexplained variability represents stochastic processes (i.e. ecological drift) and/or unmeasured environmental variables. At all regions, fine-scale variables explained more variability in βdev than coarse-scale variables. At the fine-scale in the North region, spatial variables were more significant and explained more variability in βdev than environmental variables, while at the coarse-scale spatial variables explained less variability than the (non-significant) environmental variables (Fig. 3). The Central region displayed the greatest overlap between fine and coarse-scale variables and between spatial and environmental variables, this is likely due to the Central region being in a temperate-tropical overlap zone, creating stronger spatial structuring of environmental variables. At the fine-scale in the Central region, the βdev variability explained by spatial variables was greater than that explained by environmental variables, although the environmental variables displayed greater significance. At the coarse-scale, environmental variables explained more variability than spatial variables (Fig. 3). In the South region at the fine-scale, environmental variables explained more βdev variability than spatial variables, and displayed greater significance. At the coarse scale in the South region, greater variability was explained by environmental variables, although neither environmental nor spatial variables were found to be significant (Fig. 3). Hierarchical variation partitioning of βturn provided similar results to that of βdev (Fig. S1).
Hierarchical partitioning of explainable variance of β diversity deviations attributed to space and environment at the coarse and fine scales for each region. Non-labelled components represent variation explained by a combination of either fine and coarse or spatial and environmental variables. ∙p < 0.1, *p < 0.05, **p < 0.01, ***p < 0.001
Species abundance distributions and ecological specialisation
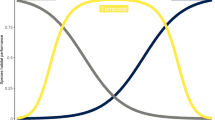
Species richness had no effect on values of δ in any of the regions (Fig. S3). The density distributions of species abundance distributions (SADs) δ values differed among regions (P < 0.001), with all pairwise comparisons being significant (North–Central, P < 0.001; North–South, P = 0.004; Central–South, P = 0.011, Fig. 4a). In all regions, more than half of the samples did not differ from the neutral expectation (North = 53.5%, Central = 52.8%, South = 60.2%) (Table S1). Values of m differed among regions (Kruskal–Wallis χ2 = 334.27, df = 2, p value < 0.001), being smallest in the North and greatest in the South regions (Fig. 4b), implying lower migration rates and thus stronger dispersal limitation in the North region. The values of θgs also differed among regions (Kruskal–Wallis χ2 = 58.172, df = 2, p value < 0.001), with species’ specialisation lowest in the North and greatest in the South region, with specialisation of Central species being intermediate (Fig. 4c).
a Density distributions of species abundance distributions (SADs) within each region. Solid portion of lines represent non-neutral SADs, and dashed portion of lines represent neutral SADs. b Values of m, with lower values of m indicating lower migration rates and thus stronger dispersal limitation, and c extent of species specialisation within each region; higher values of θgs indicate greater ecological generalisation
Discussion
The results of several analytical methods—hierarchical variance partitioning of β diversity, species abundance distributions (SADs) deviation from neutrality, estimation of degree of immigration from the regional species pool, and extent of species ecological specialisation—displayed congruence in the processes structuring Western Australian marine fish communities. As predicted, we detected (i) a large influence of neutral processes regardless of bioregion and (ii) changes in the relative importance of dispersal limitation and niche filtering along a latitudinal gradient mediating the neutral assembly of marine fish communities. Additional results included (iii) scale dependent importance of variables in structuring marine fish communities, and (iv) species’ ecological specialisation increased from fish associated with tropical regions to fish found in temperate regions. Therefore, the results of this study indicate a combination of niche and neutral processes operating in the assembly of Western Australian marine fish communities.
The processes involved in the assembly of Western Australian marine fish communities appears to operate most at the local scale, where species interactions with their biotic and abiotic environments occur. Neutral processes are a prominent process in the assembly of the marine fish of all communities studied. The relatively small β deviation values in this study indicate that changes in community composition do not differ greatly from that expected by chance. This is further supported by most SADs not deviating from that expected by a neutral model, and the large degree of unexplainable variability in the variation partitioning analyses. Unexplained variability may be due to unmeasured variables, but in concert with small β deviation values, and the SADs neutrality, the prevalence of ecological drift in structuring the fish communities is strongly suggested (Vellend 2010; Vellend et al. 2014). The nature of larval dispersal and settlement in the marine realm supports the existence of strong stochastic forces in the assembly of many marine fish communities (Cowen and Sponaugle 2009; Siegel et al. 2008). This effect is likely to be compounded by the existence of priority effects (Geange et al. 2017) and the random creation of available habitat through disturbances such as storms (Sousa 1984) or predation (Stier et al. 2014). While some of the unexplained variability in the variation partitioning analyses is likely to be due to unmeasured variables (i.e. large-scale ocean current movement or habitat features at a finer resolution than used here), the coarse-scale environment would be well represented by the variables included in the variation partitioning, with any additional environmental variables likely to be correlated with those incorporated. Furthermore, while the resolution of the fine-scale environmental variables may be too coarse to detect features such as species-specific associations between fish and benthos, temperate algal assemblages have been shown to differ along substrate topography and depth gradients (Kendrick et al. 2004), and coral communities often demonstrate turnover along depth gradients (Harborne et al. 2006). Both substrate topography and depth were included as environmental variables, therefore, while not directly measured, these species-specific associations were not completely unaccounted for.
Fine-scale variables explained greater variation in β diversity than coarse-scale variables in all regions, implying that the non-stochastic component of west Australian marine fish community assembly is strongly due to niche processes (species interactions with their abiotic and biotic environments) and dispersal limitation at a local scale. The importance of spatial variables in the variance partitioning decreased from the tropical region to the temperate region, with the importance of environmental variables demonstrating an opposing pattern. Similarly, species immigration from the regional species pool (m) increased from the tropics to the temperate zone. This implies a decrease in the importance of dispersal/recruitment limitation and an increase in the effect of niche processes in the assembly of fish communities along the latitudinal gradient (Myers et al. 2013). DiBattista et al. (2017) recently reported isolation by distance (IBD) among populations of stripey snapper (Lutjanus carponotatus) throughout Northwestern Australia, implying restricted dispersal of larvae within the region. Chust et al. (2016) compared the distance decay between community level (β diversity) and genetic levels (FST and IBD) of diversity of marine macroinvertebrate and planktonic species, finding patterns observed at the genetic level were reflected at the community level. This finding, in conjunction with the results of DiBattista et al. (2017), supports the existence of dispersal/recruitment limitation in structuring tropical Western Australian marine fishes.
Dispersal/recruitment limitation was estimated by the importance of spatial variables in the variation partitioning analysis and the calculation of m. Both measures identified dispersal limitation to be strongest in the North region and weakest in the South. Dispersal limitation appears to have greater influence in the assembly of fish communities in the tropical region, while species interactions with their environment and other fish species to be responsible for the assembly of fish communities in the temperate region. Studies have found the extent of dispersal of tropical marine species to often be less than that of their temperate counterparts (Bradbury et al. 2008; Brown 2014), although the variables often attributed to this feature are confounded with latitude (i.e. temperature and habitat fragmentation) (Leis et al. 2013). Regardless of the driving variable, decreased larval dispersal or successful recruitment by tropical species is consistent with the increased importance of spatial variables and lower values of m in the tropical region in this study. The increased importance of environmental variables in the temperate region implies stronger deterministic processes in the assembly of temperate marine fish assemblages. The analyses used in this study do not allow for the elucidation of the nature of deterministic processes involved as this result may also be due to abiotic (environmental filtering) or biotic (competition/facilitation) processes (HilleRisLambers et al. 2012; Kraft et al. 2015). Greater ecological specialisation of the temperate fishes is also congruent with stronger niche processes acting in the South region. Relative to the tropical species, the temperate species are typically observed with the same suite of species, implying the importance of habitat filtering and/or species interactions in the assembly of these fish communities. Conversely, the greater ecological generalisation of the tropical species supports the neutral theory of Hubbell (2001). In this scenario, conspecifics in species rich communities are observed with suites of different species. Over evolutionary timescales there is no consistent selective force acting upon a species, resulting in generalist species adapted to the average environmental conditions of the regions in which they are found (Hubbell 2006).
A feature of note arises from Fig. 2. The classification of locations into (bio) regions in this study is based on the bioregionalisation of Last et al. (2011), and the allocation of the Cape Naturaliste location within the South region may be due to taxonomic similarities more so than aspects of ecosystem functioning. The values of βobs, βdev, and βturn for this location (Fig. 2) are more consistent with of the Central region than the South region, suggesting processes operating within this location are more similar to that of the Central than the South region. This is also in contrast to the pattern observed by Ford et al. (2017), where the Cape Naturaliste location was included with the South locations based on species abundances. A related aspect of this feature is the similarity of these locations with respect to β diversity patterns and the orientation of the coastline where they are located. Reduced values of βobs, βturn, and, to a lesser extent, βdev are found within locations associated with the west facing coastline. Further investigations will be required to elucidate the drivers of this pattern.
To our knowledge, this study is the first to apply the suite of analytical methods used here to identify the processes assembling marine fish communities over a latitudinal gradient. This study found support for the UNTB and lottery hypotheses of Hubbell (2001) and Sale (1977, 1978) in structuring temperate and tropical west Australian marine fish communities. However, fine-scale variables (local spatial structure and reef topography and/or benthos) are more important in the assembly of communities than coarse-scale variables (i.e. gradients in sea surface temperature and primary productivity), regardless of bioregion, with dispersal limitation and niche processes structuring tropical and temperate communities, respectively. A recent study by Janzen et al. (2017) also found a dominance of neutrality in structuring cichlid communities at Lake Tanganyika, in Zambia, with secondary importance of niche processes.
When β diversity is primarily a result of species turnover, as opposed to nestedness, broader geographical scale conservation measures are more likely to be effective than focussing primarily on locations possessing greater species richness (Si et al. 2015). In addition, when dispersal limitation is a primary force at play in structuring communities, conservation and/or management activities should consider the degree of connectivity among the communities/populations of interest (DiBattista et al. 2017).
To our knowledge, this is the first study which has compared the relative importance of processes involved in the assembly of Western Australian marine fish communities among bioregions. Neutrality was found to be a major component of community structure in both tropical and temperate environments. Dispersal limitation and environmental filtering were important processes in the tropical and temperate communities, respectively, and acted strongest at the local, not regional scale. Not only do these results expand our understanding of the functioning of marine ecosystems, but also provide information which can be used in management of the marine realm for fisheries and marine protected areas.
References
Anderson MJ, Ellingsen KE, McArdle BH (2006) Multivariate dispersion as a measure of beta diversity. Ecol Lett 9:683–693
Anderson MJ, Crist TO, Chase JM, Vellend M, Inouye BD, Freestone AL, Sanders NJ, Cornell HV, Comita LS, Davies KF (2011) Navigating the multiple meanings of β diversity: a roadmap for the practicing ecologist. Ecol Lett 14:19–28
Arellano G, Tello JS, Jørgensen PM, Fuentes AF, Loza MI, Torrez V, Macía MJ (2016) Disentangling environmental and spatial processes of community assembly in tropical forests from local to regional scales. Oikos 125:326–335
Baselga A (2010) Partitioning the turnover and nestedness components of beta diversity. Glob Ecol Biogeogr 19:134–143
Baselga A, Jimenez-Valverde A, Niccolini G (2007) A multiple-site similarity measure independent of richness. Biol Let 3:642–645
Baselga A, Orme D, Villeger S, De Bortoli J, Leprieur F (2013) betapart: partitioning beta diversity into turnover and nestedness components. R package version 1.3. https://CRAN.R-project.org/package=betapart
Blanchet FG, Legendre P, Borcard D (2008) Forward selection of explanatory variables. Ecology 89:2623–2632. https://doi.org/10.1890/07-0986.1
Borcard D, Gillet F, Legendre P (2011) Numerical ecology with R. Springer, New York
Bowman AW, Azzalini A (2014) R package ‘sm’: nonparametric smoothing methods (version 2.2–5.4). http://www.stats.gla.ac.uk/~adrian/sm, http://azzalini.stat.unipd.it/Book_sm
Bradbury IR, Laurel B, Snelgrove PVR, Bentzen P, Campana SE (2008) Global patterns in marine dispersal estimates: the influence of geography, taxonomic category and life history. Proc R Soc B Biol Sci 275:1803–1809. https://doi.org/10.1098/rspb.2008.0216
Brown JH (2014) Why marine islands are farther apart in the tropics. Am Nat 183:842–846
Chase JM, Myers JA (2011) Disentangling the importance of ecological niches from stochastic processes across scales. Philos Trans R Soc B Biol Sci 366:2351–2363
Chase JM, Kraft NJB, Smith KG, Vellend M, Inouye BD (2011) Using null models to disentangle variation in community dissimilarity from variation in a-diversity. Ecosphere 2:art24. https://doi.org/10.1890/es1810-00117.00111
Chust G, Villarino E, Chenuil A, Irigoien X, Bizsel N, Bode A, Broms C, Claus S, Fernández de Puelles ML, Fonda-Umani S, Hoarau G, Mazzocchi MG, Mozetič P, Vandepitte L, Veríssimo H, Zervoudaki S, Borja A (2016) Dispersal similarly shapes both population genetics and community patterns in the marine realm. Sci Rep 6:28730
Cowen RK, Sponaugle S (2009) Larval dispersal and marine population connectivity. Annu Rev Mar Sci 1:443–466
Cushman SA, McGarigal K (2002) Hierarchical, multi-scale decomposition of species-environment relationships. Landsc Ecol 17:637–646
DiBattista JD, Travers MJ, Moore GI, Evans RD, Newman SJ, Feng M, Moyle SD, Gorton RJ, Saunders T, Berry O (2017) Seascape genomics reveals fine-scale patterns of dispersal for a reef fish along the ecologically divergent coast of Northwestern Australia. Mol Ecol. https://doi.org/10.1111/mec.14352
Dray S, Legendre P, Peres-Neto PR (2006) Spatial modelling: a comprehensive framework for principal coordinate analysis of neighbour matrices (PCNM). Ecol Model 196:483–493
Ebeling AW, Hixon MA (1991) Tropical and temperate reef fishes: comparison of community structures. In: Sale PF (ed) The ecology of fishes on coral reefs. Academic, San Diego
Etienne RS (2005) A new sampling formula for neutral biodiversity. Ecol Lett 8:253–260
Etienne RS (2007) A neutral sampling formula for multiple samples and an ‘‘exact’’ test of neutrality. Ecol Lett 10:608–618
Fajmonová Z, Zelený D, Syrovátka V, Vončina G, Hájek M (2013) Distribution of habitat specialists in semi-natural grasslands. J Veg Sci 24:616–627
Ford BM, Stewart BA, Roberts JD (2017) Species pools and habitat complexity define west Australian marine fish community composition. Mar Ecol Prog Ser 574:157–166. https://doi.org/10.3354/meps12167
Fridley JD, Vandermast DB, Kuppinger DM, Manthey M, Peet RK (2007) Co-occurrence based assessment of habitat generalists and specialists: a new approach for the measurement of niche width. J Ecol 95:707–722
Fukami T (2010) Community assembly dynamics in space. In: Verhoef HA, Morin PJ (eds) Community ecology: processes, models, and applications. Oxford University Press, Oxford, pp 45–54
Geange SW, Poulos DE, Stier AC, McCormick MI (2017) The relative influence of abundance and priority effects on colonization success in a coral-reef fish. Coral Reefs 36:151–155. https://doi.org/10.1007/s00338-016-1503-3
Gilbert B, Lechowicz MJ (2004) Neutrality, niches, and dispersal in a temperate forest understory. Proc Natl Acad Sci USA 101:7651–7656
Hankin RKS (2007) Introducing untb, an R package for simulating ecological drift under the unified nuetral theory of biodiversity. J Stat Softw 22:1–18
Harborne AR, Mumby PJ, Żychaluk K, Hedley JD, Blackwell PG (2006) Modeling the beta diversity of coral reefs. Ecology 87:2871–2881
HilleRisLambers J, Adler P, Harpole W, Levine J, Mayfield M (2012) Rethinking community assembly through the lens of coexistence theory. Annu Rev Ecol Evol Syst 43:227
Hixon MA (2011) 60 years of coral reef fish ecology: past, present, future. Bull Mar Sci 87:727–765
Hubbell SP (2001) The unified neutral theory of biodiversity and biogeography. Monographs in population biology, vol 32. Princeton University Press, Princeton
Hubbell SP (2006) Neutral theory and the evolution of ecological equivalence. Ecology 87:1387–1398
Hurtt GC, Pacala SW (1995) The consequences of recruitment limitation: reconciling chance, history and competitive differences between plants. J Theor Biol 176:1–12
Hutchinson GE (1959) Homage to Santa Rosalia or why are there so many kinds of animals? Am Nat 93:145–159
Jabot F, Chave J (2011) Analyzing tropical forest tree species abundance distributions using a nonneutral model and through approximate Bayesian inference. Am Nat 178:E37–E47
Janzen T, Alzate A, Muschick M, Maan ME, Plas F, Etienne RS (2017) Community assembly in Lake Tanganyika cichlid fish: quantifying the contributions of both niche-based and neutral processes. Ecol Evol 7:1057–1067
Jenkins DG, Ricklefs RE (2011) Biogeography and ecology: two views of one world. Philos Trans R Soc B Biol Sci 366:2331–2335. https://doi.org/10.1098/rstb.2011.0064
Johnson DW (2006) Predation, habitat complexity, and variation in density-dependent mortality of temperate reef fishes. Ecology 87:1179–1188
Jones MM, Tuomisto H, Clark DB, Olivas P (2006) Effects of mesoscale environmental heterogeneity and dispersal limitation on floristic variation in rain forest ferns. J Ecol 94:181–195
Keddy PA (1992) Assembly and response rules: two goals for predictive community ecology. J Veg Sci 3:157–164. https://doi.org/10.2307/3235676
Kendrick GA, Harvey ES, Wernberg T, Harman N, Goldberg N (2004) The role of disturbance in maintaining diversity of benthic macroalgal assemblages in southwestern Australia. Jpn J Phycol 52(Supplement):5–9
Knapp KR, Kruk MC, Levinson DH, Diamond HJ, Neumann CJ (2010) The International Best Track Archive for Climate Stewardship (IBTrACS): unifying tropical cyclone best track data. Bull Am Meteor Soc 91:363–376
Kraft NJ, Comita LS, Chase JM, Sanders NJ, Swenson NG, Crist TO, Stegen JC, Vellend M, Boyle B, Anderson MJ (2011) Disentangling the drivers of β diversity along latitudinal and elevational gradients. Science 333:1755–1758
Kraft NJ, Adler PB, Godoy O, James EC, Fuller S, Levine JM (2015) Community assembly, coexistence and the environmental filtering metaphor. Funct Ecol 29:592–599
Last PR, White WT, Gledhill DC, Pogonoski JJ, Lyne V, Bax NJ (2011) Biogeographical structure and affinities of the marine demersal ichthyofauna of Australia. J Biogeogr 38:1484–1496
Legendre P, Anderson MJ (1999) Distance-based redundancy analysis: testing multispecies responses in multifactorial ecological experiments. Ecol Monogr 69:1–24
Legendre P, Cáceres M (2013) Beta diversity as the variance of community data: dissimilarity coefficients and partitioning. Ecol Lett 16:951–963
Legendre P, Mi XC, Ren HB, Ma KP, Yu MJ, Sun IF, He FL (2009) Partitioning beta diversity in a subtropical broad-leaved forest of China. Ecology 90:663–674. https://doi.org/10.1890/07-1880.1
Legendre P, Borcard D, Blanchet FG, Dray S (2013) PCNM: MEM spatial eigenfunction and principal coordinate analyses. R package version 2.1-2/r109. https://R-Forge.R-project.org/projects/sedar/
Leis JM, Caselle JE, Bradbury IR, Kristiansen T, Llopiz JK, Miller MJ, O’Connor MI, Paris CB, Shanks AL, Sogard SM, Swearer SE, Treml EA, Vetter RD, Warner RR (2013) Does fish larval dispersal differ between high and low latitudes? Proc R Soc B Biol Sci 280:20130327. https://doi.org/10.1098/rspb.2013.0327
Levin SA (1992) The problem of pattern and scale in ecology: the Robert H. MacArthur award lecture. Ecology 73:1943–1967
Levin PS (1993) Habitat structure, conspecific presence and spatial variation in the recruitment of a temperate reef fish. Oecologia 94:176–185
Louette G, De Meester L (2007) Predation and priority effects in experimental zooplankton communities. Oikos 116:419–426
MacArthur RH, Wilson EO (1967) The theory of island biogeography. Princeton University Press, Princeton
Manthey M, Fridley JD (2009) Beta diversity metrics and the estimation of niche width via species co-occurrence data: reply to Zeleny. J Ecol 97:18–22
Matthews TJ, Whittaker RJ (2014) Neutral theory and the species abundance distribution: recent developments and prospects for unifying niche and neutral perspectives. Ecol Evol 4:2263–2277
Munoz F, Couteron P, Ramesh BR, Etienne RS (2007) Estimating parameters of neutral communities: from one single large to several small samples. Ecology 88:2482–2488
Murphy HM, Jenkins GP (2010) Observational methods used in marine spatial monitoring of fishes and associated habitats: a review. Mar Freshw Res 61:236–252. https://doi.org/10.1071/MF09068
Myers JA, LaManna JA (2016) The promise and pitfalls of β-diversity in ecology and conservation. J Veg Sci 27:1081–1083
Myers JA, Chase JM, Jiménez I, Jørgensen PM, Araujo-Murakami A, Paniagua-Zambrana N, Seidel R (2013) Beta-diversity in temperate and tropical forests reflects dissimilar mechanisms of community assembly. Ecol Lett 16:151–157
Ogle DH (2017) FSA: fisheries stock analysis. R package version 0.8.13
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2017) vegan: community Ecology Package. R package version 2.4–3. https://CRAN.R-project.org/package=vegan
Pérez-Matus A, Shima JS (2010) Disentangling the effects of macroalgae on the abundance of temperate reef fishes. J Exp Mar Biol Ecol 388:1–10
Quantum GIS Development Team (2016) QGIS Geographic Information System. Open Source Geospatial Foundation Project. http://qgis.osgeo.org
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org/
Sale PF (1977) Maintenance of high diversity in coral reef fish communities. Am Nat 111(978):337–359
Sale PF (1978) Coexistence of coral reef fishes—a lottery for living space. Environ Biol Fishes 3:85–102
Si X, Baselga A, Ding P (2015) Revealing beta-diversity patterns of breeding bird and lizard communities on inundated land-bridge islands by separating the turnover and nestedness components. PLoS One 10:e0127692
Siegel D, Mitarai S, Costello C, Gaines S, Kendall B, Warner R, Winters K (2008) The stochastic nature of larval connectivity among nearshore marine populations. Proc Natl Acad Sci 105:8974–8979
Sousa WP (1984) The role of disturbance in natural communities. Annu Rev Ecol Syst 15:353–391
Spalding MD, Fox HE, Allen GR, Davidson N, Ferdaña ZA, Finlayson M, Halpern BS, Jorge MA, Lombana A, Lourie SA (2007) Marine ecoregions of the world: a bioregionalization of coastal and shelf areas. Bioscience 57:573–583
Stegen JC, Freestone AL, Crist TO, Anderson MJ, Chase JM, Comita LS, Cornell HV, Davies KF, Harrison SP, Hurlbert AH (2013) Stochastic and deterministic drivers of spatial and temporal turnover in breeding bird communities. Glob Ecol Biogeogr 22:202–212
Stier AC, Hanson KM, Holbrook SJ, Schmitt RJ, Brooks AJ (2014) Predation and landscape characteristics independently affect reef fish community organization. Ecology 95:1294–1307
Trisos CH, Petchey OL, Tobias JA (2014) Unraveling the interplay of community assembly processes acting on multiple niche axes across spatial scales. Am Nat 184:593–608
Tucker CM, Shoemaker LG, Davies KF, Nemergut DR, Melbourne BA (2016) Differentiating between niche and neutral assembly in metacommunities using null models of β-diversity. Oikos 125:778–789
Tyberghein L, Verbruggen H, Pauly K, Troupin C, Mineur F, De Clerck O (2012) Bio-ORACLE: a global environmental dataset for marine species distribution modelling. Glob Ecol Biogeogr 21:272–281
Ulrich W, Baselga A, Kusumoto B, Shiono T, Tuomisto H, Kubota Y (2017) The tangled link between β-and γ-diversity: a Narcissus effect weakens statistical inferences in null model analyses of diversity patterns. Glob Ecol Biogeogr 26:1–5
van der Plas F, Janzen T, Ordonez A, Fokkema W, Reinders J, Etienne RS, Olff H (2015) A new modeling approach estimates the relative importance of different community assembly processes. Ecology 96:1502–1515
Vellend M (2010) Conceptual synthesis in community ecology. Q Rev Biol 85:183–206
Vellend M, Srivastava DS, Anderson KM, Brown CD, Jankowski JE, Kleynhans EJ, Kraft NJ, Letaw AD, Macdonald AAM, Maclean JE (2014) Assessing the relative importance of neutral stochasticity in ecological communities. Oikos 123:1420–1430
Viana DS, Figuerola J, Schwenk K, Manca M, Hobæk A, Mjelde M, Preston C, Gornall R, Croft J, King R (2016) Assembly mechanisms determining high species turnover in aquatic communities over regional and continental scales. Ecography 39:281–288
Zeleny D (2008) Co-occurrence based assessment of species habitat specialization is affected by the size of species pool: reply to Fridley et al. (2007). J Ecol 97:10–17
Acknowledgements
Data from Esperance, Bremer Bay, Albany, Rottnest Island, and Jurien and the Abrolhos Islands were collected through funding provided by an Australian and Western Australian Government Natural Heritage Trust Strategic Project, ‘Securing Western Australia’s Marine Futures’. We thank South Coast Natural Resource Management for access to the data and the staff of the Marine Futures team who collected the data. Dampier data was collected for Woodside Energy, who is thanked for access to this data. Ben Fitzpatrick is thanked for providing the Ningaloo data, Jock Clough is thanked for providing the Shark Bay data, and Helen Shortland-Jones is thanked for collating the data. Bronte Van Helden is thanked for comments on a draft.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Additional information
Responsible Editor: K. D. Clements.
Reviewed by Undisclosed experts.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ford, B.M., Roberts, J.D. Latitudinal gradients of dispersal and niche processes mediating neutral assembly of marine fish communities. Mar Biol 165, 94 (2018). https://doi.org/10.1007/s00227-018-3356-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-018-3356-5