Abstract
Proanthocyanidins (PAs) in the hot water extract prepared from Pinus radiata bark were isolated by a Sephadex LH-20 chromatography. The PAs were characterized using UV/Visible, FT-IR and 13C nuclear magnetic resonance (NMR) spectroscopic techniques, matrix-assisted laser desorption/ionization time of flight (MALDI-TOF) mass spectrometry (MS), and gel permeation chromatography (GPC) analysis. Bate-Smith acidic alcoholysis and FT-IR spectrum of the PAs indicated the predominant presence of the procyanidin (PC) structure. The 13C NMR spectrum revealed that the PAs comprise PC (94%) together with a small amount of prodelphinidin (PD, 6%), and also that they contain the higher trans configuration rather than the cis configuration of C2–C3. The MALDI-TOF MS results obviously proved the presence of PD units, but the maximum degree of polymerization (DP) was limited to octamer. The average molar mass (Mw, Mn), polydispersity (Mw/Mn), and DP of the pine bark PAs were 3,800 (Mw), 1,200 (Mn), 3.2, and 13, respectively.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
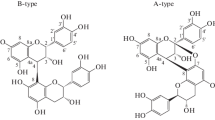
Proanthocyanidins (PAs) are distributed ubiquitously in both gymnosperms and angiosperms (Scalbert et al. 1989). They are considered as an important class of secondary metabolites in the plant kingdom. PAs belong to the category known as condensed tannins, one of the two main categories of plant tannins (De Bruyne et al. 1999). PAs are polyphenolic natural products composed of flavan-3-ol sub-units linked mainly through C4–C8 (or C6) bonds (Kennedy and Taylor 2003, Fig. 1). The structural diversity of PAs is due to the different sub-units, interflavonoid bond position, branching and the presence of non-flavonoid substituents such as gallic acid and sugars (Tanner et al. 2003). In addition, PAs can also vary markedly in molar mass distribution. The extractable PA polymers in the plant kingdom may be composed of molecular species with a wide range of molar masses up to 20,000 (∼40 flavan-3-ol units) (Haslam 1989). PAs can be categorized by molar mass as oligomeric PAs consisting of 2–6 flavan-3-ol units and polymeric PAs consisting of more than its hexamer (Haslam 1998). The bioactivity capacity of PAs is generally recognized to be largely dependent on their structure and particularly the degree of polymerization (DP) (Svedström et al. 2002).
Recently, PAs have received considerable attention in the fields of nutrition, health, and medicine largely due to their physiological activity, such as anti-oxidant activity (Bors et al. 2000), anti-microbial effects (Cowan 1999), anti-inflammatory properties (Santos-Buelga and Scalbert 2000), application in cardiovascular diseases (Maffei Facinó et al. 1996), anti-allergy activity (Bagchi et al. 2000), and inhibitory activity against some enzymes, such as phospholipase A2, cyclooxygenase and lipoxygenase, and some receptors (Zhu 1997).
We are especially interested in PAs extracted from pine bark as they constitute an abundant waste source in the wood industry, in addition to the long history of pine bark extract as a folk medicine (Drehsen 1999), which still finds therapeutic use as a dietary supplement in Europe (Karonen et al. 2004). In addition, recent evidence shows that the pine bark PAs may have health benefits (Arcangeli 2000; Grimm et al. 2004; Virgili et al. 2000). However, PAs exist in pine bark as extractable and non-extractable type with solvents depending on whether it is combined with carbohydrate matrix or not. The quantitative ratio of these types in pine bark has been considered to be due to climatic conditions. The amount of extractable PAs is a significantly affecting factor in estimating an applicability of bark among Pinus species. It was reported that Pinus radiata bark is a good resource to produce large amounts of PA-rich HWE with potent anti-oxidant activity (Mun and Ku 2006). PAs in the HWE is the most active group in anti-oxidant activity, but the chemical characteristics of the PAs are still not completely elucidated, to date. Accordingly, in the present study, the HWE was purified by a Sephadex LH-20 chromatography to isolate PAs and its structure characterized mainly by focusing on a non-destructive instrumentation.
Materials and methods
Preparation of HWE
Pinus radiata bark, obtained from Sawmilling Co. Ltd., Christchurch, New Zealand, was dried at 60°C for 48 h and ground in a Wiley mill. The bark powder (o.d. 600 g, 20–80 mesh) was extracted with 6 L of deionized water for 1 h at 100°C. The bark was immediately transferred into a cotton cloth bag and squeezed followed by washing with 16 L of deionized water. The extract was filtrated by a 3 μm-filter cartridge. The filtrate was evaporated under reduced pressure using an Eyela N-12 rotary evaporator at 65°C. The concentrate was lyophilized for 2 days and then vacuum-dried for 2 days under P2O5.
Preparation of PA
Proanthocyanidins were purified from P. radiata bark with slight modification of the extraction and isolation scheme suggested by Ohara et al. (1994). About 5 g of HWE was dissolved in 50 mL of 70% (v/v) aqueous acetone and filtered using a 0.45-μm Nylon 66 membrane followed by evaporation to remove acetone. Lipids were removed from the aqueous layer by extracting two times with 100 mL of n-hexane. The aqueous layer was again extracted five times with 100 mL of ethyl acetate to remove monomeric flavonoids and partly oligomeric PAs (DP 2–5). Subsequently, the aqueous layer was evaporated under reduced pressure at 65°C, and lyophilized to yield 4.06 g of PA-rich powder. The lyophilized powder (2.51 g) was dissolved in 50% (v/v) aqueous methanol and loaded on a Sephadex LH-20 column. Sugars and monomeric flavonoids were washed with 50% (v/v) aqueous methanol, and PAs (1.76 g) were recovered from the column by eluting with 50% (v/v) aqueous acetone. The total yield of PAs from the HWE was about 58%. The purified PAs were characterized using UV/Visible, FT-IR and 13C nuclear magnetic resonance (NMR) spectroscopy, matrix-assisted laser desorption/ionization time of flight (MALDI-TOF) mass spectrometry (MS), and gel permeation chromatography (GPC) analysis.
Instrumentation
Bate-Smith alcoholysis
Butanolysis was performed in a hydrochloric medium with ferric ammonium sulfate as a catalyst according to the method of Hagerman (2002). One milligram of PAs was added to a PTFE screw capped vial and dissolved with 1 mL of 50% (v/v) aqueous methanol. Then, 6.0 mL of the acid-butanol reagent and 0.2 mL of the iron reagent were added into the vial. The mixture was reacted in a heating block at 95–100°C for 1 h. After the reaction, its visible spectrum was obtained by a Hewlett Packard 8452A UV/Vis spectrophotometer.
Spectroscopic measurements
A bathochromic shift of PAs by addition of 2 N NaOH was observed by the UV/Vis spectrophotometer. The UV spectra were recorded in the range from 200 to 400 nm using a 1-cm path length quartz cuvette. FT-IR spectrum was recorded in the transmission mode using a Shimadzu 8201PC FT-IR spectrophotometer. The FT-IR spectrum of the pelletized KBr sample was scanned between 4,000 and 400 cm−1. 13C NMR spectrum was obtained in acetone-d 4/D2O using a JEOL JNM-EX 400 spectrometer.
MALDI-TOF MS
Acetylation of the PAs was performed according to the report by Foo et al. (1997). PAs (10 mg) were dissolved in 1 mL of pyridine to which 1 mL of acetic anhydride was added and the mixture was stored at room temperature for 48 h. One milligram of the acetylated PAs was dissolved with THF. 2,5-Dihydroxy benzoic acid (DHB) matrix was dissolved with 70% (v/v) aqueous acetone. For the enhancement of ion formation, sodium chloride was dissolved with 70% (v/v) aqueous acetone and added to the matrix. The 10:1:1 volume ratio of matrix to sample to cationization agent was vortexed prior to MALDI TOF-MS analysis. The mixed solution (1–2 μL) was deposited onto two different MALDI targets and air-dried. MALDI-TOF mass spectra were acquired using an Applied Biosystems Voyager-DE-STR MALDI-TOF-MS equipped with a pulsed N2-laser (337 nm). MALDI-TOF conditions were as follows: laser intensity 2178; accelerating voltage, 20 kV; grid voltage, 68%; calibration matrix, DHB; mode of operation, reflector; extraction delay time, 350 ns; polarity, positive; guide wire, 0% of accelerating voltage; mass range, 100–7,000 Da.
Average molar mass distribution
One milligram of the acetylated PAs was dissolved with 1 mL of THF and subjected to GPC analysis. The molar mass was estimated using a Spectra-Physics modular LC system equipped with a SP 8800 ternary HPLC pump, a Spectra 100 variable wavelength detector, and a combination of Shodex GPC KB-802.5 column (8 × 300 mm) and AM GEL GPC column (10 × 300 mm), purchased from American Polymer Standards. THF as the mobile phase was pumped into the column at the flow-rate of 1.0 mL/min. The effluent from the column was continuously monitored at 280 nm for the sample and 254 nm for standard with the UV detector. Molar mass calculations were based on a calibration curve obtained using monodisperse polystyrene standards, acetylated (+)-catechin and phenol. The molar mass of the substituted acetyl group was excluded in this calculation based on PC units.
Results and discussion
Figure 2 shows UV spectrum of the PAs isolated from HWE of P. radiata bark and a bathochromic shift produced by addition of NaOH. PAs showed a UV absorption band at 280 nm, as observed for PA-rich fractions from black wattle (Acacia mollisima Willd.), the barks of Douglas fir (Pseudotsuga menziesii), and Pinus pinaster foliage (Vàzquez et al. 1995). The addition of NaOH to the PA solution showed a bathochromic shift with 12 nm interval, suggesting the presence of phenolic compounds.
Bate-Smith alcoholysis (Bate-Smith 1973) uses an acid-catalyzed oxidative depolymerization of PAs to yield red anthocyanins such as cyanidin and delphinidin. The presence of transition metal ions during the alcoholysis reaction is a key factor to enhance color development (Dalzell and Kerven 1998). Porter et al. (1986) reported that Fe3+ was the most efficient transition metal ion in catalyzing color formation in the butanol–HCl reaction. Therefore, the butanol–HCl reaction containing Fe3+ ions was applied to approximately estimate the basic structure of the pine bark PAs. The chemical structure of the PAs could be assumed based on the fact that the number of phenolic groups in the rings A and B affects the wavelength of the absorbance maximum and the extinction coefficients of the anthocyanidin products. Particularly, PAs yield anthocyanidin pigments by the butanol–HCl reaction in which (epi)catechin-based and gallo(epi)catechin-based PAs produce cyanidins (λmax at 547 nm) and delphinidins (λmax at 558 nm), respectively (Samejima and Yoshimoto 1981). Hussein et al. (1990) suggested that variation of absorption maxima in the butanol–HCl reaction between 540 and 550 nm is due to the relative amounts of anthocyanidins (delphinidin to cyanidin ratio) formed during the acid hydrolysis. Accordingly, the maximum absorption peak at 546 nm, as shown in Fig. 3, could be considered to indicate that the main extension units of the PAs are largely composed of cyanidins rather than delphinidins.
Figure 4 shows the FT-IR spectrum of the PAs from radiata pine bark. The strong absorption bands at 3,385, 1,612, and 1,067 cm−1 were attributed to those of the characteristic functional groups of polyflavonoids (Yazaki and Hillis 1977), whereas those at 1,522 and 777 cm−1 were attributed to aromatic ring breathing mode and CH out-of-plane deformation with two adjacent free hydrogen atoms, respectively, indicating the prominent presence of procyanidin (PC) structure (Foo 1981).
13C NMR spectroscopy has been employed to examine the nature of polymeric PAs (Haslam 1989). 13C NMR spectrum of PAs presents information on the absolute and relative stereochemistry of the heterocyclic ring (C2–C3), the structure(s) of the chain terminating flavan-3-ol unit(s), the ratio of PC to PD extension units, and the number average molar mass (Mn) according to Czochanska et al. (1980).
Figure 5 shows 13C NMR spectrum of PAs from P. radiata bark. The signal assignment was made based on the publication of Czochanska et al. (1980). The spectrum shows typical signals due to PAs, containing PC and PD units. The signals at 116 (C2′, C5′), 120 (C6′), and 145 ppm (C3′, C4′) showed the presence of PC units [(+)-catechin and (−)-epicatechin]. The sharp and small signal at 146 ppm represented a typical indicative for the presence of PD units [(+)-gallocatechin and (−)-epigallocatechin]. The PC/PD ratio of PAs is usually determined from the relative ratio of the peak areas at 145 ppm (C-3′ and C-4′ of PC) and 146 ppm (C-3′ and C-5′ of PD). Although this approach is available for structural discrimination in the B ring of flavan-3-ol units, it is difficult to accurately measure the relative areas, especially when one of the two units is present in much higher proportion than the other because the signals are not completely resolved (Kraus et al. 2003). Recently, Kraus et al. (2003) attempted to utilize other features of the spectra to quantify the PC/PD ratio and used the ratio of peak heights at 116 and 107 ppm instead of peak areas. In order to determine the PC/PD ratio of the PAs, we used the value calculated by the 116/107 ppm ratio. The ratio of the signal heights at 116/107 ppm revealed that the PAs are composed of 94% PC and 6% PD units. This result was in good agreement with the ratio of 90:10 of these respective analogs suggested by Czochanska et al. (1979). In addition, the predominant production of cyanidins induced from the Bate-Smith alcoholysis of PAs and the characteristic absorption bands of FT-IR spectrum showed a reasonable accordance with the 13C NMR result. The region between 30 and 90 ppm is due to the signals of C2, C3, and C4 in flavan-3-ol units. The two signals at 76 and 83 ppm were ascribed to 2,3-cis and 2,3-trans isomers, respectively. The spectrum clearly indicated that both the stereoisomers co-exist, while the signal at 83 ppm was indicative of the majority presence of the trans form. The signals at 68 and 72 ppm were assignable to the C3 terminal and extension units, respectively.
In the mass range where single polymer chains can be resolved, MS provides the following information: the mass of the constituent repeat units and end groups, e.g., chemical heterogeneity (molar mass of by-products), and the molar mass average Mn (Pasch and Schrepp 2003). Therefore, to obtain more detailed information on the chemical composition of PAs, MALDI-TOF MS analysis was performed. Flamini (2003) and Behrens et al. (2003) reported that DHB as a matrix leads to the best analytical conditions for the detection of PCs in reflectron mode to provide the broadest mass range with the least background noise. Polymers with electronegative elements like oxygen or nitrogen are best cationized by Li+ or Na+, and non-polar polymers with π-electrons prefer polarizable cations like Ag+ or Cu+ (Pasch and Schrepp 2003). We have examined various cationizing agents in the presence of DHB matrix for MALDI-TOF MS analysis of acetylated PAs, but only Na+ ions affected the intensity of the signals on the MALDI mass spectrum. However, the pure PAs were not analyzed in the presence of DHB matrix and Na+ ions. The result was assumed to be due to unstable structure of PAs against radiation of a pulsed N2-laser (337 nm) and/or proton-donating behavior like DHB matrix. To determine the cause, the acetylation and methylation of PAs have been performed by pyridine-acetic anhydride and diazomethane reagent, respectively, but only the acetylated PAs were analyzed. Accordingly, the acetylation of hydroxyl groups in the PAs was considered to be the key factor for the successful flight under our MALDI-TOF MS conditions.
Figure 6 and Table 1 show the MALDI-TOF mass spectrum of PAs isolated from P. radiata bark. The masses of the highest peaks among the PA polymers with identical DP increased at the distance of 498 Da, corresponding to an acetylated catechin/epicatechin monomer (Nonier et al. 2004). The spectrum of the magnified tetramers in Fig. 6 clearly indicated a mass increment of 58 Da due to one -OAc group, which in turn indicated the presence of PD units, containing one more hydroxyl group at the aromatic ring B than PC units, although the sequence between PC and PD units could not be estimated. The peak intensity of mass spectra also revealed that the PAs are predominantly composed of PC and partly PD. This result further indicated that the pine bark PAs do not contain galloylated sub-units like grape PAs (Krueger et al. 2000). The sodium adduct ion of the acetylated PAs [M+Na]+ was detected up to octamer in the positive-ion reflectron mode. The mass pattern of the PAs decreased with increasing DP. The researchers (Perret et al. 2003; Flamini 2003) suggested that the lower sensitivity of the large ions may be reasonably due to collisional fragmentations during the time of flight or while the ions are extracted from the matrix. In further detail, Ohnishi-Kameyama et al. (1997) suggested that the poor intensity in the high mass region is attributed to the lability of the catechin linkage in the reflectron mode because of the post-acceleration mode. In the other point of view, Krueger et al. (2000) speculated that lower masses (from the matrix) might saturate the detector, preventing the detection of PAs with higher masses. Because of this, the peak intensities in the high mass region can not be interpreted as a concentration in the sample.
Figure 7 shows the molar mass distribution of the acetylated PAs. PAs are commonly acetylated due to their high polarity, giving rise to a strong adsorption on a GPC column. GPC is also theoretically applicable for estimating the average molar mass of polyphenolic polymers (Yanagida et al. 2003). The average molar mass (Mw, Mn), polydispersity (Mw/Mn), and DP of the PAs were 3,800 (Mw), 1,200 (Mn), 3.2, and 13, respectively. The molar mass of the substituted acetyl group based on PC units was excluded in this calculation.
Conclusions
Proanthocyanidins from P. radiata bark were isolated by Sephadex LH-20 chromatography and characterized using UV/Visible, FT-IR and 13C NMR spectroscopic techniques, MALDI-TOF MS, and GPC analysis. The PAs structure was approximately assessed according to a bathochromic shift of UV under alkaline condition, Bate-Smith alcoholysis, and FT-IR analysis. The height ratio of 116/107 ppm in 13C NMR spectrum was reasonable for quantitative interpretation of the PC/PD ratio in the pine bark PAs. The result revealed that the PAs contain PC (94%) and PD (6%) with the main constituent being the trans configuration of C2–C3. In MALDI-TOF-MS analysis, it was found that the acetylation of hydroxyl groups in the PAs successfully increased the structural stability to an extent that enabled flight into the MS detector, while the pure PAs were difficult to analyze by this technique. The MALDI-TOF MS technique was available for further accurate consideration of sub-structures of PAs, although the maximum DP was limited to octamer. GPC analysis was performed using a combination of two columns with different exclusion limit. The Mw, Mn, Mw/Mn, and DP of the PAs were 3,800 (Mw), 1,200 (Mn), 3.2, and 13, respectively.
References
Arcangeli P (2000) Pycnogenol® in chronic venous insufficiency. Fitoterapia 71:236–244
Bagchi D, Bagachi M, Stohs SJ, Das DK, Ray SD, Kuszynski CA, Joshi SS, Pruess HG (2000) Free radicals and grape seed proanthocyanidin extract: importance in human health and disease prevention. Toxicol 148:187–197
Bate-Smith EC (1973) Tannins of herbaceous leguminosae. Phytochem 12:1809–1812
Behrens A, Maie N, Knicker H, Kögel-Knabner I (2003) MALDI-TOF mass spectrometry and PSD fragmentation as means for the analysis of condensed tannins in plant leaves and needles. Phytochem. 62:1159–1170
Bors W, Michel C, Stettmaier K (2000) Electron paramagnetic resonance studies of radical species of proanthocyanidins and gallate esters. Arch Biochem Biophys 374:347–355
Cowan MM (1999) Plant products as antimicrobial agents. Clin Microbiol Rev 12:564–582
Czochanska Z, Foo LY, Newman RH, Porter LJ, Thomas WA (1979) Direct proof of a homogeneous polyflavan-3-ol structure for polymeric proanthocyanidins. JCS Chem Commun 375:377
Czochanska Z, Foo LY, Newman RH, Porter LJ (1980) Polymeric proanthocyanidins: stereochemistry, structural units and molecular weight. J Chem Soc Perkin Trans 1:2278–2286
Dalzell AS, Kerven GL (1998) A rapid method for the measurement of Leucaena spp. proanthocyanidins by the proanthocyanidin (butanol/HCl) assay. J Sci Food Agric 78:405–416
De Bruyne T, Pieters L, Deelstra H, Vlietinck A (1999) Condensed vegetable tannins: biodiversity in structure and biological activities. Biochem Syst Ecol 37:445–459
Drehsen G (1999) From ancient pine bark uses to Pycnogenol. In: Packer L, Miramatzu M, Yoshikawa T (eds) Antioxidant food supplements in human health. Academic, New York, pp 311–322
Flamini R (2003) Mass spectrometry in grape and wine chemistry. Part I: polyphenols. Mass Spectrom Rev 22:218–250
Foo LY (1981) Proanthocyanidins: gross chemical structures by infrared spectra. Phytochem 20:1397–1402
Foo LY, Mcnabb LWC, Waghorn G, Ulyatt MJ (1997) Proanthocyanidins from Lotus Pedunculatus. Phytochem 45:1689–1696
Grimm T, Schäfer A, Högger P (2004) Antioxidant activity and inhibition of matrix metalloproteinases by metabolites of maritime pine bark extract (Pycnogenol). Free Rad Biol Med 36:811–822
Hagerman AE (2002) Tannin chemistry. http://www.users.muohio.edu/hagermae/
Haslam E (1989) Plant polyphenols: vegetable tannins revisited. Cambridge university press, New York
Haslam E (1998) Practical polyphenolics from structure to molecular recognition and physiological action. Cambridge university press, New York
Hussein L, Fattah MA, Salem E (1990) Characterization of pure proanthocyanidins isolated from the hulls of faba beans. J Agric Food Chem 38:95–98
Karonen M, Loponen J, Ossipov V, Pihlaja K (2004) Analysis of procyanidins in pine bark with reversed-phase and normal-phase high-performance liquid chromatography–electrospray ionization mass spectrometry. Anal Chim Acta 522:105–112
Kennedy JA, Taylor AW (2003) Analysis of proanthocyanidins by high-performance gel permeation chromatography. J Chrom A 995:99–107
Kraus TEC, Yu Z, Preston CM, Dahlgren RA, Zasoski RJ (2003) Linking chemical reactivity and protein precipitation to structural characteristics of foliar tannins. J Chem Ecol 29:703–730
Krueger CG, Dopke NC, Treichel PM, Folts J, Reed JD (2000) Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry of polygalloyl polyfavan-3-ols in grape seed extract. J Agric Food Chem 48:1663–1667
Maffei Facinó R, Carini M, Aldini G, Berti F, Rossoni G, Bombardelli E, Morazzoni P (1996) Procyanidins from Vitis vinifera seeds protect rabbit heart from ischemia/reperfusion injury: antioxidant intervention and/or iron and copper sequestering ability. Planta Med 62:495–502
Mun SP, Ku CS (2006) Characterization of low molecular weight polyphenols from pine (Pinus radiata) bark. Food Sci Biotechnol 15:424–430
Nonier MF, Absalon C, Vivas N, Vias de Gaulejac N (2004) Application of off-line size-exclusion chromatographic fractionation-matrix assisted laser desorption ionization time of flight mass spectrometry for proanthocyanidin characterization. J Chrom A 1033:291–297
Ohara S, Suzuki K, Ohira T (1994) Condensed tainnins from Acacia mearnsii and their biological activities. Mokuzai Gakkaishi 40:1363–1374
Ohnishi-Kameyama M, Yanagida A, Kanda T, Nagata T (1997) Identification of catechin oligomers from apple (Malus pumila cv. Fuji) in matrix-assisted laser desorption/ionization time-of-flight mass spectrometry and fast-atom bombardment mass spectrometry. Rapid Commun Mass Spectrom 11:31–36
Pasch H, Schrepp W (2003) MALDI-TOF mass spectrometry of synthetic polymers. Springer, Berlin Heidelberg New York
Perret C, Pezet R, Tabacchi R (2003) Fractionation of grape tannins and analysis by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Phytochem Anal 14:202–208
Porter LJ, Hrstich LN, Chan BG (1986) The conversion of procyanidins and prodelphinidins to cyanidin and delphinidin. Phytochem 25:223–230
Samejima M, Yoshimoto T (1981) General aspects of phenolic extractives from coniferous barks. Mokuzai Gakkaishi 27:491–497
Santos-Buelga C, Scalbert A (2000) Proanthocyanidins and tannin-like compounds-nature, occurrence, dietary intake, and effects on nutrition and health. J Sci Food Agric 80:1094–1117
Scalbert A, Monties B, Janin G (1989) Tannins in wood: comparison of different estimation methods. J Agric Food Chem 37:1324–1329
Svedström U, Vuorela H, Kostiainen R, Huovinen K, Laakso I, Hiltunen R (2002) High-performance liquid chromatographic determination of oligomeric procyanidins from dimers up to the hexamer in hawthorn. J Chrom A 968:53–60
Tanner GJ, Francki KT, Abrahams S, Watson JM, Larkin PJ, Ashton AR (2003) Proanthocyanidin biosynthesis in plants: purification of legume leucoanthocyanidin reductase and molecular cloning of its cDNA. J Biol Chem 278:31647–31656
Vàzquez G, Antorrena G, Gonzàlez J, Freire S (1995) Studies on the composition of Pinus pinaster foliage. Bioresour Technol 51:83–87
Virgili F, Pagana G, Bourne L, Rimbach G, Natella F, Rice-Evans C, Packer L (2000) Ferulic acid excretion as a marker of consumption of a French maritima pine (Pinus maritima) bark extract. Free Rad Biol Med 28:1249–1256
Yanagida A., Shoji T., Shibusawa Y (2003) Separation of proanthocyanidins by degree of polymerization by means of size-exclusion chromatography and related techniques. J Biochem Biophys Methods 56:311–322
Yazaki Y, Hillis WE (1977) Polyphenolic extractives of Pinus radiata bark. Holzforschung 31:20–25
Zhu M, Phillipson JD, Greengrass PM, Bowery NE, Cai Y (1997) Plant polyphenols: biologically active compounds or non-selective binders to protein?. Phytochem 44:441–447
Acknowledgments
This study was supported by the Technology Development Program for Agricultural and Forestry, the Ministry of Agriculture and Forestry, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ku, C.S., Mun, S.P. Characterization of proanthocyanidin in hot water extract isolated from Pinus radiata bark. Wood Sci Technol 41, 235–247 (2007). https://doi.org/10.1007/s00226-006-0103-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00226-006-0103-8