Abstract
Considering the wealth of recent studies on affective touch, to date, little research addressed the role of the other sensory modalities in the modulation of hedonic tactile perception. Here, we investigated the behavioral and electrodermal signature of the interaction between simultaneously presented visual and tactile stimuli. In three experiments, participants were presented with emotional pictures (international affective picture system; IAPS), while their forearm was gently stroked by means of different tactile textures (i.e., sandpaper, satin, tinfoil, abrasive sponge, and skin-to-skin contact). In Experiment 1, the participants evaluated the pleasantness of the tactile stimulation received, while in Experiment 2 they evaluated the pictures emotional valence. In Experiment 3 the participants rated the pleasantness, the smoothness and the softness of the textures; skin conductance responses (SCRs) were also measured. In sum, the results revealed that while the visual valence ratings were not modulated by the tactile stimulation, the hedonic and sensory tactile ratings were modulated by the visual presentation of both positively and negatively valenced pictures, as well as by neutral pictures. The modulatory effects occurring during visuo-tactile interactions might thus be not necessarily reciprocal. Moreover, the SCRs were not differently affected by the visuo-tactile or tactile conditions of stimulus presentation, suggesting a dissociation between behavioral and electrodermal effects in multisensory interactions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The great emotional power of touch has only recently started to be widely acknowledged by the scientific community (Dunbar 2010; Gallace and Spence 2010, 2014, 2016; Hertenstein et al. 2006a; McGlone et al. 2014; Olausson et al. 2016). Nevertheless, widespread evidence has systematically shown that touch does convey information and emotions in social contexts (e.g., Hertenstein et al. 2006b, 2009; Kirsch et al. 2018; Morrison et al. 2010). Affective touch has indeed a fundamental role in humans’ social life. Just to mention a few examples, it is crucial for the healthy cognitive development of infants (Feldman and Eidelman 2003; Field 2010, 2014; Harlow 1958; Harlow and Zimmermann 1959), promotes the maintenance of the psychophysical well-being (Björnsdotter et al. 2010; Field 2010; Hertenstein et al. 2006a), reduces stress levels (Ditzen et al. 2007; Feldman et al. 2010; Grewen et al. 2003), and facilitates social bonding (Dunbar 2010; Feldman 2012; Nummenmaa et al. 2016; von Mohr et al. 2017).
Converging evidence suggests that pleasant tactile sensations originating from being touched on the hairy skin are mediated by Aβ afferents, a particular class of thin and unmyelinated fibers (e.g., Löken et al. 2009; Olausson et al. 2016) and C-Tactile afferents (CTs). These afferents vigorously respond to the light (force range: 0.3–2.5 mN; Vallbo et al. 1999) and slow (velocity range: 1–10 cm/s; Löken et al. 2009; Vallbo et al. 1999) skin stimulation at neutral temperatures (Ackerley et al. 2014). CTs are hypothesized to project to the posterior insula (PI; Björnsdotter et al. 2009, 2010; Morrison et al. 2011; Olausson et al. 2002, 2008). Furthermore, the CT-optimal stimulation activates an extended network of areas, including the just mentioned posterior insula (Björnsdotter et al. 2009; Gazzola et al. 2012; Gordon et al. 2013; McGlone et al. 2012; Morrison et al. 2011; Olausson et al. 2002, 2008; Morrison 2016; Perini et al. 2015), the orbitofrontal cortex (OFC; Francis et al. 1999; Gazzola et al. 2012; McCabe et al. 2008; McGlone et al. 2012), and the anterior cingulate cortex (ACC; Gazzola et al. 2012; Lindgren et al. 2012; Sliz et al. 2012). It is remarkable that all these areas are specifically involved in the hedonic and emotional processing of the stimuli (Berridge and Kringelbach 2015; Craig 2003; Etkin et al. 2011; Kringelbach 2010). Taken together, strong physiological and neural evidence has been brought forward to support the mediator role of tactile pleasantness attributed to CTs (i.e., affective touch hypothesis; McGlone et al. 2014; Olausson et al. 2016).
Differently from the laboratory settings, in daily life we rarely experience hedonic touch in isolation, as it is strongly influenced by the context of presentation. Top-down factors such as motivational state, sexual preferences, culture, and previous experience were shown to affect tactile pleasantness judgments (see Ellingsen et al. 2016, for a review; e.g.; Gazzola et al. 2012; McCabe et al. 2008; Suvilehto et al. 2015; Triscoli et al. 2014). Even when not related to the required response, time-paired information originating from the other sensory modalities still modulates hedonic touch perception. For example, CT-optimal stimulation was evaluated as less pleasant when a disgusting odor, as compared to a non-odor condition, was simultaneously presented (Croy et al. 2014). Moreover, in the domain of visuo-tactile interactions, CT-optimal touch was rated as less pleasant when presented with faces expressing negative (i.e., frowning faces) as compared to positive emotions (i.e., smiling faces; Ellingsen et al. 2014). Despite the extensive evidence, it is still not clear whether visual information interacts differently with touch depending on the emotional valence of the visual stimuli (i.e., positive, negative, neutral) and/or on the specific emotional and sensory attributes of tactile textures presented (e.g., smoothness, pleasantness). Furthermore, so far, the effects of multisensory interactions including hedonic touch were so far based on only self-reports, and not on the responses of the autonomous nervous system (ANS).
Here the goal is to first investigate whether visual emotional contexts modulate both the hedonic and sensory perception of tactile textures, as well as the autonomic responses generated by the presentation of the tactile stimuli. Furthermore, we also tested whether hedonic touch reciprocally affects the participants’ ratings of visual stimuli. In three experiments, a set of emotional pictures was simultaneously presented together with CT-optimal stimulation delivered through a range of textures. The existence of any reciprocal visuo-tactile modulatory effects on perception and the role of emotional valence (i.e., positive, neutral, negative) of the stimulus were assessed by ratings. In order to assess the autonomic arousal effects of eventual valence modulation, skin conductance responses (SCRs) were also monitored.
Considering that vision frequently affects tactile perception for both sensory and emotional evaluations (e.g., Bellan et al. 2012; Ellingsen et al. 2014; Etzi et al. 2018; Gallace et al. 2006; Gazzola et al. 2012; Hartcher-O’Brien et al. 2008, 2010; Hecht and Reiner 2009), vision is expected to modulated tactile judgments. However, this modulatory effect could be given by the pictures’ emotional valence and thus be influenced by their difference in terms of salience. Therefore, the presentation of positive and negative pictures, but not the presentation of the neutral ones, is expected to affect the tactile ratings. With regard to the evaluation of the pictures, the occurrence of a tactile modulation over vision would seem more uncertain. The frequent phenomenon of visual dominance, together with the high salience of the emotional pictures (Domínguez-Borràs et al. 2016; Ellingsen et al. 2014), should in fact argue against such an effect. Last, overall higher SCRs are expected when textures are presented with positive or negative pictures, because both vision and touch induce physiological changes in the arousal state when accompanied by emotional stimuli (Bradley et al. 2008; Lang et al. 1993; Nardelli et al. 2016; Pawling et al. 2017a, b; Ribeiro et al. 2007).
Experiment 1
Methods
Participants
Fourteen volunteers (9 females; mean age: 25 ± 2.3 years) took part in this experiment. All the participants reported to be right-handed, to have normal tactile sensitivity, and normal or corrected-to-normal vision. The study received the approval of the University of Milano-Bicocca ethical committee and was performed in accordance with the ethical standards laid down in the Declaration of Helsinki (World Medical Association 2013). All participants gave their informed consent before taking part in the study and received course credit as reward for their participation.
Stimuli
Sixty-four emotional pictures belonging to the International Affective Picture System (IAPS; Lang et al. 2008) were used as visual stimuli. The pictures were selected based on the valence mean ratings of each picture, as this was provided by the IAPS authors (Lang et al. 2008; valence range: 0–10, with 0 meaning negative and 10 positive). Half of the pictures depicted negatively valenced contents (e.g., mutilated bodies, violent attacks, war scenes; 0–3 range) and half positively valenced contents (e.g., cheerful family moments, babies, puppies; 7–10 range; see Table 1 for the reference numbers of the pictures used). Although negatively valenced pictures are rated as more arousing than positive (Lang et al. 1999), the pictures were selected to be as similarly arousing as possible (4–7.5 range on a 10-point arousal scale). The tactile stimuli were selected on the basis of a previous study by our research group, where the pleasantness and roughness of different textures were assessed (Etzi et al. 2014). Two smooth-rated (i.e., satin and tinfoil) and two rough-rated (i.e., sandpaper and abrasive sponge) materials were presented. Each tactile texture (size: 10 × 10 cm) was applied on a cardboard roll to provide a comfortable stimulation of the skin. Given the curved shape of the rolls, during the stimulation only a smaller portion of the texture (approximately 2 × 10 cm) was in contact with the skin at any given time.
Procedure
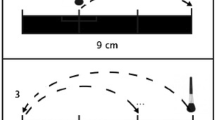
The participants were comfortably seated at a table, in front of a LCD screen (screen size: 30 cm in height and 47.5 cm in width, 1280 × 800 pixels of resolution) placed at a distance of 50 cm from the participants’ body. The experimenter sat at the opposite side of the table, partially hidden by the screen. In each trial, a picture was projected in full screen modality for 6 s and a 10-cm portion of the participants’ non-dominant ventral forearm was simultaneously stroked using one of the materials. The tactile stimulation was delivered by the same (female) experimenter at a velocity of 5 cm/s, in order to induce a high firing rate of CT fibers (preferential CT range: 1–10 cm/s; Löken et al. 2009). The proper velocity of stroking was controlled by replicating the movement of a cursor appearing on another screen only visible to the experimenter. Three 2 s-long consecutive strokes (back-and-forth, along the elbow-wrist direction) were provided. In order to prevent participants from seeing the tactile stimulation being delivered, a black curtain was placed between their trunk and non-dominant arm. At the end of each tactile stimulation the picture was replaced by a blank screen. Participants were instructed to rate the pleasantness of the tactile stimulation on paper sheets when the blank screen was presented. The ratings were expressed on visual analog scales (VASs) anchored by the words “unpleasant” and “pleasant”. Each scale had a length of 10 cm and was subsequently converted by the experimenter in measures from − 5 cm (unpleasant) to + 5 cm (pleasant). Participants were required to pay attention to the images but were explicitly told to only rate the tactile stimulation and ignore the picture content in their evaluations. During the stimulations, the participants wore earplugs to prevent the effect of any auditory information generated from the friction between the texture and their skin. Each material was presented eight times for each picture valence (i.e., positive and negative), while each picture was presented just once, for a total of sixty-four trials. The presentation order of pictures and tactile stimuli was randomized.
Results
Statistical analyses were performed with STATISTICA (Version 6.0; StatSoft, Italy). The ratings were submitted to a repeated measures analysis of variance (rmANOVA) with two within-subject factors: picture valence (positive vs. negative) and tactile stimulus (satin vs. tinfoil vs. sandpaper vs. abrasive sponge). The analysis revealed significant main effects of picture valence [F(1, 13) = 17.89, p < 0.001, \({\eta ^2}\) = 0.08] and of tactile stimulus [F(3, 39) = 24.72, p < 0.001, \({\eta ^2}\) = 0.53]. Materials presented in combination with positive pictures were rated more pleasant as compared to when presented with negative pictures (see Fig. 1). Tukey’s HSD corrected post-hoc tests on the effect of tactile stimulus indicated that utilizing satin and tinfoil for stimulation was significantly experienced as more pleasant, as compared using sandpaper and abrasive sponge for stimulation (all ps < 0.001). No differences between the stimulations delivered with satin and tinfoil (p = 0.64) or between sandpaper and abrasive sponge (p = 0.77) were found. The interaction between picture valence and tactile stimulus was not significant [F(3, 39) = 0.96, p = 0.41, \({\eta ^2}\) = 0.001].
Discussion
The results of Experiment 1 showed that the presentation of emotional pictures affected the hedonic evaluation of the tactile stimulation. Specifically, tactile pleasantness was higher when materials were presented together with positively valenced pictures, as compared to when they were combined with negatively valenced pictures. The visual modulatory effect occurred even though the participants were instructed to only rate the tactile stimulation and ignore the pictures in their evaluations. These results are clearly in line with previous research showing that vision frequently modulates tactile perception in perceptual tasks (e.g., Hartcher-O’Brien et al. 2008, 2010; Hecht and Reiner 2009). Pictures depicting emotional contents also modulate pain intensity evaluations (e.g., De Wied and Verbaten 2001; Kenntner-Mabiala and Pauli 2005; Meagher et al. 2001; Rhudy et al. 2007, 2010), the duration of tactile stimulation (Shi et al. 2012), and tactile spatial attention (Poliakoff et al. 2007). Moreover, as regards tactile pleasantness, CT-optimal touch is perceived as less pleasant when paired with frowning faces, as compared to smiling faces (Ellingsen et al. 2014). It nevertheless remains unclear whether the visual modulatory effect, as found here, was driven by the presentation of the images per se’ or by the specific emotional valence of the visual stimuli presented. Note that it is possible for the hedonic tactile ratings to be modulated by the positive and negative pictures only because of their emotional salience. The introduction of an emotional neutral visual condition should instead clarify the occurrence of this effect. For this reason, in a follow-up experiment of this study (i.e., Experiment 3), participants were presented with pictures of both positive and negative valence, as well as with neutral pictures.
With regard to the pleasantness of the tactile stimuli, as expected, smooth materials were preferred over rough materials (sandpaper and abrasive sponge, e.g., Essick et al. 1999; Etzi et al. 2014; Guest et al. 2011). Therefore, in order to assess whether a CT-optimal stimulation delivered with pleasant or unpleasant textures modulates the valence of emotional pictures, a further experiment was conducted. This experiment (Experiment 2) also assessed the potential modulatory effect of the skin-to-skin contact condition on visual valence ratings.
Experiment 2
Methods
Participants
Twenty right-handed volunteers (15 females, mean age: 22 ± 4.3 years) took part in the experiment. All the participants reported normal tactile sensitivity and normal or corrected-to-normal vision. None of the participants took part in “Experiment 1”.
Stimuli
The tactile stimuli consisted of the same materials used in Experiment 1 (i.e., satin, tinfoil, sandpaper and abrasive sponge), with the addition of a condition of experimenter’s skin-to-participant’s skin contact. Namely, the experimenter stroked the participants’ forearm with the glabrous skin of her index and middle fingers (measure: 3 × 8 cm). Further sixteen pictures (i.e., eight positive and eight negative) were added to the set used in Experiment 1 to compensate for the tactile stimulus addition (for the reference numbers of the pictures used, see Table 1). The same valence arousal criteria for picture inclusion as for Experiment 1 were adopted here.
Procedure
The procedure adopted is very similar to that of Experiment 1. Here the participants were required to rate the valence of the picture instead of the pleasantness of touch. Each texture was presented eight times for each visual valence (i.e., positive and negative), adding to a total of eighty trials.
Results
A rmANOVA, with picture valence (positive vs. negative) and tactile stimulus (satin vs. tinfoil vs. sandpaper vs. abrasive sponge vs. skin-to-skin contact) as within-subject factors, was performed. The analysis revealed a main effect of picture valence [F(1, 19) = 435.48, p < 0.001, \({\eta ^2}\) = 0.93], with positively valenced pictures being rated as more pleasant than negatively valenced pictures (see Fig. 2). The main effect of tactile stimulus [F(4, 76) = 1.53, p = 0.20, \({\eta ^2}\) = 0.001] and the interaction effect between picture valence and tactile stimulus [F(4, 76) = 1.77, p = 0.14, \({\eta ^2}\) = 0.001] were not significant.
Discussion
As expected, positively valenced pictures were rated as more pleasant than negatively valenced pictures (e.g, Lang et al. 1993, 1999, 2008). The tactile stimulation delivered here did not modulate the participants’ visual ratings, even though emotions are easily conveyed by touch (Gallace and Spence 2014, 2016; Hertenstein et al. 2006; Olausson et al. 2016), and thus interact with the visually presented emotional contents (Spence and Gallace 2011). Neither the stimulation given through the various materials, nor the skin-to-skin contact with the experimenter, affected participants’ judgments. Gigen it is a social stimulus, the skin-to-skin contact could be taken as the more salient stimulus (see Field 2014; Guest et al. 2009; Morrison et al. 2010; despite see; Triscoli et al. 2013, for similar hedonic ratings when comparing human- and robot-mediated touch), and therefore expected to be more effective in inducing a modulatory effect over vision. However, the laboratory setting adopted appears to not be optimal at facilitating the appearance of such an effect—note that the social contact was limited by preventing the participant from seeing the occurrence of the skin-to-skin contact. This was due to the need of keeping the conditions of tactile stimulation constant across the experiment.
In the perceptual domain, vision tends to extinguish simultaneously presented tactile stimuli in a number of situations (Hartcher-O’Brien et al. 2008, 2010; Hecht and Reiner 2009). Here, within a (visual) emotional domain, it seems that vision is prioritized, and cannot be modulated by touch. However, these results could be influenced by the levels of emotional salience of the specific stimuli used. Emotionally valenced pictures represent highly salient stimuli and thus their evaluation could escape the modulation by relatively low valenced, or at least less salient, tactile stimuli. On this point, future research needs to assess the effects of visuo-tactile interactions on visual ratings by paying special attention to the saliency levels of the emotional stimuli selected. In order to investigate whether the visual modulation of tactile ratings found in “Experiment 1” is due to the visual dominance power or to the saliency of positively and negatively valenced pictures, an emotional neutral visual condition was introduced in Experiment 3.
Experiment 3
Methods
Participants
Twenty right-handed volunteers (15 females; mean age: 22 ± 2.6 years) took part in this experiment. All participants reported normal tactile sensitivity and normal or corrected-to-normal vision. In order to avoid the effect of previous experience with the stimuli, none of the participants took part in “Experiments 1 and 2”.
Stimuli
One pleasant-rated (i.e., satin) and one unpleasant-rated (i.e., sandpaper) material were selected based on the results of Experiment 1, together with the skin-to-skin contact condition. Thirty-six images taken from the IAPS database were selected for visual stimulation (12 positive, 12 negative and 12 neutral). That is, the IAPS database was first split in three groups based on the picture mean valence ratings (Lang et al. 2008; 0–3: negative; 4–6: neutral; 7–10: positive), and only those pictures within the 4–7.5 range (10-point arousal scale) on the arousal dimension were kept for this experiment (Lang et al. 2008).
Procedure
The same procedure from “Experiments 1 and 2” was adopted with the following exceptions: A 9-cm portion of the participants’ dorsal forearm of the dominant limb was stimulated by delivering three consecutive strokes for 9 s at 3 cm/s, the best velocity for a CTs response (Löken et al. 2009). To provide the stimulation at the correct velocity, the (female) experimenter was trained by auditory tones emitted by a metronome. That is, two 9-cm distant points were marked on the participants’ forearm with a make-up pencil and each tone indicated the moment in which one of the two skin points had to be reached by stroking. In twelve trials, participants were blindfolded in order to have a unisensory tactile condition. Each condition was presented four times, for a total of 48 trials throughout the session. The presentation order of the conditions was pseudorandomized to prevent the same condition to be experienced twice in a row. Each trial, participants rated the pleasantness of the tactile stimulation, the smoothness, and the softness of the tactile stimuli on VASs anchored by the words “pleasant” and “unpleasant”, “smooth” and “rough”, and “soft” and “hard”, respectively. The VASs were 15.87 cm (600 pixels)-long and were implemented by using E-Prime (Version 2.0; Psychology Software Tools, Inc.). One VAS per page was presented and the presentation order of the rating scales was randomized. At the end of the experiment participants were asked to name each tactile stimulus used to deliver the tactile stimulation. Electrodermal activity was measured throughout the entire experimental session. Two Ag-AgCl electrodes (Model 1081 FG) with constant voltage (0.5 Volt) were attached to the medial phalanges of the index and the ring fingers of the non-dominant hand. Skin conductance responses (SCRs) were recorded by means of a SC2071 device (BioDerm, UFI, Morro Bay, California). Saline conductor gel was used to improve the signal-to-noise ratio. The gain parameter was set at 10 µSiemens (µS)/Volt, the analog-to-digital (A/D) resolution was 12 bits, allowing to record responses ranging from 0.1 to 100 µS, with a sample rate of 10 Hz.
Data analysis
The skin conductance (SC) data were analyzed using LEDALAB (version V3.4.7), a software implemented in MATLAB (version R2012a). A continuous decomposition analysis approach was adopted (Benedek and Kaernbach 2010). For the analysis, the average phasic driver within response window (skin conductance response; SCR) was used. The response window was set from 2 to 9 s after the onset of the stimulation. As recommended by Venables and Christie (1980), the SC data were logarithmically transformed with the following formula: y = log(1 + x) prior to the statistical analysis. The data of 6 out of 20 participants were not available for the analysis because of technical problems occurred during the recording. Both mean SCRs and VASs were submitted to rmANOVAs with tactile stimulus (sandpaper vs. satin vs. skin-to-skin contact) and picture valence (positive vs. neutral vs. negative vs. no picture) as within-participants factors. For each rating scale a separate ANOVA was performed. Three two-tailed Pearson correlations were performed to assess the relationship between SCRs and each rating scale. When significant effects were found Tukey’s HSD corrected post-hoc tests were conducted.
Results
Ratings
Pleasantness
The main effects of tactile stimulus [F(2, 38) = 20.90, p < 0.001, \({\eta ^2}\) = 0.34] and of picture valence [F(3, 57) = 12.76, p < 0.001, \({\eta ^2}\) = 0.10], and the interaction effect between tactile stimulus and picture valence [F(6, 114) = 3.33, p = 0.004, \({\eta ^2}\) = 0.01] were significant. Satin and experimenter’s skin were rated as more pleasant than sandpaper (both ps < 0.001), with no difference between them (p = 0.29; see Fig. 3a). The tactile stimulation was rated as less pleasant when associated with negative pictures as compared to the other visual conditions (respectively p < 0.001, p = 0.003, p < 0.001). No differences between tactile stimuli presented in combination with positive and neutral pictures (p = 0.13), positive and no picture (p = 0.68), as well as neutral and no picture (p = 0.69), were found. The interaction effect revealed that satin was rated more pleasant when presented with positively valenced pictures or no picture, as compared to neutral (respectively p = 0.02, p = 0.03) or negative pictures (both ps < 0.001). No differences were found when satin was presented with positive pictures or no picture at all (p = 0.1). When presented together with satin, neutral pictures also received higher ratings as compared to negative pictures (p < 0.001). The sandpaper was rated as less pleasant when presented in combination with negative pictures than positive (p < 0.001) and neutral pictures (p = 0.03), but as pleasant as with no picture (p = 0.39). Moreover, sandpaper was evaluated as more pleasant in combination with positive as compared to no picture (p = 0.03), but as pleasant as when combined with neutral pictures (p = 0.40). By contrast, there was no difference when sandpaper was presented with the neutral pictures or with no picture (p = 0.29). As regards the skin-to-skin contact, it was rated as less pleasant when presented together with negative pictures as compared to positive, neutral pictures and no picture (all ps < 0.001). No differences were found between positive and neutral pictures (p = 0.69), positive pictures and no picture (p = 0.1), and between neutral pictures and no picture (p = 0.91).
Smoothness
The analysis revealed a main effect of tactile stimulus [F(2, 38) = 438.53, p < 0.001, \({\eta ^2}\) = 0.94] and of picture valence [F(3, 57) = 6.92, p < 0.001, \({\eta ^2}\) = 0.001]. Satin was rated as being smoother than the experimenter’s skin (p = 0.01) and sandpaper (p < 0.001); experimenter’s skin was rated as smoother than sandpaper (p < 0.001; see Fig. 3). When combined with positive pictures, the tactile stimuli were rated as smoother than when combined with neutral (p = 0.03) and negative (p = 0.001) pictures, but not with no picture (p = 0.96). No differences between neutral and negative pictures (p = 0.66) or neutral pictures and no picture were found (p = 0.11). Moreover, tactile stimuli were perceived as smoother when presented alone, as compared to when presented with negative pictures (p = 0.006). The interaction between tactile stimulus and picture valence indicated only a trend toward significance [F(6, 114) = 1.96, p = 0.07, \({\eta ^2}\) <.001].
Softness
The main effects of tactile stimulus [F(2, 38) = 97.12, p < 0.001, \({\eta ^2}\) = 0.79] and of picture valence [F(3, 57) = 4.10, p = 0.01, \({\eta ^2}\) = 0.003] were significant. Sandpaper was rated as harder than satin the and experimenter’s skin (both ps < 0.001), but there was no difference between satin and the experimenter’s skin (p = 0.87; see Fig. 3c). All tactile stimuli were rated as softer when combined with positive pictures, as compared to when combined with neutral pictures (p = 0.005). No differences between the other variable levels (all ps > 0.05) were found. The interaction between picture valence and tactile stimulus was not significant [F(6, 114) = 0.89, p = 0.49, \({\eta ^2}\) = 0.001].
Skin conductance responses
The main effect of tactile stimulus was significant [F(2, 26) = 15.08, p < 0.001, \({\eta ^2}\) = 0.25]. The sandpaper induced higher SCRs than satin and skin-to-skin contact (all ps < 0.001; see Fig. 4), but there was no significant difference between satin and skin-to-skin contact (p = 0.81). The main effect of picture valence [F(3, 39) = 0.78, p = 0.50, \({\eta ^2}\) = 0.009] and the interaction between picture valence and tactile stimulus were not significant [F(6, 78) = 0.60, p = 0.72, \({\eta ^2}\) = 0.01].
Correlational analyses
No significant correlations were found between SCR and pleasantness ratings (r = − 0.001, p = 0.99), SCR and smoothness (r = − 0.13; p = 0.15), SCR and softness ratings (r = 0.06, p = 0.39). Pleasantness ratings moderately correlated with smoothness (r = 0.55, p < 0.001) and softness (r = 0.54, p < 0.001). The smoother and the softer the material was, the more pleasant it was rated. Moreover, smoothness correlated with softness ratings (r = 0.84, p < 0.001), meaning that the smoother the material was, the softer it was rated.
Recognition task
The skin-to-skin contact was recognized by the majority of the participants (70%). The sandpaper was recognized by 30% of participants, and the satin was named as “textile” by 15% of participants.
Discussion
Looking at emotional pictures while being stroked affected the participants’ hedonic and sensory evaluation of a tactile stimulation. Negatively valenced pictures induced lower pleasantness ratings for all tactile stimuli used, as compared to both positive and neutral pictures. The same results were evident when comparing negative picture to the unisensory tactile condition, but just for satin and skin-to-skin contact. As regards positive and neutral pictures, their effect on the hedonic tactile judgments, as well as that of the unisensory tactile condition, varied as a function of the specific texture presented. In some cases, positively valenced pictures led to higher pleasantness ratings as compared to neutral pictures (i.e., for satin) and the unisensory condition (i.e., for sandpaper). In another case, the unisensory condition led to higher pleasantness ratings than neutral valenced pictures (i.e., when satin was presented). Therefore, our results indicate that the hedonic evaluation of tactile stimulation can be modulated by the emotional content of concurrently presented visual stimuli, even though this modulatory effect would seem to be more consistent when visual stimuli are negatively valenced as compared to positive and neutral images.
With respect to the sensory attributes of the stimuli, the tactile stimuli were rated as smoother when presented with positive pictures as compared to negative and neutral pictures. The materials were also rated as softer when presented together with emotionally positive pictures than with neutral pictures. Hence, also perceptual evaluations of the tactile stimuli were modulated by the visual stimuli, suggesting the emotional content does also interact with sensory and discriminative perception. It is however worth noting that the effects for the sensory qualities of the stimuli were much less pronounced than those of the hedonic judgments. More generally, these results demonstrate that visual stimuli depicting both positive/negative and neutral content affect tactile judgments. Thus, the mere presentation of content-depicting pictures, regardless of their specific emotionally valenced nature, would seem to be responsible for the modulations of tactile judgments.
The skin conductance responses were not affected differently by the emotional valence of the pictures presented, nor did they vary as a function of the multisensory or unisensory condition of stimulus presentation. As regards the SCRs to the different tactile stimuli, the results showed that the sandpaper induced higher SCRs as compared to the satin and the skin-to-skin contact. More negatively valenced tactile stimuli might thus elicit stronger autonomic reactions than positive tactile stimuli. This speculation follows from evidence from other sensory domains showing that unpleasant stimuli can be more physiologically arousing relative to the pleasant ones (Eriksson et al. 2008; McDowell and Mandler 1989; Ramachandran and Brang 2008).
The textures rated as smoother and as softer than sandpaper (i.e., satin and experimenter’s skin) were also rated as more pleasant (see Etzi et al. 2014 for a study on tactile preferences for everyday materials). Satin and experimenter’s skin were evaluated as equally pleasant and soft (but with satin as smoother). It is also important to note that most of the participants were aware that the stimulation was delivered with the experimenter’s skin or with a material (even if not clearly identified). However, the skin-to-skin contact induced the same behavioral and electrodermal responses as those induced by being stroked with the satin. A greater arousal was expected in response to the skin-to-skin contact as compared to the textures. Nevertheless, as previously mentioned, the experimental setting was certainly far from a natural condition of social interaction (i.e., the real effects of a caress cannot be inferred here).
General discussion
The present study investigated the effects of visuo-tactile interactions on subjective evaluations and physiological responses to multisensory stimuli within the hedonic domain. The results highlight that the hedonic judgments of the tactile stimulation are modulated by the simultaneous presentation of emotionally valenced visual stimuli. This modulatory effect is independent of whether the picture content is relevant or not to the stroking delivered to the forearm. In “Experiment 1”, the tactile stimulation was rated as more pleasant when paired with emotionally positive pictures, as compared to when presented with negative pictures, whereas “Experiment 3” revealed emotionally neutral pictures also affect tactile evaluations as well. Even though these modulatory effects varied as a function of the tactile stimulus presented, such a result could be taken to indicate that any level of visual emotional valence (including the neutral one) has an effect on tactile ratings. Future experiments need to assess whether this effect extends to other textures and, overall, to understand its specific driver (e.g., selective or spatial attention).
While the impact of positive and neutral pictures varied as a function of the specific texture presented, in most of the cases negative pictures affected the tactile ratings for all the tactile stimuli presented. All the materials were in fact perceived as less pleasant when associated with negative pictures as compared to positive and neutral pictures. During multisensory interactions in the hedonic domain, negatively valenced and unpleasant stimuli seem to impact tactile ratings more consistently, as compared to positive stimuli (see Croy et al. 2014, on this point for olfactory-tactile interactions). This effect could be a consequence of the psychological phenomenon whereby negative contents tend to have a greater effect than positive contents in cognitive evaluations (e.g., Alves et al. 2017; Baumeister et al. 2001; Taylor 1991). People would seem in fact to be more motivated to avoid bad events instead of pursuing good ones (Baumeister et al. 2001).
The evaluations of the sensory attributes of the tactile stimuli (i.e., smoothness and softness) were also affected by the pictures presented, even though in a less pronounced way as compared to hedonic ratings. The textures were rated as rougher when presented together with negative or neutral pictures, as compared to when presented together with positive pictures. Moreover, they were evaluated as softer when presented with positive as compared to neutral pictures. Since both smoothness and softness were also moderately correlated with pleasantness (see also Etzi et al. 2014 on this point), one hypothesis could be that the hedonic value of the stimuli mediates the occurrence of these interactions. The role of the hedonic value and emotional valence is in fact often taken into consideration in order to account, at least in part, for the existence of some crossmodal correspondences and interactions found in the extant literature on human perception (Crisinel and Spence 2010; Demattè et al. 2006; Etzi et al. 2016; Velasco et al. 2015).
The effect of the hedonic interaction between the visual and tactile stimuli was further assessed by monitoring the autonomic nervous system (ANS) activity, which is highly responsive to emotional stimuli. Surprisingly, the arousal response assessed through the measure of skin conductance was not affected by the visual stimulation, irrespective of the emotional salience of positive and negative pictures. This result would seem to suggest the presence of a dissociation between the explicit and implicit effects of visuo-tactile interactions. That is, emotional visual stimuli would seem to modulate hedonic and sensory ratings but not body arousal responses. It is, however, possible that any potential visual effect on the autonomic measure was overcome by the greater arousing power of being stroked with textures (Etzi and Gallace 2016).
The results revealed that the evaluation of the pictures was not affected by the attributes of the tactile stimulation. Specifically, in Experiment 2, the evaluation of the pictures varied only as a function of their emotional valence (i.e., positive and negative). This result is not surprising, given that the presentation of visual stimuli has often been shown to dominate over touch and not vice-versa (e.g., Hatcher-O’ Brien et al. 2008, 2010; Hecht and Reiner 2009). Visual stimuli are infrequently prioritized when presented together with stimuli from other sensory modalities (e.g., Hecht and Reiner 2009; Posner et al. 1976; Sinnett et al. 2007). Note, however, that multiple source of evidence suggests that sensory dominance depends on the specific task performed by the participant (Ernst and Banks 2002; Ernst and Bülthoff 2004; Welch and Warren 1980). The lack of tactile modulation over vision in this study could thus reflect the specific set of visual and tactile stimuli used. In particular, the emotional pictures adopted in our study were likely more salient than the tactile stimuli used. It is also worth noting that, although the relevance of the emotional aspects conveyed by touch is now widely recognized (e.g., Gallace and Spence 2010, 2014, 2016; Hertenstein et al. 2006a; Löken et al. 2009; McGlone et al. 2014; Morrison et al. 2010; Olausson et al. 2016), it is still not clear whether the slow stimulation of the skin by means of tactile textures can effectively induce variations in the emotional individual state.
“Experiment 3” demonstrates that participants in most of the cases correctly identify the skin of the experimenter as one of the tactile stimuli presented. However, with the sole exception of smoothness, no differences between skin-to-skin contact and satin were found, neither for pleasantness ratings nor for SCRs. This is likely due to the fact that here the higher salience of the skin-to-skin contact as compared to the stimulation by means of the everyday materials was limited by the poor emotional context of the laboratory setting. More ecological situations, such as real social interactions, are needed to carefully assess whether affective touch is salient enough to modulate visual emotional judgments.
With regard to tactile preferences, the pleasantness ratings of the textures varied as a function of both the material presented (differing in terms of microgeometric properties) and the valence of the pictures simultaneously shown. Moreover, the skin stimulation performed by means of the smooth and soft-rated textures (tinfoil, skin and satin) was preferred over the stimulation performed with the rough and hard-rated textures (sandpaper and abrasive sponge), thus providing additional evidence about the role of smoothness and softness dimensions in modulating tactile pleasure (Essick et al. 1999; Etzi et al. 2014; Guest et al. 2011).
Sandpaper induced a higher electrodermal arousal response as compared to satin and skin-to-skin contact. Such a result extends our knowledge about the autonomic reactions to textures presented on the hairy skin and suggests that tactile stimulation duration, as a function of the stimulus used, can affect people’s arousal responses (see Triscoli et al. 2017 for the effects of prolonged CT-optimal stimulation on heart rate variability -HRV-). Note that this previous study delivered 9 s strokes. By contrast, the results of our recent study revealed that the same kind of tactile stimulation (by means of satin and sandpaper), lasting only for 2 s, did not induce any variation in the arousal response (Etzi and Gallace 2016). It is thus possible that a higher arousal response to sandpaper emerges only later in time, possibly due to the realization of the potential threat represented by such material to the skin.
Taken together, the present study describes the behavioral and physiological link from the multisensory interactions occurring between vision and touch to the hedonic and sensory evaluations of tactile textures. In particular, our results support the view that the hedonic value attributed to stroking can be easily modulated by other information concurrently presented (see Croy et al. 2014; Ellingsen et al. 2014, 2016; Gazzola et al. 2012; McCabe et al. 2008). However, the strong visual modulatory effect affects only the explicit evaluations of tactile stimuli, and crucially, not the arousal response generated by them (see Etzi and Gallace 2016). Future research needs to address the role of the social context in visuo-tactile interactions (e.g., including interactive dyads).
References
Ackerley R, Wasling HB, Liljencrantz J, Olausson H, Johnson RD, Wessberg J (2014) Human C-tactile afferents are tuned to the temperature of a skin-stroking caress. J Neurosci 34:2879–2883
Alves H, Koch A, Unkelbach C (2017) Why good is more alike than bad: Processing implications. Trends Cognit Sci 21:69–79
Baumeister RF, Bratslavsky E, Finkenauer C, Vohs KD (2001) Bad is stronger than good. Rev Gen Psychol 5:323–370
Bellan V, Reverberi C, Gallace A (2012) Evidence for ‘Visual Enhancement of Touch’ mediated by visual displays and its relationship with body ownership. Haptics: perception, devices, mobility, and communication. lecture notes in computer science, 7282, 58–66
Berridge KC, Kringelbach ML (2015) Pleasure systems in the brain. Neuron 86:646–664
Björnsdotter M, Löken L, Olausson H, Vallbo Å, Wessberg J (2009) Somatotopic organization of gentle touch processing in the posterior insular cortex. J Neurosci 29:9314–9320
Björnsdotter M, Morrison I, Olausson H (2010) Feeling good: on the role of C fiber mediated touch in interoception. Exp Brain Res 207:149–155
Bradley MM, Miccoli L, Escrig MA, Lang PJ (2008) The pupil as a measure of emotional arousal and autonomic activation. Psychophysiology 45:602–607
Craig AD (2003) Interoception: the sense of the physiological condition of the body. Curr Opin Neurobiol 13:500–505
Crisinel AS, Spence C (2010) As bitter as a trombone: synesthetic correspondences in nonsynesthetes between tastes/flavors and musical notes. Atten Percept Psychophys 72:1994–2002
Croy I, D’Angelo S, Olausson H (2014) Reduced pleasant touch appraisal in the presence of a disgusting odor. PLoS One 9:e92975
De Wied M, Verbaten M, N (2001) Affective picture processing, attention, and pain tolerance. Pain 90:13–172
Dematte ML, Sanabria D, Sugarman R, Spence C (2006) Cross-modal interactions between olfaction and touch. Chem Senses 31:291–300
Ditzen B, Neumann ID, Bodenmann G, von Dawans B, Turner RA, Ehlert U, Heinrichs M (2007) Effects of different kinds of couple interaction on cortisol and heart rate responses to stress in women. Psychoneuroendocrinology 32:565–574
Domínguez-Borràs J, Rieger SW, Corradi-Dell’Acqua C, Neveu R, Vuilleumier P (2016) Fear spreading across senses: visual emotional events alter cortical responses to touch, audition, and vision. Cereb Cortex 27:68–82
Dunbar RI (2010) The social role of touch in humans and primates: behavioural function and neurobiological mechanisms. Neurosci Biobehav Rev 34:260–268
Ellingsen DM, Wessberg J, Chelnokova O, Olausson H, Laeng B, Leknes S (2014) In touch with your emotions: oxytocin and touch change social impressions while others’ facial expressions can alter touch. Psychoneuroendocrinology 39:11–20
Ellingsen DM, Leknes S, Løseth G, Wessberg J, Olausson H (2016) The neurobiology shaping affective touch: expectation, motivation, and meaning in the multisensory context. Front Psychol 6:1986
Eriksson M, Storm H, Fremming A, Schollin J (2008) Skin conductance compared to a combined behavioural and physiological pain measure in newborn infants. Acta Paediatr 97:27–30
Ernst MO, Banks MS (2002) Humans integrate visual and haptic information in a statistically optimal fashion. Nature 415:429–433
Ernst MO, Bülthoff HH (2004) Merging the senses into a robust percept. Trends Cognit Sci 8:162–169
Essick G, James A, McGlone FP (1999) Psychophysical assessment of the affective components of non-painful touch. Neuroreport 10:2083–2087
Etkin A, Egner T, Kalisch R (2011) Emotional processing in anterior cingulate and medial prefrontal cortex. Trends Cognit Sci 15:85–93
Etzi R, Gallace A (2016) The arousing power of everyday materials: an analysis of the physiological and behavioral responses to visually and tactually presented textures. Exp Brain Res 234:1659–1666
Etzi R, Spence C, Gallace A (2014) Textures that we like to touch: an experimental study of aesthetic preferences for tactile stimuli. Conscious Cogn 29:178–188
Etzi R, Spence C, Zampini M, Gallace A (2016) When sandpaper is ‘kiki’ and satin is ‘bouba’: an exploration of the associations between words, emotional states, and the tactile attributes of everyday materials. Multisens Res 29:133–155
Etzi R, Ferrise F, Bordegoni M, Zampini M, Gallace A (2018) The effect of visual and auditory information on the perception of pleasantness and roughness of virtual surfaces. Multisens Res 31:501–522
Feldman R (2012) Oxytocin and social affiliation in humans. Horm Behav 61:380–391
Feldman R, Eidelman AI (2003) Skin-to-skin contact (Kangaroo Care) accelerates autonomic and neurobehavioural maturation in preterm infants. Dev Med Child Neurol 45:274–281
Feldman R, Singer M, Zagoory O (2010) Touch attenuates infants’ physiological reactivity to stress. Dev Sci 13:271–278
Field T (2010) Touch for socioemotional and physical well-being: a review. Dev Rev 30:367–383
Field T (2014) Touch. MIT press
Francis S, Rolls ET, Bowtell R, McGlone F, O’Doherty J, Browning A, Smith E (1999) The representation of pleasant touch in the brain and its relationship with taste and olfactory areas. Neuroreport 10:453–459
Gallace A, Spence C (2010) The science of interpersonal touch: an overview. Neurosci Biobehav Rev 34:246–259
Gallace A, Spence C (2014) In touch with the future: The sense of touch from cognitive neuroscience to virtual reality. University Press, Oxford
Gallace A, Spence C (2016) Social touch. In: Olausson H, Wessberg J, Morrison I, McGlone F (eds) affective touch and the neurophysiology of CT afferents. Springer, London
Gallace A, Auvray M, Tan HZ, Spence C (2006) Visual transients impair the detection of tactile changes: a novel case of crossmodal change blindness? Neurosci Lett. 34:246–259
Gazzola V, Spezio ML, Etzel JA, Castelli F, Adolphs R, Keysers C (2012) Primary somatosensory cortex discriminates affective significance in social touch. Proc Natl Acad Sci 109:E1657–E1666
Gordon I, Voos AC, Bennett RH, Bolling DZ, Pelphrey KA, Kaiser MD (2013) Brain mechanisms for processing affective touch. Hum Brain Mapp 34:914–922
Grewen KM, Anderson BJ, Girdler SS, Light KC (2003) Warm partner contact is related to lower cardiovascular reactivity. Behav Med 29:123–130
Guest S, Essick G, Dessirier JM, Blot K, Lopetcharat K, McGlone F (2009) Sensory and affective judgments of skin during inter-and intrapersonal touch. Acta Physiol (Oxf) 130:115–126
Guest S, Dessirier JM, Mehrabyan A, McGlone F, Essick G, Gescheider G, Fontana A, Xiong R, Ackerley R, Blot K (2011) The development and validation of sensory and emotional scales of touch perception. Atten Percept Psychophys 73:531–550
Harlow HF (1958) The nature of love. Am Psychol 13:673–685
Harlow HF, Zimmermann RR (1959) Affectional responses in the infant monkey. Science 130:421–432
Hartcher-O’Brien J, Gallace A, Krings B, Koppen C, Spence C (2008) When vision ‘extinguishes’ touch in neurologically-normal people: extending the Colavita visual dominance effect. Exp Brain Res 186:643–658
Hartcher-O’Brien J, Levitan C, Spence C (2010) Extending visual dominance over touch for input off the body. Brain Res 1362:48–55
Hecht D, Reiner M (2009) Sensory dominance in combinations of audio, visual and haptic stimuli. Exp Brain Res 193:307–314
Hertenstein MJ, Verkamp JM, Kerestes AM, Holmes RM (2006a) The communicative functions of touch in humans, nonhuman primates, and rats: a review and synthesis of the empirical research. Genet Soc Gen Psychol Monogr 132:5–94
Hertenstein MJ, Keltner D, App B, Bulleit BA, Jaskolka AR (2006b) Touch communicates distinct emotions. Emotion 6:528–533
Hertenstein MJ, Holmes R, McCullough M, Keltner D (2009) The communication of emotion via touch. Emotion 9:566–573
Kenntner-Mabiala R, Pauli P (2005) Affective modulation of brain potentials to painful and nonpainful stimuli. Psychophysiology 42:559–567
Kirsch LP, Krahé C, Blom N, Crucianelli L, Moro V, Jenkinson PM, Fotopoulou A (2018) Reading the mind in the touch: Neurophysiological specificity in the communication of emotions by touch. Neuropsychologia 116:136–149
Kringelbach ML (2010) The hedonic brain: a functional neuroanatomy of human pleasure. In: Kringelbach ML, Berridge KC (eds) Pleasures of the brain. Oxford University Press, Oxford, pp 202–221
Lang PJ, Greenwald MK, Bradley MM, Hamm AO (1993) Looking at pictures: affective, facial, visceral, and behavioral reactions. Psychophysiology 30:261–273
Lang PJ, Bradley MM, Cuthbert BN (1999) International affective picture system (IAPS): Technical manual and affective ratings, 2. The Center for Research in Psychophysiology, University of Florida, Gainesville
Lang PJ, Bradley MM, Cuthbert BN (2008) International affective picture system (IAPS): affective ratings of pictures and instruction manual. Technical Report A-8, University of Florida, Gainesville, FL
Lindgren L, Westling G, Brulin C, Lehtipalo S, Andersson M, Nyberg L (2012) Pleasant human touch is represented in pregenual anterior cingulate cortex. Neuroimage 59:3427–3432
Löken LS, Wessberg J, McGlone F, Olausson H (2009) Coding of pleasant touch by unmyelinated afferents in humans. Nat Neurosci 12:547–548
McCabe C, Rolls ET, Bilderbeck A, McGlone F (2008) Cognitive influences on the affective representation of touch and the sight of touch in the human brain. Soc Cognit Affect Neurosci 3:97–108
McDowell KA, Mandler G (1989) Constructions of emotion: discrepancy, arousal, and mood. Motivation Emotion 13:105–124
McGlone F, Olausson H, Boyle JA, Jones-Gotman M, Dancer C, Guest S, Essick G (2012) Touching and feeling: Differences in pleasant touch processing between glabrous and hairy skin in humans. Eur J Neurosci 35:1782–1788
McGlone F, Wessberg J, Olausson H (2014) Discriminative and affective touch: Sensing and feeling. Neuron 82:737–755
Meagher MW, Arnau RC, Rhudy JL (2001) Pain and emotion: Effects of affective picture modulation. Psychosom Med 63:79–90
Morrison I (2016) ALE meta-analysis reveals dissociable networks for affective and discriminative aspects of touch. Hum Brain Mapp 37:1308–1320
Morrison I, Löken LS, Olausson H (2010) The skin as a social organ. Exp Brain Res 204:305–314
Morrison I, Björnsdotter M, Olausson H (2011) Vicarious responses to social touch in posterior insular cortex are tuned to pleasant caressing speeds. J Neurosci 31:9554–9562
Nardelli M, Greco A, Bianchi M, Scilingo EP, Valenza G (2016) On the pleasantness of a haptic stimulation: How different textures can be recognized through heart rate variability nonlinear analysis. In: Engineering in Medicine and Biology Society, 2016 IEEE 38th Annual International Conference, pp. 3560–3563.
Nummenmaa L, Tuominen L, Dunbar R, Hirvonen J, Manninen S, Arponen E, Sams M (2016) Social touch modulates endogenous µ-opioid system activity in humans. Neuroimage 138:242–247
Olausson HW, Lamarre Y, Backlund H, Morin C, Wallin BG, Starck G, Ekholm S, Strigo I, Worsley K, Vallbo ÅB, Bushnell MC (2002) Unmyelinated tactile afferents signal touch and project to insular cortex. Nat Neurosci 5:900–904
Olausson HW, Cole J, Vallbo ÅB, McGlone F, Elam M, Krämer HH, Rylander K, Wessberg J, Bushnell MC (2008) Unmyelinated tactile afferents have opposite effects on insular and somatosensory cortical processing. Neurosci Lett 436:128–132
Olausson H, Wessberg J, Morrison I, McGlone F (2016) Affective touch and the neurophysiology of CT afferents. Springer, New York
Pawling R, Cannon PR, McGlone FP, Walker SC, Shirinyan D, Lieberman M (2017a) C-tactile afferent stimulating touch carries a positive affective value. PLoS One 12:e0173457
Pawling R, Trotter PD, McGlone FP, Walker SC (2017b) A positive touch: C-tactile afferent targeted skin stimulation carries an appetitive motivational value. Biol Psychol 129:186–194
Perini I, Olausson H, Morrison I (2015) Seeking pleasant touch: neural correlates of behavioral preferences for skin stroking. Front Behav Neurosci 9:8
Poliakoff E, Miles E, Li X, Blanchette I (2007) The effect of visual threat on spatial attention to touch. Cognition 102:405–414
Posner MI, Nissen MJ, Klein RM (1976) Visual dominance: an information-processing account of its origins and significance. Psychol Rev 83:157–171
Ramachandran VS, Brang D (2008) Tactile-emotion synesthesia. Neurocase 14:390–399
Rhudy JL, McCabe KM, Williams AE (2007) Affective modulation of autonomic reactions to noxious stimulation. Int J Psychophysiol 63:105–109
Rhudy JL, Bartley EJ, Williams AE (2010) Habituation, sensitization, and emotional valence modulation of pain responses. Pain 148:320–327
Ribeiro RL, Teixeira-Silva F, Pompéia S, Bueno OFA (2007) IAPS includes photographs that elicit low-arousal physiological responses in healthy volunteers. Physiol Behav 91:671–675
Shi Z, Jia L, Müller HJ (2012) Modulation of tactile duration judgments by emotional pictures. Front Integr Neurosci 6:24
Sinnett S, Spence C, Soto-Faraco S (2007) Visual dominance and attention: the Colavita effect revisited. Percep Psychophys 69:673–686
Sliz D, Smith A, Wiebking C, Northoff G, Hayley S (2012) Neural correlates of a single-session massage treatment. Brain Imaging Behavior 6:77–87
Spence C, Gallace A (2011) Multisensory design: Reaching out to touch the consumer. Psychol Market 28:267–308
Suvilehto JT, Glerean E, Dunbar RI, Hari R, Nummenmaa L (2015) Topography of social touching depends on emotional bonds between humans. Proc Natl Acad Sci 112:13811–13816
Taylor SE (1991) Asymmetrical effects of positive and negative events: the mobilization-minimization hypothesis. Psychol Bull 110:67–85
Triscoli C, Olausson H, Sailer U, Ignell H, Croy I (2013) CT-optimized skin stroking delivered by hand or robot is comparable. Front Behav Neurosci 7:208
Triscoli C, Ackerley R, Sailer U (2014) Touch satiety: differential effects of stroking velocity on liking and wanting touch over repetitions. PLoS One 9:e113425
Triscoli C, Croy I, Steudte-Schmiedgen S, Olausson H, Sailer U (2017) Heart rate variability is enhanced by long—lasting pleasant touch at CT-optimized velocity. Biol Psychol 128:71–81
Vallbo ÅB, Olausson H, Wessberg J (1999) Unmyelinated afferents constitute a second system coding tactile stimuli of the human hairy skin. J Neurophysiol 81:2753–2763
Velasco C, Woods AT, Deroy O, Spence Ch (2015) Hedonic mediation of the crossmodal correspondence between taste and shape. Food Qual Preference 41:151–158
von Mohr M, Kirsch LP, Fotopoulou A (2017) The soothing function of touch: affective touch reduces feelings of social exclusion. Sci Rep 7:13516
Welch RB, Warren DH (1980) Immediate perceptual response to intersensory discrepancy. Psychol Bull 88:638–667
World Medical Association (2013) Declaration of Helsinki world medical association declaration of Helsinki: ethical principles for medical research involving human subjects. J Am Med Assoc 210:2191–2194
Acknowledgements
We would like to thank Dr. Clara Piliego for help with data collection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Etzi, R., Zampini, M., Juravle, G. et al. Emotional visual stimuli affect the evaluation of tactile stimuli presented on the arms but not the related electrodermal responses. Exp Brain Res 236, 3391–3403 (2018). https://doi.org/10.1007/s00221-018-5386-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-018-5386-0