Abstract
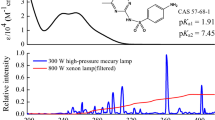
The objective of this research was to study the effect of pH on the photodegradation of aqueous solutions of 5×10–5 M acesulfame-K (λmax, 226 nm;ε, 11 400 mol–1 L cm–1). Photodegradation followed first-order kinetics and was found to be pH-dependent. The degradation rate constant was calculated to be 6.7×10–2 min–1, 4.5×10–2 min–1, 3.4×10–2 min–1 and 17.4×10–2 min–1, respectively, at pH 3, pH 9 and pH 12.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 21 April 1998 / Revised version: 9 June 1998
Rights and permissions
About this article
Cite this article
Coiffard, C., Coiffard, L. & De Roeck-Holtzhauer, Y. Photodegradation kinetics of acesulfame-K solutions under UV light: effect of pH. Z Lebensm Unters Forsch 208, 6–9 (1999). https://doi.org/10.1007/s002170050367
Issue Date:
DOI: https://doi.org/10.1007/s002170050367