Abstract
The derivatives of 1,2,4- triazole have attracted great attention among medicinal chemists due to their wide range of biological activity, good pharmacodynamic and pharmacokinetic profiles, and low toxicity, that necessitates the development of various synthesis methods and a comprehensive study of their reaction mechanisms. A detailed investigation of possible pathways for formation of new spiro-condensed [1,2,4]triazolo[1,5-c]quinazolines, that combine two structural domains with different biological properties, was performed by computational study at the SMD/B3lyp/6-31+G(d) theory level. The mechanism of interaction between [2-(3-hetaryl-1,2,4-triazol-5-yl)phenyl]amine and cyclohexanone in methanol involves three main processes: formation of carbinolamine by addition of an amine to double bond C=O, elimination of a water molecule, and intramolecular cyclization leading to formation of spiro compounds. Results show increase in reactivity of reactants during acid-catalyzed reaction compared to uncatalyzed one. The nature of the heterocyclic substituent on the triazole ring has little effect on the reaction energy, while the mechanism is unchanged.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
One of the most challenging tasks in medicinal chemistry is the search for new biologically active compounds, that may be used in efficient drug discovery. Derivatives of 1,2,4-triazole usually possess a series of pharmacological properties such as antibacterial, antiviral, antifungal, antitubercular, and anticancer activities [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15]. Combination of two or more structural domains, which possess different biological properties, can overcome drug resistance and reduce toxicity. The presence of 1,2,4-triazole moiety can influence dipole moment, polarity, solubility, lipophilicity, and hydrogen bonding capacity of molecule, improving physicochemical, pharmacological, and pharmacokinetic properties of the compounds [1,2,3]. Synthesis of compounds that combine 1,2,4-triazole and quinazoline moieties in one structure leads to the discovery of new effective agents with antibacterial, antifungal, and anticancer capabilities [16,17,18,19,20,21,22,23]. Spiro compounds are particularly interesting, as spiro[5,6-dihydro-(1,2,4)triazolo[4,3-c]quinazoline-5,1ʹ-cyclohexan]-3(2H)-one showed high inhibitory activity on poly (ADP-ribose) polymerase 1 (PARP-1) [16]. Inhibition of PARP-1 is necessary to cell survival when PARP-1 is overactivated and induces depletion of cellular NAD+ and ATP levels leading to cell dysfunction or necrotic cell death.
A group of new spiro-condensed [1, 2, 4]triazolo[1,5-c]quinazolines was synthesized by reaction of [(1,2,4-triazol-5-yl)phenyl]amines with ketones in high yields (Scheme 1) [17, 24]. Synthesized compounds displayed moderate to good affinity for the nociception (NOP) receptor and behaved as partial agonists [17]. NOP receptor is an important biological target for a number of potential therapeutic applications due to induction of analgesia, hypotension, bradycardia, and diuresis, stimulation of food intake, production of anxiolysis, inhibition of memory processes, and neurotransmitter release at central and peripheral sites [19]. The reaction of synthesis of the spiro derivatives can be considered as a binucleophilic addition of [(1,2,4-triazol-5-yl)phenyl]amines to cyclic ketones with sequential elimination of a water molecule [17, 24]; however, the mechanism of the reaction process is still unknown. To shed light on the issue, the mechanism of interaction between [2-(3-hetaryl-1,2,4-triazol-5-yl)phenyl]amines and cyclohexanone was modelled in the present paper.
There are computational studies on mechanism of interaction of amines with oxo compounds [25,26,27,28,29]. Usually, the mechanism consists of the addition of an amine to carbonyl group with formation of a carbinolamine intermediate, and the sequential elimination of a water molecule, that leads to the formation of an imine (Schiff base). The rate-limiting step was reported to be the dehydration of the carbinolamine with an activation energy of more than 40 kcal/mol [25,26,27]. The activation barriers for the both steps can be reduced by assisting of proton transfer with water molecules or sterically approached group bearing acidic hydrogen [26, 29, 30], as well as using basic or acidic catalyst [26, 28]. In the acid-catalyzed mechanism, the protonation of amines has more stabilizing effect than the protonation of carbonyl group [28, 31]. The presence of two amino groups in one structure may lead to the formation of spiro intermediate in reaction with oxo compounds [31].
The present work aimed to model mechanism for reaction of [2-(3-hetaryl-1,2,4-triazol-5-yl)phenyl]amines with cyclohexanone. The possibility of unassisted as well as acid-catalyzed mechanisms was considered.
1.1 Computational methods
The geometries of all structures were optimized within the Gaussian 09 program [32], using the B3lyp functional of the density functional theory (DFT) and 6–31 + G(d) basis set [33, 34]. The influence of methanol (ε = 32.613) bulk was simulated within SMD model [35]. Stationary points for modeled reactions were characterized as either local minima (reactant, intermediate, product) having all real frequencies, or as transition states possessing only one imaginary frequency, by calculation of the analytic harmonic vibrational frequencies at the SMD/B3lyp/6–31 + G(d) theory level.
2 Results and discussion
Modeled mechanisms for reaction between [2-(3-(fur-2-yl)-1,2,4-triazol-5-yl)phenyl]amine and cyclohexanone are shown in Figs. 1, 2, 3, 4, 5, while the mechanisms for interaction of [2-(3-(thiophen-2-yl)-1,2,4-triazol-5-yl)phenyl]amine and [2-(3-(pyrrol-2-yl)-1,2,4-triazol-5-yl)phenyl]amine with cyclohexanone are presented in Figs. S1 and S2 (SI), respectively. Calculated relative Gibbs free energies are discussed throughout the text.
SMD/B3lyp/6–31 + G(d) modeled pathways for reaction of [2-(3-(fur-2-yl)-1,2,4-triazol-5-yl)phenyl]amine with cyclohexanone in methanol along with the corresponding Gibbs free energy diagram. Numbers along with each of the arrow indicate the corresponding Gibbs free activation energies, while numbers under structures indicate Gibbs free energies for reactions in kcal/mol. Gibbs free activation energy is in italics in the diagram
SMD/B3lyp/6–31 + G(d) modeled pathways for reaction of [2-(3-(fur-2-yl)-2H-1,2,4-triazol-5-yl)phenyl]amine (R1o1) and [2-(3-(fur-2-yl)-2H-1,2,4-triazol-5-yl)phenyl]amine (R1o2) with protonated cyclohexanone in methanol along with the corresponding Gibbs free energy diagram. Numbers along with each of the arrow indicate the corresponding Gibbs free activation energies, while numbers under structures indicate Gibbs free energies for reactions in kcal/mol. Gibbs free activation energy is in italics in the diagram
SMD/B3lyp/6–31 + G(d) modeled pathways for reaction of protonated [2-(3-(fur-2-yl)-2H-1,2,4-triazol-5-yl)phenyl]amine (R1o1) with cyclohexanone in methanol along with the corresponding Gibbs free energy diagram. Numbers along with each of the arrow indicate the corresponding Gibbs free activation energies, while numbers under structures indicate Gibbs free energies for reactions in kcal/mol. Gibbs free activation energy is in italics in the diagram
SMD/B3lyp/6–31 + G(d) modeled pathways for reaction of protonated [2-(3-(fur-2-yl)-1H-1,2,4-triazol-5-yl)phenyl]amine (R1o2) with cyclohexanone in methanol along with the corresponding Gibbs free energy diagram. Numbers along with each of the arrow indicate the corresponding Gibbs free activation energies, while numbers under structures indicate Gibbs free energies for reactions in kcal/mol. Gibbs free activation energy is in italics in the diagram
SMD/B3lyp/6–31 + G(d) modeled pathways for acid-catalyzed reaction of [2-(3-(fur-2-yl)-1,2,4-triazol-5-yl)phenyl]amine with cyclohexanone in methanol along with the corresponding Gibbs free energy diagram. Numbers along with each of the arrow indicate the corresponding Gibbs free activation energies, while numbers under structures indicate Gibbs free energies for reactions in kcal/mol. Gibbs free activation energy is in italics in the diagram
In methanol solution [2-(3-(fur-2-yl)-1,2,4-triazol-5-yl)phenyl]amine exists as an equilibrium mixture of two tautomers [36]. The [2-(3-(fur-2-yl)-1H-1,2,4-triazol-5-yl)phenyl]amine (R1o2) is slightly more stable than the [2-(3-(fur-2-yl)-2H-1,2,4-triazol-5-yl)phenyl]amine (R1o1) (Fig. 1). The activation barrier for the isomerization catalyzed by three methanol molecules is 21.7 kcal/mol, that allows the process to occur at ambient temperature. Therefore, the reaction of [2-(3-(fur-2-yl)-1,2,4-triazol-5-yl)phenyl]amine with ketone was studied for both isomers.
2.1 Uncatalyzed mechanism
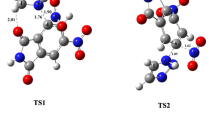
The addition of cyclohexanone to [2-(3-(fur-2-yl)-2H-1,2,4-triazol-5-yl)phenyl]amine (R1o1) requires a high activation energy and leads to unstable carbinolamine intermediates INT1 and INT2 (Fig. 1). The formation of a new C-N bond between the carbon of carbonyl group and the nitrogen N1 of triazole cycle is energetically more favorable, than the formation of C-N bond involving the nitrogen of amino group due to more stability of the five-membered structure of the transition state as compared with the four-membered one (Fig. S1). The reaction between [2-(3-(fur-2-yl)-1H-1,2,4-triazol-5-yl)phenyl]amine (R1o2) and cyclohexanone also has high activation barriers for formation of C-N bond between the carbon of carbonyl group and the nitrogen of amino group or the nitrogen N2 of triazole cycle leading to carbinolamine intermediates INT3 and INT4, respectively. If the proton transfer is assisted by methanol molecule, the activation energy decreases from 53.8 to 46.9 kcal/mol in case of formation of INT3. The decrease of activation energy occurs due to more stability of the six-membered structure of the transition state compared with the four-membered one (Fig. S1). Close location of NH2 and N1H groups in R1o2 allows them both to be involved in the reaction with R2. If the carbon of double bond C=O forms a new C-N bond with the nitrogen of amino group, and the oxygen takes proton from the nitrogen N1 of triazole, the activation barrier is significantly reduced to 30.6 kcal/mol. The decrease of activation barrier occurs due to lower steric tension of the formed transition state as compared with the four-membered one (Fig. S1). In addition, the interatomic distance C-N of newly formed bond is longer in transition state leading to INT5 (1.67 Å) than in one leading to INT3 (1.58 Å). Although the formed INT5 is 29.7 kcal/mol less stable than the reactants, the subsequent proton transfer between the two nitrogen atoms occurs quickly and increases stability of INT5. The elimination of water molecule from INT6 proceeds easy if the hydrogen from the triazole ring participates in the formation of the water molecule. Formed zwitterionic intermediate INT7 can decompose in two ways: a proton transfer between two nitrogen atoms leading to Schiff base INT8, and an attack of triazole nitrogen onto carbon of cyclohexane moiety that yields spiro compounds. Formation of C-N bond between the nitrogen atoms N1 and N4 of triazole cycle and the carbon of cyclohexane moiety requires activation energy of 7.8 and 12.3 kcal/mol, respectively, and leads to spiro compounds P1o and P2o, which are 12.3 and 0.6 kcal/mol, respectively, more stable, than INT7.
The difference in activation energy values can be explained by a higher nucleophilicity of nitrogen N1 compared to nitrogen N4. Obtained results show that the formation of Schiff base INT8 has a lower activation barrier than the formation of spiro compound P1o, while P1o is more stable than INT8. One can conclude that INT8 can be considered as a kinetic product, while P1o is a thermodynamic product of the studied reaction. This means that INT8 is formed and then rearranged into more stable P1o via INT7. The rate-limiting step is the addition of cyclohexanone to [2-(3-(fur-2-yl)-1H-1,2,4-triazol-5-yl)phenyl]amine. The activation barrier can be overcome under reaction condition (heating) [24].
2.2 Acid-catalyzed mechanism
In the presence of a strong acid, the protonation of [2-(3-(fur-2-yl)-1,2,4-triazol-5-yl)phenyl]amine as well as cyclohexanone can occur at the nitrogen atoms and the oxygen of carbonyl group, respectively (Figs. 2, 3, 4). The protonation of cyclohexanone is 5.2 kcal/mol exergonic. The addition of protonated cyclohexanone to [2-(3-(fur-2-yl)-2H-1,2,4-triazol-5-yl)phenyl]amine (R1o1) requires a significantly lower activation energy as compared with the addition of neutral cyclohexanone (Figs. 1 and 2). The formation of INT1h is the dominant pathway due to the lower energy barrier and higher stability of product as compared with the formation of INT2h and INT3h. The situation for reaction of protonated cyclohexanone with [2-(3-(fur-2-yl)-1H-1,2,4-triazol-5-yl)phenyl]amine (R1o2) is the same as for R1o1. The formation of a new C-N bond occurs more easily for the nitrogen of amino group than for the nitrogen atoms of triazole cycle. The activation energy for formation of INT4h is only 9.4 kcal/mol, that is considerably lower compared to the reaction for unprotonated cyclohexanone (Figs. 1 and 2). The decrease of activation energy can be explained by the increase of electrophilicity of the carbon atom of carbonyl group after protonation, that facilitates formation of a new C-N bond. Structures of transition states for reactions of R1o1 and R1o2 with protonated cyclohexanone (R2h) show a longer C-N bond formation distance (2.53, 2.41 Å) than in case of reaction with unprotonated cyclohexanone (1.57, 1.58 Å) (Fig. S1). According to the obtained results, the most energetically favorable pathway for reaction of [2-(3-(fur-2-yl)-1,2,4-triazol-5-yl)phenyl]amine with protonated cyclohexanone is the formation of INT1h and INT4h.
Protonation of [2-(3-(fur-2-yl)-2H-1,2,4-triazol-5-yl)phenyl]amine (R1o1) and [2-(3-(fur-2-yl)-1H-1,2,4-triazol-5-yl)phenyl]amine (R1o2) are highly acceptable (Figs. 3 and 4). The R1o1 and R1o2 protonated at the amino group are more stable than the protonated at the nitrogen atoms of triazole cycle. The addition of R2 to R1o1h1 occurs by a nucleophilic attack of nitrogen atoms N1 or N4 onto carbon of C=O bond with the simultaneous proton transfer from the protonated amino group to oxygen. The required activation energy is 33.5 and 41.1 kcal/mol, respectively. There are two pathways for the addition of R2 to R1o1h. The first pathway involves formation of a new C-N bond between the nitrogen N1 of triazole ring and the carbonyl carbon that is accompanied by a proton transfer from nitrogen N2 to oxygen. The second one consists of an attachment of nitrogen of amino group to carbon of C=O bond and a proton transfer from nitrogen N4 to oxygen. The activation barrier of the second pathway leading to INT1h is considerably lower than of the first one. The intermediate INT1h is also formed by the addition of R2 to R1oh. The reaction occurs by the formation of a C-N bond between the nitrogen of amino group and the carbonyl carbon, and the simultaneous proton transfer from nitrogen N1 to oxygen of C=O bond.
The attachment of R2 to nitrogen atoms N2 and N4 of R1o2h1 requires high activation energy of 38.3 and 40.5 kcal/mol to form high-lying intermediates INT8h and INT6h, respectively (Fig. 4). The same energy is needed for an addition of R2 to nitrogen N2 of R1o2h. Considerably lower activation barrier is calculated for an attachment of R2 to nitrogen of amino group in R1o2h with simultaneous proton transfer from nitrogen N4 to oxygen, that leads to INT4h. Protonated R1oh is formed from both isomers R1o1 and R1o2. Structures of the transition states for the formation of INT1h from R1oh and INT4h from R1o2h are asynchronous (Fig. S1). Proton transfer from nitrogen atom of triazole cycle to oxygen atom of cyclohexanone occurs before the formation of C-N bond. Earlier proton transfer leads to increasing of electrophilicity of carbon atom of carbonyl group, that promotes C-N bond formation.
According to the obtained results, the most energetically favorable pathway for reaction of protonated [2-(3-(fur-2-yl)-2H-1,2,4-triazol-5-yl)phenyl]amine (R1o1) and [2-(3-(fur-2-yl)-1H-1,2,4-triazol-5-yl)phenyl]amine (R1o2) with cyclohexanone (R2) is the formation of INT1h and INT4h (Figs. 3 and 4). These intermediates are also dominant in the case of interaction of [2-(3-(fur-2-yl)-1,2,4-triazol-5-yl)phenyl]amine with protonated cyclohexanone (Fig. 2). The activation energy for the acid-catalyzed addition of cyclohexanone to [2-(3-(fur-2-yl)-1,2,4-triazol-5-yl)phenyl]amine is significantly lower than for the uncatalyzed reaction (Figs. 1, 2, 3, 4).
The mechanism of further transformation of INT1h and INT4h during the studied reaction is shown in Fig. 5. It should be noted that INT1h and INT4h are tautomers with the close stability and exist simultaneously in the reaction mixture. The activation energy for the isomerization of INT1h into INT4h is 15.7 kcal/mol.
Dehydration of protonated carbinolamine INT1h occurs easier by a two-step mechanism than by a one-step transformation. Firstly, a proton is transferred between two nitrogen atoms, then a water molecule is eliminated with the participation of a hydroxy group and a proton at nitrogen N1. Intramolecular cyclization in INT3o occurs via bond formation between the nitrogen N1 or N4 of triazole and the carbon of cyclohexane moiety. A lower activation energy is required for formation of C-N1 bond. Deprotonation of INT5o by treatment with sodium acetate leads to product P1o.
Dehydration of INT4h also occurs in two steps: a proton transfer to nitrogen N4 leading to INT7o, and an elimination of a water molecule involving proton transfer from N1 or N4, that results in formation of INT8o or INT10o, respectively (Fig. 5). Intramolecular cyclization for INT8o occurs rapidly with an activation barrier of 7.4 kcal/mol. Deprotonation of INT9o yields P1o. Intermediate INT10o can undergo cyclization leading to INT11o or tautomerizes into INT3o, which transforms into spiro intermediate INT5o. The activation energy for both processes of INT10o transformation is similar, while tautomerization leads to more stable intermediate INT3o. Deprotonation of intermediates INT3o, INT8o, and INT10o leading to formation of Schiff bases INT12o, INT13o, and INT14o, respectively, is hard to achieve due to the high endergonicity of the process. Obtained results reveal that the dominant product for the transformation of INT1h and INT4h is P1o (Fig. 5). The result is consistent with the main product for uncatalyzed mechanism (Fig. 1) and with experimental data for interaction between [2-(3-R-1,2,4-triazol-5-yl)phenyl]amines and oxo compounds [24].
Comparative analysis of modeled reaction pathways for the acid-catalyzed interaction of [2-(3-(fur-2-yl)-1,2,4-triazol-5-yl)phenyl]amine, [2-(3-(thiophen-2-yl)-1,2,4-triazol-5-yl)phenyl]amine, and [2-(3-(pyrrol-2-yl)-1,2,4-triazol-5-yl)phenyl]amine with cyclohexanone showed that all three heterocycles react according to the same mechanism, and the corresponding activation energies differ insignificantly (Figs. 5, S2 and S3).
3 Conclusions
Triazole, a heterocyclic core, has attracted great attention among medicinal chemists in search of new therapeutic molecules. Increased amount of methods for synthesis of new biologically active compounds with several structural domains causes necessity to study the mechanism of the whole synthetic process. Reaction pathways of interaction between [2-(3-hetaryl-1,2,4-triazol-5-yl)phenyl]amine and cyclohexanone in methanol were modeled at the SMD/B3lyp/6–31 + G(d) theory level. Computational study showed that the mechanism consists of three main processes: addition of an amine to double bond C=O of cyclohexanone, elimination of a water molecule, and intramolecular cyclization leading to formation of a spiro compound. The ortho-position of the triazole substituent relative to the amino group on benzene ring enables intramolecular catalysis during the reaction process, that promotes the reaction. Acidic catalysis also facilitates the reaction by reducing activation barriers. The nature of the heterocyclic substituent on the triazole ring has little effect on the reaction energy, while the mechanism is unchanged.
Data availability
The data supporting the findings of this study are available within its supplementary material and from the corresponding author on reasonable request.
References
Kazeminejad Z, Marzi M, Shiroudi A, Kouhpayeh SA, Farjam M, Zarenezhad E (2022) Novel 1, 2, 4-triazoles as antifungal agents. Biomed Res Int 2022:1–39. https://doi.org/10.1155/2022/4584846
Maddila S, Pagadala R, Jonnalagadda BS (2013) 1, 2, 4-triazoles: a review of synthetic approaches and the biological activity. Lett Org Chem 10:693–714. https://doi.org/10.2174/157017861010131126115448
Kaur P, Chawla A (2017) 1, 2, 4-triazole: a review of pharmacological activities. Int Res J Pharm 8:10–29. https://doi.org/10.7897/2230-8407.087112
Kapron B, Luszczki JJ, Plaziska A, Siwek A, Karcz T, Grybos A, Bowak G, Makuch-Kocka A, Walczak K, Langner E, Szalast K, Marciniak S, Paczkowska M, Cielecka J, Ciesla LM, Plech T (2019) Development of the 1, 2, 4-triazole-based anticonvulsant drug candidates acting on the voltage-gated sodium channels. Insights from in-vivo, in-vitro, and in-silico studies. Eur J Pharm Sci 129:42–57. https://doi.org/10.1016/j.ejps.2018.12.018
Gao F, Wang T, Xiao J, Huang G (2019) Antibacterial activity study of 1, 2, 4-triazole derivatives. Eur J Med Chem 173:274–281. https://doi.org/10.1016/j.ejmech.2019.04.043
Othman AA, Kihel M, Amara S (2019) 1, 3, 4-oxadiazole, 1, 3, 4-thiadiazole and 1, 2, 4-triazole derivatives as potential antibacterial agents. Arab J Chem 12:1660–1675. https://doi.org/10.1016/j.arabjc.2014.09.003
Shahzad SA, Yar M, Khan ZA, Shahzadi L, Naqvi SAR, Mahmood A, Ullah S, Shaikh AJ, Sherazi TA, Bale AT, Kukulowicz J, Bajda M (2019) Identification of 1, 2, 4-triazoles as new thymidine phosphorylase inhibitors: Future anti-tumor drugs. Bioorg Chem 85:209–220. https://doi.org/10.1016/j.bioorg.2019.01.005
El-Sherief HAM, Youssif BGM, Bukhari SNA, Abdelazeem AH, Abdel-Aziz MA-RHM (2018) Synthesis, anticancer activity and molecular modeling studies of 1, 2, 4-triazole derivatives as EGFR inhibitors. Eur J Med Chem 156:774–789. https://doi.org/10.1016/j.ejmech.2018.07.024
Wittine K, Babic MS, Makuc D, Plavec J, Pavelic SK, Sedic M, Pavelic K, Leyssen P, Neyts J, Balzarini J, Nintas M (2012) Novel 1, 2, 4-triazole and imidazole derivatives of L-ascorbic and imino-ascorbic acid: synthesis, anti-HCV and antitumor activity evaluations. Bioorg Med Chem 20:3675–3685. https://doi.org/10.1016/j.bmc.2012.01.054
Zhang S, Xu Z, Gao C, Ren QC, Chang L, Lv ZS, Feng LS (2017) Triazole derivatives and their anti-tubercular activity. Eur J Med Chem 138:501–513. https://doi.org/10.1016/j.ejmech.2017.06.051
Jin RY, Zeng CY, Liang XH, Sun XH, Liu YF, Wang YY, Zhou S (2018) Design, synthesis, biological activities and DFT calculation of novel 1, 2, 4-triazole Schiff base derivatives. Bioorg Chem 80:253–260. https://doi.org/10.1016/j.bioorg.2018.06.030
Xu J, Cao Y, Zhang J, Yu S, Chai X, Wu Q, Zhang D, Jiang Y, Sun Q (2011) Design, synthesis and antifungal activities of novel 1, 2, 4-triazole derivatives. Eur J Med Chem 46:3142–3148. https://doi.org/10.1016/j.ejmech.2011.02.042
Fan YL, Ke X, Li M (2018) Coumarin–triazole hybrids and their biological activities. J Heterocycl Chem 55:791–802. https://doi.org/10.1002/jhet.3112
Mishra SS, Singh P (2016) Hybrid molecules: the privileged scaffolds for various pharmaceuticals. Eur J Med Chem 124:500–536. https://doi.org/10.1016/j.ejmech.2016.08.039
Hu YQ, Gao C, Zhang S, Xu L, Xu Z, Feng LS, Wu X, Zhao F (2017) Quinoline hybrids and their antiplasmodial and antimalarial activities. Eur J Med Chem 139:22–47. https://doi.org/10.1016/j.ejmech.2017.07.061
Amin KM, Anwar MM, Syam YM, Khedr M, Kamel MM, Kassem EMMA (2013) A novel class of substituted spiro[quinazoline-2,1’-cyclohexane] derivatives as effective ppar-1 inhibitors: molecular modeling, synthesis, cytotoxic and enzyme assay evaluation. Acta Pol Pharm Drug Res 7:687–708
Mustazza C, Borioni A, Sestili I, Sbraccia M, Rodomonte A, Ferretti R, Rosaria Del Giudice M (2006) Synthesis and evaluation as NOP ligands of some spiro[piperidine-4,2’(1’H)-quinazolin]-4’(3’H)-ones and spiro[piperidine-4,5’(6’H)-[1,2,4]triazolo[1,5-c]quinazolines]. Chem Pharm Bull 54:611–622. https://doi.org/10.1248/cpb.54.611
Antypenko OM, Kholodnyak SV, Schabelnyk KP, Antypenko LM, Kovalenko SI (2017) 2-(Azolyl) anilines: methods of synthesis, cyclocondensations, and biological properties. Chem Heterocycl Comp 53:292–309. https://doi.org/10.1007/s10593-017-2051-7
Kehler J, Ritzen A, Langgard M, Petersen SL, Farah MM, Bundgaard C, Christoffersen CT, Nielsen J, Kilburn JP (2011) Triazoloquinazolines as a novel class of phosphodiesterase 10A (PDE10A) inhibitors. Bioorg Med Chem Lett 21:3738–3742. https://doi.org/10.1016/j.bmcl.2011.04.067
Antipenko LN, Karpenko AV, Kovalenko SI, Katzev AM, Komarovska-Porokhnyavets EZ (2009) Synthesis, cytotoxicity by bioluminescence inhibition, antibacterial and antifungal activity of ([1,2,4]Triazolo[1,5-c]quinazolin-2-ylthio)carboxylic acid amides. Arch Pharm Chem Life Sci 342:651–662. https://doi.org/10.1002/ardp.200900077
Wesseler F, Lohmann S, Riege D, Halver J, Roth A, Pichlo C, Weber S, Takamiya M, Muller E, Ketzel J, Flegel J, Gihring A, Rastegar S, Bertrand J, Baumann U, Knippschild U, Peifer C, Sievers S, Waldmann H, Schade D (2022) Phenotypic discovery of Triazolo[1,5-c]quinazolines as a first-in-class bone morphogenetic protein amplifier chemotype. J Med Chem 65:15263–15281. https://doi.org/10.1021/acs.jmedchem.2c01199
Zeydi MM, Montazeri N, Fouladi M (2017) Synthesis and evaluation of novel [1,2,4]Triazolo[1,5-c]quinazoline derivatives as antibacterial agents. J Heterocycl Chem 54:3549–3553. https://doi.org/10.1002/jhet.2979
Kovalenko SI, Antypenko LM, Bilyi AK, Kholodnyak SV, Karpenko OV, Antypenko OM, Mykhaylova NS, Los TI, Kolomoets OS (2013) Synthesis and anticancer activity of 2-(alkyl-, alkaryl-, aryl-, hetaryl-)-[1, 2, 4] triazolo [1, 5-c] quinazolines. Sci Pharm 81:359–391. https://doi.org/10.3797/scipharm.1211-08
Kholodnyak SV, Schabelnyk KP, Voskoboynik OYu, Antypenko OM, Кovalenko SI, Palchykov VO, Okovyty SI, Shishkina SV (2016) 5,6-dihydro-[1,2,4]triazolo[1,5-c]quinazolines. message 4. Spirocompounds with [1,2,4]triazolo[1,5-c] quinazolines moieties. The synthesis and spectral characteristics. J Org Pharm Chem 14:24–31
Cmikiewicz A, Gordon AJ, Berski S (2018) Characterisation of the reaction mechanism between ammonia and formaldehyde from the topological analysis of ELF and catastrophe theory perspective. Struct Chem 29:243–255. https://doi.org/10.1007/s11224-017-1024-x
Hall NE, Smith BJ (1998) High-level ab initio molecular orbital calculations of imine formation. J Phys Chem A 102:4930–4938. https://doi.org/10.1021/jp9810825
Ding Y, Cui Y, Li T (2015) New views on the reaction of primary amine and aldehyde from DFT study. J Phys Chem A 119:4252–4260. https://doi.org/10.1021/acs.jpca.5b02186
Silva PJ (2020) New insights into the mechanism of Schiff base synthesis from aromatic amines in the absence of acid catalyst or polar solvents. PeerJ Org Chem 2:1–4. https://doi.org/10.7717/peerj-ochem.4
Solís-Calero C, Ortega-Castro J, Hernàndez-Laguna A, Muñoz F (2012) Reaction mechanism of covalent modification of phosphatidylethanolamine lipids by reactive aldehydes 4-hydroxy-2-nonenal and 4-oxo-2-nonenal. Theor Chem Acc 131:1–12. https://doi.org/10.1021/acs.chemrestox.6b00443
Ortega-Castro J, Adrover M, Frau J, Salvà A, Donoso J, Muñoz F (2010) DFT studies on Schiff base formation of vitamin B6 analogues. Reaction between a pyridoxamine-analogue and carbonyl compounds. J Phys Chem A 114:4634–4640. https://doi.org/10.1021/jp909156m
Qu ZW, Zhu H, Zhukova NA, Katsyuba SA, Mamedov VA, Grimme S (2021) Mechanistic insights for acid-catalyzed rearrangement of quinoxalin-2-one with diamine and enamine. ChemCatChem 13:1503–1508
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima,T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JrJA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski,VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ( 2009) Gaussian 09, Revision A.01. Gaussian Inc., Wallingford
Becke ADJ (1993) Density-functional thermochemistry.3.The role of exact exchange. Chem Phys 98:5648–5652
Hehre WJ, Radom L, Schleyer PR, Pople JA (1986) Ab initio molecular orbital theory. Wiley, New York
Marenich AV, Cramer CJ, Truhlar DG (2009) Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J Phys Chem B 13:6378–6396. https://doi.org/10.1021/jp810292n
Pylypenko OO, Okovytyy SI, Sviatenko LK, Voronkov EO, Shabelnyk KP, Kovalenko SI (2023) Tautomeric behavior of 1, 2, 4-triazole derivatives: combined spectroscopic and theoretical study. Struct Chem 34:181–192. https://doi.org/10.1007/s11224-022-02057-0
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
OP calculated and interpreted data and was a contributor in writing the manuscript. LS calculated and interpreted data and was a contributor in writing the manuscript. KS was a contributor in writing the manuscript. SK was a contributor in writing the manuscript. SO interpreted data and was a contributor in writing and editing the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
214_2024_3110_MOESM1_ESM.docx
SMD/B3lyp/6-31+G(d) modeled pathways for acid-catalyzed reaction of 2-(3-(thiophen-2-yl)-1,2,4-triazol-5-yl)anilines and 2-(3-(pyrrol-2-yl)-1,2,4-triazol-5-yl)anilines with cyclohexanone in methanol along with the corresponding Gibbs free energy diagram (DOCX 4789 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pylypenko, O.O., Sviatenko, L.K., Shabelnyk, K.P. et al. Reaction of [2-(3-hetaryl-1,2,4-triazol-5-yl)phenyl]amines with ketones: a density functional theory study. Theor Chem Acc 143, 35 (2024). https://doi.org/10.1007/s00214-024-03110-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-024-03110-3