Abstract
Summary
The risk factors for atypical femur fracture in patients exposed to bisphosphonates for at least 1 year were examined. Prolonged and continuous use of bisphosphonates, long-term use of glucocorticoids, and a higher body mass index were associated with increased risk of atypical femur fracture.
Introduction
The purpose of the present study is to determine whether rheumatoid arthritis (RA) and other clinical factors are associated with an increased risk of bisphosphonate (BP)-related atypical femur fracture (AFF).
Methods
A retrospective nested case-control study of patients who had taken BPs for at least 1 year was conducted. Patients with AFF were identified by reviewing surgical and radiographic records. Three controls with no history of AFFs were randomly selected and age- and sex-matched to each patient with AFFs. Cox proportional hazard models were used to analyze the independent contribution of risk factors to BP-related AFF.
Results
Among the 35,104 patients prescribed BPs for at least 1 year, 43 females (mean age, 68 years) suffered AFFs (0.12%). Patients with AFFs were exposed to BPs for a mean of 7.3 years. Patients with AFFs were exposed to BPs for longer than those without AFFs and continued treatment without a drug holiday. More patients with AFF than controls had taken glucocorticoids and disease-modifying anti-rheumatic drugs. Multivariate Cox regression analyses estimated that long-term use of glucocorticoids, prolonged exposure to BP without cessation, and every 1 kg/m2 increase in the body mass index (BMI) increased the hazard ratio for AFFs by 3.0, 5.2, and 1.2, respectively.
Conclusions
Prolonged and continuous use of BPs, long-term use of glucocorticoids, and a higher BMI increase the risk of AFFs. Switching long-term BP and glucocorticoid users to other bone-protective agents should be considered.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bisphosphonates (BPs) are widely used to treat and prevent osteoporosis. BPs are adsorbed to the bone mineral surface and internalized by bone-resorbing osteoclasts [1]. By inhibiting bone resorption, BPs increase bone mineral density (BMD) and reduce the risk of fracture [2, 3].
The age-adjusted incidence of hip fracture has declined (a 24% decrease in women and a 20% decrease in men) since 1995, which corresponds to the approval and widespread use of BPs [4, 5]. These drugs are generally safe, with the most common adverse events occurring in the upper gastrointestinal tract. However, since BPs were linked to osteonecrosis of the jaw (ONJ) [6] and atypical femur fracture (AFF) [7], concerns over potential serious adverse events (AEs) have increased. BP-related ONJ is rare, and most cases occur in patients with metastatic bone disease receiving high doses of drugs [8, 9]. BP-associated AFF is also rare: 1.6 to 18.7 cases per 100,000 patient-years [10, 11]. Despite this, a US Food and Drug Administration announcement regarding the safety of BPs led to a reduction in their use [12].
The incidence of AFFs among postmenopausal women taking BPs for at least 5 years is 0.22% [13]. In a previous study, we found that 1.8% of patients with rheumatoid arthritis (RA) and exposed to BP for least 1 year suffered AFF [14]. This incidence, although relatively low, is higher than that reported by other population studies. The risk of AFF increases with long-term BP treatment; however, no other risk factors have been identified [15]. There are few data on the relationship between RA and the risk of AFF. Furthermore, the relative contribution of oral glucocorticoids (which are often used to treat RA) to the increased risk of AFF remains unclear. Therefore, the aims of this study were to identify subgroups of patients on long-term BP that may suffer AFF.
Methods
Study population
This was a retrospective, nested case-control study of patients recruited from Seoul St. Mary’s Hospital, which is a 1339-bed teaching, tertiary referral/general hospital in Seoul, Republic of Korea. Patients treated with BPs for at least 1 year between 2008 and 2015 were identified from the hospital electronic health record (EHR). Then, possible AFF subjects were identified from the EHR using the Korean Classification of Disease seventh edition (KCD-7) diagnosis codes (National Clinical Modification of ICD-10) for subtrochanteric and diaphyseal femoral fractures (S722, S723, S724, and S728). The lower extremity radiographs and surgical records of all possible AFF subjects were retrospectively reviewed.
In accordance with the previously described definition of AFF, the fractures had to include at least four of the following features: (1) atraumatic or low-trauma fracture of the subtrochanteric or femoral shaft; (2) the fracture is non-comminuted or minimally comminuted; (3) the fracture line originates at the lateral cortex and is substantially transverse in orientation, although it may become oblique as it passes medially across the femur; (4) a medial spike in cases of a complete fracture and a fracture in the lateral cortex only in cases of incomplete fracture; and (5) localized periosteal or endosteal thickening of the lateral cortex [15].
During the 8-year study period, 35,104 patients received BPs (alendronate, risedronate, ibandronate, or zoledronate) for at least 1 year. Of these, 198 patients in the EHR were identified as having a diagnosis code for subtrochanteric and diaphyseal femoral fracture. Patients with a history of cancer during the preceding 5 years and those with conditions associated with pathologic fracture (e.g., hyperparathyroidism, renal osteodystrophy, or osteomalacia) were excluded. During this time, 43 postmenopausal women suffered AFF (0.12%). Controls were women who received BPs but did not experience AFF. AFF cases were frequency matched according to 10-year age intervals, at a case/control ratio of 1:3.
Age at the time of first AFF, body mass index (BMI), comorbid conditions, and documented presence of prodromal pain were recorded. The duration of BP exposure was assessed by recording the length of time for which patients were prescribed oral alendronate, risedronate, ibandronate, intravenous ibandronate, or zoledronate. Patients prescribed BPs for 5 years or more were considered to be on long-term BP treatment. A drug holiday was defined as temporary discontinuation of BPs for a period of at least 6 months to 5 years after at least 1 year of treatment. The use of other anti-osteoporotic agents such as selective estrogen receptor modulators, calcitonin, or teriparatide was also taken into account. The daily dose of prednisolone (or equivalent) within the 6 months prior to AFF, and the total accumulated dose of glucocorticoids, was recorded. Long-term glucocorticoid therapy was defined as glucocorticoid therapy lasting for at least 1 year. Any prior history of vertebral or non-vertebral fracture (VF/NVF) was identified by chart review. Fracture bilateralism, fracture site, and occurrence of delayed healing after surgical treatment were reviewed. Concomitant medications considered to affect bone and mineral metabolism were also reviewed.
Measurement of BMD
BMD data were obtained using Lunar Prodigy dual-energy x-ray absorptiometry (DXA) (GE Healthcare Lunar, Madison, WI, USA). For patients with AFFs, the BMD of the femur prior to fracture was noted. For patients without AFFs, the latest BMD data were assessed. Available serial BMD data from the preceding 3 years were used to track changes in BMD. The lumbar spine trabecular bone score (TBS) was calculated after reanalysis of the latest DXA lumbar spine scans using the TBS iNsight® software (Version 2.2.0.0, Med-Imaps, Bordeaux, France).
Radiographic measurement
A previous report suggests that the site of an AFF is affected by the alignment of the lower limb under weight-bearing conditions [16]. Standing knee radiographs of 78 patients (23 patients with AFFs and 55 controls) were available for analysis. The femorotibial angle (FTA) on anteroposterior standing knee radiographs was measured by two independent and trained rheumatologists blinded to the patient characteristics. The FTA, which is the lateral angle formed at the intersection between the femoral axis and the tibial axis, was measured as previously described [17]. The mean standing FTA for the Korean population is 177 ± 2.4° [18]. We defined a valgus deformity of the knee as a FTA less than 175° and a varus deformity of the knee as a FTA greater than 181°. To assess intraobserver repeatability and interobserver agreement, all radiographic measurements were repeated twice by both rheumatologists: the interclass correlation coefficients (ICC) for intraobserver repeatability were 0.939 (95% confidence interval [CI], 0.892–0.965) and 0.968 (95% CI, 0.950–0.980). The ICC for interobserver agreement was 0.828 (95% CI, 0.729–0.890). Thus, the average value was used in the final analysis.
Statistical analyses
The normality of the parameters was assessed using the Shapiro-Wilk test. Comparisons between groups were made using the Student’s t test or the Mann-Whitney U test as appropriate. Categorical data were compared using the χ 2 test or Fisher’s exact test. A P value <0.05 was considered statistically significant. The ICC was used to assess the intraobserver reliability and the interobserver agreement between FTA measurements.
Cox proportional hazard regression models were used to examine the association between potential contributors and AFF occurrence, with observed time from the start of BP treatment as the time-dependent variable. The first AFF was taken as the failure time and the last visit (if no AFF occurred) as the censor time. The models were run in a univariate manner and then repeated after adjusting for potential multicollinearity between RA, glucocorticoids, and disease-modifying anti-rheumatic drugs (DMARDs). The AFF group and control group were age-matched with controls, and the matched numbers were considered when performing multiple analyses. To avoid multicollinearity between RA, glucocorticoids, and DMARDs, each variable was added respectively into the Cox proportional hazard regression model. The most relevant regression model was selected based on minimization of the Aikake information criterion (AIC) and parsimony. Statistical analysis of pooled data was performed using the SAS software (Version 9.4; SAS Institute Inc., Cary, NC, USA).
Ethics statement
The study was approved by the Institutional Review Board of Seoul St. Mary’s Hospital (KC14RISI0555). The requirement for informed consent was waived due to the retrospective nature of the study.
Results
Demographic characteristics
The mean age of the 43 patients with AFF was 68.1 years. Comorbidities included osteoarthritis (34.9%), hypertension (41.9%), RA (27.9%), and diabetes mellitus (18.2%). Patients with AFF had a higher BMI than control subjects who had taken BPs for at least 1 year but did not suffer AFF (Table 1). The mean duration of BP treatment was 7.3 years for patients with AFF and 5.2 years for patients without. Seven patients with AFF (16.3%) had a drug holiday, whereas 46 (35.7%) patients without AFF discontinued treatment. The median duration of BP cessation was 1.6 years (IQR, 0.8–2.8).
Patients with AFF tended to suffer from RA and so were also prescribed DMARDs and glucocorticoids prior to AFF. More patients with AFF received glucocorticoids for at least 1 year than those without. The cumulative glucocorticoid dose was also higher in patients with AFF than in those without, although the duration of glucocorticoid treatment was similar. There were no differences in the use of other medications such as non-steroidal anti-inflammatory drugs, calcium/vitamin D supplements, and proton pump inhibitors between the groups.
Fracture characteristics
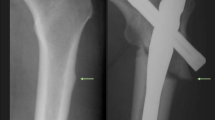
Three quarters of AFFs occurred in the proximal one third of the femoral shaft (32 cases), and the most common type was a complete fracture (81.4%). Focal lateral cortical thickening with a radiolucent transverse incomplete fracture line was observed in the other eight patients. Focal cortical thickening before AFF was detected on plain radiographs or DXA images from 12 patients (27.9%) (Fig. 1a, b). Bilateral femur involvement was noted in 16 patients (37.2%), four of whom showed bilateral focal cortical thickening, seven of whom showed unilateral complete fracture and contralateral focal cortical thickening, and five of whom showed sequential fracture (Fig. 1c–e). The median interval between sequential bilateral fractures was 26.1 months.
Subtrochanteric, sequential bilateral atypical femur fracture. a The radiograph was obtained from a patient complaining of hip pain. A small area of periosteal and endosteal thickening of the lateral cortex is visible (white arrow). b Note the transverse cortical thickening of the lateral cortex on the three-dimensional computed tomography (CT) reconstruction. c Radiograph obtained 2 years after prophylactic intramedullary rod placement. Note the localized periosteal thickening of the lateral cortex at the contralateral fracture site (white arrow). d, e Radiograph and three-dimensional CT scan obtained immediately after fracture. A transverse line in the lateral cortex becomes oblique as it progresses medially across the femur
Osteoporotic fracture after initiating BP treatment was more common in patients with AFFs than in patients without. Half of osteoporotic fractures (nine) occurred 2.0 ± 1.3 years (mean ± SD) after discontinuing BPs, and half occurred during continued use of BPs for 6.4 ± 2.9 years (mean ± SD). Clinical vertebral compression fracture was the most common type of osteoporotic fracture in this study (12 patients). The forearm was the next most common site of osteoporotic fracture (four patients). One patient suffered a femoral neck fracture and one suffered a shoulder fracture. Among the 12 patients with AFF, ten suffered vertebral fracture and two suffered a forearm fracture. Five patients experienced osteoporotic fracture after 4.0 ± 2.1 years (mean ± SD) from occurrence of AFF. The other five patients experienced osteoporotic fracture before AFF but during treatment with BPs. Two patients experienced osteoporotic fracture during a drug holiday before AFF.
Subgroup analysis of osteoporotic fracture in patients with AFF revealed that the mean BMD and T-score for the first to fourth lumbar vertebra were significantly lower in AFF patients with osteoporotic fracture than in patients with only AFF. The BMD of the total hip, femoral neck, and shaft was not different between patients with and without osteoporotic fracture (Supplementary Table 1). Other clinical parameters such as the duration of BP treatment, comedications, BMI, and age were not significantly different.
BMD of patients with and without AFF
The medial time between occurrence of AFF and the latest DXA scan before fracture was 8.1 months (IQR, 5.9–10.9). There was no difference in the BMD or T-score for the total hip, trochanter, femoral neck, and lumbar spine between patients with AFF and the control group (Table 2). There was no difference in the lumbar TBS scores between the two groups. There was no difference in the percentage change in femoral neck BMD during the last 3 years between patients with and without AFFs.
Predictors of AFF in patients treated with BPs
Variables with a P value <0.1 from univariate analyses are listed in Table 3. BMI and continued BP treatment without a drug holiday were common factors. As multicollinearity between RA, DMARDs, and long-term use of glucocorticoids was a possibility, each variable was added respectively to the Cox proportional hazard model (Table 3; models 1 to 3). Cox proportional hazard model 1 was the most relevant and identified BMI (per kg/m2; HR, 1.24; 95% CI, 1.109–1.391), long-term use of glucocorticoids (HR, 3.036; 95% CI, 1.1403–6.568), and prolonged BP treatment without a drug holiday (HR, 5.170; 95% CI, 2.0–13.362) as factors related to increased risk of AFF after initiating BP treatment.
Subgroup analysis of patients according to the measured FTA
FTA was measured in 23 patients with AFF and 55 patients without AFF. The mean FTA was not different between the two groups. However, patients with AFF were more likely to have genu valgus (FTA <175°) than those without AFFs (47.8 vs. 14.6%, respectively; P = 0.002). Abnormal lower limb alignment, including genu varus and genu valgus (OR, 4.036; 95% CI, 1.183–13.765), was significantly associated with AFF (Supplementary Table 2).
Discussion
Here, we found that 0.12% of Korean female patients exposed to BPs for at least 1 year suffered AFF. Regardless of the presence of RA as a comorbidity, long-term use of glucocorticoids increased the risk of AFF after initiating BPs. In addition, prolonged BP treatment without a drug holiday and a higher BMI were associated with a higher risk of AFF.
The association between use of glucocorticoids and AFF is controversial [19,20,21]; however, the data presented herein suggest that glucocorticoids increase the risk of BP-related AFF. Concurrent long-term administration of BP and glucocorticoids may lead to marked suppression of bone turnover. The main mechanism underlying glucocorticoid-induced osteoporosis (GIOP) is the direct action of the drugs on osteoblasts and osteoclasts. Glucocorticoids inhibit osteoblast differentiation and proliferation and induce apoptosis of osteoblasts and osteocytes [22, 23]. They also prolong the life span of osteoclasts and reduce the number of osteoclast precursors [24]. Thus, treatment with glucocorticoids leads to a loss of cortical and cancellous bone and increases fractures due to bone fragility [25]. BPs improve BMD and reduce the risk of fracture and are recommended for the prevention and treatment of GIOP (treatment lasts for at least 24 months) [26,27,28]. However, long-term use of glucocorticoids suppresses bone turnover by inhibiting the activity of both osteoblasts and osteoclasts [29]. The additive effect of glucocorticoids plus BPs with respect to suppression of bone turnover is supported by an in vivo study demonstrating that a steroid-releasing pellet implanted into mice injected daily with a BP significantly reduced the mineralizing surface, the mineral apposition rate, and bone formation [30]. Profound suppression of bone turnover could lead to accumulation of microcracks, which is one possible explanation for AFF [31, 32]; this may explain the increased risk of AFF in those on long-term concurrent treatment with BPs and glucocorticoids.
Prolonged BP treatment without a drug holiday was a major risk factor for AFF. Reversal of this effect after discontinuing BPs varies according to individual clinical factors [1]. Although the mechanism underlying the reduced rate of AFFs after a drug holiday remains unknown, the risk falls by 70% per year after the last dose [33]. Recently published recommendations for postmenopausal osteoporosis patients on long-term BP therapy suggest that for patients at low risk of osteoporotic fracture, a drug holiday of 2 to 3 years can be considered after they have been taking BPs for 3 to 5 years [34]. However, we observed more osteoporotic fractures in patients with AFF than in those without. Half of the patients sustained a fracture after discontinuing BPs. Patients with osteoporotic fracture had a significantly lower BMD and TBS in the lumbar spine than those without. Therefore, women at high risk of osteoporotic fracture (i.e., older women and those with a low hip T-score or high fracture risk score) should be considered for extended BP treatment [34].
In addition, there is no consensus with respect to the timing and duration of discontinuation in patients with GIOP. Substantial loss of BMD is observed in patients who discontinue BPs while taking glucocorticoids [35]. Thus, it is usually recommended that BPs are continued for as long as glucocorticoids are prescribed [36]. Unfortunately, glucocorticoids are often prescribed for several years to many patients with RA and other autoimmune diseases. Given that the efficacy of BPs in preventing bone loss is greater in the setting of “high-turnover” than in the setting of “low-turnover” osteoporosis [37], switching long-term glucocorticoid users to anabolic anti-osteoporotic agents could be considered after prolonged treatment with BPs. Further large and long-term follow-up studies are needed to examine osteoporotic fracture and AEs and to develop strategies involving the use of bone-protective agents in patients with GIOP.
We have demonstrated that development of AFF is associated with abnormal lower limb alignment. This result is consistent with those of an earlier study showing that patients with varus alignment suffered diaphyseal fracture, whereas those with valgus alignment tended to suffer subtrochanteric fracture [16, 38]. Moreover, 37% of patients that suffered AFF had bilateral involvement at the same location on the contralateral femur. Patients suffering AFF were also shorter and heavier. These results indicate that abnormal loading can be intensified by altered lower limb alignment and an increase in BMI. Thus, the proximal femur may be subjected to abnormal and sustained mechanical loading in the proximal femur, which may contribute to the development of AFF. The incidence of femoral bowing is significantly greater in Asian women than in Caucasian women [18]. While no definitive conclusions can be reached regarding the role of altered lower limb alignment in the development of AFF, the association would explain the high incidence of AFF in Asian women [11].
This study has several limitations. First, it was a single-center study that included only female patients (because only female patients experienced fracture). Second, because this was a retrospective study, we did not evaluate bone absorption markers and vitamin D in the whole population at the time of AFF. Available data suggested that there were no differences in the expression of bone absorption markers between patients with and without AFF. Third, it is not clear whether patients with RA have a higher risk of AFF. As RA itself affects osteoclast differentiation [39], the question was whether it is the RA itself or glucocorticoids that affected the occurrence of AFF. As DMARDs and glucocorticoids are widely used therapeutic agents for RA, multicollinearity exists between those factors. Indeed, DMARDs were prescribed to 90% of RA patients and long-term glucocorticoids to 81% of RA patients, in the present study. To clarify this hypothesis, prospective long-term cohort studies that include an adequate number of AFF cases and controls are required. Fourth, weight-bearing whole-leg radiography is ideal for estimating lower limb alignment; however, we used the FTA calculated from standing knee radiographs. This is because standing knee radiographs were the most readily available weight-bearing radiographs for the lower limb before AFF.
Despite these limitations, the present study has several strengths. First, it included patients treated with BPs for at least 1 year and followed them after treatment began. We obtained long-term follow-up data (including BMD and detailed medication histories) from all patients. No comparative study of BPs versus placebo in patients on prolonged GC treatment is available; therefore, long-term safety data with respect to BP therapy in those taking glucocorticoids are lacking. This study suggests that long-term administration of BPs has an indirect impact on bone development in those taking long-term glucocorticoids. Second, we demonstrated that AFF is affected by mechanical limb alignment. To identify mechanical factors that predispose to AFF, many studies measured cortical thickness or bone microarchitecture; however, they found no evidence that thick femoral cortices and deterioration of bone microarchitecture placed patients at higher risk of AFF [40,41,42].
A drug holiday in selected subjects after 3 to 5 years of treatment may reverse the reduction in bone turnover. Such patients should be selected according to osteoporotic fracture risk, BMI, and lower limb alignment. Clinicians should exercise vigilance with respect to patients on long-term glucocorticoids and undergoing prolonged treatment with BPs to identify AEs due to profound suppression of bone turnover. Although there is an association between AFF and long-term BP administration without a drug holiday, AFF is still rare; thus, the benefit of BPs outweighs the potential risk.
References
Russell RG, Watts NB, Ebetino FH, Rogers MJ (2008) Mechanisms of action of bisphosphonates: similarities and differences and their potential influence on clinical efficacy. Osteoporos Int 19(6):733–759
Hochberg MC, Ross PD, Black D, Cummings SR, Genant HK, Nevitt MC, Barrett-Connor E, Musliner T, Thompson D (1999) Larger increases in bone mineral density during alendronate therapy are associated with a lower risk of new vertebral fractures in women with postmenopausal osteoporosis. Fracture Intervention Trial Research Group. Arthritis Rheum 42(6):1246–1254
Hochberg MC, Greenspan S, Wasnich RD, Miller P, Thompson DE, Ross PD (2002) Changes in bone density and turnover explain the reductions in incidence of nonvertebral fractures that occur during treatment with antiresorptive agents. J Clin Endocrinol Metab 87(4):1586–1592
Brauer CA, Coca-Perraillon M, Cutler DM, Rosen AB (2009) INcidence and mortality of hip fractures in the united states. JAMA 302(14):1573–1579
Leslie WD, O’Donnell S, Jean S et al (2009) TRends in hip fracture rates in canada. JAMA 302(8):883–889
Marx RE (2003) Pamidronate (Aredia) and zoledronate (Zometa) induced avascular necrosis of the jaws: a growing epidemic. J Oral Maxillofac Surg 61(9):1115–1117
Odvina CV, Zerwekh JE, Rao DS, Maalouf N, Gottschalk FA, Pak CY (2005) Severely suppressed bone turnover: a potential complication of alendronate therapy. J Clin Endocrinol Metab 90(3):1294–1301
Solomon DH, Mercer E, Woo SB, Avorn J, Schneeweiss S, Treister N (2013) Defining the epidemiology of bisphosphonate-associated osteonecrosis of the jaw: prior work and current challenges. Osteoporos Int 24(1):237–244
Suresh E, Pazianas M, Abrahamsen B (2014) Safety issues with bisphosphonate therapy for osteoporosis. Rheumatology (Oxford) 53(1):19–31
Pedrazzoni M, Giusti A, Girasole G, Abbate B, Verzicco I, Cervellin G (2016) Atypical femoral fractures in Italy: a retrospective analysis in a large urban emergency department during a 7-year period (2007–2013). J Bone Miner Metab. doi:10.1007/s00774-016-0790-7
Lo JC, Hui RL, Grimsrud CD, Chandra M, Neugebauer RS, Gonzalez JR, Budayr A, Lau G, Ettinger B (2016) The association of race/ethnicity and risk of atypical femur fracture among older women receiving oral bisphosphonate therapy. Bone 85:142–147
Kim SC, Kim DH, Mogun H, Eddings W, Polinski JM, Franklin JM, Solomon DH (2016) Impact of the US Food and Drug Administration’s safety-related announcements on the use of bisphosphonates after hip fracture. J Bone Miner Res 31(8):1536–1540
Park-Wyllie LY, Mamdani MM, Juurlink DN, Hawker GA, Gunraj N, Austin PC, Whelan DB, Weiler PJ, Laupacis A (2011) Bisphosphonate use and the risk of subtrochanteric or femoral shaft fractures in older women. JAMA 305(8):783–789
Koh JH, Myong JP, Jung SM, Lee J, Kwok SK, Park SH, Ju JH (2016) Atypical femoral fracture in rheumatoid arthritis patients treated with bisphosphonates: a nested case-control study. Arthritis Rheumatol 68(1):77–82
Shane E, Burr D, Abrahamsen B, Adler RA, Brown TD, Cheung AM, Cosman F, Curtis JR, Dell R, Dempster DW, Ebeling PR, Einhorn TA, Genant HK, Geusens P, Klaushofer K, Lane JM, McKiernan F, McKinney R, Ng A, Nieves J, O'Keefe R, Papapoulos S, Howe TS, van der Meulen MC, Weinstein RS, Whyte MP (2014) Atypical subtrochanteric and diaphyseal femoral fractures: second report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res 29(1):1–23
Saita Y, Ishijima M, Mogami A, Kubota M, Baba T, Kaketa T, Nagao M, Sakamoto Y, Sakai K, Kato R, Nagura N, Miyagawa K, Wada T, Liu L, Obayashi O, Shitoto K, Nozawa M, Kajihara H, Gen H, Kaneko K (2014) The fracture sites of atypical femoral fractures are associated with the weight-bearing lower limb alignment. Bone 66:105–110
Koshino T, Takeyama M, Jiang LS, Yoshida T, Saito T (2002) Underestimation of varus angulation in knees with flexion deformity. Knee 9(4):275–279
Shetty GM, Mullaji A, Bhayde S, Nha KW, Oh HK (2014) Factors contributing to inherent varus alignment of lower limb in normal Asian adults: role of tibial plateau inclination. Knee 21(2):544–548
Giusti A, Hamdy NA, Dekkers OM, Ramautar SR, Dijkstra S, Papapoulos SE (2011) Atypical fractures and bisphosphonate therapy: a cohort study of patients with femoral fracture with radiographic adjudication of fracture site and features. Bone 48(5):966–971
Schilcher J, Michaelsson K, Aspenberg P (2011) Bisphosphonate use and atypical fractures of the femoral shaft. N Engl J Med 364(18):1728–1737
Feldstein AC, Black D, Perrin N, Rosales AG, Friess D, Boardman D, Dell R, Santora A, Chandler JM, Rix MM, Orwoll E (2012) Incidence and demography of femur fractures with and without atypical features. J Bone Miner Res 27(5):977–986
O'Brien CA, Jia D, Plotkin LI, Bellido T, Powers CC, Stewart SA, Manolagas SC, Weinstein RS (2004) Glucocorticoids act directly on osteoblasts and osteocytes to induce their apoptosis and reduce bone formation and strength. Endocrinology 145(4):1835–1841
Henneicke H, Gasparini SJ, Brennan-Speranza TC, Zhou H, Seibel MJ (2014) Glucocorticoids and bone: local effects and systemic implications. Trends Endocrinol Metab 25(4):197–211
Jia D, O'Brien CA, Stewart SA, Manolagas SC, Weinstein RS (2006) Glucocorticoids act directly on osteoclasts to increase their life span and reduce bone density. Endocrinology 147(12):5592–5599
Weinstein RS (2011) Clinical practice. Glucocorticoid-induced bone disease. N Engl J Med 365(1):62–70
Grossman JM, Gordon R, Ranganath VK, Deal C, Caplan L, Chen W, Curtis JR, Furst DE, McMahon M, Patkar NM, Volkmann E, Saag KG (2010) American College of Rheumatology 2010 recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res (Hoboken) 62(11):1515–1526
Saag KG, Emkey R, Schnitzer TJ, Brown JP, Hawkins F, Goemaere S, Thamsborg G, Liberman UA, Delmas PD, Malice MP, Czachur M, Daifotis AG (1998) Alendronate for the prevention and treatment of glucocorticoid-induced osteoporosis. Glucocorticoid-Induced Osteoporosis Intervention Study Group. N Engl J Med 339(5):292–299
Saag KG, Shane E, Boonen S, Marin F, Donley DW, Taylor KA, Dalsky GP, Marcus R (2007) Teriparatide or alendronate in glucocorticoid-induced osteoporosis. N Engl J Med 357(20):2028–2039
Teitelbaum SL, Seton MP, Saag KG (2011) Should bisphosphonates be used for long-term treatment of glucocorticoid-induced osteoporosis? Arthritis Rheum 63(2):325–328
Plotkin LI, Bivi N, Bellido T (2011) A bisphosphonate that does not affect osteoclasts prevents osteoblast and osteocyte apoptosis and the loss of bone strength induced by glucocorticoids in mice. Bone 49(1):122–127
Iwata K, Mashiba T, Hitora T, Yamagami Y, Yamamoto T (2014) A large amount of microdamages in the cortical bone around fracture site in a patient of atypical femoral fracture after long-term bisphosphonate therapy. Bone 64:183–186
Mashiba T, Turner CH, Hirano T, Forwood MR, Johnston CC, Burr DB (2001) Effects of suppressed bone turnover by bisphosphonates on microdamage accumulation and biomechanical properties in clinically relevant skeletal sites in beagles. Bone 28(5):524–531
Schilcher J, Koeppen V, Aspenberg P, Michaëlsson K (2014) Risk of atypical femoral fracture during and after bisphosphonate use. N Engl J Med 371(10):974–976
Adler RA, El-Hajj Fuleihan G, Bauer DC, Camacho PM, Clarke BL, Clines GA, Compston JE, Drake MT, Edwards BJ, Favus MJ, Greenspan SL, McKinney R Jr, Pignolo RJ, Sellmeyer DE (2016) Managing osteoporosis in patients on long-term bisphosphonate treatment: report of a Task Force of the American Society for Bone and Mineral Research. J Bone Miner Res 31(1):16–35
Emkey R, Delmas PD, Goemaere S, Liberman UA, Poubelle PE, Daifotis AG, Verbruggen N, Lombardi A, Czachur M (2003) Changes in bone mineral density following discontinuation or continuation of alendronate therapy in glucocorticoid-treated patients: a retrospective, observational study. Arthritis Rheum 48(4):1102–1108
Devogelaer JP, Goemaere S, Boonen S, Body JJ, Kaufman JM, Reginster JY, Rozenberg S, Boutsen Y (2006) Evidence-based guidelines for the prevention and treatment of glucocorticoid-induced osteoporosis: a consensus document of the Belgian Bone Club. Osteoporos Int 17(1):8–19
Sambrook PN, Kotowicz M, Nash P, Styles CB, Naganathan V, Henderson-Briffa KN, Eisman JA, Nicholson GC (2003) Prevention and treatment of glucocorticoid-induced osteoporosis: a comparison of calcitriol, vitamin D plus calcium, and alendronate plus calcium. J Bone Miner Res 18(5):919–924
Taormina DP, Marcano AI, Karia R, Egol KA, Tejwani NC (2014) Symptomatic atypical femoral fractures are related to underlying hip geometry. Bone 63:1–6
Gravallese EM, Manning C, Tsay A, Naito A, Pan C, Amento E, Goldring SR (2000) Synovial tissue in rheumatoid arthritis is a source of osteoclast differentiation factor. Arthritis Rheumatism 43(2):250–258
Napoli N, Jin J, Peters K, Wustrack R, Burch S, Chau A, Cauley J, Ensrud K, Kelly M, Black DM (2012) Are women with thicker cortices in the femoral shaft at higher risk of subtrochanteric/diaphyseal fractures? The study of osteoporotic fractures. J Clin Endocrinol Metab 97(7):2414–2422
Kim D, Sung YK, Cho SK, Han M, Kim YS (2016) Factors associated with atypical femoral fracture. Rheumatol Int 36(1):65–71
Zanchetta MB, Diehl M, Buttazzoni M, Galich A, Silveira F, Bogado CE, Zanchetta JR (2014) Assessment of bone microarchitecture in postmenopausal women on long-term bisphosphonate therapy with atypical fractures of the femur. J Bone Miner Res 29(4):999–1004
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Ethics statement
The study was approved by the Institutional Review Board of Seoul St. Mary’s Hospital (KC14RISI0555). The requirement for informed consent was waived due to the retrospective nature of the study.
Electronic supplementary material
Supplementary table 1
(DOCX 18.4 kb)
Supplementary table 2
(DOCX 17.1 kb)
Rights and permissions
About this article
Cite this article
Koh, J.H., Myong, J.P., Yoo, J. et al. Predisposing factors associated with atypical femur fracture among postmenopausal Korean women receiving bisphosphonate therapy: 8 years’ experience in a single center. Osteoporos Int 28, 3251–3259 (2017). https://doi.org/10.1007/s00198-017-4169-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-017-4169-y