Abstract
The aim of this study was to clarify and compare the temporal course of bone mineral density (BMD) between fast bone losers and normal residents in Miyama Village, a rural Japanese community. BMD was measured over a 10-year period in a cohort study in Miyama Village, Wakayama Prefecture, Japan, to provide information on rate of bone loss in the mature and elderly population. Subjects (n=400) were selected by sex and age stratum from the full list of residents born in 1910–1949, with 50 men and 50 women in each age decade. Baseline BMD of the lumbar spine and proximal femur was measured using dual energy X-ray absorptiometry in 1990, 1993, 1997 and 2000. In the cohort, 171 men and 189 women completed the follow-up survey performed in 1993. After calculating the rate of bone loss between 1990 and 1993, the greatest tertile from the distribution of bone loss was categorized as fast bone losers, with the remainder considered as normal subjects. Changes in BMD were compared between normal subjects and fast bone losers over the 10-year period. Mean rate of change for BMD at both lumbar spine and femoral neck in fast bone losers recovered to levels similar to those in normal subjects over 7 years of observation. By contrast, BMD at the lumbar spine and femoral neck decreased steeply over the 10-year period in both groups, and mean BMD for fast bone losers was significantly lower than that of normal subjects (P<0.05). These differences were apparent only at the lumbar spine in both men and women, even after adjusting for age. These results indicate that fast bone loss is a transient phenomenon rather than a fixed status, although individuals who have been categorized as fast bone losers at some stage continue to display low BMD in the lumbar spine.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporotic fracture is one of the leading causes of elderly individuals becoming bedridden in Japan [1, 2]. Incidence of hip fracture has increased sharply over the last decade, concomitant with the rapid increase in the proportion of elderly people in the population [3]. Prevention of osteoporosis is therefore a matter of urgency if quality of life is to be maintained among elderly individuals and costs for medical care are to be contained.
Although fast bone losers have been recognized as a high-risk group for osteoporosis and osteoporotic fractures [4], few epidemiological studies of this group have been undertaken in Japan [5]. As bone loss is typically asymptomatic, longitudinal population surveys are required to clarify the epidemiological characteristics of fast bone losers.
To assess BMD and bone loss in the general population, two cohorts were established in Wakayama Prefecture, Japan. We have already reported results from a cohort selected from a seaside area, Taiji Town, in a study initiated in 1993 [6, 7, 8]. In that study, 3-year bone loss rates were measured in subjects randomly selected by sex and year of birth [6]. These data provided estimates of rate of bone loss among 40- to 79-year-old Japanese men and women. Rates of change of BMD at the lumbar spine in men increased slightly in all age strata, while rates in women decreased in all age strata. Our findings suggested that body build and menstrual function in women represent important determinants of bone loss [8].
In a second longitudinal study reported elsewhere, BMD was measured 4 times over the course of a decade. The study was initiated in 1990 among residents in a mountain area, Miyama Village, also in Wakayama Prefecture [9, 10]. In previous reports of the Miyama study, rates of bone loss for the lumbar spine and proximal femur were ascertained in men and women over a 10-year period, and secular trends regarding bone loss at these sites in Japan were clarified [5, 11].
In the present study, we observed changes to BMD in participants in the Miyama study, and compared bone changes between fast bone losers and normal subjects, as defined by the rate of bone change during the initial 3 years. This study aimed to address characteristics of fast bone losers and to observe changes in BMD predictive of prognosis for fast bone losers.
Materials and methods
This population-based epidemiological study, beginning in 1990, was conducted in Miyama, a mountain village in Wakayama Prefecture, Japan. As the Miyama cohort has been profiled in detail elsewhere [9, 10], subject characteristics are only summarized briefly here. A list of all inhabitants born in this village between 1910 and 1949 was compiled from the register of residents as of the end of 1989. A cohort of 1543 inhabitants (716 men, 827 women) was identified, all of whom completed a 125-item self-administered questionnaire covering daily activities such as dietary habits, smoking habits, alcohol consumption and physical exercise.
From the above total cohort, a BMD cohort was recruited, comprising 400 subjects, divided into four groups of 50 men and 50 women each, stratified for age according to decade of birth (1910–1919, 1920–1929, 1930–1939, 1940–1949). No differences were observed between BMD sub-cohorts and the total cohort with regard to distribution of items such as smoking, alcohol consumption, hours of sleep, exercise, walking, dietary habits or stress, as measured by the initial questionnaire [9]. In addition, mean values for weight and height, and distribution of smokers, drinkers, and hypertensives in the study population did not differ significantly from those reported in a nationwide study [12].
A second questionnaire was administered to the 400 participants, covering items of past and family history, calcium intake, dietary habits, physical exercise, occupational activities, sun exposure, and reproductive variables in women. All subjects provided informed consent to participate in the study.
Baseline measurement of BMD was undertaken in 1990. Dual energy X-ray absorptiometry (DXA; Lunar DPX, GE Medical Systems LUNAR, Madison Wisc., USA) was used to measure BMD, providing antero-posterior images at both lumbar vertebrae L2–L4 and the proximal femur. In addition to BMD measurement, physical measurements of height (cm) and body weight (kg) were made, and body mass index (BMI, kg/m2) was calculated. BMD measurements were repeated on the same participants after 3, 7 and 10 years (1993, 1997 and 2000).
To control for precision of DXA, the apparatus was checked at every examination in 1990, 1993, 1997 and 2000 using the same phantom, and the BMD of the phantom was regulated to within 1.270±0.025 g/cm2 (2%) during examinations. All participants were examined by the same medical doctor, to remove inter-observer variability. Intra-observer variability of DXA (Lunar DPX) had been measured in vitro and in vivo for the previous study [13], using the same doctor. CV% for L2–L4 in vitro was 0.35%, while in vivo values of CV% for L2–L4, proximal femur and total hip, examined in five male volunteers, were 0.61–0.90% and 1.02–2.57%, respectively.
Annual rates of change in BMD over the 3-, 7- and 10-year periods were calculated and classified by sex and age stratum. Fast bone losers were defined as those subjects within the greatest tertile for the distribution of bone loss in the 1990–1993 period. To explore the influence of bone loss on subsequent BMD, bone loss rate and BMD for the lumbar spine and proximal femur in 1997 and 2000 were assessed and compared between fast bone losers and normal subjects.
Statistical analysis
Statistical analyses were performed using SPSS statistical software (SPSS, Chicago, Ill., USA) and the STATA statistical package (STATA Corp., College Station, Tex., USA). Differences were tested for statistical significance using analysis of variance (ANOVA) for comparisons between multiple groups and Scheffe’s least significant difference test between pairs of groups. To control the confounding factors in the assessment of rates of change, analysis of variance covariance (ANCOVA) was utilized.
Results
Of the 400 participants at the time of the initial survey performed in 1990, evaluation of changes to BMD over 3 years was completed in 360 (171 men, 189 women; 90.0%). Forty participants were ineligible for investigation of annual change in BMD between 1990 and 1993; ten had died (eight men, two women), five had moved (three men, two women), and 25 declined to participate or were away from the area at the time of follow-up (18 men, seven women). Five men and 16 women had undergone treatment for osteoporosis in the past, but in all cases, these individuals had been treated medically with calcium and/or vitamin D tablets only. Therefore, these subjects were included into analysis.
The greatest tertile of the final 360 subjects in the distribution of bone loss, as calculated by BMD obtained in the 1990 and 1993 surveys, was categorized as fast bone losers (57 men, 63 women). The remaining subjects were considered normal subjects. Table 1 shows sex and age distributions of subjects for each group. Mean values for rates of bone change at the lumbar spine and femoral neck in normal subjects were significantly higher than those of fast bone losers (P<0.001) among both men and women.
Mean age of men displaying fast bone loss at the lumbar spine was 61.8 years, 4.8 years older than normal subjects (P<0.01). Mean age of men demonstrating fast bone loss at the femoral neck was 60.0 years, 2.0 years older than normal subjects. However, this difference was not significant. Mean age of women did not differ significantly between those displaying fast bone loss at the lumbar spine (59.2 years) and normal subjects (58.8 years). By contrast, mean age of women demonstrating fast bone loss at the femoral neck was 62.9 years, 6.0 years older than that of normal subjects (P<0.001; Table 1).
Table 1 also demonstrates the characteristics of fast bone losers and normal subjects at the baseline study. Regarding body build, mean values for weight and BMI in men and women displaying fast bone loss at the lumbar spine were significantly lower than those of normal subjects (P<0.001). Similarly, mean weight for female fast bone losers at the femoral neck was significantly less than that of normal women (P<0.05). Menstrual status of fast bone losers, including age at menarche, age at menopause, and years since menopause, was compared to that of normal subjects. Although mean age at menarche was higher in fast bone losers at the femoral neck compared to normal women (P<0.05), no significant difference in mean age at menopause was observed between the two groups (Table 1).
Of the 360 subjects who participated in both the initial survey and the 1993 follow-up, 326 subjects (149 men, 177 women; 90.6%) participated in the 1997 follow-up survey, and 291 (130 men,161 women; 80.8%) completed the 2000 follow-up survey. Among the men, 11 normal subjects (9.7%) and 11 fast bone losers at the lumbar spine (19.3%) could not be measured for BMD in 1997, while 25 normal subjects (21.9%) and 16 fast bone losers (28.1%) could not be measured in 2000. In women, seven normal subjects (5.6%) and five fast bone losers (7.9%) could not be measured in 1997, while 21 normal subjects (16.7%) and seven fast bone losers (11.1%) could not be measured in 2000. Drop-out rates for each site did not differ significantly between normal subjects and fast bone losers in either men or women.
Over the course of the 7 years between 1993 and 2000, 23 men and 12 women died. Deaths among men included 12 normal subjects (10.5%) and 11 fast bone losers (19.3%) at the lumbar spine, and 17 normal subjects (14.9%) and six fast bone losers (10.5%) at the femoral neck. No differences in cumulative mortality were observed between normal subjects and fast bone losers. Among women, deaths comprised seven normal subjects (5.6%) and five fast bone losers (7.9%) at the lumbar spine, and eight normal subjects (6.4%) and four fast bone losers (6.4%) at the femoral neck. Again, no difference in cumulative mortality was observed between normal subjects and fast bone losers.
Figures 1 and 2 compare annual rates for bone loss at the lumbar spine (L2–L4; Fig. 1) and femoral neck (Fig. 2) between fast bone losers and normal subjects. To avoid “regression to the mean”, figures illustrate results after adjustment for rate of change for 1990–1993. Mean rates (SD) of lumbar BMD change in men were 0.56 (1.4) %/yr in fast bone losers and 0.38 (1.3) %/yr in normal subjects for 1993–1997, and 0.26 (1.5) %/yr in fast bone losers and 0.04 (1.4) %/yr in normal subjects for 1997–2000. In women, rates were –0.59 (1.8) %/yr in fast bone losers and –0.51 (1.8) %/yr in normal subjects for 1993–1997, and –0.58 (2.3) %/yr in fast bone losers and –0.90 (2.3) %/yr in normal subjects for 1997–2000. Mean rates (SD) of femoral neck BMD change in men were –0.14 (1.4) %/yr in fast bone losers and –0.48 (1.4) %/yr in normal subjects for 1993–1997, and –0.88 (2.8) %/yr in fast bone losers and –1.1 (2.8) %/yr in normal subjects for 1997–2000. In women, rates were –0.93 (1.7) %/yr in fast bone losers and –0.52 (1.7) %/yr in normal subjects for 1993–1997, and –1.2 (2.4) %/yr in fast bone losers and –0.45 (2.4) %/yr in normal subjects for 1997–2000. These figures demonstrate that fast bone losers as defined in the 1990–1993 period no longer met the criteria for fast bone losers in 1993–1997 or 1997–2000 at any site.
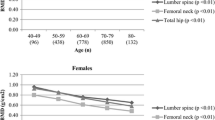
Figures 3 and 4 show comparisons between fast bone losers and normal subjects with regard to changes in BMD at the lumbar spine (L2–L4; Fig. 3) and femoral neck (Fig. 4) over the 10-year period, after adjustment for age. For men, mean lumbar BMD(SD) in 1993, 1997 and 2000 was 1.00 (0.20), 1.01 (0.21), and 1.02 (0.22) g/cm2, respectively, in fast bone losers; and 1.14 (0.20), 1.16 (0.21) and 1.15 (0.22) g/cm2, respectively, in normal subjects. For women, mean lumbar spine BMD(SD) in 1993, 1997 and 2000 was 0.84 (0.18), 0.82 (0.18) and 0.81 (0.18) g/cm2, respectively, in fast bone losers; and 0.98 (0.18), 0.94 (0.18) and 0.93 (0.18) g/cm2, respectively, in normal subjects. For men, mean femoral BMD(SD) in 1993, 1997 and 2000 was 0.84 (0.12), 0.84 (0.14) and 0.81 (0.15) g/cm2, respectively, in fast bone losers; and 0.88 (0.12), 0.86 (0.14) and 0.83 (0.15) g/cm2, respectively, in normal subjects. For women, mean femoral BMD(SD) in 1993, 1997 and 2000 was 0.70 (0.10), 0.70 (0.11) and 0.68 (0.12) g/cm2, respectively, in fast bone losers; and 0.74 (0.10), 0.71 (0.11) and 0.70 (0.13) g/cm2, respectively, in normal subjects. Mean BMD for fast bone losers was significantly lower than that for normal subjects in both men and women at the lumbar spine, but no significant differences were observed at the femoral neck except in 1993. Differences in lumbar spine BMD between normal subjects and fast bone losers remained in both men and women even after adjusting for age, body weight, height and history of osteoporosis at the 1990 baseline.
Discussion
In the present study, age-based cohorts from a single village were categorized as either fast bone losers or normal subjects according to annual rate of change of BMD between 1990 and 1993. BMD changes over the following 7 years were monitored. The characteristics of fast bone losers and the courses of BMD in both fast bone losers and normal subjects were clarified.
Fast bone losers were first reported by Christiansen et al. [4] in 1987, and have been recognized as a high-risk group for osteoporosis. As bone loss is typically asymptomatic, population surveys are needed to estimate prevalence of fast bone losers and clarify associated characteristics. However, few longitudinal studies have been performed in Japan [5]. In the aforementioned study by Christiansen et al. [4], radial bone mass was determined in 178 female volunteers using single photon absorptiometry. Since selection criteria for participants, measurement instruments, and sites of measurement differed between the Christiansen study and ours, the use of identical criteria to define fast bone losers might be unfeasible. The present study therefore categorized fast bone losers as those subjects in the greatest tertile of bone loss distribution. Characteristics and temporal course of BMD were compared between fast bone losers and normal subjects.
Before evaluating rate of bone loss, the phenomenon of “regression to the mean” should be considered. “Regression to the mean” refers to the phenomenon by which a variable that is extreme on first measurement will tend to be closer to the centre of distribution on later measurements [14]. To avoid this confounding factor in the assessment of rates of change, ANCOVA was utilized and rate of bone change was obtained after adjusting for the initial (from 1990 to 1993) rate of bone change.
We demonstrated differences in physical characteristics between fast bone losers and normal subjects. Mean BMI and body weight were significantly lower in fast bone losers than in normal subjects, except when comparing results for the femoral neck in men. Body composition of fast bone losers is thus significantly lighter than that of normal subjects. This relationship remained after adjustment for age. Body composition represents an important determinant of BMD and BMD change in women [5, 7, 8]. Tsunenari et al. found that changes in body weight contributed to the rate of change in BMD in postmenopausal women [15]. Tremollieres et al. demonstrated that excess weight protects against postmenopausal weight loss [16]. Hannan et al. noted in the Framingham Osteoporosis Study that thinness and weight loss were associated with bone loss in elderly individuals [17]. Dennison et al. indicated that decreased anthropometric measurements were not only associated with bone loss in women, but also in men [18]. The results of the present study, indicating that fast bone losers display lower body weight, are consistent with earlier studies.
The course of annual rates of bone loss showed, for all sites and genders, that fast bone losers defined in the 1990–1993 period no longer met the criteria for fast bone losers in later examinations. This means that fast bone loss apparently represents a transient phenomenon, rather than a fixed status. Conversely, BMD for fast bone losers at the lumbar spine was always lower than that of normal subjects, in both men and women. This result was observed only at the lumbar spine, not at the femoral neck. Although fast bone loss might represent a transient phenomenon, individuals who have been categorized as fast bone losers at some stage continue to display low BMD. This tendency remained even after adjusting for age, weight, height and history of treatment for osteoporosis. This might indicate that BMD values at the lumbar spine of fast bone losers display tracking. Tracking is defined as the persistence over time of rank for a variable in an individual compared to peers [19]. The term is sometimes used in epidemiological studies in blood pressure, and the hypothesis that high blood pressure in adulthood is a direct result of high blood pressure in childhood might represent an example of tracking [20]. In the present study, the period of observation was only 10 years, which may well be too short to identify tracking. However, the concept of tracking has been utilized in predicting future values of variables from early measurements, so the results of the present study might be useful for future prevention of osteoporosis. At the very least, once a subject is categorized as a fast bone loser at the lumbar spine, they should be considered at high risk of osteoporosis and followed carefully.
While fast bone losers at the lumbar spine displayed continued low BMD, fast bone losers at the femoral neck tended to display higher BMD than normal subjects in follow-ups. We have previously reported trends for bone loss at the lumbar spine and femoral neck in the same subcohort of the Miyama study [11]. In that report, we concluded that bone loss at the femoral neck progressed with age, while BMD change at the lumbar spine was greatest among women in their 40s and 50s. These results show that bone loss differs with the site involved. The results of the present study suggest that fast bone losers in the femoral neck may be able to stop losing BMD up to the threshold for hip fractures. We have also compared rates of bone loss rate between British and Japanese populations, and concluded that rate of bone loss among the Japanese population was less than that in the British population [21]. These results may help to explain the relatively low incidence of hip fracture in the Japanese population.
Our study identified no differences in cumulative mortality between fast bone losers and normal subjects over the 7 years of follow-up. However, observations should be continued to ascertain whether fast bone loss influences incidence of bone fractures, changes to quality of life or mortality.
References
Hosoda Y, Fujiwara S (1992) The epidemiology of osteoporosis in Japan. J Epidemiol 2:205–213
Hashimoto T, Sakata K, Yoshimura N (1997) Epidemiology of osteoporosis in Japan. Osteoporos Int 7:99–102
Orimo H, Hashimoto T, Sakata K, Yoshimura N, Suzuki T, Hosoi T (2000) Trends of incidence of hip fracture in Japan, 1987–1997—the third nation-wide survey. J Bone Miner Metabol 18:126–131
Christiansen C, Riis BJ, Rodbro P (1987) Prediction of rapid bone loss in postmenopausal women. Lancet 1:1105–1108
Yoshimura N (1996) Incidence of fast bone losers and factors affecting changes in bone mineral density: a cohort study in a rural Japanese community. J Bone Miner Metab 14:171–177
Kasamatsu T, Yoshimura N, Morioka S, Sugita K, Hashimoto T (1996) A population survey on bone mineral density in a fishing village in Wakayama Prefecture (part I). Distribution of bone mineral density by sex and age on a representative sample of the community. Jpn J Hyg 50:1084–1092 (In Japanese)
Yoshimura N, Kasamatsu T, Morioka S, Hashimoto T (1996) A population survey on bone mineral density in a fishing village in Wakayama Prefecture (part 2). The analysis of the risk factors affecting the bone mineral density. Jpn J Hyg 51:677–684 (In Japanese)
Yoshimura N, Hashimoto T, Morioka S, Sakata K, Kasamatsu T, Cooper C (1998) Determinants of bone loss in a rural Japanese community: the Taiji Study. Osteoporos Int 8:604–610
Kasamatsu T, Morioka S, Hashimoto T, Kinoshita H, Yamada H, Tamaki T (1991) Epidemiological study on bone mineral density of inhabitants in Miyama Village, Wakayama Prefecture (part 1). Background of study population and sampling method. J Bone Miner Metab 9:50–55
Kinoshita H, Danjoh S, Yamada H et al. (1991) Epidemiological study on the bone mineral density of inhabitants in Miyama Village, Wakayama Prefecture (part II) Bone mineral density of the spine and proximal femur. J Bone Miner Metab 9:56–60
Yoshimura N, Kinoshita H, Danjoh S et al. (2002) Bone loss at the lumbar spine and proximal femur in a rural Japanese community, 1990–2000: the Miyama Study. Osteoporos Int 13:803–808
Ministry of Health and Welfare of Japan (1990) Results of National Nutrition Survey 1988. Daiichi-shuppan, Tokyo
Yoshimura N, Kakimoto T, Nishioka M et al. (1997) Evaluation of reproducibility of bone mineral density measured by dual energy X-ray absorptiometry (DPX-L). Wakayama Igaku 48:461 6.14
Everitt BS (1995) Cambridge dictionary of statistics in the medical sciences. Cambridge University Press, Cambridge
Tsunenari T, Yamada S, Kawakatsu M, Negishi H, Tsutsumi M (1995) Menopause-related changes in bone mineral density in Japanese women. A longitudinal study. Calcif Tissue Int 56:5–10
Termollieres FA, Poulles JA, Ribot C (1993) Vertebral postmenopausal bone loss is reduced in overweight women: a longitudinal study in 155 early postmenopausal women. J Clin Endocrinol Metab 77:683–686
Hannan MT, Felson DT, Dawson-Hughes B et al. (2000) Risk factors for longitudinal bone loss in elderly men and women: the Framingham Osteoporosis Study. J Bone Miner Res 15:710–720
Dennison E, Eastell R, Fall CHD, Kellingray S, Wood PJ, Cooper C (1999) Determinants of bone loss in elderly men and women: a prospective population-based study. Osteoporos Int 10:384–391
Foulkes MA, Davis CE (1981) An index of tracking for longitudinal data. Biometrics 37:439–448
Burke V, Beikin LJ, Dunbar D (2001) Tracking of blood pressure in Australian children. J Hypertens19:1185–1192
Dennison E, Yoshimura N, Hashimoto T, Cooper C (1998) Bone loss in Great Britain and Japan: a comparative longitudinal study. Bone 23:379–382
Acknowledgements
This work was supported by Grant-in-Aids for Scientific Research A09770272 and C2 13670383 from the Ministry of Education, Culture, Sports, Science and Technology and H12-Chojyu-025 from the Ministry of Health, Labour and Welfare in Japan. Noriko Yoshimura received a Grant for Young Investigators in 1999 from the Japanese Society of Bone Morphometry. The authors wish to thank members of the public office of Miyama Village for assistance in arranging the location and schedule for examinations. We are grateful to Dr. S. Tokunaga, Department of Preventive Medicine, Kyushu University, for his valuable advice concerning the analysis of the data in the present study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yoshimura, N., Takijiri, T., Kinoshita, H. et al. Characteristics and course of bone mineral densities among fast bone losers in a rural Japanese community: the Miyama Study. Osteoporos Int 15, 139–144 (2004). https://doi.org/10.1007/s00198-003-1518-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-003-1518-9