Abstract
Purpose
Time-dependent surgical instrument contamination and the effect of covering during arthroplasty have not been investigated. This study aimed to evaluate time-dependent contamination of surgical instruments and the effect of covering on contamination as well as to perform bacterial typing of contaminated samples. The hypothesis was that covering the surgical instruments would decrease contamination rates.
Methods
Sixty patients who underwent total knee arthroplasty were randomized and divided into two groups: surgical instruments covered with a sterile towel or surgical instruments left uncovered. K-wires were used to extract microbiological samples. The K-wires were placed in a liquid culture medium at 0, 15, 30, 60, 90, and 120 min. After 24-h incubation period, samples from liquid cultures were cultured on blood agar using swabs. Samples with growth after 48 h were considered contaminated. Microscopic, staining, and biochemical properties were used for bacterial typing.
Results
Bacterial growth started after 30 and 60 min in the uncovered and covered groups, respectively. An increase in the number of K-wires contaminated with time was detected. At least 10,000 CFU/mL bacterial load was observed in the culture samples. Contamination was more significant in the uncovered group. A statistically significant difference in contamination was found between the uncovered and covered groups at 30-, 60-, 90-, and 120 min (p = 0.035, p = 0.012, p = 0.024, and p = 0.037, respectively). The most common bacteria on the contaminated instruments were coagulase-negative Staphylococci (60.4%), Staphylococcus aureus (22.9%), and Streptococcus agalactia (16.7%), respectively.
Conclusion
The risk of contamination increases with time. However, it may decrease if surgical instruments are covered. In the clinical practice, empiric antibiotic regimens based on the type of identified microorganisms in this study may be developed for postoperative periprosthetic joint infection prophylaxis.
Level of evidence
Prognostic, Level II.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Periprosthetic joint infection (PJI) is a significant burden to the patient and economically to the health care system [1]. Requirement of total joint arthroplasty and associated PJI is expected to increase [11]. Therefore, it is important to control risk factors to decrease infection rates.
Several risk factors which influence PJI were evaluated previously [1, 10]. These risk factors may be classified as patient and perioperative environment-related risk factors [7]. Many of these risk factors are modifiable [1]. Modifiable risk factors have been reported as antibiotic prophylaxis, diabetes mellitus, obesity, malnutrition, smoking, vitamin d deficiency, methicillin-resistant Staphylococcus aureus colonization, inflammatory arthropathies, surgical site preparation, wound irrigation, operative room environment, and aggressive anticoagulation [2]. As intraoperative risk factors, effect of bleeding, body temperature, room trafficking, air filtration, gloving, irrigation, and suture materials on PJI have been evaluated [7]. However, the effect of contamination of surgical instruments during arthroplasty has not been investigated. Therefore, the primary aim of this study was to evaluate time-dependent contamination of surgical instruments and the effect of covering surgical instruments on contamination. In addition, this study aimed to perform bacterial typing of the contaminated samples. The hypothesis of this study was that covering the surgical instruments would decrease contamination rates.
Methods
After obtaining the local ethics committee’s approval, contamination of surgical instruments was assessed prospectively during TKA surgery. All operations were performed by one surgeon and assisted by the same operating team in the same operating room with laminar flow. Room temperature and humidity were kept between 20 ℃ and 24 ℃, and 20% and 40%, respectively. The same operating team assisted in all operations. Movement in the room was strictly controlled by the circulating nurse. The door was closed before the start of the operation and no entry or exit was allowed until the wound was completely closed. A total of 60 patients who underwent primary TKA for unilateral varus OA between October 2018 and January 2019 were included in the study. Patients with a history of previous knee surgery (14 patients) or prior septic arthritis (three patients) of the native joint were not included in the study. Patients were randomly allocated to two groups. Randomization was performed by the “two-choice closed envelope method” with the envelope being chosen by the patient. Six pieces of Kirschner wires (TST, Istanbul, Turkey), 2 cm long and 1.5 mm thick, were added to the surgical trays before sterilization to collect samples at different times. A total of 60 surgical trays, 30 uncovered and 30 covered, were included. Demographic data of the 60 patients are summarized in Table 1.
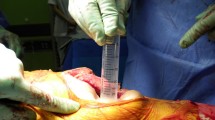
All trays were opened 5 min before the surgeon was ready to use them. The circulating nurse first opened the top wrapper flap and then opened the flaps to each side. The scrubbed nurse took out the instruments and K-wires with sterile forceps. Sterilization of the contents was confirmed using indicator strips. Wet and damaged packages were considered contaminated and were excluded from the study. In the uncovered group, the surface of the back table including all surgical instruments was left uncovered. In the covered group, the surface of the back table including surgical instruments was immediately covered with a sterile towel (Fig. 1). It was repeatedly covered after every instance of removal of an instrument from the tray. K-wires were placed in contact with the surgical instruments.
The K-wire samples were obtained and placed in the liquid culture medium immediately after opening (time zero) and at 15, 30, 60, 90, and 120 min after opening. Then, all culture media were placed in the incubator. Patients were followed up for a minimum of 6 months postoperatively.
K-wires were placed in sterile tryptic soy broth (TSB) liquid culture mediums (RTA, Kocaeli, Turkey) using aseptic techniques and incubated at 37 °C for 24 h. At the end of the incubation period, the samples were retrieved from the liquid culture medium using sterile loops (Fig. 2). Each plastic calibrated loop was removed from its package aseptically. A 0.001-mL loop was inserted vertically into the TSB, allowing it to adhere to the loop. Then, a loopful of liquid was spread on the surface of a 5% sheep blood agar plate (RTA, Kocaeli, Turkey), and the plates were incubated at 37 °C. Microbiological procedures and evaluations were performed by an expert microbiologist, and the presence of growth on the plaque at the end of 48 h was recorded (Fig. 2). Microscopic, staining, and biochemical properties of the microorganisms were evaluated by the expert microbiologist. The number of colony-forming units (CFUs) was multiplied by 1000, because the 0.001-mL loop was used to determine the number of CFU/mL in the original specimen.
Data were analyzed with IBM SPSS V23 (IBM, Armonk, NY, USA). Distribution of the data was examined using the Shapiro–Wilk test. Independent samples t test was used for comparison of normal distribution data based on contamination, and the Mann–Whitney U test was used for comparison of non-matching data. The Chi-square test was used to examine qualitative data. A post hoc power analysis was performed to detect statistical power. Results of the analysis were presented as the mean ± standard deviation for quantitative data and as frequency (percentage) for qualitative data. A p value < 0.05 was considered significant.
Results
Growth was detected in 6 of 30 (20%) sets in the uncovered group at 30 min, whereas growth was detected in 2 of 30 (6.7%) sets in the covered group at 60 min (p = 0.024) (Table 2). Contamination increased with time. The contamination rates at 120 min were 43.4% in the uncovered group and 16.7% in the covered group. A statistically significant difference in contamination was detected at the 30-, 60-, 90-, and 120 min (Table 1). The most common bacteria detected on the contaminated instruments were coagulase-negative Staphylococci (60.4%), Staphylococcus aureus (22.9%), and Streptococcus agalactia (16.7%), respectively. Early PJI developed in one patient (1.6%) 1 week postoperatively. The patient was among those with contaminated instruments in the uncovered group. Coagulase-negative Staphylococcus was isolated from the debridement material. Infection regressed following debridement, insert exchange, and intravenous antibiotic therapy. At the postoperative 1-year follow-up visit, the patient was symptom-free.
In the post hoc power analysis, between-groups comparison effect size was calculated as d = 0.324. Approximately 104 patients would be needed to obtain statistical power at the recommended 0.80 power level.
Discussion
The most important finding of the present study was that surgical instrument contamination was found to begin at 30 min in the uncovered surgical instrument group. However, contamination was detected after 60 min in the covered surgical instrument group. The contamination increased with time.
Although there are significant advances in techniques for the prevention of infection along with modifying patient and surgical risk factors, periprosthetic infection is still a major problem in total hip and knee arthroplasty [9]. This study attempted to evaluate the potential source of periprosthetic infection in TKA.
Prolonged operative duration has been associated with increased complications and infection risk [3, 5, 13, 14]. It may probably be due to a combination of factors, including bleeding, increased tissue damage, and contamination [3]. In their study, Wang et al. [16] reported nearly 25% increased risk of PJI for each 20-min increase in operative time. They found significantly higher surgical site and PJI rates with more than 90 min of operative time than 60–90 min and below 60 min of operation time. The results of our study showed that patients are at risk of infection even if the surgical procedure is not prolonged.
The presence of implant material in the surgical field is a predisposing factor for the tissues to become more sensitive to infections [12]. Development of biofilm on the implant surface reduces the effect of antibiotics on bacteria [8]. Thus, the treatment of infection becomes complicated. Therefore, reducing contamination as far as possible is important for achieving successful surgical treatment, increasing prosthetic survival, and preventing additional morbidities among patient in arthroplasty operations [15]. The results of this study may contribute to the reduction of contamination in patients with arthroplasty. The contamination and frequency of contamination-associated periprosthetic infections can be reduced by the simple method of covering surgical instruments during surgery.
In the literature, contamination rates of various implant sets in covered and uncovered trays have been evaluated. Bible et al. [4] reported 9.5% of general contamination rate in cultures taken after placing the last implant. They found no association between contamination and the number of scrubbed personnel, types of instruments, and the duration of exposure. However, in a similar study, Dalstrom et al. [6] reported no contamination in the covered group and a contamination rate of 30% after 4 h in the uncovered group. They also reported that controlling room traffic was not effective in contamination. Dalstrom et al. [6] performed their study in a real operation but without removing the tray. This study was performed in real operations. In addition, the K-wires were placed among the surgical instruments that were actively used on the nurse’s desk. That enabled us to assess the real intraoperative environment, similar to that in clinical practice. Published literature does provide limited data concerning the clinical relevance of contaminated surgical instruments. In addition, a correlation between the occurrence of infection and contamination was not reported previously. Similarly, an important correlation was not demonstrated between contamination and infection in our study.
Menekse et al. [12] evaluated time-dependent contamination of spinal implants using a liquid culture medium. They reported that growth started after 30 min in the uncovered implants. However, the growth started after 60 min in the covered group. In addition, they showed an increase in growth with time in both groups, but a more significant increase in the uncovered group. In our study, the growth started after 30 min in the uncovered group and after 60 min in the covered group. In addition, a time-dependent increase in growth in both groups, but a more significant increase in the uncovered group, was observed.
The technique of obtaining culture samples using swabs and then culturing them on a solid medium was previously used in various studies. The whole material in the liquid culture medium was cultured to more effectively detect contamination as previously noted by Menekse et al. [12]. After the 24-h incubation, the samples were cultured on blood agar using swabs under sterile conditions. This technique allowed us to evaluate the entire surface of the K-wire. Considering the various shape characteristics of surgical instruments, the liquid culture technique that used is superior to the samples taken with the swabs.
This study has several limitations. The relatively small sample size may have affected our results. Early PJI developed in only one patient. It was not sufficient to detect a clinically meaningful difference between the covered and uncovered groups. The duration of operation was limited to 120 min. More complex or revision cases may require more than 120 min. As the pathogenesis of periprosthetic infection is multifactorial, an association between contamination and periprosthetic infection could not be demonstrated. Incubation time limited to 24 h. More microorganisms might even grow after 24 h. Although bacterial typing was performed, the antibiotic susceptibility test could be useful in selecting certain postoperative empirical antibiotics. Due to our limited budget, only 30 patients per group could be included. According to post hoc power analysis, a limited statistical power was detected due to modest sample size (n = 60) that may have played a role in limiting the significance of comparisons conducted.
Conclusion
Contamination of surgical instruments increases with time. Contamination rates may be decreased by covering surgical instruments. Empiric antibiotic regimens may be developed for postoperative PJI prophylaxis on the basis of the type of microorganisms identified in this study. The knowledge of time-dependent surgical instrument contamination and the preventive effect of covering on contamination may help to reduce contamination-related postoperative periprosthetic joint infection in total hip and knee arthroplasty patients. In the clinical practice, a simple method of covering the surgical instruments with a sterile towel during TKA may help to decrease bacterial contamination of surgical instruments and the related PJI risk.
References
Alamanda VK, Springer BD (2018) Perioperative and modifiable risk factors for periprosthetic joint infections (PJI) and recommended guidelines. Curr Rev Musculoskelet Med 11(3):325–331
Alamanda VK, Springer BD (2019) The prevention of infection 12: modifiable risk factors. Bone Joint J 101-B(1 Suppl A):3–9
Badawy M, Espehaug B, Fenstad AM, Indrekvam K, Dale H, Havelin LI, Furnes O (2017) Patient and surgical factors affecting procedure duration and revision risk due to deep infection in primary total knee arthroplasty. BMC Musculoskelet Disord 18(1):544
Bible JE, O’Neill KR, Crosby CG, Schoenecker JG, McGirt MJ, Devin DJ (2013) Implant contamination during spine surgery. Spine J 13(6):637–640
Campbell DA Jr, Henderson WG, Englesbe MJ, Hall BL, O’Reilly M, Bratzler D, Dellinger EP, Neumayer L, Bass BL, Hutter MM, Schwartz J, Ko C, Itani K, Steinberg SM, Siperstein A, Sawyer RG, Turner DJ, Khuri SF (2008) Surgical site infection prevention: the importance of operative duration and blood transfusion—results of the first American College of Surgeons–National Surgical Quality Improvement Program Best Practices Initiative. J Am Coll Surg 207:810–820
Dalstrom DJ, Venkatarayappa I, Manternach AL, Palcic MS, Heyse BA, Prayson MJ (2008) Time-dependent contamination of opened sterile operating-room trays. J Bone Joint Surg Am 90(5):1022–1025
George DA, Drago L, Scarponi S, Gallazzi E, Haddad FS, Romano CL (2017) Predicting lower limb periprosthetic joint infections: a review of risk factors and their classification. World J Orthop 8(5):400–411
Gristina AG, Costerton JW (1984) Bacterial adherence and the glycocalyx and their role in musculoskeletal infection. Orthop Clin N Am 15:517–535
Kapadia BH, Berg RA, Daley JA, Fritz J, Bhave A, Mont MA (2016) Periprosthetic joint infection. Lancet 387(10016):386–394
Kong L, Cao J, Zhang Y, Ding W, Shen Y (2017) Risk factors for periprosthetic joint infection following primary total hip or knee arthroplasty: a meta-analysis. Int Wound J 14(3):529–536
Kurtz SM, Lau E, Watson H, Schmier JK, Parvizi J (2012) Economic burden of periprosthetic joint infection in the United States. J Arthroplast 27(8 Suppl):61–65.e1
Menekse G, Kuscu F, Suntur BM, Gezercan Y, Ates T, Ozsoy KM, Okten AI (2015) Evaluation of the time-dependent contamination of spinal implants: prospective randomized trial. Spine (Phila Pa 1976) 40(16):1247–1251
Namba RS, Inacio MC, Paxton EW (2013) Risk factors associated with deep surgical site infections after primary total knee arthroplasty: an analysis of 56,216 knees. J Bone Joint Surg Am 95:775–782
Peersman G, Laskin R, Davis J, Peterson MG, Richart T (2006) Prolonged operative time correlates with increased infection rate after total knee arthroplasty. HSS J 2:70–72
Tsai DM, Caterson EJ (2014) Current preventive measures for health-care associated surgical site infections: a review. Patient Saf Surg 8(1):42
Wang Q, Goswami K, Shohat N, Aelirezaie A, Manrique J, Parvizi J (2019) Longer operative time results in a higher rate of subsequent periprosthetic joint infection in patients undergoing primary joint arthroplasty. J Arthroplast 34(5):947–953
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Erdal Uzun, Abdulhamit Misir, Mustafa Ozcamdalli, Emine Eylul Kizkapan, Alper Cirakli, and Mustafa Kerem Calgin declare that they have no actual or potential conflict of interest including any financial, personal, or other relationships with other people or organizations within 3 years of beginning the submitted work that could inappropriately influence, or be perceived to influence, their work.
Ethical approval
Ordu University, Faculty of Medicine clinical investigations ethics committee approved the study protocol (Approval date/number:18.10.2018/2018-215).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Uzun, E., Misir, A., Ozcamdalli, M. et al. Time-dependent surgical instrument contamination begins earlier in the uncovered table than in the covered table. Knee Surg Sports Traumatol Arthrosc 28, 1774–1779 (2020). https://doi.org/10.1007/s00167-019-05607-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-019-05607-y