Abstract
Purpose
To evaluate the feasibility of forced oscillation technique (FOT) measurements at the bedside and to describe the relationship between positive end-expiration pressure (PEEP) and lung mechanics in different groups of ventilated infants.
Methods
Twenty-eight infants were studied: 5 controls, 16 newborns with respiratory distress syndrome (RDS) and 7 chronically ventilated newborns that developed bronchopulmonary dysplasia. An incremental/decremental PEEP trial was performed by changing PEEP in 1-min steps of 1 cmH2O between 2 and 10 cmH2O. Forced oscillations at 5 Hz were superimposed on the ventilator waveform. Pressure and flow, measured at the inlet of the ETT, were used to compute resistance (Rrs) and reactance (Xrs).
Results
In controls Rrs and Xrs were on average 41 ± 21 and −22 ± 6 cmH2O s/l respectively and were almost unaffected by PEEP. RDS infants presented similar Rrs (48 ± 25 cmH2O s/l) and reduced Xrs (−71 ± 19 cmH2O s/l) at the beginning of the trial. Two behaviours were observed as PEEP was increased: in extremely low birth weight infants Xrs decreased with PEEP with marked hysteresis; in very low and low birth weight infants Xrs and Rrs were less PEEP dependent. Chronically ventilated infants had very high Rrs and very negative Xrs values at very low PEEPs (121 ± 41 and −95 ± 13 cmH2O s/l at PEEP = 2 cmH2O) that markedly changed as PEEP exceeded 3–4 cmH2O.
Conclusions
Rrs and Xrs measurement in preterm newborns is feasible, and data are representative of the lung mechanics and very sensitive to its changes with PEEP, making FOT a promising technique for the non-invasive bedside titration of mechanical ventilation in preterm newborns.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Surfactant deficiency combined with the poor outward elastic recoil of the chest wall makes premature infants susceptible to alveolar instability, airways collapse and atelectasis, leading to lung volume derecruitment and increased lung opening pressure.
The application of positive end-expiration pressure (PEEP) is widely used to prevent the collapse of alveoli and terminal airways at end-expiration in respiratory distress syndrome (RDS). Unfortunately, there is evidence that an inappropriate use of PEEP may cause alveolar overdistension, with reduced compliance, carbon dioxide retention [1] and an increased risk of developing VILI [2].
Nowadays PEEP titration in clinical practice in newborns relies on monitoring oxygen saturation, which is an indirect indicator of lung recruitment.
In the pioneering study of Suter et al. [3], the authors argued that PEEP should be tailored to each individual patient, identifying the lowest pressure providing the highest lung compliance.
Recent studies have provided further evidence that PEEP can be successfully optimised in acute lung injury or acute RDS (ALI/ARDS) at the bedside by either maximising dynamic compliance (Cdyn) [4, 5] or minimising dynamic elastance (Edyn) [6, 7] during a decremental PEEP trial. However, the estimation of Cdyn (and Edyn) is affected by the respiratory muscle activity, which prevents its use in non-paralysed patients [8].
Moreover, preterm infants are prone to develop bronchopulmonary dysplasia (BPD) and require mechanical ventilation for long periods in the phase of evolving BPD. While the rationale for PEEP titration in RDS is to identify the lowest level of PEEP able to counteract lung volume de-recruitment, in evolving BPD PEEP should be set at the minimum level that counteracts the airways collapse, which is likely to occur because of airway wall remodelling, interstitial fluids accumulation and reduced elastic recoil of the lung. Therefore, the assessment of lung mechanics would also be of great value in evolving BPD but with a different aim, i.e., identifying airways collapse.
The lack of adequate bedside tools for the assessment of respiratory mechanics in ventilated infants is nowadays the major limiting factor for the individualised tailoring and continuous adjustment of PEEP in these patients.
The forced oscillation technique (FOT) is a non-invasive method for the assessment of respiratory mechanics, which can be applied during mechanical ventilation at the bedside with spontaneously breathing patients. Briefly, it consists of analysing the response of the respiratory system to high frequency (>5 Hz) and low amplitude (~2 cmH2O amplitude) oscillatory pressures. It provides the so-called respiratory system impedance (Zrs), a variable made of two components, the resistance (Rrs) and the reactance (Xrs). Zrs can be used to identify lung volume recruitment-derecruitment during both conventional [9, 10] and high-frequency oscillatory ventilation [11]. Moreover, it has recently been shown that Xrs, which accounts for the elastic and inertial properties of the system, can guide PEEP titration by identifying the lowest PEEP that maintains lung volume recruitment, minimising lung mechanical stress in an experimental model of ALI [12, 13] and leading to a more protective ventilation strategy compared to an oxygenation-based approach [13].
To the best of our knowledge, FOT has been applied to ventilated preterm newborns in very few studies [14, 15], and none of them studied Zrs at different PEEP settings.
The aim of the present study is to apply a new setup designed to perform FOT measurements on mechanically ventilated newborns during an incremental/decremental PEEP trial in order to evaluate the feasibility of bedside FOT measurements and to characterise the impedance-PEEP relationships in newborns with healthy lungs, with RDS and chronically ventilated infants that developed BPD.
Methods
All measurements were performed in the Neonatal Intensive Care Unit of Fondazione IRCCS Ca’ Granda, Ospedale Maggiore Policlinico in Milan. The study had been approved by the local Ethics Committee, and informed parental consent was obtained prior to carrying out the studies.
Study population
All infants assisted by conventional mechanical ventilation, in haemodynamically stable conditions, regardless of the gestational age, postnatal age and the cause of respiratory failure, were eligible for the study. Infants with severe intracranial haemorrhage (III and IV) and/or malformations were excluded.
Study protocol
All the infants were studied in the supine position. Infants were intubated with uncuffed endotracheal tubes (size 2.5–3.5 mm i.d.) and treated with synchronised intermitted positive pressure (S-IPPV) ventilation (Babylog 8000 plus, Drager, Lübeck, Germany, or Leoni Plus, Heinen and Lowenstein, Germany) with volume guarantee (Vt = 5 ml/kg) and PIP = 24 ± 2 cmH2O. All ventilatory parameters but PEEP were kept constant at the clinically set values, while the FiO2 was adjusted in order to maintain oxygen saturation (SpO2) between the clinical limits (86–94 %). At first PEEP was lowered to 2 cmH2O for 1 min, then it was increased to 10 cmH2O and subsequently decreased to 2 cmH2O in steps of 1 cmH2O. Preliminary measurements showed that 1 min allows the major impedance changes to stabilise, but we cannot exclude the occurrence of dynamic phenomena with longer time scales. Since we wanted to minimise the duration of the manoeuvre and especially the time spent at sub-optimal PEEP levels, each step lasted 1 min.
Forced oscillations at 5 Hz were delivered for the whole duration of the trial and superimposed on the ventilator waveform. The frequency of 5 Hz was used as it has been previously shown to be highly sensitive to peripheral phenomena, such as alveolar recruitment and distension, without being affected by the breathing rate [10, 12, 16] (see Online Resource for more details). The total duration of the study never exceed 30 min.
Experimental setup
A detailed description of the measurement setup is reported in the online data supplement. Briefly ~2 cmH2O peak-to-peak sinusoidal oscillations at 5 Hz were generated by oscillating the piston of a 20-ml glass syringe with a linear motor. The syringe was connected to the inspiratory line of the mechanical ventilator close to the inlet of the endotracheal tube. Pressure (Pao) and airflow (V’ao) were measured at the airway opening by a pressure transducer and a mesh-type heated pneumotachograph, digitised at 600 Hz and stored on a personal computer (see Fig. 1).
Measurements and monitoring
SpO2, blood pressure and heart rate were continuously monitored non-invasively to evaluate how the manoeuvre was tolerated by the patient.
Data analysis
Zrs was computed using a least squared method [17]. Impedance data were corrected by subtracting Rrs and Xrs values for the ETTs [14]. For each protocol step the last 2–4 breaths were selected, and their end-expiratory Rrs and Xrs values were averaged providing a single data point for each PEEP level (Fig. 2). Figure 2 shows the pressure and flow recordings and the corresponding Rrs and Xrs tracings.
Experimental tracings. Right panel: pressure, flow, resistance and reactance. For each PEEP step the last five breaths are reported. Resistance and reactance are reported as a mean and SD of these five breaths. Left panel: enlargement of one PEEP step (5 cmH2O). The gray areas represent the data that were discarded
Oscillatory elastance (EFOT) was obtained from Xrs as follows:
Edyn was estimated by fitting Pao and V’ao to the equation of motion of the respiratory system:
where V is volume obtained by integration of V’ao, Rrs is the resistance of the respiratory system, and EEP is the end-expiratory pressure.
Statistic analysis
Significance of differences in population characteristics and clinical parameters among groups was tested by Mann-Whitney test. Two-way ANOVA for repeated measurements was used to test the significance of differences in impedance data among groups and within each group among PEEP levels. Multiple comparisons after ANOVA were performed using the Holm-Sidak method. Differences were considered statistically significant for p < 0.05. Spearman’s correlation was performed to test whether there was a statistical dependence between impedance and clinical parameters, and ρ was used to test the correlation strength.
Results
A total of 28 newborns were studied (Table 1). Infants were divided into three groups: infants with RDS (n = 16), chronically ventilated infants (for more than 20 consecutive days at the time of measurement) that developed BPD (n = 7; BPD was defined as in [18]) and infants ventilated for surgical pathologies that did not affect the respiratory system (controls, n = 5).
The setup and the manoeuvre were well tolerated by all the infants and did not induce clinically relevant changes in SpO2, heart rate or arterial blood pressure. At the highest PEEP, the peak inspiratory pressure was limited by the set value in nine patients, but without affecting Vt significantly (see Online Resource).
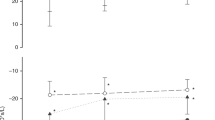
Figure 3 shows the relationships of PEEP with Rrs and Xrs for the three groups of patients.
Respiratory system resistance (Rrs) and reactance (Xrs) in the control (a), RDS (b) and evolving BPD (c) groups. Data are expressed as mean and SD. Open symbols represent the incremental PEEP trial and closed symbols the decremental one. * Indicates significant difference versus PEEP = 2 cmH2O on the inflation limb (p < 0.05, **p < 0.01)
In the control group Rrs was 36 ± 15 cmH2O s/l at PEEP = 2 cmH2O and slowly decreased with increasing PEEP. Xrs was −27 ± 7 cmH2O s/l at PEEP = 2 cmH2O, significantly higher than in both BPD and IRDS (p < 0.01) infants, and it was almost unaffected by PEEP.
In infants with evolving BPD, Rrs was not significantly different than in controls (p = 0.213), while Xrs was significantly lower than in controls at all PEEP levels. At low PEEPs Rrs was highest (Rrs at PEEP = 2 cmH2O during increasing PEEPs measured 121 ± 42 cmH2O s/l) and Xrs most negative (Xrs at PEEP = 2 cmH2O during increasing PEEP measured −95 ± 13 cmH2O s/l). As PEEP was increased, a sudden change was observed with a rapid drop in Rrs and a steep increase in Xrs. As PEEP was further increased, Rrs showed a smaller and gradual decrease while Xrs was stable with no negative swings.
In the RDS group, Rrs values were not significantly different than in the control group and were independent from PEEP, while Xrs values were significantly lower than in controls and they decreased with increasing PEEP. As can be observed in Fig. 3b, in this group Xrs presented large error bars that increased with PEEP. This is due to the fact that in different infants Xrs decreased differently with PEEP. Since the difference between the maximum in Xrs and the value of Xrs at PEEP = 10 cmH2O displayed a strong hyperbolic correlation with body weight (see Online Resource), we applied a commonly used threshold to body weight (1,000 g) to further divide this group into: extremely low birth weight (ELBW, n = 8) and very low or low birth weight (VLBW/LBW, n = 8). ELBW infants had a significantly lower GA and a significantly better PaO2 at baseline compared with VLBW/LBW infants. Rrs was not significantly different between the two groups (p = 0.379), while Xrs was significantly higher in the VLBW/LBW than in the ELBW group at each PEEP (see Fig. 4). In VLBW/LBW newborns Xrs presented quite a flat pattern, similar to that observed in the control group, but with significantly lower values. In ELBW infants Xrs decreased with increasing PEEP, suggesting that these infants may be more prone to lung tissue distension. In addition, for these infants the Xrs-PEEP relationship presented a marked hysteresis, with higher Xrs values during the decremental than during the incremental PEEP series. Moreover, we found a significant correlation between baseline values of Xrs and oxygenation (ρ = −0.44 vs. FiO2), which was particularly strong in ELBW infants (ρ = −0.843 vs. FiO2 and ρ = 0.762 vs. PaO2/FiO2).
Respiratory system resistance (Rrs) and reactance (Xrs) in ELBW (triangles) and VLBW/LBW (squares) subgroups. Open symbols, dotted line: inflation trial; closed symbols, dashed line: deflation trial. * Indicates significant difference versus PEEP = 2 cmH2O on the inflation limb (p < 0.05, **p < 0.01)
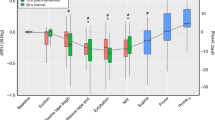
Figure 5 shows elastance estimated from Xrs (EFOT) and intratidal elastance (Edyn). Good agreement can be observed between EFOT and Edyn in identifying the differences among groups and in describing the relationship between lung mechanics and PEEP.
Comparison between respiratory system elastance measured by FOT (EFOT, left panel) and dynamic elastance (Edyn, right panel) in controls (a), RDS infants (b) and the evolving BPD group (c). Open symbols, dotted line: inflation trial; closed symbols, dashed line: deflation trial. For RDS infants, triangles indicate ELBW, squares indicate VLBW/LBW
Discussion
This study reports respiratory system resistance and reactance during an incremental/decremental PEEP trial in infants subjected to mechanical ventilation. Both measurements and procedures were well tolerated by all infants indicating that FOT is feasible even in very small and severe infants. The main findings were: (1) impedance in preterm newborns is representative of lung mechanics and very sensitive to its changes with PEEP in ventilated infants; (2) the relationship between impedance and PEEP differed in infants with different lung diseases, suggesting that oscillatory mechanics during mechanical ventilation can provide useful information for tailoring the ventilator settings according to the pathophysiological characteristics of the patient.
Comparison with other studies
To the best of our knowledge, this is the first time that Zrs has been measured during mechanical ventilation as a function of PEEP in a population of small and severe newborns. Only one study reported Zrs at different PEEP settings [19] but in infants affected by bronchiolitis.
Dorkin et al. [14] studied six ventilated infants with RDS in the first week of life over the range of 4–40 Hz at zero end-expiratory pressure. They reported impedance data for only four infants who showed an average Rrs of about 40 cmH2O*s/l at 4 Hz. This value is very similar to the one that we found (40 ± 20 cmH2O s/l at the lowest PEEP) in our VLBW/LBW patients who were also very similar in terms of GA, weight at measurement and days of life.
Effects of PEEP on respiratory input impedance
Positive end-expiration pressure affected Rrs and Xrs differently in different groups of infants. In the control group Rrs decreased slightly with PEEP, likely as a consequence of the increased lung volume at higher end-expiratory pressures. Lung volume and airway resistance are related by an inverse relationship resulting from the effect of the elastic recoil of lung tissue, which increases with increasing lung volumes and dilates the airways, reducing airflow resistance [20]. The relationship between Xrs and PEEP was quite flat, suggesting that, for the range of PEEPs applied in this study, the lungs did not either collapse or reach the upper flat part of the pressure-volume relationship. In this group Xrs was higher compared with the other groups, likely because these infants have more mature lungs with larger aerated volumes, and Xrs is positively related to body size and lung maturation [21, 22].
In the RDS group Rrs showed a negative PEEP dependence, similar to the one of controls, while Xrs presented two peculiar features: a significant reduction with increasing PEEP and hysteresis between increasing and decreasing PEEPs.
This shape of the relationship between Xrs and PEEP is very similar to the one already observed in a surfactant depletion model of RDS [12], and it has been associated with the occurrence of lung volume recruitment and de-recruitment and with an increasing distension applied to the lung tissues with increasing pressures. The maximum value of Xrs in the deflation limb of the PEEP trial was associated with the lowest PEEP able to maximise alveolar recruitment, as confirmed by CT scan [12].
Interestingly, in this group, the point of maximum Xrs was, on average, 5 cmH2O, the most commonly applied value for these infants.
The average Xrs pattern is the result of different individual behaviours. In ELBW infants Xrs was significantly lower than in VLBW/LBW infants and markedly decreased with PEEP, being significantly higher at the lowest PEEP level than at several steps between 5 and 10 cmH2O, possibly because at these PEEPs they were ventilated in the upper flat part of their lung pressure-volume curve.
A further different behaviour was found in infants with evolving BPD. In these patients Rrs was significantly higher and Xrs significantly more negative at PEEP = 2 cmH2O than at higher PEEP levels with a sudden and marked change occurring on average at PEEP = 4 cmH2O.
The combination of the high Rrs and the negative Xrs values at low PEEP levels are compatible with central airway narrowing, which can be reversed by the application of a relatively low level of PEEP. Even if the small number of patients suggests caution in interpreting the data, this behaviour may be related to the physio-pathological features of BPD, which mostly affect the mechanics of peripheral airways with the thickening of the mucosa layer and the accumulation of interstitial fluids due to the inflammatory processes and with the lower elastic recoil due to the alteration of the alveolarization processes. All these factors reduce the transmural pressure across airway walls, leading to excessive airway narrowing at low lung volumes. If this interpretation is correct, the role of PEEP in these patients would be to keep the airways dilated. Even though further studies of a larger population of patients are needed to confirm these results, the continuous assessment of the effects of PEEP on impedance data might provide useful information for the management of these patients.
Comparison between EFOT and Edyn/Cdyn
Previous studies have used tidal elastance, or its inverse Cdyn, to identify the optimal PEEP in ventilated subjects [5, 7]. These parameters have the advantage that they are displayed by most mechanical ventilators, but they are unreliable in triggered ventilation modalities because they are strongly affected by the spontaneous breathing of the patients. On the contrary FOT is independent from the patient respiratory efforts.
When Edyn was computed selecting breaths showing minimal patient efforts, we found good agreement between EFOT and Edyn. However, some differences exist between the two parameters: (1) in the present study EFOT was computed at end-expiration, while Edyn reflects the average properties over the large volume variation required to perform the measurement, (2) elastance is frequency dependent, especially in presence of heterogeneities; therefore, some of the differences in the absolute values can be explained by the difference between the frequencies at which elastance was evaluated: 5 Hz for EFOT and the breathing frequency (roughly 1 Hz) for Edyn.
Limitations of the study
It is possible that the duration of each PEEP step (1 min) was not always enough to allow for the lung volume to equilibrate. However, based on preliminary measurements, we found that 1 min is a good compromise between allowing enough time for major changes to be completed and keeping the manoeuvre short without exposing the infants to non-optimal PEEP levels for too long. This short time prevented us from measuring changes in SpO2 and FiO2 given the longer time needed by these parameters to stabilise. For this reason, future studies are required to relate changes in lung mechanics to changes in blood oxygenation.
Finally, a greater number of subjects would be necessary to establish whether FOT is representative of lung mechanics, in particular in healthy and evolving BPD infants.
In conclusion, FOT has recently been validated in animal models for the detection of lung volume recruitment/derecruitment and the identification of the optimal level of PEEP, defined as the lowest value that maintains lung recruitment [9, 12]. This study supports the use of FOT for monitoring and optimising mechanical ventilation at bedside for ventilated newborns. Considering that it would be easy to implement FOT in a modern mechanical ventilator by modifying the software without the need of external dedicated hardware, this technique could provide a useful tool for improving individualisation and tailoring of mechanical ventilation, allowing a better implementation of the concept of protective lung ventilation in preterm newborns.
References
Field D, Milner AD, Hopkin IE (1985) Effects of positive end expiratory pressure during ventilation of the preterm infant. Arch Dis Child 60:843–847
Jobe AH, Hillman N, Polglase G, Kramer BW, Kallapur S, Pillow J (2008) Injury and inflammation from resuscitation of the preterm infant. Neonatology 94:190–196
Suter PM, Fairley B, Isenberg MD (1975) Optimum end-expiratory airway pressure in patients with acute pulmonary failure. N Engl J Med 292:284–289
Dargaville PA, Rimensberger PC, Frerichs I (2010) Regional tidal ventilation and compliance during a stepwise vital capacity manoeuvre. Intensive Care Med 36:1953–1961
Suarez-Sipmann F, Böhm SH, Tusman G, Pesch T, Thamm O, Reissmann H et al (2007) Use of dynamic compliance for open lung positive end-expiratory pressure titration in an experimental study. Crit Care Med 35:214–221
Carvalho AR, Jandre FC, Pino AV, Bozza FA, Salluh JI, Rodrigues R et al (2006) Effects of descending positive end-expiratory pressure on lung mechanics and aeration in healthy anaesthetized piglets. Crit Care 10:R122
Carvalho AR, Jandre FC, Pino AV, Bozza FA, Salluh J, Rodrigues R et al (2007) Positive end-expiratory pressure at minimal respiratory elastance represents the best compromise between mechanical stress and lung aeration in oleic acid induced lung injury. Crit Care 11:R86
De LD, Piastra M, Conti G (2012) Technical problems with dynamic compliance evaluation in neonates and infants. Intensive Care Med 38:1082–1083
Bellardine Black CL, Hoffman AM, Tsai LW, Ingenito EP, Suki B, Kaczka DW et al (2007) Relationship between dynamic respiratory mechanics and disease heterogeneity in sheep lavage injury. Crit Care Med 35:870–878
Dellacà RL, Andersson OM, Zannin E, Kostic P, Pompilio PP, Hedenstierna G et al (2009) Lung recruitment assessed by total respiratory system input reactance. Intensive Care Med 35:2164–2172
Pillow JJ, Sly PD, Hantos Z (2004) Monitoring of lung volume recruitment and derecruitment using oscillatory mechanics during high-frequency oscillatory ventilation in the preterm lamb. Pediatr Crit Care Med 5:172–180
Dellacà RL, Zannin E, Kostic P, Olerud MA, Pompilio PP, Hedenstierna G et al (2011) Optimisation of positive end-expiratory pressure by forced oscillation technique in a lavage model of acute lung injury. Intensive Care Med 37:1021–1030
Kostic P, Zannin E, Andersson OM, Pompilio PP, Hedenstierna G, Pedotti A et al (2011) Positive end-expiratory pressure optimization with forced oscillation technique reduces ventilator induced lung injury: a controlled experimental study in pigs with saline lavage lung injury. Crit Care 15:R126
Dorkin HL, Stark AR, Werthammer JW, Strieder DJ, Fredberg JJ, Frantz ID (1983) Respiratory system impedance from 4 to 40 Hz in paralyzed intubated infants with respiratory disease. J Clin Invest 72:903–910
Sullivan KJ, Durand M, Chang HK (1991) A forced perturbation method of assessing pulmonary mechanical function in intubated infants. Pediatr Res 29:82–88
Dellacà RL, Santus P, Aliverti A, Stevenson N, Centanni S, Macklem PT et al (2004) Detection of expiratory flow limitation in COPD using the forced oscillation technique. Eur Respir J 23:232–240
Kaczka DW, Ingenito EP, Lutchen KR (1999) Technique to determine inspiratory impedance during mechanical ventilation: implications for flow limited patients. Ann Biomed Eng 27:340–355
Jobe AH, Bancalari E (2001) Bronchopulmonary dysplasia. Am J Respir Crit Care Med 163:1723–1729
Gauthier R, Beyaert C, Feillet F, Peslin R, Monin P, Marchal F (1998) Respiratory oscillation mechanics in infants with bronchiolitis during mechanical ventilation. Pediatr Pulmonol 25:18–31
Jordan C, Lehane JR, Jones JG, Altman DG, Royston JP (1981) Specific conductance using forced airflow oscillation in mechanically ventilated human subjects. J Appl Physiol 51:715–724
Pillow JJ, Hall GL, Willet KE, Jobe AH, Hantos Z, Sly PD (2001) Effects of gestation and antenatal steroid on airway and tissue mechanics in newborn lambs. Am J Respir Crit Care Med 163:1158–1163
Pillow JJ, Stocks J, Sly PD, Hantos Z (2005) Partitioning of airway and parenchymal mechanics in unsedated newborn infants. Pediatr Res 58:1210–1215
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dellacà, R.L., Veneroni, C., Vendettuoli, V. et al. Relationship between respiratory impedance and positive end-expiratory pressure in mechanically ventilated neonates. Intensive Care Med 39, 511–519 (2013). https://doi.org/10.1007/s00134-012-2795-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-012-2795-6