Abstract
Objectives
A prospective survey of French intensive care units (ICUs) in 1997 showed moderate and variable use of noninvasive ventilation (NIV). This study examined changes in NIV use in French ICUs after the intervening 5 years.
Settings
Patients were enrolled in a prospective survey in 70 French ICUs.
Methods
Three-week survey, with prospective inclusion of all patients requiring ventilatory support.
Measurements and results
Overall 1,076 patients received ventilatory support (55% of admissions). First-line NIV was significantly more common than 5 years earlier, overall (23% vs. 16%) and especially in patients not intubated before ICU admission (52% vs. 35%). Reasons for respiratory failure were coma (33%), cardiogenic pulmonary edema (8%), acute-on-chronic respiratory failure (17%), and de novo respiratory failure (41%). Significant increases in NIV use were noted for acute-on-chronic respiratory failure (64% vs. 50%) and de novo respiratory failure (22% vs. 14%). Among patients given NIV, 38% subsequently required endotracheal intubation (not significantly different). Independent risk factors for NIV failure were high SAPS II and de novo respiratory failure, whereas factors associated with success were good NIV tolerance and high body mass index.
Conclusions
NIV use has significantly increased in French ICUs during the past 5 years, and the success rate has remained unchanged. In patients not previously intubated, NIV is the leading first-line ventilation modality. The proportion of patients successfully treated with NIV increased significantly over the 5-year period (13% vs. 9% of all patients receiving ventilatory support).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Noninvasive ventilation (NIV) offers major benefits in selected patients with acute respiratory failure requiring intensive care unit (ICU) admission. NIV was introduced for critically ill patients in the 1990s when randomized controlled trials showed that NIV reduced the risk for endotracheal intubation (ETI) in patients with acute on chronic respiratory failure [1, 2, 3]. A similar effect was then found in patients with cardiogenic pulmonary edema [4] or hypoxemic respiratory failure [5]. To determine whether the benefits observed in clinical trials occurred also in everyday practice we evaluated NIV use in 1997 in 42 French ICUs [6]. We included all patients requiring ventilatory support for acute respiratory failure. The results showed that NIV was used in 16% of patients overall and in 35% of patients without ETI at admission. NIV success significantly decreased the risk of pneumonia and death. Since 1997 several studies have further documented the benefits of NIV and have expanded the indications for NIV to patients with various forms of hypoxemic respiratory failure [5, 7], immunosuppression [8, 9, 10, 11], and specific postoperative conditions [12]. NIV was found superior over conventional oxygen therapy in patients with cardiogenic pulmonary edema [13] and benefited patients with acute-on-chronic respiratory failure in various situations [14, 15, 16, 17]. Finally, the use of NIV in acute respiratory failure was discussed at an international consensus conference [18].
To determine whether changes have occurred in the use of NIV we repeated another prospective multicenter survey in 2002, 5 years after the initial survey. We compared the results to those of the earlier survey. This study has been presented in abstract form [19].
Methods
Study centers
A prospective observational study was conducted in French ICUs. In the name of a task force on mechanical ventilation we contacted the 376 ICUs whose directors were members of the French Society for Intensive Care (Société de Réanimation de Langue Française, SRLF); participation was on a voluntary basis, and we specified that we were equally interested by centers performing or not performing NIV; 102 ICU directors initially agreed to participate.
Patients
We included all patients admitted between 4 and 24 March 2002 who required endotracheal or noninvasive ventilation for acute respiratory failure at any time during the ICU stay. The total number of admissions during the study period was recorded. In each ICU the study investigators included each patient prospectively and completed a standardized form supplying data from admission to ICU discharge or death in patients receiving ventilatory support. Completed forms were mailed to the coordinating study center at the end of the 3-week study period or when the last included patient had died or been discharged from the ICU, whichever occurred first.
Data on demographics and history consisted of age, gender, and location before hospital and ICU admission. The Simplified Acute Physiology Score (SAPS) II [20] was used to assess severity of illness and the Logistic Organ Dysfunction (LOD) score to evaluate organ dysfunction [21] at admission. Underlying diseases were scored 0 if not present or not fatal, 1 if expected to cause death within 5 years, and 2 if expected to cause death within 1 year [22]. Previous ICU stays, previous experience with ventilatory assistance, and history of home oxygen therapy were recorded, as well as history of chronic pulmonary disease (COPD) and other causes of chronic respiratory failure, chronic heart failure (NYHA III or IV), and cancer or immunosuppression (defined as neutropenia below 1000/mm3 after bone marrow transplantation or anticancer chemotherapy, immunosuppressive therapy for solid organ transplantation, corticosteroid therapy of 20 mg or more per day for at least 3 weeks, or AIDS). Immediately before initiating ventilatory assistance the followings were recorded: reason for respiratory failure, arterial blood gas values, respiratory rate, heart rate and systolic arterial blood pressure, ability of the patient to mobilize secretions (yes, 1; and no, 2); Glasgow Coma Scale score; severity of encephalopathy scored from 0–4 (0, no abnormalities; 1, hypertonia or cogwheel rigidity with normal consciousness; 2, tremor, asterixis, or sleepiness; 3, confusion or agitation; and 4, prostration or obtundation), and body temperature. The characteristics of noninvasive ventilatory support were recorded throughout the first week or until ETI was required, as well as the patient-to-nurse ratio, type of ventilator (ICU type, NIV ventilator defined as a ventilator that has been designed for the specific purpose of in-hospital NIV, and home device defined as a ventilator designed primarily for home ventilation), mode of ventilation, main ventilator settings, type of mask (facial, nasal, integral mask or helmet), airway humidification system (heat-moisture exchanger, heated humidifier, no humidification device), use of nasal protection (local nasal skin protection with a hydrocolloid gel that prevents the occurrence of skin irritation and breakdown), and arterial blood gas values on the first ventilation day. We also recorded: clinical tolerance by the patient (from 1, very good to 4, bad) assessed by the clinician, extent of air leaks during NIV (from 1, very small or none to 5, massive), side effects of NIV, reasons for discontinuing NIV, and whether ETI was performed subsequently.
Data quality
All patient forms were reviewed by the principal investigator (A.D.) for compliance with inclusion criteria, correct calculation of severity and organ failure scores, and completeness and accuracy of daily data recording. Investigators were contacted about suspected errors or missing data. A double data entry procedure was used, with a system that automatically alerted keyboarding personnel to aberrant, outlier, or contradictory values. Data that triggered the alert system or showed discrepancies were checked and corrected.
A comparison was performed with the survey performed in 1997 in 42 French ICUs [6]. Of note, more centers were included in this survey; only 28 centers participated in both surveys, and the 1997 survey also included one center each from Spain, Switzerland, Belgium, and Tunisia.
Statistical analysis
The median and the interquartile range were calculated for continuous variables and the absolute and relative frequencies for categorical variables, as well as the 95% confidence intervals (95%CI) where appropriate. Each potential risk factor for NIV failure was evaluated in a univariate model; for continuous data, we used the Student's t test or the Mann-Whitney U test as appropriate and for categorical variables the χ2 test or Fisher's exact test. Factors yielding p values of 0.05 or less were entered into a logistic regression model, except those redundant with other factors (NIV during the first 24 h with NIV tolerance and LOD score with SAPS II) or having a high missing value rate (Glasgow Coma Scale score). Goodness-of-fit was assessed using the Hosmer-Lemeshow χ2 test and discrimination using the area under the receiver operating characteristic (ROC) curve. Accuracy was considered good when the area under the ROC curve ranged from 0.70–0.80 and excellent when it was greater than 0.80. Continuous variables were introduced either without correction or after dichotomization according to the median value. The adjusted odds ratio and 95% confidence interval were calculated for each factor that was significant in the logistic regression model. Differences with p values less than 0.05 were considered statistically significant. All tests were two-tailed. Statistical tests were performed using Intercooled STATA 8.2 software (StatCorp, Texas, USA).
Results
ICUs
Complete data were obtained for 70 ICUs, including 38 (54%) in nonuniversity hospitals and 32 (46%) in university hospitals. There were 24 (34%) medical ICUs, 42 (60%) medical-surgical ICUs, and 4 respiratory ICUs. These 70 ICUs had medians of 11 beds (interquartile range, IQR, 8–14), declared having 458 admissions per year (IQR, 350–618) and 52% of patients requiring mechanical ventilation (IQR, 40–61). Among the 70 participating units, 34 had used NIV in more than 50 patients during the year preceding the survey, 31 centers used NIV in 10–50 patients, and only 1 center reported using NIV in 1–10 patients (4 missing data). No unit declared not using NIV.
Demographics
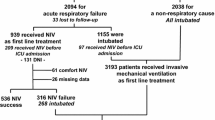
Of the 1943 patients admitted during the 3-week study period 1,076 (55%; 95%CI, 53–58) required mechanical ventilation, as compared to 51% (95%CI, 49–54) in 1997 (p = 0.03). Figure 1 shows the distribution of patients treated with NIV and with ETI. Overall NIV was the first-line ventilation modality in 249 (23%) of 1,076 patients (95%CI, 21–26), a significant increase compared to 1997 (16%, 95%CI, 13–19, p < 0.0001; Fig. 2). NIV use in patients admitted to the ICU without ETI was also significantly increased compared to 1997 (52%; 95%CI, 46–58; vs. 35%; 95%CI, 30–41; p< 0.0001; Fig. 2). Variations in NIV use among the ICUs of the study are shown on Fig. 3. In 2002 33 patients received NIV before ICU admission, compared to none in 1997. In the 28 centers that participated in both surveys 498 patients were ventilated and the rate of NIV use in 2002 was 34%.
The causes of acute respiratory failure were classified into four groups: 172 (16%) patients had acute-on-chronic respiratory failure, including 11% with obstructive disease and 4.5% with restrictive disease; 366 (34%) patients had coma of various causes (e.g., stroke, severe drug overdose) resulting in ventilatory failure as the main reason for the need of ventilatory support: in this group, intubation was performed to protect upper airways and allow achievement of satisfactory gas exchanges through mechanical ventilation; patients with coma secondary to respiratory acidosis were classified in the previous group; 86 (8%) patients had cardiogenic pulmonary edema; and 452 (42%) patients had de novo respiratory failure, including postoperative failure (14%), pneumonia (13%), extrapulmonary sepsis (6%) and others. NIV was used in one patient with coma. Compared with 1997, NIV use increased significantly for acute-on-chronic respiratory failure (64%, 95%CI, 56–71, vs. 50%, 95%CI, 40–60; p = 0.03) and for de novo respiratory failure (22%, 95%CI, 19–26 vs. 14%, 95%CI, 10–18; p = 0.003) (Fig. 4). In the 28 centers that participated in both surveys, the use of NIV in 2004 was comparable, 69% for acute-on-chronic respiratory failure, 29% for de novo respiratory failure, and 52% for cardiogenic pulmonary edema.
Among the 172 patients who received ventilatory support for an acute-on-chronic respiratory failure, 24% were intubated outside the ICU, 64% received NIV, and only 12% were intubated (as a first line treatment) in the ICU. Among those, 60% had a severe ventilatory acidosis (pH< 7.20), 60% had an extrarespiratory organ failure, and 60% had a severe encephalopathy.
NIV characteristics
Pressure support was the most widely used ventilatory mode (83%). Assist-control ventilation was used in 7% of patients and spontaneous breathing with continuous positive airway pressure in 8%. An ICU-type ventilator was used in 79% of cases, an NIV ventilator in 12%, and a home device in 5% (4% missing data). Airway humidification was achieved using a heat-moisture exchanger in 48% of patients and a heated humidifier in 19%; no humidification device was used in 30% of patients. NIV was delivered via a face mask in 83% of patients, a total face mask (but no helmet) in 9%, and a nasal mask in 6%. A nasal protection was used in 45% of the patients ventilated through a face mask.
Response to NIV
For the effectiveness analysis we excluded the single patient with coma who was given NIV as this patient was switched to ETI upon evaluation by the senior physician, whose decision was independent of the effect of NIV and based on the need to protect the airway. Of the 248 remaining patients 117 (47%) had an early discontinuation of NIV, i.e., while continued indications for ventilatory assistance were present. Reasons for discontinuation were inadequate efficacy [59%, with absence of clinical improvement (22%), lack of arterial blood gas improvement (6%), or both (31%)], full dependence on ventilatory support contrasting with the need to interrupt NIV (26%), inability to manage copious secretions (14%), agitation (13%), intercurrent event (12%), and patient refusal (8%) (some patients had more than one reason). The premature NIV discontinuation rate was similar to that seen in 1997 (47% vs. 48%, respectively), but significant differences were found regarding reasons for discontinuation: decreases were seen for inadequate efficacy (from 84% to 59%), inability to manage copious secretions (from 32% to 14%), and patient refusal (from 22% to 8%); whereas a significant increase was noted for full dependence (from 14% to 26%). Of the 117 patients with premature NIV discontinuation 94 (80%, representing 38% of all NIV patients) received ETI, and 15 died within 24 h following NIV discontinuation without receiving ETI because of do-not-intubate orders. These 109 patients were classified as NIV failures. The 8 patients with premature NIV discontinuation who survived without receiving ETI and the 131 patients treated with NIV until they no longer required assistance were classified as NIV successes. This 56% success rate (95%CI, 50–62) was similar to the rate in 1997 (60%, 95%CI, 50–69), using the same definition. Due to the increased use of NIV, the proportion of patients successfully treated by NIV in the overall population requiring mechanical ventilation was higher in the present study than in 1997 (13%, 95%CI 11–15; vs. 9%, 95%CI 7–12; p = 0.03).
Predictors of successful NIV
Table 1 reports the main characteristics in the groups with successful NIV and failed NIV. The patient with coma who received NIV, was excluded from this analysis. Severity of illness, reasons for acute respiratory failure, preexisting immunosuppression, and several physiological characteristics at NIV initiation, including PaO2/FIO2 were significantly different between the two groups. NIV outcome was not influenced by the type of ventilation (i.e., type of ventilator, humidification device, PEEP or pressure support level), neither by the patient to nurse ratio. The scores for air leaks and NIV tolerance were significantly different between the two groups.
As shown in Table 2, a high SAPS II score and de novo respiratory failure were the two independent factors associated with failed NIV. Good NIV tolerance score and high body mass index were independently associated with successful NIV. The same analysis was conducted separately in patients with de novo respiratory failure [risk factors were SAPS II, odds ratio (OR) 1.05, CI 1.01–1.09, p = 0.007 and NIV leaks, OR 2.63, CI 1.03–6.71, p = 0.044], and patients with acute-on-chronic respiratory failure or cardiogenic pulmonary edema (risk factors were SAPS II, OR 1.07, CI 1.03–1.10, p < 0.0001 and good NIV tolerance, OR 0.30, CI 0.11–0.77, p = 0.012).
Discussion
Increased NIV use
NIV use increased from 16% to 24% in all ICU patients requiring ventilatory support for acute respiratory failure and from 35% to 52% in those without ETI before or at ICU admission. A retrospective, observational, cohort study showed a steady rise in NIV use from 1994–2001 in a single ICU in France [23]. The present results suggest a similar trend in French ICUs.
Before discussing the results, some limitations of the present study warrant mentioning. The present survey was conducted in 2002, and it may not even reflect present-day practices since further changes may have occurred before publication of the results. Also, this 2002 survey may have underestimated the use of NIV since patients in whom NIV was entirely performed into the emergency room or in the ward were not included in the survey. Another limitation is the fact that it was conducted in only a single country. To evaluate the course of NIV use over years, however, we wished to compare the use of NIV in the present study with the use of NIV in a precedent survey conducted in France. Only a part of French ICUs participated in this survey, which could have introduced a bias, physician being more likely to participate if they were “NIV believer.” When intensivists were asked to participate, we clearly specified that we were similarly interested if they use or did not use NIV. Finally, the centers that participated in the two surveys were not exactly the same, as indicated in the methods section, but 28 centers that were part of the 1997 survey participated in the 2002 survey. The NIV use and its increase between the two surveys among the centers that participated in the two surveys were comparable to what was observed overall. We increased the number of centers to make it more representatives of French ICUs with more nonuniversity centers.
That NIV has gained ground so rapidly is remarkable, as many medical advances have been slow to take root in clinical practice. Although it differs in many aspects, the example of the tidal volume range in acute lung injury does not seem to offer the example of such a rapid change in practice. Weinert et al. [24] investigated trends in ventilatory assistance in 398 patients with acute lung injury treated in three teaching hospitals between 1994 and 2001 and found that mean prescribed tidal volume was still 10.1 ± 1.9 ml/kg actual body weight in the 2 years following the release of the NIH study, in which 6 ml/kg predicted body weight was superior over 12 ml/kg. Our survey was not designed to identify the reasons for increased NIV use. Nevertheless, the above example suggests that the accumulation of data on benefits from NIV may not be the only reason. The factors that govern acceptance of new treatments by physicians in everyday practice have been investigated [25, 26], and the trigger that motivates a change in prescription practices has emerged as more powerful than exposure to new information [26]. In a study evaluating the NIH Consensus Development Program [27] Kanouse and Jacoby [26] suggested that a motivating trigger could be the belief by the physician that “his or her patients are not experiencing a satisfactory outcome.” Intensivists have noted poor outcomes in patients intubated for acute-on-chronic obstructive respiratory failure, most notably prolonged need for mechanical ventilation and difficulties during the weaning process. This clinical reality may have prompted a readiness for change among intensivists. Greco and Eisenberg [25] also suggested that physicians change their practice only when they perceive the change as beneficial to their patients. Thus the major and readily perceptible benefits of NIV may explain the rapid growth of NIV use documented in the present study. Other factors may facilitate rapid adoption of NIV: the resources needed for NIV are under the control of the physician; NIV failure leads to ETI, which is simply the standard treatment [26]. Furthermore, giving physicians information about their practices may influence their readiness to adopt changes [25]. The 1997 survey provided information on practices regarding NIV. Finally, the 1999 consensus conference may have played a role [18]. The impact of consensus conferences on medical practices is controversial [28] and may be limited in some cases [29]; nevertheless, consensus conferences may hold potential for improving medical practices [30].
NIV failure
Despite this large increase in NIV use the failure rate remained unchanged between 1997 and 2002. This steady failure rate was not associated with a major increase in the severity of patients receiving NIV, and the SAPS II of patients treated with NIV was comparable between the two surveys (39 ± 16 in the present survey vs. 36 ± 20 in 1997). In our univariate analysis several variables differed between the success and failure groups. Many of these variables have been shown to predict NIV failure, such as level of consciousness [6, 31], PaO2/FIO2 [32], presence of leaks [33], and NIV tolerance score [6]. The univariate analysis found that immunosuppression was associated with a higher rate of NIV failure, but this did not remain in the multivariate analysis. NIV has been found beneficial in immunosuppressed patients [8, 10, 11]. The univariate analysis also found that do-not-intubate orders did not differ between the success and failure groups. This result is consistent with those of previous studies that found NIV success rates averaging 60% in these patients [34, 35]. Nevertheless, the present survey did not assess the long-term prognosis of these patients. The present study identified four independent factors, namely, worse SAPS II, de novo respiratory failure, low body mass index, and poor NIV tolerance. A worse SAPS II or severity score was associated with NIV failure in other studies [32, 36] including our 1997 survey [6]. NIV use in patients with de novo respiratory failure was initially controversial until recent trials [5, 8, 37]. We observed a high rate of NIV failure leading to ETI in patients with de novo respiratory failure (54%). Pneumonia was the leading cause of hypoxemia in patients requiring ETI, and similar or higher rates of NIV failure have been reported in patients with pneumonia [5, 38, 39, 40]. This survey also indirectly suggests that most of the patients treated with NIV in this survey needed ventilatory support. Indeed, among the 117 patients in whom NIV was discontinued prematurely only 8 survived without further ventilatory support. Therefore our results should not be construed as evidence that NIV is not beneficial in patients with de novo respiratory failure; instead, they suggest that physicians should be aware of the high risk of failure in this patient subgroup.
Good NIV tolerance, as assessed using a semiquantitative score, independently predicted success. This score, however, does not allow identification of a specific reason for poor NIV tolerance, which can be related to patient, equipment, or settings. The outcome of NIV seemed independent of the mode of ventilation, type of ventilator, and humidification device. This result should be interpreted with caution, as the study was probably not adequately powered to detect a role for each of these factors. Although of marginal significance, a high body mass index was associated with successful NIV. Because the median value was 25 kg/m2, this result does not indicate a protective effect of obesity. Among the possible explanations, a higher rate of upper airway obstruction responsive to mask positive pressure ventilation or a different level of nutrition could play a role [36].
Among the patients who received ventilatory support for an acute-on-chronic respiratory failure, 24% were intubated outside the ICU, and 12% were intubated as a first line treatment in the ICU. Among those, 60% had a severe ventilatory acidosis (pH< 7.20), 60% had an extrarespiratory organ failure, and 60% had severe encephalopathy. Some of these patients might possibly have been intubated because of insufficient familiarity with the technique, but a majority were probably considered as having contraindication to the technique, namely an organ failure other than respiratory or an encephalopathy that was considered too severe to allow the technique to be initiated.
In conclusion, compared to 1997, NIV use has significantly increased in French ICUs, up to 24% overall and 52% of patients admitted without ETI. The number of patients successfully treated by NIV among all patients requiring mechanical ventilation was higher in the present study. Future efforts should focus on improving patient selection and on enhancing patient acceptance via better adjustment of NIV settings [33, 41, 42].
References
Brochard L, Isabey D, Piquet J, Amaro P, Mancebo J, Messadi AA, Brun-Buisson C, Rauss A, Lemaire F, Harf A (1990) Reversal of acute exacerbations of chronic obstructive lung disease by inspiratory assistance with a face mask. N Engl J Med 323:1523–1530
Bott J, Keilty SE, Elliott MW, Moxham J, Carroll MP, Conway JH, Brown AM, Godfrey RC, Ward EM, Paul EA, Wedzicha JA (1993) Randomised controlled trial of nasal ventilation in acute ventilatory failure due to chronic obstructive airways disease. Lancet 341:1555–1557
Brochard L, Mancebo J, Wysocki M, Lofaso F, Conti G, Rauss A, Simonneau G, Benito S, Gasparetto A, Lemaire F (1995) Noninvasive ventilation for acute exacerbations of chronic obstructive pulmonary disease. N Engl J Med 333:817–822
Bersten AD, Holt AW, Vedig AE, Skowronski GA, Baggoley CJ (1991) Treatment of severe cardiogenic pulmonary edema with continuous positive airway pressure delivered by face mask. N Engl J Med 325:1825–1830
Antonelli M, Conti G, Rocco M, Bufi M, De Blasi RA, Vivino G, Gasparetto A, Meduri GU (1998) A comparison of noninvasive positive-pressure ventilation and conventional mechanical ventilation in patients with acute respiratory failure. N Engl J Med 339:429–435
Carlucci A, Richard JC, Wysocki M, Lepage E, Brochard L (2001) Noninvasive versus conventional mechanical ventilation. An epidemiologic survey. Am J Respir Crit Care Med 163:874–880
Confalonieri M, Potena A, Carbone G, Porta RD, Tolley EA, Umberto Meduri G (1999) Acute respiratory failure in patients with severe community-acquired pneumonia. A prospective randomized evaluation of noninvasive ventilation. Am J Respir Crit Care Med 160:1585–1591
Hilbert G, Gruson D, Vargas F, Valentino R, Gbikpi-Benissan G, Dupon M, Reiffers J, Cardinaud JP (2001) Noninvasive ventilation in immunosuppressed patients with pulmonary infiltrates, fever, and acute respiratory failure. N Engl J Med 344:481–487
Azoulay E, Moreau D, Alberti C, Leleu G, Adrie C, Barboteu M, Cottu P, Levy V, Le Gall JR, Schlemmer B (2000) Predictors of short-term mortality in critically ill patients with solid malignancies. Intensive Care Med 26:1817–1823
Azoulay E, Alberti C, Bornstain C, Leleu G, Moreau D, Recher C, Chevret S, Le Gall JR, Brochard L, Schlemmer B (2001) Improved survival in cancer patients requiring mechanical ventilatory support: impact of noninvasive mechanical ventilatory support. Crit Care Med 29:519–525
Antonelli M, Conti G, Bufi M, Costa MG, Lappa A, Rocco M, Gasparetto A, Meduri GU (2000) Noninvasive ventilation for treatment of acute respiratory failure in patients undergoing solid organ transplantation: a randomized trial. JAMA 283:235–241
Auriant I, Jallot A, Herve P, Cerrina J, Le Roy Ladurie F, Fournier JL, Lescot B, Parquin F (2001) Noninvasive ventilation reduces mortality in acute respiratory failure following lung resection. Am J Respir Crit Care Med 164:1231–1235
Masip J, Betbese AJ, Paez J, Vecilla F, Canizares R, Padro J, Paz MA, de Otero J, Ballus J (2000) Non-invasive pressure support ventilation versus conventional oxygen therapy in acute cardiogenic pulmonary oedema: a randomised trial. Lancet 356:2126–2132
Girault C, Daudenthun I, Chevron V, Tamion F, Leroy J, Bonmarchand G (1999) Noninvasive ventilation as a systematic extubation and weaning technique in acute-on-chronic respiratory failure: a prospective, randomized controlled study. Am J Respir Crit Care Med 160:86–92
Hilbert G, Gruson D, Portel L, Gbikpi-Benissan G, Cardinaud JP (1998) Noninvasive pressure support ventilation in COPD patients with postextubation hypercapnic respiratory insufficiency. Eur Respir J 11:1349–1353
Nava S, Ambrosino N, Clini E, Prato M, Orlando G, Vitacca M, Brigada P, Fracchia C, Rubini F (1998) Noninvasive mechanical ventilation in the weaning of patients with respiratory failure due to chronic obstructive pulmonary disease. A randomized, controlled trial. Ann Intern Med 128:721–728
Plant PK, Owen JL, Elliott MW (2000) Early use of non-invasive ventilation for acute exacerbations of chronic obstructive pulmonary disease on general respiratory wards: a multicentre randomised controlled trial. Lancet 355:1931–1935
Evans TW (2001) International Consensus Conferences in Intensive Care Medicine: non-invasive positive pressure ventilation in acute respiratory failure. Intensive Care Med 27:166–178
Demoule A, Girou E, Taille S, Brochard L, SRLF Collaborative Group on Mechanical Ventilation (2003) Current use of non-invasive ventilation: results from a 2002 multicenter French survey. Intensive Care Med 29:S64
Le Gall JR, Lemeshow S, Saulnier F (1993) A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. JAMA 270:2957–2963
Le Gall JR, Klar J, Lemeshow S, Saulnier F, Alberti C, Artigas A, Teres D (1996) The Logistic Organ Dysfunction system. a new way to assess organ dysfunction in the intensive care unit. ICU Scoring Group. JAMA 276:802–810
McCabe WR, Jackson GG (1962) Gram negative bacteriemia. I. Etiology and ecology. Arch Intern Med 110:845–847
Girou E, Brun-Buisson C, Taille S, Lemaire F, Brochard L (2003) Secular trends in nosocomial infections and mortality associated with noninvasive ventilation in patients with exacerbation of COPD and pulmonary edema. JAMA 290:2985–2991
Weinert CR, Gross CR, Marinelli WA (2003) Impact of randomized trial results on acute lung injury ventilator therapy in teaching hospitals. Am J Respir Crit Care Med 167:1304–1309
Greco PJ, Eisenberg JM (1993) Changing physicians' practices. N Engl J Med 329:1271–1273
Kanouse DE, Jacoby I (1988) When does information change practitioners' behavior? Int J Technol Assess Health Care 4:27–33
Kanouse DE, Brook RH, Winkler JD, Kosecoff JB, Berry SH, Carter GM, Kahan JP, McCloskey L, Rogers WH, Winslow CM, Anderson GM, Brodsley L, Fink A, Meredith LS (1989) Changing medical practice through technology assessment: an evaluation of the NIH Consensus Development Program. Health Administration Press, Ann Arbor
Cabana MD, Rand CS, Powe NR, Wu AW, Wilson MH, Abboud PA, Rubin HR (1999) Why don't physicians follow clinical practice guidelines? A framework for improvement. JAMA 282:1458–1465
Misset B, Artigas A, Bihari D, Carlet J, Durocher A, Hemmer M, Langer M, Nicolas F, de Rohan-Chabot P, Schuster HP, Tensillon A (1996) Short-term impact of the European Consensus Conference on the use of selective decontamination of the digestive tract with antibiotics in ICU patients. Intensive Care Med 22:981–984
Durand-Zaleski I, Bonnet F, Rochant H, Bierling P, Lemaire F (1992) Usefulness of consensus conferences: the case of albumin. Lancet 340:1388–1390
Anton A, Guell R, Tarrega J, Sanchis J (2002) Non-invasive ventilation. Thorax 57:919
Antonelli M, Conti G, Moro ML, Esquinas A, Gonzalez-Diaz G, Confalonieri M, Pelaia P, Principi T, Gregoretti C, Beltrame F, Pennisi MA, Arcangeli A, Proietti R, Passariello M, Meduri GU (2001) Predictors of failure of noninvasive positive pressure ventilation in patients with acute hypoxemic respiratory failure: a multi-center study. Intensive Care Med 27:1718–1728
Navalesi P, Fanfulla F, Frigerio P, Gregoretti C, Nava S (2000) Physiologic evaluation of noninvasive mechanical ventilation delivered with three types of masks in patients with chronic hypercapnic respiratory failure. Crit Care Med 28:1785–1790
Chu CM, Chan VL, Wong IW, Leung WS, Lin AW, Cheung KF (2004) Noninvasive ventilation in patients with acute hypercapnic exacerbation of chronic obstructive pulmonary disease who refused endotracheal intubation. Crit Care Med 32:372–377
Meduri GU, Fox RC, Abou-Shala N, Leeper KV, Wunderink RG (1994) Noninvasive mechanical ventilation via face mask in patients with acute respiratory failure who refused endotracheal intubation. Crit Care Med 22:1584–1590
Mehta S, Hill NS (2001) Noninvasive ventilation. Am J Respir Crit Care Med 163:540–577
Ferrer M, Esquinas A, Leon M, Gonzalez G, Alarcon A, Torres A (2003) Noninvasive ventilation in severe hypoxemic respiratory failure: a randomized clinical trial. Am J Respir Crit Care Med 168:1438–1444
Jolliet P, Abajo B, Pasquina P, Chevrolet JC (2001) Non-invasive pressure support ventilation in severe community-acquired pneumonia. Intensive Care Med 27:812–821
Wysocki M, Tric L, Wolff MA, Millet H, Herman B (1995) Noninvasive pressure support ventilation in patients with acute respiratory failure. A randomized comparison with conventional therapy. Chest 107:761–768
Domenighetti G, Gayer R, Gentilini R (2002) Noninvasive pressure support ventilation in non-COPD patients with acute cardiogenic pulmonary edema and severe community-acquired pneumonia: acute effects and outcome. Intensive Care Med 28:1226–1232
Calderini E, Confalonieri M, Puccio PG, Francavilla N, Stella L, Gregoretti C (1999) Patient-ventilator asynchrony during noninvasive ventilation: the role of expiratory trigger. Intensive Care Med 25:662–667
Prinianakis G, Delmastro M, Carlucci A, Ceriana P, Nava S (2004) Effect of varying the pressurisation rate during noninvasive pressure support ventilation. Eur Respir J 23:314–320
Acknowledgements
Physicians and centers participating in the study: Angers, Centre Hospitalier Universitaire: A. Kouatchet MD, A. Mercat MD; Argenteuil, Hôpital Victor Dupouy: G. Bleichner MD, H. Mentec MD; Arles, Centre Hospitalier J Imbert: C. Crombé MD, D. Selzer MD; Aubenas, Centre Hospitalier: P. Fernandez MD; Beaune, Hôpital Philippe le Bon: P. Noël MD; Belfort, Centre Hospitalier de Belfort-Montbéliard: O. Ruyer MD; Bordeaux, Hôpital Pellegrin Tripode: G. Hilbert MD, F. Vargas MD; Boulogne-Billancourt, Hôpital Ambroise-Paré: S. Prin MD, A. Vieillard-Baron MD; Brest, CHU la Cavale Blanche: A. Renault MD, E. l'Her MD; Briis Sous Forges, CMC Bligny: P. Andrivet MD; Calais, Centre Hospitalier de Calais: K. Guego MD, H. Peneau MD; Clamart, Hôpital Antoine Béclère: F. Brivet MD, F. Jacobs MD; Colombes, Hôpital Louis Mourier: D. Dreyfuss MD, J.-D. Ricard MD; Corbeil-Essonnes, Centre Hospitalier Sud Francilien: D. Caen MD, D. Larbi MD; Créteil, Centre Hospitalier Intercommunal: O. Gilhodes MD; Dieppe, Centre Hospitalier de Dieppe: M. Pinsard MD, F. Bougerol MD; Dourdan, Centre Hospitalier Général: J.-P. Colin MD; Draguignan, Centre Hospitalier: P. Brofferio MD; Elbeuf, Centre Hospitalier: M. Canonne MD, O. Delastre MD; Etampes, Centre Hospitalier d'Etampes: S. Percheron MD, S. Siami MD; Fréjus, CHI Fréjus Saint-Raphaël: M. Kaidomar MD, A. Besançon MD; Garches, Hôpital Raymond Poincaré: D. Annane MD, D. Orlikowski MD; Helfaut, Centre Hospitalier de la Région de. Saint-Omer: B. Sergent MD; La Roche sur Yon, CH Départemental: E. Clementi MD, L. Martin Lefèvre MD; Le Kremlin-bicêtre, Hôpital de Bicêtre: V. Zarka MD, C. Richard MD; Le Plessis Robinson, Centre Chirurgical Marie Lannelongue: I. Auriant MD, F. Parquin MD; Lyon, Hôpital de la Croix Rousse: C. Guerin MD; Mâcon, Centre Hospitalier de Mâcon: D. Debatty MD; Marseille, CHU La Conception: J. Billot MD; Maubeuge, Hôpital Sambre-Avesnois, W. Joos MD; Meaux, Centre Hospitalier: A. Combes MD; Metz, Hôpital Bon Secours: T. Jacques MD, J.-F. Poussel MD; Montpellier, Hôpital Gui de Chauliac: L. Landreau MD; Montpellier, Hôpital Lapeyronie: P. Chardon MD, JP. Roustan MD; Montreuil, Centre Hospitalier André Grégoire: E. Obadia MD, J.-L. Pallot MD; Moulins, Centre Hospitalier Moulins-Yzeure: F. Nicolas MD; Nancy, Hôpital Central: A. Cravoisy MD; Nantes, Hôpital Laennec: S. Chollet MD, J.-R. Ordronneau MD; Niort, Centre Hospitalier de. Niort: F. Barraud MD; Oloron Sainte Marie, Centre Hospitalier Général: J.-M. Mazou MD, Y. Gauthier MD; Orléans, Centre Hospitalier Régional: T. Boulain MD, I. Runge MD; Orsay, Centre Hospitalier Général d'Orsay: D. Benichou MD; Paris, Fondation Hôpital St Joseph: J. Carlet MD, W. Marie MD; Paris, Groupe Hospitalier Pitié-Salpêtrière: A. Duguet MD, H. Prodanovic MD, T. Similowski; Paris, Hôpital Bichat Claude Bernard: F. Schortgen MD, J.-F. Timsit MD; Paris, Hôpital Cochin: J.-D. Chiche MD, N. Nciri MD; Paris, Hôpital Européen Georges Pompidou: J.-L. Diehl MD, E. Guerot MD; Paris, Hôpital Saint-Antoine: E. Maury MD, G. Offenstadt MD; Paris, Hôpital Saint-Louis: E. Azoulay MD, G. Thiery MD; Paris, Hôpital Tenon: M. Fartoukh MD, F. Vincent MD; Paris, Hôtel Dieu: A. Lefebvre MD, A. Rabbat MD; Pessac, Clinique Mutualiste: R. Bedry MD; Poissy, CHI Poissy-Saint-Germain: S. Delerme MD, J.-C. Lacherade MD; Poitiers, Centre Hospitalier Universitaire: D. Chatellier MD; Pontoise, Centre Hospitalier René Dubos: J. Richecoeur MD; Rambouillet, Centre Hospitalier de. Rambouillet: N. Fadel MD; Reims, Hôpital Robert Debré: J. Cousson MD; Reims, Polyclinique Saint-André: J.-C. Farkas MD, B. Lafon MD; Roanne, Centre Hospitalier: P. Beuret MD; Roubaix, Hôpital Victor Provo: P. Herbecq MD, M. Nyunga Makenga MD; Rouen, CHU, Hôpital Charles Nicolle: J. Aboab MD, G. Minaret MD; Saint-Germain en Laye, CHI Poissy-Saint-Germain: J.-L. Ricome MD; Saintes, Centre Hospitalier: Y. Lefort MD; Suresne, Hôpital Foch: M. Djibre MD; Tulle, Centre Hospitalier: O. Hirtz MD; Valence, Centre Hospitalier: J.-Y. Petit MD; Villefranche sur Saone, Centre Hospitalier Général: L. Liron MD; Villejuif, Institut Gustave Roussy: F. Blot MD.
Author information
Authors and Affiliations
Corresponding author
Additional information
For SRLF Task Force on Mechanical Ventilation
Rights and permissions
About this article
Cite this article
Demoule, A., Girou, E., Richard, JC. et al. Increased use of noninvasive ventilation in French intensive care units. Intensive Care Med 32, 1747–1755 (2006). https://doi.org/10.1007/s00134-006-0229-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-006-0229-z