Abstract
Objective
Prolonged controlled mechanical ventilation (MV) is known to induce diaphragmatic oxidative stress that seems to be an important factor reducing force-generating capacity. To better understand the cellular mechanisms involved, this work examined the effect of short vs. prolonged MV on antioxidant defense in the diaphragm.
Design and setting
Prospective, randomized, controlled animal study in a university laboratory.
Methods
Eleven piglets (15–20 kg) were assigned to one of two groups: a long-MV group (n=6) ventilated for 3 days or a short-MV group (n=5) ventilated for 3 h. Force frequency curves of the transdiaphragmatic pressure (Pdi) were obtained in vivo by phrenic nerve pacing. Oxidative stress was evaluated by thiobarbituric reactive substance (TBARs) content and the enzymatic antioxidant activity of both superoxide dismutase (SOD) and glutathione peroxidase (GPx) in samples of diaphragm.
Results
Pdi decreased in the long-MV group by 30–35% over the 3 days at all frequencies compared to the short-MV group. Diaphragm TBARs content was significantly higher and SOD activity lower in long-MV animals than in short-MV animals after 72 h. GPx activity tended to be lower in diaphragms from long-MV animals, but this difference was not significant.
Conclusions
This study shows that 3 days of MV in piglets is associated with a decrease in antioxidant activity which could emphasize oxidative stress and both contribute to the diaphragm dysfunction caused by MV
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mechanical ventilation (MV) is currently used in the management of respiratory failure. Difficult weaning from mechanical ventilation occurs in 20–50% of patients who receive mechanical ventilation [1]. Alterations in intrinsic properties of respiratory muscles is one of the main factors responsible for this difficulty. The question of how MV affects the respiratory muscles in patients is of great clinical relevance but is difficult to assess in patients. Over the past few years several recent animal studies [2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16] have reported that controlled MV is associated with significant diaphragmatic atrophy and contractile dysfunction. However, the exact mechanism(s) responsible for this MV-induced diaphragmatic dysfunction remains unclear and is likely multifactorial [17]. Reduction in diaphragm mass [2], decrease in cross-sectional area of all the diaphragm fibers [11, 13, 16], myofibril damage [5], or decreased efficiency of mitochondrial oxidative phosphorylation coupling [10] have been reported. Recently the team of Powers [7, 14] showed that controlled MV increases oxidative stress in the diaphragm. Indeed, local production of free radicals has been shown to play an important role in muscle weakness in in vitro studies and this without myofibrillaire atrophy [18]. These results have recently been confirmed by an antioxidant treatment that prevented MV-induced contractile impairments in rats [19]. To our knowledge, only one study has evaluated the body’s antioxidant defense change under MV [9]. These authors found that 18 h of MV in rats increased superoxide dismutase (SOD) activity in diaphragm (one of the most important enzymes in the antioxidant defense system), which could reduce oxidative stress. However, previous studies showed that this adaptive mechanism may be exceeded by overproduction of reactive oxygen species (ROS) [20]. As duration of MV is an important factor which may increase the negative effect of oxidative stress, to better understand the cellular mechanisms involved in oxidative stress induced by MV, the diaphragm’s antioxidant defense after longer period of MV must be assessed. Moreover, studies on animal models such as pigs, which are very similar to humans in term of respiratory muscle anatomy and ventilator used (same device with the same adjustment), are necessary before use in humans. Therefore, using the pig model, the purpose of this study was to compare the effects of short-duration vs. prolonged-duration of MV on diaphragm antioxidant defense in association with the diaphragm contractility properties and oxidative burden involved in the pathogenesis of the respiratory muscles dysfunction caused by MV.
Methods
Animal preparation and experimental procedures
Eleven piglets weighing 15–20 kg were assigned to one of two groups, one with a prolonged period of controlled MV (long-MV group, n=6) ventilated for 3 days, and one with a short period of controlled MV (short-MV group n=5) ventilated for 2–4 h and then killed.
Protocol for short-MV and long-MV animals
After orotracheal intubation the animals were mechanically ventilated with volume-controlled ventilation. Continuous infusion of ketamine and propofol was used for anesthesia. Neuromuscular blocking agents were not used. Bipolar transvenous pacing catheters were introduced via each internal jugular vein and adjusted to achieve stimulation of the phrenic nerve and subsequent contraction of the diaphragm. Double air-filled balloon-tipped catheters were placed transorally in the distal one-third of the esophagus and in the stomach for measurement of transdiaphragmatic pressure (Pdi) (see Electronic Supplementary Material).
Care for long-MV animals
The inhibition of spontaneous diaphragm activity was confirmed by intermittent qualitative measurement of the electromyographic diaphragm activity during controlled MV with surface electrodes and by the lack of triggering of the ventilator (see Electronic Supplementary Material).
Animal monitoring in short-MV and long-MV groups
Heart rate, systolic and mean arterial blood pressures, and oxygen saturation were continuously monitored. Functional residual capacity was evaluated after stopping the sedation with the animal under spontaneous breathing under similar conditions [21].
In vivo measurement of diaphragm contractile properties
A force frequency curve was obtained on days 1 and 3 by stimulating diaphragm contractions at end-expiration with the airways occluded, using supramaximal phrenic nerve stimulation with stimulus duration of 0.15 ms and train duration of 2000 ms; two contractions were obtained at fixed frequencies of 20, 30, 50, 80, 100, and 120 Hz.
Tissue preparation
All morphometric (histochemical) and biochemical assays were conducted on the costal region of the diaphragm. Diaphragm samples were removed from the entire midcostal muscle spanning from the costal margin to the central tendon.
Biochemical analysis
To determine the effects of MV on both bioenergetics and the markers of oxidative stress we measured the activities of citrate synthase (CS), SOD, glutathione peroxidase (GPx), and thiobarbituric reactive substance (TBARs) content in the diaphragm samples [7, 8, 14]. The diaphragm antioxidant activity of GPx and SOD was quantified spectrophotometrically according to a previously reported method [22, 23] (see Electronic Supplementary Material).
Lung and peritoneal infection surveillance
Sepsis is associated with diaphragm contractile dysfunction [24]. Therefore aseptic techniques were used throughout the long-MV procedures. After the animals were killed, macroscopic examination of the lungs and peritoneal cavity revealed no abnormalities. The serum C-reactive protein (CRP) concentration was measured on the first and third days to rule out any infectious or inflammatory process.
Statistical analysis
Analyzed parameters for the short and long-MV groups during the study period are summarized in Table 1. Values are given as mean ±SEM. The two groups were compared on each dependent variable using nonparametric tests (Mann-Whitney). Repeated-measure analysis of variance was used to examine the data changes over time. Nonparametric paired Wilcoxon tests were used to compare data from days 1 and 3 for each animal in the long-MV group. The significance level for all statistics was fixed at p≤0.05.
Results
Systemic and biological response to MV
The average body weights did not differ significantly between the short-MV (17±2 kg) and long-MV group (18±2 kg). Throughout the study the animals of the long-MV group had normal intestinal transit and diuresis. They were afebrile. No animal suffered disturbed sodium, potassium, or calcium concentrations in serum (Table 2). There was no clinical or laboratory evidence of infection, the macroscopic examination of the lungs and the peritoneal cavity was normal, and the CRP was in the normal range at the 72nd hour (Table 2). Together these results indicate that the short-MV and long-MV groups were free of significant infection. All animals achieved normal fluid balance over 3 days. Hemoglobin levels decreased slightly between days 1 and 3, but this did not reach statistical significance (Table 2). Values for blood pressure and arterial blood gases showed no significant differences between days 1 and 3 in the long-MV group or between the long-MV and short-MV groups on day 1 (Table 3). Moreover, arterial blood pressure and blood gas/pH homeostasis were well maintained during the period of MV. Minute ventilation was reduced slightly over time on the first day (Table 3). Minor adjustments to ventilator settings were needed and oxygenation and ventilation variables showed little change over time on the last 2 days. No electrical activity of the diaphragm was observed in animals over time. Functional residual capacity remained stable, and no significant changes were noted between days 1 and 3, which is interpreted as evidence of stable lung volumes over the course of the study (Table 3).
Contractile properties
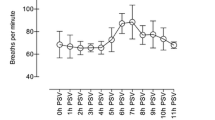
Pdi decreased at all frequencies tested between days 1 and 3 (Fig. 1). The fall in Pdi was statistically significant on day 3 compared with third hour in the long-MV group for all frequencies of stimulation except 20 Hz (Fig. 1). No significant differences were observed in Pdi values between long-MV and short-MV groups on day 1 (Fig. 1).
Diaphragmatic force-frequency curves (mean ±SEM). Transdiaphragmatic pressure (Pdi) after supramaximal phrenic nerve stimulation at 20, 30, 50, 80, 100, and 120 Hz over 3 days obtained in short-MV animals after 2–4 h of MV (D1) and in prolonged long-MV animals at 2–4 h (D1) and after 3 days of MV (D3). No significant difference was observed for the values obtained at all frequencies between short-MV and long-MV on day 1. NS No significant. *p<0.05 day 1 vs. day 3
Muscular biochemical analysis
Measurements of diaphragm total protein concentrations and CS activity showed no significant difference between the short-MV and long-MV groups obtained at the time at which the animals were killed (Figs. 2, 3).
Measurement of diaphragm oxidative injury
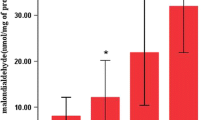
TBARs content was significantly higher in diaphragms from long-MV animals than in those from short-MV animals (Fig. 4). Total SOD activity was significantly lower in long-MV animals than in short-MV animals (Fig. 5a). Similarly, GPx activities tended to be lower in diaphragms from long-MV animals than in those from short-MV animals, but this difference did not reach statistical significance (Fig. 5b).
Discussion
The present study is the first to demonstrate in a piglet model of long-MV (72 h) that the reduction in diaphragm force-generating capacity is associated with both oxidative stress injury and decreased enzymatic antioxidant defenses. Indeed, contrary to a previous study using different animal model (rat) and duration of MV (18 h) [9], we found that 72 h of MV induces a decrease in antioxidant enzymatic system that may contribute to ventilator-induced diaphragm muscle weakness.
Reduction in diaphragm force
This study supports previous findings that prolonged MV results in a significant reduction in maximal Pdi [2, 3, 4, 7, 8, 9]. Indeed, Pdi decreased by 30–35% over the 3 days at all frequencies obtained in vivo. We did not continue MV after 3 days, considering a previous work [4] which reported in a similar animal model the same diaphragm force frequency relationships over 3 and 5 days of MV. This reduction in diaphragm force is time-dependent, with the force decline becoming evident quite early after 18 h in rats [8, 9]. This decrease is not due to changes in lung volume or abdominal compliance, as determined in the present study (Table 2) and others [3, 4]. Moreover, neither the phrenic nerve nor alteration in the neuromuscular junction seem to be involved; Radell et al. [4] reported that this parameter remains intact after 3 and 5 days of MV in an animal model similar to ours. However, it should be noted that although we used different anesthetic protocols (use of neuromuscular blocking agents or not), different ventilatory settings (ventilatory modes, positive end-expiratory pressure), and different animal species, the adverse effects of MV on diaphragmatic function were similar.
Oxidative stress and diaphragm function
Oxidative stress can be defined most simply as the imbalance between the production of free radicals capable of causing peroxidation of the lipid layer of cells and the body’s antioxidant defenses. Previous experimental studies have reported that a relatively short duration of MV (18–24 h) results in diaphragmatic force deficits associated with oxidative injury and increased proteolytic activity [8].
In the present study a global method was first used to assess the oxidant-mediated lipid damage: lipid peroxidation by measurement of TBARs, which is a nonspecific oxidative stress index that should be used with caution [25]. We used a fluorometric method, however, that was sufficiently sensitive and reproducible to provide a valid estimation of oxidative stress [26]. Our results show, as noted above, that MV induces oxidative stress in the diaphragm, as reflected by higher TBARs observed in the long-MV group (Fig. 4).
Our study brings to light new information about the change in enzymatic antioxidant activity with ventilator-induced diaphragm dysfunction. Indeed, alteration in enzymatic antioxidant systems has been previously involved in the oxidative stress genesis [27]. Catalase, SOD, and GPx are the three main antioxidant systems. Our study focuses on the latter two of these because the intramuscular catalase level is comparatively weak [28]. We found that SOD activity (one of the most important enzymes in the antioxidant defense system) significantly decreased (Fig. 5a), and GPx activity tended to be lower in diaphragms from long-MV animals, but this difference was not statistically significant (Fig. 5b). Very few studies have investigated the effect of MV on the adaptation of these defenses. Immobilization, which is a model close to MV, induced an increase in one isoform of SOD (Cu-Zn SOD) after several days [29]. The other isoform (Mn SOD) and GPx seemed to be unaffected or were even decreased by immobilization. Only one study [9] has reported increased SOD activity after 18 h of MV. After 3 days of ventilation we found for the first time a decrease in SOD activity associated with oxidative stress. The discrepancy between the findings of Shanely et al. [9] and those of the present study suggest a time course effect of the antioxidant defenses of skeletal muscle immobilization. We could speculate that the initial response is an increase within the first 24 h of MV, but as the oxidative stress continues, oxidant defenses begins to be exhausted, such as seen in our study. Moreover, in our study we compared in piglets after a few hours of MV (2–4 h) with 72 h of MV, and Shanely et al. [9] compared no MV with 18 h of MV in rats. To confirm this hypothesis, the time course of antioxidant defenses needs to be studied to understand the opposing effect between the present study and that of Shanely et al. [9]. The discrepancy between the two studies may be due in part to the differences in antioxidant response to MV related to differences in fiber susceptibility to oxidative stress or fiber composition. Thus extrapolation of these data to patient situation needs to be made with caution. However, this result could be interpreted as increased proteolytic activity induced by chronic exposure to ROS that could deteriorate the cellular pathway described previously [30] and decrease the adaptive capacity of the diaphragmatic antioxidant system. Since we did not find a statistically significant difference in GPx activity, we could speculate that this is probably due to a type II error, and/or that this system is less susceptible to oxidative stress than SOD. It thus seems that by high exposure to ROS 3 days of MV had a negative effect on an adaptive mechanism of diaphragmatic antioxidant capacity that was observed after 18 h of MV [9]; this would increase the risk of difficult weaning from MV in some cases.
Other enzymatic systems also seem to be affected by the alteration observed after 3 days of MV. Indeed, our results are in accordance with those of Le Bourdelles et al. [2] who reported that prolonged MV is associated with a significant reduction in diaphragm CS activity, CS being the main enzyme of the Krebs cycle and used as a marker of oxidative capacity.
Protein content did not differ significantly between the two groups, although the values in the diaphragms from long-MV animals tended to be lower than those in the short-MV group (Fig. 2). A previous study showed that insoluble proteins are primarily targeted by MV-induced oxidative stress [14], thus global assessment of protein content as a marker of oxidative stress has low sensitivity and our result is not surprising.
Limitations
Although piglet respiratory muscles are similar to those of humans, a limitation of this study is that we studied the effects of 3 days of MV on healthy diaphragm muscles. Although the evidence for MV-induced diaphragm dysfunction in animal models is convincing, it is considerably more difficult to obtain conclusive proof of diaphragm dysfunction in mechanically ventilated critically ill patients. However, two trials [31, 32] strongly suggest that diaphragm dysfunction is extremely prevalent among mechanically ventilated patients.
Because sepsis is associated with diaphragmatic contractile dysfunction, strict aseptic techniques were followed throughout the experiments. Our results indicate that the animals included in our analysis did not develop infections. This was supported by microscopic examination of blood, which revealed no detectable bacteria, and postmortem gross examination of the lungs and peritoneal cavity did not demonstrate abnormalities. Furthermore, both our long-MV and short-MV animals were afebrile (38–39°) during the investigation.
The anesthetic agents used in our protocol do not promote diaphragmatic contractile dysfunction or oxidative stress [8, 14]. The oxidative stress in the diaphragm reported in our study was obtained after only 3 days of MV. The kinetics and sources of oxidant production and antioxidant defense within the diaphragm during MV are unknown.
Clinical implications and future directions
Our study show that prolonged MV (3 days) induces a decrease in enzymatic antioxidant system, which may contribute to decreased force generating capacity by the diaphragm. Recent work in rats shows that the administration of antioxidant agent prevents MV-induced contractile impairments and proteolysis in the diaphragm during MV [19] by improving the antioxidant capacity of the diaphragm with a beneficial effect on diaphragmatic contractile deficit induced by MV. Studies evaluating effects of pharmacological antioxidant therapy on enzymatic antioxidant defense are needed to better understand the cellular mechanisms involved, in the beneficial impact observed with such a therapy. In addition, it is important to determine how varying degrees of spontaneous respiratory effort and other aspects of the patient-ventilator interaction are able to preserve diaphragmatic force and prevent oxidative stress. Recently Sassoon et al. [15], using a rabbit model with 3 days of MV, reported that partial diaphragm muscle activation associated with assisted MV is sufficient to attenuate the force loss induced by complete inactivity. Moreover, the role of ventilatory strategy such as respiratory rate, tidal volume, and use of positive end-expiratory pressure on the inactive and passively shortened respiratory muscles is unknown. Further studies are needed to determine more precisely the mechanisms involved in the long-MV induced oxidative stress and to confirm its implication in diaphragmatic dysfunction.
References
Laghi F, Tobin M (2003) Disorders of the respiratory muscles. Am J Respir Crit Care Med 168:10–48
Le Bourdelles G, Viires N, Boczkowski J, Seta N, Pavlovic D, Aubier M (1994) Effects of mechanical ventilation on diaphragmatic contractile properties in rats. Am J Respir Crit Care Med 149:1539–1544
Anzueto A, Peters JI, Seidner SR, Cox WJ, Schroeder W, Coalson JJ (1997) Effects of continuous bed rotation and prolonged mechanical ventilation on healthy, adult baboons. Crit Care Med 25:1560–1564
Radell PJ, Remahl S, Nichols DG, Eriksson LI (2002) Effects of prolonged mechanical ventilation and inactivity on piglet diaphragm function. Intensive Care Med 28:358–364
Sassoon CS, Caiozzo VJ, Manka A, Sieck GC (2002) Altered diaphragm contractile properties with controlled mechanical ventilation. J Appl Physiol 92:2585–2595
Yang L, Luo J, Bourdon J, Lin MC, Gottfried SB, Petrof BJ (2002) Controlled mechanical ventilation leads to remodeling of the rat diaphragm. Am J Respir Crit Care Med 166:1135–1140
Shanely RA, Zergeroglu MA, Lennon SL, Sugiura T, Yimlamai T, Enns D, Belcastro A, Powers SK (2002) Mechanical ventilation-induced diaphragmatic atrophy is associated with oxidative injury and increased proteolytic activity. Am J Respir Crit Care Med 166:1369–1374
Powers SK, Shanely RA, Coombes JS, Koesterer TJ, McKenzie M, Van-Gammeren D, Cicale M, Dodd SL (2002) Mechanical ventilation results in progressive contractile dysfunction in the diaphragm. J Appl Physiol 92:1851–1858
Shanely RA, Coombes JS, Zergeroglu AM, Webb AI, Powers SK (2003) Short-duration mechanical ventilation enhances diaphragmatic fatigue resistance but impairs force production. Chest 123:195–201
Bernard N, Matecki S, Py G, Lopez S, Mercier J, Capdevila X (2003) Effects of prolonged mechanical ventilation on respiratory muscle ultrastructure and mitochondrial respiration in rabbits. Intensive Care Med 29:111–118
Capdevila X, Lopez S, Bernard N, Rabischong E, Ramonatxo M, Martinazzo G, Prefaut C (2003) Effects of controlled mechanical ventilation on respiratory muscle contractile properties in rabbits. Intensive Care Med 29:103–110
Racz GZ, Gayan-Ramirez G, Testelmans D, Cadot P, De Paepe K, Zador E, Wuytack F, Decramer M (2003) Early changes in rat diaphragm biology with mechanical ventilation. Am J Respir Crit Care Med 168:297–304
Gayan-Ramirez G, de Paepe K, Cadot P, Decramer M (2003) Detrimental effects of short-term mechanical ventilation on diaphragm function and IGF-I mRNA in rats. Intensive Care Med 29:825–833
Zergeroglu M, McKenzie M, Shanely R, Van Gammeren D, DeRuisseau K, Powers S (2003) Mechanical ventilation-induced oxidative stress in the diaphragm. J Appl Physiol 95:1116–1124
Sassoon C, Zhu E, Caiozzo V (2004) Assist-control mechanical ventilation attenuates ventilator-induced diaphragmatic dysfunction. Am J Respir Crit Care Med 170:626–632
Radell P, Edstrom L, Stibler H, Eriksson L, Ansved T (2004) Changes in diaphragm structure following prolonged mechanical ventilation in piglets. Acta Anaesthesiol Scand 48:430–437
Vassilakopoulos T, Petrof B (2004) Ventilator-induced diaphragmatic dysfunction. Am J Respir Crit Care Med 169:336–341
Reid M (2001) Redox modulation of skeletal muscle contraction: what we know and what we don’t. J Appl Physiol 90:724–731
Betters J, Criswell D, Shanely R, Van Gammeren D, Falk D, Deruisseau K, Deering M, Yimlamai T, Powers S (2004) Trolox attenuates mechanical ventilation-induced diaphragmatic dysfunction and proteolysis. Am J Respir Crit Care Med 170:1179–1184
Sen C (1995) Oxidants and antioxidants in exercise. J Appl Physiol 79:675–686
Miller J, Law A, Parker R, Sundell H, Lindstrom D, Cotton R (1995) Validation of a nitrogen washout system to measure functional residual capacity in premature infants with hyaline membrane disease. Pediatr Pulmonol 20:403–409
Chiu D, Stults F, Tappel A (1976) Purification and properties of rat lung soluble glutathione peroxidase. Biochim Biophys Acta 445:558–566
Sun M, Zigman S (1978) An improved spectrophotometric assay for superoxide dismutase based on epinephrine autoxidation. Anal Biochem 90:81–89
Divangahi M, Matecki S, Dudley R, Tuck S, Bao W, Radzioch D, Comtois A, Petrof B (2004) Preferential diaphragmatic weakness during sustained Pseudomonas aeruginosa lung infection. Am J Respir Crit Care Med 169:679–686
Halliwell B, Chirico S (1993) Lipid peroxidation: its mechanism, measurement, and significance. Am J Clin Nutr 57:715S–724S
Yagi K (1976) A simple fluorometric assay for lipoperoxide in blood plasma. Biochem Med 15:212–216
Ji L (2001) Exercise at old age: does it increase or alleviate oxidative stress? Ann N Y Acad Sci 928:236–247
Gutteridge J, Halliwell B (1989) Iron toxicity and oxygen radicals. Baillieres Clin Haematol 2:195–256
Lawler J, Song W, Demaree S (2003) Hindlimb unloading increases oxidative stress and disrupts antioxidant capacity in skeletal muscle. Free Radic Biol Med 1:9–16
Hollander J, Fiebig R, Gore M, Ookawara T, Ohno H, Ji L (2001) Superoxide dismutase gene expression is activated by a single bout of exercise in rat skeletal muscle. Pflugers Arch 442:426–434
Watson A, Hughes P, Louise Harris M, Hart N, Ware R, Wendon J, Green M, Moxham J (2001) Measurement of twitch transdiaphragmatic, esophageal, and endotracheal tube pressure with bilateral anterolateral magnetic phrenic nerve stimulation in patients in the intensive care unit. Crit Care Med 29:1325–1331
Laghi F, Cattapan S, Jubran A, Parthasarathy S, Warshawsky P, Choi Y, Tobin M (2003) Is weaning failure caused by low-frequency fatigue of the diaphragm? Am J Respir Crit Care Med 167:120–127
Acknowledgements
The authors are grateful to Pr. Peter Dodek for his very useful comments and thank Cathy Carmeni and Caryne Digena for reviewing the English.
Author information
Authors and Affiliations
Corresponding author
Additional information
S. Jaber and M. Sebbane contributed equally to this study.
This article refers to the editorial http://dx.doi.org/10.1007/s00134-005-2693-2
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Jaber, S., Sebbane, M., Koechlin, C. et al. Effects of short vs. prolonged mechanical ventilation on antioxidant systems in piglet diaphragm. Intensive Care Med 31, 1427–1433 (2005). https://doi.org/10.1007/s00134-005-2694-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-005-2694-1