Abstract
Objective
We examined whether mechanical ventilation with low tidal volume induces polymorphonuclear infiltration and proinflammatory and profibrogenic responses in rat lungs compared dependent and nondependent lung region to expression of interleukin-1β (IL-1β) and α-1 procollagen III (PC III) mRNA.
Design
An experimental, randomized and controlled protocol with previously normal rats.
Interventions
Three groups of ten animals were studied. Two groups were ventilated (FIO2=0.3) in supine position for 1 h without positive end expiratory pressure, one group with a low tidal volume (6 ml/kg), and the other with a high tidal volume (24 ml/kg). In the third group animals were kept in spontaneous ventilation for 1 h.
Measurements and results
After ventilation the right lung was used to quantify polymorphonuclear infiltration. The left lung was divided into dependent and nondependent regions, and expression of IL-1β and PC III mRNA was quantified by northern blot analysis. The group ventilated with low tidal volume had greater polymorphonuclear infiltration IL-1β and PC III mRNA expression than the nonventilated group. Similar results were observed with high tidal volumes. There was no difference between low and high tidal volume ventilation. Expression levels of IL-1β and PC III mRNA were higher in the nondependent region of ventilated groups and equal in the nonventilated group.
Conclusions
Even a low tidal volume mode of mechanical ventilation induces proinflammatory and profibrogenic response, with a nondependent predominance for IL-1β and PC III mRNA expression in supine, ventilated, previously normal rats.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Experimental studies have shown that mechanical ventilation (MV) with high tidal volume and high inflation pressure causes edema, tissue damage, neutrophil infiltration, and production of inflammatory and profibrogenic mediators [1]. The injury induced by MV is characterized by a dependent (posterior) distribution of pulmonary collapse and edema [2]. However, the association of these alterations in MV with low tidal volume is not established. It has been shown in patients with acute lung injury and acute respiratory distress syndrome that MV with a low tidal volume results in decreased mortality [3], and therefore an increased use of MV with low tidal volume is expected.
We examined whether MV with low tidal volume in previously normal supine rats induces polymorphonuclear infiltration and production of inflammatory and profibrogenic mediators. Proinflammatory and profibrogenic responses were represented by their key mediators interleukin-1β (IL-1β) and α-1 procollagen III (PC III) mRNA expression. In addition, we compared dependent and nondependent lung regions to IL-1β and PC III mRNA expression.
Methods
Male Wistar rats were sedated (ketamine, 80 mg/kg and xylazine, 8 mg/kg) and paralyzed (succinylcholine, 3 mg/kg). Tracheotomy was performed and a catheter inserted into the right carotid artery. Groups of ten animals were randomly assigned to 1 h under the following conditions: (a) NV group: animals were tracheotomized, not paralyzed, superficially sedated and kept in spontaneous ventilation; (b) LV group: low tidal volume of 6 ml/kg and zero positive end-expiratory pressure (ZEEP); (c) HV group: high tidal volume of 24 ml/kg and ZEEP.
Ventilation was performed in supine position using a volume-controlled ventilator (Inter 3, Intermed, Brazil) with FIO2 at 0.3 and a frequency of 90 breaths/min. To assure the same inspiratory time we used distinct inspiratory flows (LV=10 ml/min and HV=40 ml/min). Airway pressure was measured using a transducer (142PC05D, Honeywell, Freeport, Ill., USA) connected to the endotracheal tube. Flows were measured by a pneumotachograph (Hans-Rudolph 8420, Kansas City, Mo., USA) connected to a transducer (163PC01D36, Honeywell). Signals were sampled at 200 Hz (Anadat 4.0, Montreal, Calif., USA). At the end of the MV period animals were bled to death. To avoid collapse artifacts the lungs were recruited with a continuous positive airway pressure of 30 cmH2O for 10 s and kept statically inflated with 5 cmH2O. The right lung was sectioned in a sagittal plane, and 3-µm-thick sections were stained with hematoxylin and eosin for the histomorphometric study. The left lung was divided into three regions: anterior, hilar, and posterior. Nondependent and dependent tissues were immediately frozen for mRNA quantification.
Based on the point-counting method [4], the polymorphonuclear infiltration index was the ratio between the numbers of points that fell on polymorphonuclears and the numbers of lines intersection with the alveolar septum. Measurements were performed in 20 fields per slide, using a magnification of ×1000, by two investigators blinded to the region of sampling. IL-1β and PC III expressions in lungs were determined by northern blot analysis using total RNA as previously described [5]. As probes we used cDNA fragments from IL-1β and PC III (positions 5′ CTAATGCCTTCCCCAGGA 3′ and 5′ GCTGTGCACTGGTCCA 3′ and 5′ TACACAGTTCTAGAGGATGG 3′ and 5′ CTGTATTGGTGGGTGAAACAG 3′, respectively) and GAPDH as control for RNA loading. Filters were scanned by a phosphorimager (Storm 840, Molecular Dynamics, Sunnyvale, Calif., USA).
The groups of animals were similar regarding weight, inspiratory time, and dose of drugs (Table 1). Mean arterial blood pressure was kept above 60 mmHg. No animal died, nor did any develop air leaks or intrinsic positive end-expiratory pressure during MV.
Data are expressed as the ratio probe/GAPDH. General characteristics, mRNA expression, and histomorphometric values were compared by one-way analysis of variance. Dunnett's post hoc test was used to determine statistically significant differences between specific pairs of groups. Nondependent and dependent mRNA expressions were compared using the Wilcoxon test. A p value less than 0.05 was considered statistically significant. Values are reported as median and interquartile range.
Results
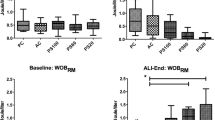
The polymorphonuclear infiltration index was significantly higher in LV and HV than in NV (p<0.001); there was no difference between LV and HV (Table 1). In both dependent and nondependent lung regions there was higher IL-1β mRNA expression in LV and HV than in NV (p<0.05) while there was no statistically significant difference between LV and HV in either region (Fig. 1). In the nondependent lung region PC III mRNA expression in LV was higher than in NV (p=0.01); HV showed a trend toward higher expression than NV (p=0.08). In the dependent lung region PC III mRNA expression in LV was higher (p=0.05) and HV similar to that in NV (p=0.77; Fig. 2).
LV and HV showed higher levels of IL-1β mRNA in the nondependent than the dependent lung region, while in NV there was no difference between regions (Fig. 1). PC III mRNA expression showed no difference between nondependent and dependent lung regions in the NV, while in LV there was a significantly higher level in the nondependent than in the dependent region, and in HV there was a trend toward a higher expression in the nondependent region (Fig. 2).
Comparison of interleukin-1β (IL-1β) mRNA expression between nondependent and dependent regions in each group. Low tidal volume: 6 ml/kg and ZEEP; high tidal volume: 24 ml/kg and ZEEP. IL-1β mRNA expression is expressed as IL-1β mRNA/GAPDH arbitrary units. Graphics are clustered boxplot summarizing the median and quartiles *p=0.96, **p=0.02, ***p=0.04, dependent vs. nondependent regions
Comparison of procollagen III (PC III) mRNA expression between nondependent and dependent regions in each group. Low tidal volume: 6 ml/kg and ZEEP; high tidal volume; 24 ml/kg and ZEEP. PC III mRNA expression is expressed as PC III mRNA/GAPDH arbitrary units. Graphics are clustered boxplot summarizing the median and quartiles. The low volume group had significantly higher PC III mRNA expression in the nondependent region. *p=0.39, **p=0.04, ***p=0.07, dependent vs. nondependent regions
Discussion
In our study the use of low (respiratory rate and tidal volume within physiological range) or high tidal volume caused an increase in polymorphonuclear infiltration and in inflammatory and profibrogenic mediators. A possible explanation for the increased mediator expression with low tidal volume could be the lung heterogeneity, since general anesthesia and supine position are associated with dependent atelectasis [6]. This lung heterogeneity may have caused alveolocapillar distention in the nondependent region and repetitive opening and closing of distal lung in the dependent lung region, rendering the lung more susceptible to ventilator induced lung injury [1]. In addition to heterogeneity, passive MV is not a physiological condition (even within physiological ranges) and may induce a proinflammatory and profibrogenic response. Infants without preexisting lung disease and mechanically ventilated for 2 h present in the tracheal aspirates an immune balance favoring a proinflammatory response pattern [7]. However, in patients with normal pulmonary function plasma cytokines, which may not entirely mirror the lungs, do not change with MV [8].
An interesting finding in our study was the equal induction of IL-1β and PC III mRNA between LV and HV. The use of a moderately aggressive MV mode in HV with a peak inspiratory pressure kept below that customary for injury induction in animal experiments [1] and the fact that previously healthy lungs are less prone to injury [9] could explain the results. According to the protocol design, arterial CO2 differed between LV and HV, and it is known that hypercapnia attenuates pulmonary inflammation [10]. Notwithstanding this protective effect of hypercapnia, proinflammatory and profibrogenic mediators were induced in LV. The intriguing result was the nondependent predominance of mediators mRNA expression in contrast to the dependent predominance of edema and collapse described in previously normal animals subjected to MV [2]. A possible explanation for this finding is the different distribution of the injury mechanisms. If the alveolocapillar distention generates more mediators, a higher induction in the non-dependent region is to be expected. Experimental models with high inflation pressures, a probable cause of alveolocapillar distention, show extensive ultrastructural failure of the alveolocapillar barrier, histological damage, and production of inflammatory mediators [1]. Moreover, experimental models designed to study repetitive opening in previously healthy lungs show disturbance of lung mechanics and blood gases while the alveolar microstructure remains preserved and inflammation was absent [11]. Despite this, to reexpand a collapsed region the surrounding alveoli are exposed to a higher distending pressure thus combining the two mechanisms of ventilator-induced lung injury in the same region. Finally, as positive end-expiratory pressure alters lung gas distribution, its use may alter the distribution of injury mechanisms and mediator production.
We measured IL-1β because it is an early-phase cytokine, and its level increase during injurious ventilatory strategies [12]. Moreover, most of the proinflammatory activity from patients with acute respiratory distress syndrome is attributable to biologically active IL-1β [13]. We also measured PC III since its level increases during injurious ventilatory strategies in normal animals [14]. We decided to determine mRNA values rather than protein values because our MV duration might be not sufficient for the production of an expressive amount of collagen; however, short periods of MV are enough for increasing procollagen mRNA expression [15].
In conclusion, we demonstrated that a proinflammatory and profibrogenic response is induced in previously normal rats even at low tidal volumes, with a predominance of IL-1β and PC III mRNA expression in the nondependent lung region. MV is essential in severe respiratory failure and alters mortality [3]. Since many aspects of injury induced by MV are currently unknown, this knowledge is essential to design less aggressive ventilatory strategies.
References
Dreyfuss D, Saumon G (1998) Ventilator-induced lung injury: lessons from experimental studies. Am J Respir Crit Care Med 157:294–323
Broccard A, Shapiro RS, Schmitz LL, Adams AB, Nahum A, Marini JJ (2000) Prone positioning attenuates and redistributes ventilator-induced lung injury in dogs. Crit Care Med 28:295–303
Amato MB, Barbas CS, Medeiros DM, Magaldi RB, Schettino GP, Lorenzi-Filho G, Kairalla RA, Deheinzelin D, Munoz C, Oliveira R, Takagaki TY, Carvalho CR (1998) Effect of a protective-ventilation strategy on mortality in the acute respiratory distress syndrome. N Engl J Med 338:347–354
Weibel ER (1963) Principles and methods for the morphometric study of the lung and other organs. Lab Invest 12:131–155
Dias AA, Goodman AR, Dos Santos JL, Gomes RN, Altmeyer A, Bozza PT, Horta MF, Vilcek J, Reis LF (2001) TSG-14 transgenic mice have improved survival to endotoxemia and to CLP-induced sepsis. J Leukoc Biol 69:928–936
Hedenstierna G, Lundquist H, Lundh B, Tokics L, Strandberg A, Brismar B, Frostell C (1989) Pulmonary densities during anaesthesia. An experimental study on lung morphology and gas exchange. Eur Respir J 2:528–535
Plotz FB, Vreugdenhil HA, Slutsky AS, Zijlstra J, Heijnen CJ, van Vught H (2002) Mechanical ventilation alters the immune response in children without lung pathology. Intensive Care Med 28:486–492
Wrigge H, Zinserling J, Stuber F, von Spiegel T, Hering R, Wetegrove S, Hoeft A, Putensen C (2000) Effects of mechanical ventilation on release of cytokines into systemic circulation in patients with normal pulmonary function. Anesthesiology 93:1413–1417
Dreyfuss D, Soler P, Saumon G (1995) Mechanical ventilation-induced pulmonary edema. Interaction with previous lung alterations. Am J Respir Crit Care Med 151:1568–1575
Laffey JG, Tanaka M, Engelberts D, Luo X, Yuan S, Tanswell AK, Post M, Lindsay T, Kavanagh BP (2000) Therapeutic hypercapnia reduces pulmonary and systemic injury following in vivo lung reperfusion. Am J Respir Crit Care Med 162:2287–2294
Taskar V, John J, Evander E, Wollmer P, Robertson B, Jonson B (1995) Healthy lungs tolerate repetitive collapse and reopening during short periods of mechanical ventilation. Acta Anaesthesiol Scand 39:370–376
Tremblay L, Valenza F, Ribeiro SP, Li J, Slutsky AS (1997) Injurious ventilatory strategies increase cytokines and c-fos m-RNA expression in an isolated rat lung model. J Clin Invest 99:944–952
Pugin J, Ricou B, Steinberg KP, Suter PM, Martin TR (1996) Proinflammatory activity in bronchoalveolar lavage fluids from patients with ARDS, a prominent role for interleukin-1. Am J Respir Crit Care Med 153:1850–1856
Berg JT, Fu Z, Breen EC, Tran HC, Mathieu-Costello O, West JB (1997) High lung inflation increases mRNA levels of ECM components and growth factors in lung parenchyma. J Appl Physiol 83:120–128
Deheinzelin D, Jatene FB, Saldiva PH, Brentani RR (1997) Upregulation of collagen messenger RNA expression occurs immediately after lung damage. Chest 112:1184–1188
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Caruso, P., Meireles, S.I., Reis, L.F.L. et al. Low tidal volume ventilation induces proinflammatory and profibrogenic response in lungs of rats. Intensive Care Med 29, 1808–1811 (2003). https://doi.org/10.1007/s00134-003-1908-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-003-1908-7