Abstract
Although designed to control pests selectively, there is some evidence that environmental contamination by pesticides increases risks for humans and wildlife. In the present study, we evaluated biomarkers of oxidative stress in Astyanax jacuhiensis exposed to (5, 15 and 30 µg L−1) of carbamate Propoxur (PPX) for 96 h. Glutathione S-transferase (GST) in liver and gills showed reduced activity in all PPX concentrations tested. Acetylcholinesterase (AChE) activities reduced in brain and muscle at concentrations 15 and 30 µg L−1 of PPX. Lipid peroxidation (LPO) and hydrogen peroxide (HP) had no significant differences. In the brain, protein carbonyl (PC) increased in all groups treated with PPX. Although PPX is a selective pesticide, it causes oxidative damage and enzyme alteration in fish. This study pointed out some biomarkers that could be used to assess effects of environmentally relevant concentrations of pesticides, and infer about studies using fish as bioindicator.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Pesticides are chemical compounds used in agricultural crops to eliminate pests selectively. However, many studies have already shown that pesticides are potentially released into the environment, triggering immediate or long-term impacts on non-target organisms, even at sub-lethal doses (Clasen et al. 2014; Loro et al. 2015). Carbamate is a group of insecticide widely used because of its rapid action and selectivity, as well as short persistence in the environment. It acts by inhibiting acetylcholinesterase (AChE) non-permanently (Schmuck and Mihail 2004). Carbamate has been associated to acute and chronic intoxication of non-target organisms, resulting in neurological and behavioral changes (Grue et al. 1997). Additionally, carbamate pesticides cause oxidative stress by overproduction of reactive oxygen species (ROS) and induce lipid peroxidation in fish exposed (Clasen et al. 2014). In aerobic organisms, ROS are generated in cells via numerous endogenous and exogenous processes. However, when the redox homeostasis is disturbed, oxidative stress may lead to harmful effects to cellular macromolecules such as nucleic acids, carbohydrates, proteins, and lipids, causing several alterations and disease development (Schmuck and Mihail 2004; Trachootham et al. 2008). Propoxur (Baygon®) (PPX, 2-isopropoxyphenyl methylcarbamate) is a widely broad-spectrum carbamate insecticide and acaricide used both for agricultural and public health purposes. Agricultural crop applications include sugar cane, maize, rice, vegetables, cotton, forestry, and ornamentals (Wang et al. 2009). In a general trend, the commercial carbamate PPX displays less potent AChE inhibition than other carbamates, e.g. bendiocarb and carbofuran (Jiang et al. 2013). Although considered moderately hazardous to humans and domestic animals (class II), PPX is highly toxic to birds and fish, and it is a potential contaminant of the aquatic environment due to its high solubility in water (WHO 2009). Pesticides contaminate groundwater and drinking waters in areas with intensive agriculture. PPX has been detected in the environment at a concentration ranging from 0.04 to 29.1 µg L−1 (Loro et al. 2015; Amaral et al. 2018). The presence of PPX has been linked to several disorders such as abnormal neurodevelopment (Ostrea Jr. et al. 2012), energy-producing mechanisms injury, mitochondrial inner membrane potential disrupt (Schmuck and Mihail 2004), histopathological lesions, oxidative and genotoxic effects, as well lipid peroxidation (Maran et al. 2010; Tsitsimpikou et al. 2013). Although PPX is highly toxic to fish, scarce studies of its effects have been conducted on these animals especially considering environmental relevant concentrations of pesticides found in freshwaters. Astyanax jacuhiensis (Characidae), known as “lambari”, is a native Brazilian freshwater fish widely distributed in Brazilian ecosystems. The Astyanax species are bioindicator for ecological investigations due to their characteristics as wide distribution, omnivorous feeding habits and small size (Bueno-Krawczyk et al. 2015; Loro et al. 2015). As fish are animals in constant exposure to pesticides released into the environment, and considering that PPX carbamate is still scarcely investigated in fish, this study aimed to investigate the effects of acute exposure to PPX on selected oxidative stress parameters in Astyanax jacuhiensis. The parameters evaluated are adequate indicators of fish exposure to xenobiotics. The biomarkers tested at present study may increase knowledge about the toxicity of PPX in fish.
Materials and Methods
Propoxur (PPX) up to 99% purity was purchased from Sigma-Aldrich (St. Louis, MO, USA). The standard solution prepared by LARP (laboratory for the analysis of pesticide residues) and after was diluted in water to fish exposure. 100 juvenile Astyanax jacuhiensis of both sexes (weight 7.8 ± 1.1 g; length 8.3 ± 0.5 cm) were obtained from a fish farm (Rio Grande do Sul, Brazil). They were maintained in a static system in 40 L boxes of fiberglass, with continuously aerated tap water and physical and biological filters. The fishes were acclimated for ten days to laboratory conditions, with natural photoperiod (14 h light/10 h dark). Water quality parameters were quantified daily. Fishes were fed twice a day with commercial feed during the acclimation period. After acclimation, the animals were distributed into four experimental groups (n = 25, per group) as follows: CON (control), PPX I (5 µg L−1), PPX II (15 µg L−1) and PPX III (30 µg L−1). The concentration of PPX in water was measured at the beginning and at the end of exposure time according to Sabin et al. (2009). Final concentrations of PPX were 100%, 94% and 80% of the initial concentrations in the PPX I, PPX II and PPX III groups, respectively. The methodology to verify PPX in water used: Liquid chromatography Equipment with C18 reverse phase and ultraviolet detector with 5 µm granulometry and 30 cm equivalent length. The LOD was 0.05 µg L−1 with recovery % of 99%.
After 96 h, fishes were euthanized by a section of spinal cord behind the operculum. Brain, gill, liver, and muscle were removed for biochemical analysis (n = 13). The study was in accordance with the Ethics Committee on Animal Use of Universidade Federal do Pampa-UNIPAMPA (protocol no 004-2014). Hydrogen peroxide (HP) content was determined in brain, gills, liver, and muscle according to Loreto and Velikova (2001), considering the n = 12. The amount of H2O2 was expressed in µmol mg protein−1. AChE activity was measured according to Ellman et al. (1961) in brain and muscle. AChE activity was expressed as µmol of AcSCh hydrolyzed min−1 mg protein−1. Glutathione S-transferase (GST) activity was measured in all tissues according to Habig et al. (1974). The activity was expressed as µmol GS-DNB min mg protein−1. Lipid peroxidation (LPO) was estimated by TBARS assay (thiobarbituric acid-reactive substances) in brain, gills, liver, and muscle by malondialdehyde (MDA) reaction with the 2-thiobarbituric acid (TBA), according to Draper and Hadley (1990) and expressed as nmol TBARS mg−1 protein. Protein carbonyl (PC) content was evaluated in the same tissues of lipid peroxidation according to Yan et al. (1995) and was expressed as nmol carbonyl mg protein−1. Protein was determined using bovine serum albumin as standard (Bradford 1976). Normality and homogeneity were determined by Shapiro–Wilk and Kolmogorov–Smirnov tests. Results are reported as mean ± standard deviation (S.D.). Comparisons between groups were evaluated by one-way ANOVA followed by Tukey’s test. p < 0.05 was the significant level considered. Statistical analyses were performed with GraphPad Prism 6.01.
Results and Discussion
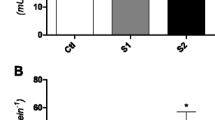
The increased use of pesticides worldwide is a concern since the 1960s, although they contribute greatly to pest control and agricultural improvement. However, environmental contamination by pesticides results in serious implications to non-target fauna and human health (Rodrigues et al. 2017). The model of pesticide poisoning using fishes to verify toxic effects is common in ecotoxicological studies specially considering environmental relevant concentrations (Loro et al. 2015; Bueno-Krawczyk et al. 2015; Amaral et al. 2018). In this study, the HP content was not different between control and the PPX exposed groups in all tissues analyzed (Table 1). Probably the activities of antioxidant enzymes as CAT, SOD, and GPX (not evaluated in this study) could be the cause of the absence of results for this parameter. Lipid peroxidation is an oxidative damage biomarker used in studies considering pesticides exposure. In the present work, there was no significant difference in LPO between the treatments (Table 1). Increased LPO levels were show in different tissues of Cyprinus carpio after carbofuran exposure for 7 and 30 days in laboratory condition (Clasen et al. 2014). Samanta et al. (2014) also showed LPO increased levels in the same tissues evaluated in this study after glyphosate exposure. The absence of LPO between treatments could be attributed to high activities of antioxidant enzymes and other antioxidants as glutathione (GSH) (not evaluated in this study). In relation to GST, the results found in this work suggest that PPX exposure negatively affected its activity. The activity of GST was reduced in all groups and tissues exposed to PPX (Fig. 1a). GST is a phase II detoxification enzyme producing less toxic substances that can be easily excreted from the organism. In the present study, fish cannot eliminate PPX through GST activity. Several studies have already suggested the inhibition of GST activity by exposure to pesticide as carbofuran (Clasen et al. 2014), organochlorine pesticides (Bacchetta et al. 2014) and glyphosate-based herbicide (Samanta et al. 2014). Being a carbamate insecticide PPX is a cholinesterase inhibitor and although less persistent and not susceptible to bioaccumulation like organochlorines, they are toxic to several animals. In our study, AChE activity reduced in groups exposed to PPX, in both tissues analyzed (Fig. 1b). This inhibition has already been found in fish exposed to other carbamate insecticides (Bacchetta et al. 2014; Clasen et al. 2014). AChE inhibition leads to accumulation of acetylcholine at cholinergic synapses and consequently to the overstimulation of the post-synaptic membrane. This phenomenon is responsible for impairment of the central nervous system and many others physiological functions (Menéndez-Helman et al. 2012). The results of present study support the disruption of AChE activity as shown in the literature.
a GST (µmol GS-DNB min mg µmol−1) activity in brain, muscle, gills and liver. b AChE (µmol of AcSCh hydrolyzed min−1 mg protein−1) activity in brain and muscle of Astyanax sp. exposed to 5, 10 or 30 µg L−1 of propoxur (n = 13). Different letters indicate significant statistical difference among the treatments (p < 0.05)
Carbonylation disturbs protein function and leads to cell dysfunction. The results of present work showed an increase in protein carbonyl in the brain for all groups treated with PPX (Fig. 2). This PC increase could confirm the protein oxidation induction when fish were exposed to PPX. In gills and liver, PPX increased PC at two higher doses (Fig. 2), and at the highest in muscle. The PC increase in tissues of the Astyanax jacuhiensis and the absence of LPO formation pointed out the preference of PPX to oxidize proteins. Moura et al. (2017) has already reported PC in fish exposed to a glyphosate-based herbicide, and corroborates with the theory that pesticides are able to trigger oxidative damage in fish, representing a threat to fish health.
The widespread use of pesticides worldwide has resulted in contamination of certain ecosystems. This contamination can be attributed to the absence of adequate pesticide management. It is estimated that only 0.1% of the applied pesticide reaches the target pest, the remaining end up polluting water, soil, and air through drift, flow, volatilization and leaching (Pimentel 1995). The increase of oxidative stress and the suppression of antioxidant defenses in response to exposure to pesticides have been widely investigated as a possible mechanism for fish toxicity. The current investigation suggests that PPX exposure may cause significant oxidative stress and suppression of antioxidant defense in Astyanax jacuhiensis under laboratory conditions. Another study has already investigated a genotoxic potential of PPX (Gul et al. 2012), however, this is the first work that investigates the potential of PPX to cause oxidative damage in fish and highlights the importance to screen the toxic potentials of carbamates to wildlife. The present work recognizes the need for a comprehensive assessment of the ecological risk of PPX since its use as a household and commercial pesticide has increased. We also documented the importance of using reliable bioindicators as a tool in assessing toxic effects of pesticides, considering that their management is not adequate, especially in Brazil.
References
Amaral A, Gomes J, Weimer G, Marins A, Loro V, Zanella R (2018) Seasonal implications on toxicity biomarkers of Loricariichthys anus (Valenciennes, 1835) from a subtropical reservoir. Chemosphere 191:876–885
Bacchetta C, Rossi A, Ale A, Campana M, Parma MJ, Cazenave J (2014) Combined toxicological effects of pesticides: a fish multi-biomarker approach. Ecol Indic 36:532–538
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bueno-Krawczyk ACD, Guiloski IC, Piancini LDS, Azevedo JC, Ramsdorf WA, Ide AH, Guimarães ATB, Cestari MM, Silva AHC (2015) Multibiomarker in fish to evaluate a river used to water public supply. Chemosphere 135:257–264
Clasen B, Leitemperger J, Murussi C, Pretto A, Menezes C, Dalabona F, Marchezan E, Adaime MB, Zanella R, Loro VL (2014) Carbofuran promotes biochemical changes in carp exposed to rice field and laboratory conditions. Ecotoxicol Environ Saf 101:77–82
Draper HH, Hadley M (1990) Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol 186:421–431
Ellman GL, Courtney KD, Andres VJR (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Grue CE, Gibert PL, Seeley ME (1997) Neurophysiological and behavioral changes in non-target wildlife exposed to organophosphate and carbamate pesticides: thermoregulation, food consumption, and reproduction. Am Zool 37:369–388
Gül A, Benli ÇK, Ayhan A, Memmi BK, Selvi M, Sepici-Dinçel A, Çakiroğullari G, Erkoç F (2012) Sublethal propoxur toxicity to juvenile common carp (Cyprinus carpio L., 1758): biochemical, hematological, histopathological, and genotoxicity effects. Environ Chem Toxicol 31(9):2085–2092
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferases, the first enzimatic step in mercapturic acid formation. J Biol Chem 249:7130–7139
Jiang Y, Swale D, Carlier PR, Hartsel JA, Ma M, Ekström F, Bloomquist JR (2013) Evaluation of novel carbamate insecticides for neurotoxicity to non-target species. Pestic Biochem Physiol 106:156–161
Loreto F, Velikova V (2001) Isoprene produced by leaves protects the photosynthetic apparatus against ozone damage, quences ozone products, and reduces lipid peroxidation of cellular membranes. Plant Physiol 127:1781–1787
Loro VL, Murussi C, Menezes C, Leitemperger J, Severo E, Guerra L, Costa M, Perazzo XG, Zanella R (2015) Spatial and temporal biomarkers responses of Astyanax jacuhiensis (Cope, 1894) (Characiformes: Characidae) from the middle rio Uruguai, Brazil. Neotrop Ichthyol 13:569–578
Maran E, Fernández-Franzón M, Font G, Ruiz MJ (2010) Effects of aldicarb and propoxur on cytotoxicity and lipid peroxidation in CHO-K1 cells. Food Chem Toxicol 48:1592–1596
Menéndez-Helman RJ, Ferreyroa GV, dos Santos Afonso M, Salibián A (2012) Glyphosate as an acetylcholinesterase inhibitor in Cnesterodon decemmaculatus. Bull Environ Contam Toxicol 88:6–9
Moura FR, Brentegani KR, Gemelli A, Sinhorin AP, Sinhorin VDG (2017) Oxidative stress in the hybrid fish jundiara (Leiarius marmoratus × Pseudoplatystoma reticulatum) exposed to Roundup Original®. Chemosphere 185:445–451
Ostrea EM Jr, Reyes A, Villanueva-Uy E, Pacifico R, Benitez B, Ramos E, Bernardo RC, Bielawski DM, Delaney-Black V, Chiodo L, Janisse JJ, Ager JW (2012) Fetal exposure to propoxur and abnormal child neurodevelopment at 2 years of age. Neurotoxicology 33(4):669–675
Pimentel (1995) Amounts of pesticides reaching target pests: environmental impacts and ethics. J Agric Environ Ethics 8(1):17–29
Rodrigues AO, Souza LC, Rocha CCS, Costa ACG, Mendes RA (2017) Assessment of DDT and metabolites in soil and sediment of potentially contaminated areas of Belem, Amazon Region, Brazil. Bull Environ Contam Toxicol 99:125–130
Sabin GP, Prestes OD, Adaime MB, Zanella R (2009) Multiresidue determination of pesticides in drinking water by gas chromatography-mass spectrometry after solid-phase extraction. J Braz Chem Soc 20:918–925
Samanta P, Pal S, Mukherjee AK, Ghosh AR (2014) Biochemical effects of glyphosate based herbicide, ExcelMera71 on enzyme activities of acetylcholinesterase (AChE), lipid peroxidation (LPO), catalase (CAT), glutathione-S-transferase (GST) and protein content on teleostean fishes. Ecotoxicol Environ Saf 107:120–125
Schmuck G, Mihail F (2004) Effects of the carbamates fenoxycarb, propamocarb and propoxur on energy supply, glucose utilization and SH-groups in neurons. Arch Toxicol 78:330–337
Trachootham D, Lu W, Ogasawara MA, Valle NR, Huang P (2008) Comprehensive invited review redox regulation of cell survival. Antioxid Redox Signal 10(8):1343–1374
Tsitsimpikou C, Tzatzarakis M, Fragkiadaki P, Kovatsi L, Stivaktakis P, Kalogeraki A, Kouretas D, Tsatsakis AM (2013) Histopathological lesions, oxidative stress and genotoxic effects in liver and kidneys following long term exposure of rabbits to diazinon and propoxur. Toxicology 10:109–114
Wang C, Lu G, Cui J, Wang P (2009) Sublethal effects of pesticide mixtures on selected biomarkers of Carassius auratus. Environ Toxicol Pharmacol 28:414–419
World Health Organization - WHO (2009) The WHO recommended classification of pesticides by hazard and guidelines to classification. WHO, Stuttgart
Yan LJ, Traber MG, Packer L (1995) Spectrophotometric method for determination of carbonyls in oxidatively modified apolipoprotein B of human low-density lipoproteins. Anal Biochem 228:349–351
Acknowledgements
We would like to thank Universidade Federal de Santa Maria (UFSM) for the support and the financial support and fellowships from the Brazilian agencies CAPES (Coordination for the Improvement of Higher Education Personnel) and CNPq (National Council for Scientific and Technological Development) for research´s fellowship to Vania Lucia Loro.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gonçalves, C.R., Marins, A.T., do Amaral, A.M.B. et al. Biochemical Responses in Freshwater Fish Exposed to Insecticide Propoxur. Bull Environ Contam Toxicol 100, 524–528 (2018). https://doi.org/10.1007/s00128-018-2285-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-018-2285-9