Abstract
Cryptosporidium spp. has been recognized as an important pathogen. As bivalve mollusks are noted as potential sources of several pathogens due to their consumption as foodstuffs, the aim of this study was to investigate the occurrence of Cryptosporidium spp. oocysts in Perna perna mussels and in seawater samples from a mussel farm in Southeastern Brazil, where mussels are grown directly in the sea, attached to ropes. Oocysts were observed by microscopy and confirmed by an enzyme-linked immunosorbent assay. Oocysts were present in mussel gills and GI tracts, as well as in the seawater. Of the 100 females, 10% and 11% showed contaminated GI tracts and gills, respectively, while this rate was lower in males, at 5% and 8.9%. Oocysts were present in higher amounts in the GI tract compared to gills and water. Contamination of the study area is apparent, leading to public health risks. More in-depth studies are needed, including molecular investigations, to identify Cryptosporidium species in mussels, as well as the implementation of monitoring actions in animals destined for human consumption.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
The risk of infectious diseases resulting from the consumption of bivalve mollusks has been widely recognized for several years, both by the food industry and public health agencies. Many microorganisms are considered as being public health risks, such as pathogenic bacteria (Salmonella spp., Shigella spp., Campylobacter spp., and Escherichia coli, among others), protozoa (Cryptosporidium spp., Entamoeba spp.) and viruses (noroviruses, rotaviruses and the hepatitis A virus, among others) (Coelho et al. 2003; Lee et al. 2003; Lee and Younger 2002).
Cryptosporidium species, in particular, have been increasingly recognized as an important pathogen and widespread cause of enteric disease (Fayer et al. 2004; Smith and Rose 1998; Solo-Gabriele and Neumeister 1996), transmitted faeco–orally by the ingestion of sporulated oocysts, primarily by consumption of contaminated food and water (Srisuphanunt et al. 2009; Ugwoke et al. 2013). This enteric water-borne coccidian zoonotic protozoan parasite is often discharged into rivers and seas by rain and sewage treatment effluents, while its oocysts are discharged with feces and may enter surface waters through discharges of untreated or inadequately treated wastewater and surface runoff, as well as by anthropogenic and livestock fecal discharges (Da Fonseca et al. 2006; Fayer et al. 2003). Oocysts can persist for a long time in the environment (Ugwoke et al. 2013), up to 7 months in aquatic environments, and up to a year in marine environments (Jenkins et al. 2000; McOliver 2009). They are also resistant to chemical disinfection, including chlorination and most agents used in water treatments (McOliver 2009).
Among Cryptosporidium species, Cryptosporidium parvum is the main agent responsible for the zoonotic diarrhoeic illness known as cryptosporidiosis (Fayer et al. 2000). However, it has been recently recognized that other species of the Cryptosporidium genera are also capable of infecting humans, particularly immunocompromised individuals, who are at higher risk since there is no cure for the disease (Cacciò et al. 2002; Fayer et al. 1997; Pedraza-Díaz et al. 2001, 2000). In fact, the infective dose causative of illness of some Cryptosporidium species for immunocompromised individuals has been suggested as being a single oocyst (Rose 1997). Additionally, the mortality rates for this sector of the population due to infection by Cryptosporidium are extremely high, ranging between 52% and 68% (Cacciò et al. 2002). Thus, it is paramount to monitor public health risks associated to exposure to this protozoan.
Bivalve mollusks, such as clams and mussels, have been noted as potential sources of human Cryptosporidium infections (Melo et al. 2006). They are filter-feeders, able to retain and concentrate many different pathogens in their tissues, thus, being considered adequate sentinel organisms regarding water quality and contamination (Graczyk et al. 2004; Ugwoke et al. 2013).
Bivalve shellfish culture and production in sea farms for commercial purposes is a substantial contribution to the economic development of many countries. In Brazil, mariculture activities are significant, with over 479,000 tons of seafood produced per year. Over 90% of bivalve mollusk production is concentrated in Southeastern Brazil, due to geographical localization, climate and water quality, in the states of Santa Catarina, São Paulo, Rio de Janeiro and Espírito Santo (National Aquaculture Sector Overview 2011). However, bivalve aquaculture in Brazil is not conducted by large companies, but by artisanal farmers organized in associations and cooperatives, with a lack of sanitary control of bivalve growing waters being the norm (Suplicy 2008).
In this context, the aim of the present study was to investigate the occurrence of Cryptosporidium oocysts in the bivalve mollusk Perna perna collected from a mussel farm located in Angra dos Reis Bay, Rio de Janeiro, Southeastern Brazil, where mussels are grown directly in the sea, attached to ropes.
Materials and Methods
Perna perna mussels were sampled from a mariculture farm located in Tarituba, a small town in the municipality of Angra dos Reis, Southeastern Rio de Janeiro (23°03″51″, 23°05″58″; 44°35″51″ and 44°36″45″W) (Fig. 1). No sanitary treatment is available at most cities in this municipality, so untreated domestic effluents are discharged directly into the sea without prior treatment, through direct dumping into the water by homemade pipes, tunnels or holes, with no standardization regarding depth or distance from shore. Environmental monitoring programs have indicated high counts of fecal coliforms present in both drinking water and seawater samples from Tarituba (Choi et al. 2003; Skinner et al. 2016).
Samplings were conducted in January (50 females and 46 males) and July (50 females and 33 males) of 2009, in rope-grown mussel cultivations immersed directly in the sea. The samples were immediately placed on ice and taken to the Protein and Peptide Biochemistry Laboratory at the Oswaldo Cruz Foundation (FIOCRUZ). Arriving at the laboratory, the mollusks were rinsed under a gentle ultra-pure water flow to remove external detritus and the gills and gastrointestinal (GI) tracts of each animal were removed.
In addition, subsurface seawater from the sampling site was also collected (20 mL from each sampling point). The water samples were kept at 4 °C until arriving at the laboratory and then filtered and concentrated through sterile cellulose membranes at a 4 L min−1 rate through a vacuum pump. Filtering was conducted through 0.22 µm Millipore filters (47 mm diameter), with 0.45 µm pre-filters, prior to Cryptosporidim spp. determinations. Subsequently, each membrane was transferred to a Petri dish containing an elution solution. The material adsorbed onto the filters was then extracted by surface scraping with sterile plastic instruments.
Pooled mussels samples (for females, n = 10 in both January and July, and for males, 4 groups of 10 individuals and one of 6 in January, and 3 groups of 11 individuals in July) were separately placed in polypropylene tubes containing 7 mL of a 10% formaline solution, according to Ritchtie (Ritchie 1948). Four mililiters of ether were added, and the samples were then manually homogenized and subsequently centrifuged at 2500×g for 10 min (Universal 320 R, Hettich-Zentrifugen). The supernatants were discarded and the slides were prepared using the precipitated pellets.
The water samples were transferred to 15 mL polypropylene tubes and centrifuged at 1250×g for 15 min, twice, in order to concentrate the samples. The supernatants were then discarded, also using the formaline-ether technique to eliminate sample residues, facilitating oocyst detection (Ritchie 1948). The resulting precipitated pellets were then used to prepare the slides (duplicates for each sample).
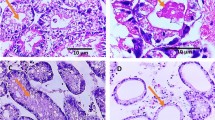
The Ziehl-Neelsen stain was used to identify Cryptosporidim spp. oocysts (Henriksen and Pohlenz 1981) in both the water and mussel samples under an optical microscope (L-2000 B-AC-CE, Bioval). This method was chosen because it the best-suited for the detection of Cryptosporidim spp. oocysts, since these structures do not stain by usual methods (Smith et al. 1989).
The diagnosis of the presence of Cryptosporidim spp. oocysts was confirmed by an enzyme-linked immunosorbent assay (ELISA), using the Microplate Assay KIT 96 TS SOB EM, Remel (OXOID, USA) (Meireles 2010). Briefly, 100 mL of the gastrointestinal tract samples were homogenized in 1 mL of a 1 mmol/L phosphate buffer. Two hundred microliters of this mixture were then placed on a 96-well microplate sensibilized with Cryptosporidim spp. antigen. After adding the appropriate reagents, the microplate was incubated for 40 min and absorbances were determined at 450 nm on an ND-1000 NanoDrop spectrophotometer. All assays (both water and mussels) were conducted in triplicate and repeated 5 times to ensure reproducibility.
Cryptosporidim spp. oocyte frequency was compared among organs and sexes by a One-way variance test (ANOVA), since data was normally distributed. Statistical significance was set at p < 0.05.
Results and Discussion
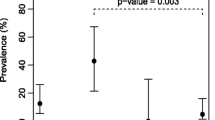
A total of 179 samples were examined between January and July 2009, and Cryptosporidium oocysts were detected by microscopy and ELISA in 33 of the samples (18.43%). The occurrence of Cryptosporidium spp. oocytes in male and female Perna perna GI tracts and gills is displayed in Table 1.
Cryptosporidium spp. was present in more gill samples compared to GI tract samples, although this difference was statistically non-significant (p > 0.05). Counts were higher in females than males for both organs, although this difference was non-significant (p > 0.05). The number of Cryptosporidium spp. oocysts observed was, however, significantly higher in the GI tract compared to both gills and water (p < 0.05). Counts in gills and water were similar (p > 0.05).
Water bodies can show Cryptosporidium oocyst contamination due to the presence of both anthropogenic and livestock fecal discharges (Srisuphanunt et al. 2009). Water contamination is then passed on to bivalves, since these organisms are filter feeders, making these organisms a direct transmission route for protozoan infections to other organisms, including humans (Melo et al. 2006). This was corroborated in the present study, since oocyst counts in water were similar to oocysts counts in gills, indicating direct uptake of oocysts from the water, also confirming sources of contamination in the mussel farming area.
Bivalve aquaculture in Brazil shows lack of control of harvesting areas and the lack of traceability of the products. The mollusks are cooked and handled in unsuitable conditions, generally leading to severe risks for consumers (Suplicy 2008). Corroborating this, the observed contamination rates in the present study were considerably high from a public health perspective (Srisuphanunt et al. 2009), similar to other reports on Cryptosporidium oocysts in marine bivalve mollusks (Giangaspero et al. 2005; Gomez-Bautista et al. 2000; Lowery et al. 2001). This indicates high public health risks, since, in addition to high contamination rates, multiple organ contamination was also observed, corroborating previous reports in the literature (Fayer et al. 1997; Ugwoke et al. 2013) and indicating greater risks, since cryptosporidial oocyst clearance time in mussels has been shown as being of at least 1 month, perhaps more (Fayer et al. 1997). As mussels are usually ingested whole, this increases contamination risks for both biota and humans. In addition, Cryptosporidium. oocysts can survive in seawater for long periods (Fayer et al. 1998) at relatively low temperatures (Freire-Santos et al. 2000) and still be infective.
Cryptosporidiosis epidemics caused by ingestion of different contaminated foodstuffs and water have been previously reported, and this type of contamination has, increasingly, gained attention (Girdwood and Smith 1999; Griffiths 1998; Robertson and Gjerde 2000; Srisuphanunt et al. 2009), since several Cryptosporidium species and genotypes are known to cause serious opportunistic infections in immunocompromised individuals (Melo et al. 2006) and susceptible populations such as newborn infants, the elderly, patients on immunosuppressive drugs and people infected with HIV are at higher risks of infection (Ugwoke et al. 2013). Thus, the occurrence of Cryptosporidium oocysts in seafood represents an important public health concern (Srisuphanunt et al. 2009).
Cryptosporidium oocysts have been reported worldwide in mussels, in different European countries (Gomez-Bautista et al. 2000; Gómez-Couso et al. 2003; Li et al. 2006; Lowery et al. 2001; MacRae et al. 2005), the United States (Graczyk et al. 1999), Thailand (Srisuphanunt et al. 2009), and Canada (Graczyk et al. 2001). The first study on the occurrence of contamination by Cryptosporidium oocysts in shellfish from the coastal region of Brazil and, indeed, Latin America, was reported by Leal (Leal et al. 2008), in oysters (Crassostrea rhizophorae) and cockles (Tivela mactroides). In Brazil, however, only two reports are available regarding the presence of this protozoa specifically in mussels, both in Perna perna from Mangaratiba Bay, also located in Rio de Janeiro (Cardoso et al. 2012; Oliveira et al. 2014), at a distance of 110 km to the sampling site of the present study. Even though those sampling sites evaluated in the aforementioned literature and the one investigated herein are relatively far from each other, it seems that the entire area surrounding these mussel farms shows contamination by Cryptosporidium spp.
The lack of reports of human cryptosporidiosis cases resulting from the specific consumption of bivalve mollusks has been postulated as being due to the long incubation period of 7–10 days before symptoms appear in humans, making it difficult to associate infection with a particular food source, thus under-diagnosing or under-reporting cryptosporidiosis cases (Graczyk and Schwab 2000). However, increasing studies on the presence of Cryptosporidium oocysts in bivalve mussels are being undertaken (Gomez-Bautista et al. 2000; Schets et al. 2007), indicating rising concerns in this regard.
Cryptosporidium spp. oocytes were observed by both microscopy and confirmed by ELISA in mussels and water from a mussel aquaculture farm in Southeastern Brazil. The results presented herein indicate that Perna perna mussels are adequate bioindicators for the presence of protozoan contamination by Cryptosporidium spp. The presence of Cryptosporidium spp. contamination in these organisms and in the water sampled from the mussel farm suggests contamination of the area due to sewage treatment effluents and/or anthropogenic and livestock fecal discharges, leading to public health risks regarding the ingestion of these animals. More in-depth studies are needed, including molecular investigations, to verify the presence and identify the Cryptosporidium species in Brazilian mussels, as well as the implementation of routine pathogen-monitoring in animals destined for human consumption.
Change history
14 February 2018
The original version of this article unfortunately contained a mistake.
References
Cacciò S, Pinter E, Fantini R, Mezzaroma I, Pozio E (2002) Human infection with Cryptosporidium felis: case report and literature review. Emerg Infect Dis 8:85–86
Cardoso TSF, Santos IRS, Berto BP, Cardoso SV, Mesquita EFM, Lopes CWG (2012) Correlation between biometric and parasitologic data of mussels (Perna perna) from Ponta do Tinguí, Mangaratiba, RJ. Brazil Revista Brasileira de Medicina Veterinária 34:9–13
Choi N, Kfouri C, Kweon HJ, You EC, Chagnon FJF, Harleman DRF (2003) Potable water and wastewater infrastructure planning for the city of Paraty, Brazil Global Water at MIT 264
Coelho C, Heinert AP, Simões CM, Barardi CR (2003) Hepatitis A virus detection in oysters (Crassostrea gigas) in Santa Catarina State, Brazil, by reverse transcription-polymerase chain reaction. J Food Prot 66:507–511
Da Fonseca IP, Ramos PS, Ruano FA, Duarte AP, Costa JC, Almeida AC, Falca ML, Fazendeiro MI (2006) Efficacy of commercial cleansing procedures in eliminating Cryptosporidium parvum oocysts from bivalves. J Eukaryot Microbiol 53:S49-S51
Fayer R, Speer CA, Dubey JP (1997) The general biology of Cryptosporidium. Cryptosporidium and Cryptosporidiosis. FL CRC Press, Boca Raton, pp 1–42
Fayer R, Graczyk TK, Lewis EJ, Trout JM, Farley CA (1998) Survival of infectious Cryptosporidium parvum oocysts in seawater and eastern oysters (Crassostrea virginica) in the Chesapeake Bay. Appl Environ Microbiol 64:1070–1074
Fayer R, Morgan U, Upton SJ (2000) Epidemiology of Cryptosporidium: transmission, detection and identification International. J Parasitol 30:1305–1322
Fayer R, Trout JM, Lewis EJ, Santin M, Zhou L, Lal AA, Xiao L (2003) Contamination of Atlantic coast commercial shell-fish with Cryptosporidium. Parasitol Res 89:141–145
Fayer R, Dubey JP, Lindsay DS (2004) Zoonotic protozoa: from land to sea. Trends Parasitol 20:531–536
Freire-Santos F, Oteiza-López AM, Vergara-Castiblanco CA, Ares-Mazás ME (2000) Study of the combined influence of environmental factors on viability of Cryptosporidium parvum oocysts in water evaluated by fluorogenic vital dyes and excystation techniques. Vet Parasitol 89:253–259
Girdwood RWA, Smith HV (1999) Cryptosporidium. In: Encyclopaedia of food microbiology. London Academic Press, pp 487–497
Giangaspero A, Molini U, Iorio R, Traversa D, Paoletti B, Giansante C (2005) Cryptosporidium parvum oocysts in sea-water clams (Chameleagallina) in Italy. Prev Vet Med 69:203–312
Gomez-Bautista M, Ortega-Mora LM, Tabares E, Lopez-Rodas V, Costas E (2000) Detection of infectious Cryptosporidium parvum oocysts in mussels (Mytilus galloprovincialis) and cockles (Cerastoderma edule). Appl Environ Microbiol 66:1866–1870
Gómez-Couso H, Freire-Santos F, Martínez-Urtaza J, Ares-Mazás ME (2003) Contamination of bivalve molluscs by Cryptosporidium oocysts: the need for new quality control standards. Int J Food Microbiol 87:97–105
Graczyk TK, Schwab KJ (2000) Food-borne infections vectored by molluscan shellfish. Curr Gastroenterol Rep 2:305–309
Graczyk TK, Fayer R, Lewis EJ, Trout JM, Farley CA (1999) Cryptosporidium oocysts in Bent mussels (Ischadium recurvum) in the Chesapeake Bay. Parasitol Res 85:518–521
Graczyk TK, Marcogliese DJ, Delafontaine Y, Dasilva AJ, Mhagami-Ruwende B, Pieniazek NJ (2001) Cryptosporidium parvum oocysts in zebra mussels (Dreissena polymorpha): evidence from the St Lawrence River. Parasitol Res 87:231–234
Graczyk TK, Conn DB, Lucy F, Minchin D, Tamang L, Moura L, Da Silva A (2004) Human waterborne parasites in zebra mussels (Dreissena polymorpha) from the Shannon river drainage area, Ireland. Parasitol Res 93:385–391
Griffiths JK (1998) Human cryptosporidiosis: epidemiology, transmission, treatment and diagnosis. Adv Parasitol 40:37–85
Henriksen SA, Pohlenz JF (1981) Staining of Cryptosporidia by a modified Ziehl-Neelsen technique. Acta Vet Scand 22:594–596
Jenkins MC, Trout JM, Abrahamsen MS, Lancto CA, Higgins J, Fayer R (2000) Estimating viability of Cryptosporidium parvum oocysts using reverse transcriptase-polymerase chain reaction (RT-PCR) directed at mRNA encoding amyloglucosidase. J Microbiol Methods 43:97–106
Leal DAG, Pereira MA, Franco RMB, Branco N, Cantusio-Neto R (2008) First report of Cryptosporidium spp. oocysts in oysters (Crassostrea rhizophorae) and cockles (Tivela mactroides) in Brazil. Braz J Water Health 6:527–532
Lee JR, Younger AD (2002) Developing microbiological risk assessment for shellfish purification. Int Biodeterior Biodegrad 50:177–183
Lee CY, Panicker G, Bej AK (2003) Detection of pathogenic bacteria in shellfish using multiplex PCR followed by CovaLinkTM NH microwell plate sandwich hybridization. J Microbiol Methods 53:199–209
Li X, Guyot K, Dei-Cas E, Mallard JP, Ballet JJ, Brasseur P (2006) Cryptosporidium oocysts in mussels (Mytilus edulis) from Normandy (France). Int J Food Microbiol 108:321–325
Lowery CJ, Nugent P, Moore JE, Millar BC, Xiru X, Dooley JS (2001) PCR-IMS detection and molecular typing of Cryptosporidium parvum recovered from a recreational river source and an associated mussel (Mytilus edulis) bed in Northern Ireland. Epidemiol Infect 127:545–553
MacRae M, Hamilton C, Strachan NJC, Wright S, Ogden ID (2005) The detection of Cryptosporidium parvum and Escherichia coli O157 in UK bivalve shellfish. J Microbiol Methods 60:395–401
McOliver CC (2009) Recreational water contact as a risk factor for Cryptosporidium exposure among persons with HIV/AIDS in Baltimore, MD. The Johns Hopkins University, Baltimore, p 234
Meireles MV (2010) Cryptosporidium infection in Brazil: implications for veterinary medicine and public health Revista. Brasileira de Parasitologia Veterinária 19:197–204
Melo PC, Teodisio J, Reis J, Duarte A, Costa JC, Fonseca IP (2006) Cryptosporidium spp. in freshwater bivalves in Portugal. J Eukaryot Microbiol 53:S28-S29
National Aquaculture Sector Overview (2004) Brazil. National Aquaculture Sector Overview Fact Sheets. Text by Suplicy, F.M. In: FAO Fisheries and Aquaculture Department [online]. Rome
Oliveira GFM, Lima MF, Bomfim TCB (2014) Perna perna mussels as bioindicators of aquatic contamination by Cryptosporidium spp. Acta Scientiae Veterinariae 42:1–6
Pedraza-Díaz S, Amar C, McLauchlin J (2000) The identification and characterization of an unusual genotype of Cryptosporidium from human faeces as Cryptosporidium meleagridis. FEMS Microbiol Lett 189:189–194
Pedraza-Díaz S, Amar C, Iversen AM, Stanley PJ, McLauchlin J (2001) Unusual Cryptosporidium species recovered from human faeces: first description of Cryptosporidium felis and Cryptosporidium ‘‘dog type’’ from patients in England. J Med Microbiol 50:293–296
Ritchie LS (1948) An ether sedimentation technique for routine stool examination. Bull US Army Med Dep 8:326–334
Robertson LJ, Gjerde B (2000) Isolation and enumeration of Giardia cysts, Cryptosporidium oocysts, and Ascaris eggs from fruits and vegetables. J Food Prot 63:775–877
Rose JB (1997) Environmental ecology of Cryptosporidium and public health implications. Annu Rev Public Health 18:135–161
Schets FM, Harold HJM, Berg VD, Engels GB, Lodder WJ, Husman AMR (2007) Cryptosporidium and Giardia in commercial and non-commercial oysters (Crassostrea gigas) and water from the Oosterschelde, The Netherlands. Int J Food Microbiol 113:189–194
Skinner LF, Barboza DF, Rocha RM (2016) Rapid assessment survey of introduced ascidians in a region with many marinas in the Southwest Atlantic Ocean, Brazil. Manag Biol Invasions 7:13–20
Smith HV, Rose JB (1998) Waterborne cryptosporidiosis: current status. Parasitol Today 14:14–22
Smith HV, McDiarmid A, Smith AL, Hinson AR, Gilmour RA (1989) An analysis of staining methods for the detection of Cryptosporidium spp. oocysts in water-related samples. Parasitol Res 3:323–327
Solo-Gabriele H, Neumeister M (1996) U.S. outbreaks of Cryptosporidiosis. J Am Water Works Assoc 88:76–86
Srisuphanunt M, Saksirisampant W, Karanis P (2009) Detection of cryptosporidium oocysts in green mussels (Perna viridis) from shell-fish markets of Thailand. Parasite 16:235–239
Suplicy FM (2008) Legal aspects and governmental actions for the development of mollusc farming in Brazil. In: FAO Actas de Pesca y Acuicultura, Rome FAO, pp 205–208
Ugwoke EV, Umoh JU, Okolocha EC, Lawai IA (2013) Cryptosporidium oocysts in Anodonta sp. (bivalve mollusc) as indicators of pollution of Tiga Lake ecosystem in Kano State, Nigeria. J Parasitol Vector Biol 5:77–82
Author information
Authors and Affiliations
Corresponding author
Additional information
A correction to this article is available online at https://doi.org/10.1007/s00128-018-2284-x.
Rights and permissions
About this article
Cite this article
Santos, A.L., de Oliveira, L.T.F., Souza, A.L.A. et al. Cryptosporidium spp. Contamination in Perna perna Mussels Destined for Human Consumption in Southeastern Rio de Janeiro, Brazil. Bull Environ Contam Toxicol 100, 240–244 (2018). https://doi.org/10.1007/s00128-017-2223-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-017-2223-2