Abstract
The distribution of butyltins (BTs) and polycyclic aromatic hydrocarbons (PAHs) were studied in surface sediments to assess how relevant is Mar del Plata port (Argentina) as a source of contamination to the surrounding environments. Within the port, TBT concentrations ranged from 24.2 to 150 ng Sn g−1 and PAHs (Σ16) from 180 to 17,094 ng g−1. At the surrounding beaches, PAHs were detected at low concentrations and TBT concentrations reached 10.9 ng Sn g−1. Although those low levels indicate that the Port might not be an important source of contamination to the surrounding beaches, the very low TOC content and the coarse grain size of the beaches sediments could explain the sedimentary levels. The results show a reduction in TBT levels in Mar del Plata port after national and international use restrictions of TBT-based antifouling paints.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Shipping ports often represent significant anthropogenic sources of contamination for coastal environments, contributing to inputs of polycyclic aromatic hydrocarbons (PAHs), metals, antifouling biocides, and plastic debris (Darbra et al. 2005). Ports are often situated in estuaries, bays or artificial enclosed systems where circulation normally is limited and, thus, sediments may contain high concentrations of contaminants (Chen and Chen 2011). As a consequence benthic communities from local and surrounding areas can be degraded (Martínez-Lladó et al. 2007). Sandy beaches shelter a great biodiversity and are ecologically relevant to the whole ecosystem (Schlacher et al. 2008). However, despite butyltin compounds from antifouling paints and PAHs are concerning contaminants (Delucchi et al. 2011; Ünlü et al. 2013) they have been less studied in sandy beaches than trace-elements (Nagarajan et al. 2013) and oil spills (Veiga et al. 2010).
Tributyltin (TBT) has been used since the 1960s as an antifouling biocide paint in the maritime industry. Due to its high environmental persistence, lipophilicity and toxicity, the International Marine Organization (IMO) banned the use of TBT-based antifouling paints in September 2008. PAHs are organic contaminants that reach the aquatic environment following the combustion of organic matter, petroleum spills or natural and diagenetic processes (Srogi 2007). Their carcinogenic and mutagenic properties (Nisbet Ian and LaGoy 1992) justifies their inclusion in priority pollutant lists (i.e. European Union and US EPA).
Mar del Plata port (MDPP) holds about 60 % of the Argentinian fishing fleet and it has one of the most important shipyard facility in Argentina. Approximately 110 ocean vessels, 330 coastal vessels and 442 international destinations vessels per year pass through the port. It is surrounded by several sandy and rocky beaches where many intertidal benthic communities can be found and human recreational activities take place. Although BTs and PAHs have been detected in surface sediments of Mar del Plata port (Goldberg et al. 2004; Cledón et al. 2006; Bigatti et al. 2009; De Waisbaum et al. 2010; Albano et al. 2013), they have never been measured in the surrounding beaches (except Popular beach for TBT). In addition, TBT contamination have not been assessed in the area since its banning in 2008. Thus, it is important to update information about BT levels in Mar del Plata port in order to verify the effectiveness of local and international regulations since this area was identified as a hot spot of TBT contamination (Castro et al. 2012a). Further, fresh inputs of TBT have been recently identified in different areas of South America (i.e. Paz-Villarraga et al. 2015).
In this context, the present study evaluated the spatial distribution of BTs and PAHs in areas under the influence of Mar del Plata port in order to assess whether the port represents a significant source of contamination to the surrounding intertidal environments. Considering the predominant coastal sand drift direction in the area (south-north), the hypothesis of a contamination gradient from Mar del Plata port towards northern tourist beaches was appraised.
Materials and Methods
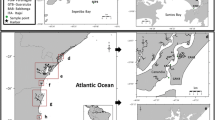
Surface sediments (upper 5 cm) were collected using a stainless steel Van-Veen grab at nine sites during December 2012. Five of these sites were sampled inside Mar del Plata port (S1–S5), and three in northern locations (Popular (P), Alfonsina (A) and Constitución C)) and one in southern beach (Punta Mogotes (PM)) (Fig. 1). Since sand predominated in beach sediments, nine sediment samples were taken randomly at each site, then mixed and sieved in situ using a 120 µm mesh to make two single composite samples (100–200 g wet weight). In the laboratory, sediments were oven dried (45°C) until constant weight and then stored at −20°C for subsequent analysis. The concentration of total organic carbon (%TOC) were determined by an elemental analyzer (CHNS Perkin Elmer 2400 Series II) after decarbonation in a desiccator containing HCl (37 %).
Butyltin levels were analyzed according to Castro et al. (2012b). Briefly, 5 g of dried sediments were extracted by sonification in tropolone solution (0.05 % w/v) in methanol. The extracts were then derivatized using 2 mL of n-pentyl magnesium bromide in diethyl ether solution (2 M), according to Morabito et al. (2000), and again extracted with 3 × 5 mL of hexane. A cleanup was then performed using a silica-gel column (3.5 g) eluted with 15 mL of hexane/toluene (1:1). Finally, 100 ng of tetrabutyltin solution was added (internal standard). Extracts were analyzed by gas chromatography using a Perkin Elmer Clarus 500MS equipped with mass spectrometer detector. The analytical curves were made using the matrix addition technique and quantification limits (LQ) were 2, 3 and 3 ng Sn g−1 for TBT, DBT and MBT, respectively. The quality assurance and quality control was based on regular analyses of blanks, spiked matrices and certified reference material (PACS-2/National Research Council of Canada, Ottawa, Canada). Results obtained for the PACS-2 (TBT – 852 ± 47 ng Sn g−1; DBT – 1035 ± 35 ng Sn g−1 and MBT – 557 ± 38 ng Sn g−1) were in good agreement with the certified (TBT – 890 ± 105 ng Sn g−1 and DBT – 1047 ± 64 ng Sn g−1) and reported values (MBT – 600 ng Sn g−1). The samples recoveries were between 79 % and 98 % and RSD (relative standard deviation) below 20 % (IUPAC 2002). In order to assess the degree of TBT degradation and to predict if the contamination in coastal areas of Mar del Plata is recent or not, the butyltin degradation index (BDI) was calculated based on the following equation: Butyltin Degradation Index (BDI) = [MBT] + [DBT]/[TBT] (Díez et al. 2006). Old TBT inputs are normally associated with values of BDI > 1.
The analytical procedure is described in Niencheski and Fillmann (2006). An exact mass of dried sediments (~15 g) was spiked with p-Terphenyl-d14 a surrogate standard and then Soxhlet-extracted with n-hexane/dichloromethane (1:1) for 12 h. The extract was concentrated down to 1 mL using a Büchi Syncore® Polyvap R-24 system parallel evaporator and activated copper was added to remove any sulfur. Then, the extract was fractionated into aliphatic and polyaromatic hydrocarbons by liquid chromatography adsorption using a column of silica gel (6 g) and neutral aluminum oxide (8 g). The identification was based on the retention time and mass spectra. Analyte quantification was performed comparing the sample results with standard curves built using deuteron naphthalene-d8, acenaphthylene-d10, phenanthrene-d10, chrysene-d12 and perylene-d12 for PAH (Sigma Aldrich, São Paulo, SP, Brazil). The extracts were analyzed using a gas chromatograph equipped with a mass spectrophotometry detector (Perkin Elmer® Clarus 500 – GC–MS). All chemicals used were pesticide residue grade and the LQ was 0.125 ng L−1. The sample recoveries varied between 82 % and 97 %.
Results and Discussion
Sediment total organic carbon contents ranged from 0.06 % (Popular and Punta Mogotes beaches) to 3.31 % (S5 – Port) (Table 1). Levels of organic carbon were always higher in port than beach sediment, which is consistent with the smaller grain size and the low hydrodynamics of the port due to the wave breakers (Rivero et al. 2005). Butyltin residues were detected at all samples ranging from <2 to 150.4 ng Sn g−1 for TBT < 3–11.8 ng Sn g−1 for DBT and <3–258.8 ng Sn g−1 for MBT (Table 1). The high levels of BTs inside MDPP indicate that this zone could be a source of TBT to the surrounding areas. BDI > 1 (Table 1) indicates chronic contamination with BTs in MDPP. However, the high levels of TBT (mainly at S3, S4 and S5) suggest that fresh inputs are also taking place inside the Port.
Total organic carbon (TOC) were significantly correlated with TBT (r 2 = 0.76; p < 0.01), DBT (r 2 = 0.76; p < 0.01) and MBT (r 2 = 0.58; p < 0.05) concentrations. Although some studies have suggested that TOC binds BTs (i.e. Pinochet et al. 2009), a straightforward relationship is not always seen (i.e. Shim et al. 1999; Paz-Villarraga et al. 2015) since other factors such as BT sources and characteristics, organic matter composition, and biological activity of the sediment (Town and Filella 2002) also plays a role in this association.
In 2001, the highest TBT concentration found in South America was reported for MDPP (6500 ng Sn g−1, Goldberg et al. 2004). Later, in 2002, Cledón et al. (2006) reported 2.45 ng Sn g−1, whereas in 2006 Bigatti et al. (2009) measured 728.3 ng Sn g−1 and in 2007 De Waisbaum et al. (2010) found 700 ng Sn g−1. Thus, a clear decreasing trend of TBT contamination is evident for Mar del Plata Port since present levels were much lower (<2–150 ng Sn g−1). Several recent studies have reported a decrease in BTs levels in ports, marinas and shipyards around the world (Castro et al. 2012c) after the global banning on the use of TBT-based antifouling paints in 2008 (Choi et al. 2009; Sousa et al. 2009). Moreover, a reduction of imposex ratios in gastropods, which indicates a reduction of TBT contamination since it is a specific biomarker, has been also observed (Galante-Oliveira et al. 2011; Castro et al. 2012c). In the case of Mar del Plata, since the TBT levels reduction was observed before the global 2008 banning, it is clear that the national legislation of 1998 (DPMA 1998) has also contributed to the reduction. The importance of national legislations has also been addresses by Undap et al. (2013) who observed a decrease in TBT levels in Japan, a country with national TBT regulation, and an increase in Indonesia, without such restriction, despite both being members of IMO.
According to Waite et al. (1991), TBT concentrations ranging between 10 and 50, 60 and 200 and 300 and 1000 ng Sn g−1 indicate sites under low, moderate and high TBT contamination, respectively. Therefore, MDPP sediments can be considered as low to moderately contaminated by TBT. However, the current TBT concentrations observed in sediments of Mar del Plata are in the same range as those reported for harbors from Brazil (Oliveira et al. 2010), Ecuador (Castro et al. 2012b), and two other Argentinian ports (Bigatti et al. 2014). Thus, the present TBT levels for MDPP are still high and represent a risk to marine life. On a global scale, TBT levels in sediments of MDPP are lower than those found in ports from countries without national restrictions on the use of TBT-based antifouling paints (Undap et al. 2013) or some major harbors (Kim et al. 2015), but still comparable to levels considered as unsafe to biota (Okoro et al. 2013; Kim et al. 2014) (Table 2).
Butyltin residues were detected in all sites outside MDPP (Popular, Alfonsina, Constitución, and Punta Mogotes), although only Alfonsina was above the quantification limit (Table 1). However, the influence from the Port contamination inputs (maybe sediment remobilization) cannot be ruled out. Some BTs might be associated to the circulating suspended matter and cannot reach the sediments due to local dynamics. Felizzola et al. (2008) suggested that the current driven dispersion in the water phase prior to sedimentation would explain low BTs values in areas of Todos os Santos Bay far away from major sources. However, degradation and loss processes of sedimentary BTs may occur along the way from the port to the beaches. In the present study, the very low TOC content and the coarse grain size of the beaches sediments could explain the low BTs sedimentary levels. Nonetheless, another source of BTs at Alfonsina Beach cannot be excluded. The predominance of TBT at this site in comparison to other beaches could be an indication of punctual sources. Even though maritime activities are the principal sources, BTs can also be released from industrial activity (Hoch 2001), urban runoff (18.4 ng Sn g−1 of TBT in sediments; David et al. 2012) and sewage sludge (175 ng Sn L−1 of TBT; Fent and Müller 1991). There are two pluvial effluents near Alfonsina beach that could be sources of TBT. Even though, sediments in the surrounding areas of MDPP could be considered as non to low contaminated, according to Waite et al. (1991). Although BTs levels in marine intertidal sediments are poorly explored, some authors have detected values similar to those found here at beaches under the influence of port/harbor (Table 2). However, the Environmental Quality Guidelines (EQG) proposed by the Dutch RIKZ (National Institute for Coastal and Marine Management) establish the Maximum Permissible Concentrations (MPC: above which the possible adverse effects are considered unacceptable) and the Negligible Concentrations (NC: rarely associated with adverse effects) for TBT as 1.9 and 0.019 ng Sn g−1 d.w., respectively (Crommentuijn et al. 2000). Therefore, although the levels of TBT found in Alfonsina beach could be considered as low, based on this EQG it cannot be considered safe.
PAHs were found at all sites, which is consistent with the ubiquity of these compounds. As expected, PAHs concentrations were much higher in the port than in the beach sites. PAHs levels (Σ16 priority PAHs recognized by the USEPA) ranged from 179.6 to 17,093.7 ng g−1 in MDPP sediments whereas in the beaches they varied between 4.9 and 17.3 ng g−1 (Table 3). Concentrations of PAHs within Mar del Plata port were significantly higher than those found by Albano et al. (2013) in 2009 (Σ16PAHs: 25.2–684.7 ng g−1), which suggests either a recent increase in hydrocarbon contamination or some sediment spatial variability within the port. According to the classification adapted by Baumard et al. (1998), MDPP sediments can be considered as moderate (S1) to very highly contaminated (other sediments), whereas beach sediments can be considered as low contaminated by PAHs. No significant correlation was found between sediment organic carbon and PAHs levels at any site (r 2 = 0.32; p > 0.05). Thus, PAHs distribution is more likely to be associated to sources of contamination than sediment characteristics (Commendatore et al. 2012).
The main sources of hydrocarbons (biogenic, petrogenic, pyrogenic and diagenetic) can be distinguished by analyzing multiple parameters including parental and alkyl-substituted PAHs (Simoneit 2005) and some of their diagnostic ratios (Readman et al. 2002; Yunker et al. 2002). The following four PAH ratios were used to identify possible sources of PAH in sediments: benzo(a)anthracene/(benzo(a)-anthracene + chrysene) – BaA/228; fluoranthene/(fluoranthene + pyrene) – Fl/(Fl + Py); antracene/(antracene + phenanthrene) – Ant/(Ant + Phe); and indene(1,2,3-c,d)pyrene/(indene(1,2,3-c,d)pyrene + benzo(g,h,i)perylene) – IP/(IP + BghiP). According to Yunker et al. (2002), the ranges of PAH ratios for different sources are: (a) Fl/(Fl + Py) ratio < 0.40 indicates dominance of petroleum, 0.40–0.50 indicates petroleum combustion, and >0.50 combustion of coal, grasses and wood; (b) BaA/228 ratio < 0.20 indicates petroleum, 0.20–0.35 petroleum and combustion, and >0.35 combustion; and, (c) IP/(IP + BghiP) < 0.20 indicates petroleum, 0.20–0.50 petroleum combustion, and >0.50 combustion of coal, grasses and wood. Ratios were only calculated for samples where the PAH concentrations exceeded 10 ng g−1 since concentrations approaching detection limits could produce misinterpretation (Martins et al. 2010). The present ratios indicate a mixture of pyrolitic and petrolitic sources for sites S2, S3 and S5. In sites S1, S4 and at the beaches a pyrolytic source predominates. High molecular weight PAHs predominated indicating pyrogenic sources in all sites (Table 3), what was also seen by Albano et al. (2013) for some MDPP sediments.
Putting the PAH contamination in perspective, concentrations found in the present study were higher than other ports of Argentina, Brazil and Chile (Table 4). On the other hand, they are comparable with Guanabara Bay in Brazil (Silva et al. 2007), Naples Harbor, one of the largest and most important commercial port of the Mediterranean (Sprovieri et al. 2007), Kaohsiung Harbor, the largest international port in Taiwan (Chen and Chen 2011) and Alexandria Eastern Harbor (Barakat et al. 2011), one of the two harbors forming Alexandria Port, considered the main Egyptian port (Table 4).
The PAH concentrations measured in this study were compared to the threshold effect concentrations (TELs) specified by the National Oceanic and Atmospheric Administration (U.S. NOAA) sediment quality guidelines under the National Status & Trends Program (NS&T) as indicators of biological effects (MacDonald 1994). The stations S2 and S5 presented concentrations above the PEL – Probable Effect Level (Σ PAH < 7110 ng g−1), above which effects on organisms are frequently expected. Stations S3 and S4 showed levels above TEL – Threshold Effect Level (Σ PAH > 468 ng g−1) indicating possible effects. Finally, S1 and beaches presented concentrations below TEL, where effects on organisms are rarely expected (Table 3).
The remobilization of sediments in port areas may eventually transport PAHs to adjacent areas, especially during dredging events. This process could represent a risk to the marine community (benthonic and pelagic) and potentially to people when these sediments are highly contaminated. In 1998, the Popular beach was filled with 1.670,000 m3 of dredged sediments from MDPP and, consequently, high TBT concentrations were found there (240 ng Sn g−1 in 2001 – Goldberg et al. 2004, and 175 ng Sn g−1 in 2007 – de Waisbaum et al. 2010). The results of the present study indicate that 14 years after these activities much of the contaminants have dissipated with very low BT and PAH contamination still present at Popular and adjacent beaches.
In conclusion, MDPP sediments can be considered as low to moderately contaminated by TBT, whereas nearby beaches are non to low contaminated. A clear reduction in MDPP contamination over the last few years suggests that local and international laws controlling TBT-based antifouling paints are being effective. On contrary, PAHs levels were very high implying a hazard to invertebrates living in MDPP. A contamination gradient in beaches sediments was not detected either for BTs or PAHs and the levels of these compounds were low and below the guidance levels (except for TBT at Alfonsina beach). The low PAH and TBT levels at beach sediments indicate that either MDPP in not acting as an important source of contamination to the surrounding environments or contaminants are being degraded on the way or kept on the water column and transported further away. Thus, it is important to study other compartments (water and suspended matter) to see whether remobilization and transport of MDPP sedimentary contaminants is indeed an issue to be considered or not.
References
Albano MJ, Lana PC, Bremec C, Elías R, Martins CC, Venturini N, Muniz P, Rivero S, Vallarino EA, Obenat S (2013) Macrobenthos and multi-molecular markers as indicators of environmental contamination in a South American port (Mar del Plata, Southwest Atlantic). Mar Pollut Bull 73(1):102–114
Barakat AO, Mostafa A, Wade TL, Sweet ST, El Sayed NB (2011) Distribution and characteristics of PAHs in sediments from the Mediterranean coastal environment of Egypt. Mar Pollut Bull 62:1969–1978
Baumard P, Budzinski H, Garrigues P (1998) Polycyclic aromatic hydrocarbons in sediments and mussels of the Western Mediterranean Sea. Env Toxicol Chem 7:765–776
Bigatti GI, Primost MA, Cledón M, Averbuj A, Theobald N, Gerwinski W, Arntz W, Morriconi E, Penchaszadeh PE (2009) Contamination by TBT and imposex biomonitoring along 4700 km of Argentinean shoreline (SW Atlantic: from 38°S to 54°S). Mar Pollut Bull 58:695–701
Bigatti GI, Averbuj A, Castro IB, Primost M, Carranza A, Commendatore M, Fillmann G, Delgado E, Scarabino F, del Brío F, Franco M, Letamendía M, Martínez G, Sauco S, Penchaszadeh PE (2014) Situación de contaminación en las costas del sur de Brasil, Uruguay y Argentina, luego de la prohibición mundial de TBT. In: Goso C (ed) Nuevas miradas a la problemática de los ambientes costeros: Sur de Brasil, Uruguay y Argentina, vol 1. DIRAC, Montevideo, pp 277–301
Castro IB, Perina FC, Fillmann G (2012a) Organotin contamination in South American coastal areas. Env Monit Assess 184:1781–1799
Castro IB, Arroyo MF, Gomes Costa P, Fillmann G (2012b) Butyltin compounds and imposex levels in Ecuador. Arch Env Contam Toxicol 62:68–77
Castro IB, Rossato M, Fillmann G (2012c) Imposex reduction and residual butyltin contamination in southern Brazilian harbors. Env Toxicol Chem 31(5):947–954
Chen CW, Chen CF (2011) Distribution, origin, and potential toxicological significance of polycyclic aromatic hydrocarbons (PAHs) in sediments of Kaohsiung Harbor, Taiwan. Mar Pollut Bull 63:417–423
Choi M, Choi HG, Moon HB, Kim GY (2009) Spatial and temporal distribution of tributyltin (TBT) in seawater, sediments and bivalves from coastal areas of Korea during 2001–2005. Env Monit Assess 151:301–310
Cledón M, Theobal N, Gerwinski W, Penchaszadeh PE (2006) Imposex and organotin compounds in marine gastropods and sediments from the Mar del Plata coast, Argentina. J Mar Biol Assoc UK 86:751–755
Commendatore MG, Esteves JL (2007) An assessment of oil pollution in the coastal zone of Patagonia, Argentina. Env Manage 40:814–821
Commendatore MG, Nievas ML, Amin O, Esteves JL (2012) Sources and distribution of aliphatic and polyaromatic hydrocarbons in coastal sediments from the Ushuaia Bay (Tierra del Fuego, Patagonia, Argentina). Mar Env Res 74:20–31
Crommentuijn T, Sijm D, de Bruijn J, van Leeuwen K, van de Plassche E (2000) Maximum permissible and negligible concentrations for some organic substances and pesticides. J Env Manage 58:297–312
Darbra RM, Ronza A, Stojanovic TA, Wooldridge C, Casal J (2005) A procedure for identifying significant environmental aspects in seaports. Mar Pollut Bull 50:866–874
David A, Bancon-Montignya C, Salles C, Rodier C, Tournoud MG (2012) Contamination of riverbed sediments by hazardous substances in the Mediterranean context: influence of hydrological conditions. J Hydrol 468–469:76–84
De Waisbaum RG, Rodriguez C, Sbarbati Nudelman N (2010) Determination of TBT in water and sediment samples along the Argentine Atlantic coast. Env Technol 31(12):1335–1342
Delucchi F, Narvarte MA, Amín O, Tombesi NB, Freije H, Marcovecchio J (2011) Organotin compounds in sediments of three coastal environments from the Patagonian shore, Argentina. Int J Env Waste Manage 8(1–2):3–17
Deng L, Liu G, Zhang H, Xu H (2015) Levels and assessment of organotin contamination at Futian Mangrove Wetland in Shenzhen, China. Reg Stu Mar Sci 1:18–24
Díez S, Jover E, Albaiges J, Bayona JM (2006) Occurrence and degradation of butyltins and wastewater marker compounds in sediments from Barcelona Harbor, Spain. Env Int 32:858–865
DPMA (1998) Prefectura Naval Argentina. Ordenanza 4/98 Tomo 6, Régimen para la protección del medio ambiente. Anexo 1. 1998
Felizzola JF, Wagener ALR, Almeida AC, Lin WO (2008) Butyltin speciation in sediments from Todos os Santos bay (Bahia, Brazil) by GC-PFPD. Quim Nova 31(1):89–93
Fent K, Müller MD (1991) Occurrence of organotins in municipal wastewater and sewage sludge and behavior in a treatment plant. Environ Sci Technol 25:489–493
Froehner S, Maceno M, Da Luz EC, Souza DB, Machado KS (2010) Distribution of polycyclic aromatic hydrocarbons in marine sediments and their potential toxic effects. Env Monit Assess 168:205–213
Galante-Oliveira S, Oliveira I, Ferreira N, Santos JA, Pacheco M, Barroso C (2011) Nucella lapillus L. imposex levels after legislation prohibiting TBT antifoulants: temporal trends from 2003 to 2008 along the Portuguese coast. J Env Monit 13:304–312
Goldberg RN, Averbuj A, Cledón M, Luzzato D, Nudelman NS (2004) Search for TOTs in Mar del Plata (Argentine) marine coast. Finding of TBT in egg capsules of a snail Adelomenlon brasiliana (Lamarck, 1822) population showing imposex effects. Appl Organomet Chem 18:117–123
Hoch M (2001) Organotin compounds in the environment – an overview. Appl Geochem 16:719–743
IUPAC, International Union of Pure and Applied Chemistry (2002) Analytical, Applied, clinical, inorganic, and physical chemistry divisions’ interdivisional working party for harmonization of quality assurance schemes for analytical laboratories. Pure Appl Chem 74:835–855
Kim NS, Hong SH, Yim UH, Shin KH, Shim WJ (2014) Temporal changes in TBT pollution in water, sediment, and oyster from Jinhae Bay after the total ban in South Korea. Mar Pollut Bull 86:547–554
Kim NS, Hong SH, An JG, Shin KH, Shim WJ (2015) Distribution of butyltins and alternative antifouling biocides in sediments from shipping and shipbuilding areas in South Korea. Mar Pollut Bull. doi:10.1016/j.marpolbul.2015.03.010
MacDonald DD (1994) Approach to the assessment of sediment quality in Florida coastal waters. Volume 1-Development and evaluation of sediment quality assessment guidelines. Report prepared for Florida Department of Environmental Protection, Tallahassee, FL
Martínez-Lladó X, Gibert O, Martí V, Díez S, Romo J, Bayona JM, de Pablo J (2007) Distribution of polycyclic aromatic hydrocarbons (PAHs) and tributyltin (TBT) in Barcelona port sediments and their impact on benthic communities. Env Pollut 149:104–113
Martins CC, Bicego MC, Mahiques MM, Figueira RCL, Tessler MG, Montone RC (2010) Depositional history of sedimentary linear alkylbenzenes (LABs) in a large South American industrial coastal area (Santos Estuary, Southeastern Brazil). Env Pollut 158:3355–3364
Morabito R, Massanisso P, Quevauviller P (2000) Derivatization methods for the determination of organotin compounds in environmental samples. Trends Anal Chem 19:113–119
Nagarajan R, Jonathan MP, Roy PD, Wai-Hwa L, Prasanna MV, Sarkar SK, Navarrete-López M (2013) Metal concentrations in sediments from tourist beaches of Miri City, Sarawak, Malaysia (Borneo Island). Mar Pollut Bull 73:369–373
Niencheski LFH, Fillmann G (2006) Contaminantes: Metais, Hidrocarbonetos e Organoclorados. In: Lana PC, Bianchini A, Ribeiro CAO, Niencheski LFH, Fillmann G, Santos CSG (eds) Avaliação Ambiental de estuários brasileiros: diretrizes metodológicas. Museu Nacional, Rio de Janeiro, pp 63–118
Nisbet Ian CT, LaGoy PK (1992) Toxic equivalency factors (TEFs) for polycyclic aromatic hydrocarbons (PAHs). Regul Toxicol Pharmacol 16(3):290–300
Okoro HK, Fatoki OS, Adekola FA, Ximba BJ, Snyman RG (2013) Spatio-temporal variation of organotin compounds in seawater and sediments from Cape Town harbour, South Africa using gas chromatography with flame photometric detector (GC-FPD). Arab J Chem. doi:10.1016/j.arabjc.2013.05.014
Oliveira CR, Santos DM, Santos Madureira LA, Rodrigues de Marchi MR (2010) Speciation of butyltin derivatives in surface sediments of three southern Brazilian harbors. J Hazard Mate 181:851–856
Palma-Fleming H, Asencio PAJ, Gutierrez H (2004) Polycyclic aromatic hydrocarbons in sediments and mussels of Corral Bay, south central Chile. J Env Monit 6:229–233
Paz-Villarraga CA, Castro IB, Miloslavich P, Fillmann G (2015) Venezuelan Caribbean Sea under the threat of TBT. Chemosphere 119:704–710
Pinochet H, Tessini C, Bravo M, Quiroz W, De Gregori I (2009) Butyltin compounds and their relation with organic matter in marine sediments from San Vicente Bay, Chile. Env Monit Assess 155:341–353
Readman JW, Fillmann G, Tolosa I, Bartocci J, Villeneuve J-P, Catinni C, Mee LD (2002) Petroleum and PAH contamination of the Black Sea. Mar Pollut Bull 44:48–62
Rivero MS, Elías R, Vallarino EA (2005) First survey of macroinfauna in the Mar del Plata Harbour (Argentina), and the use of polychaetes as pollution indicators. Revista de Biología Marina y Oceanografía 40(2):101–108
Schlacher TA, Schoeman DS, Dugan J, Lastra M, Jones A, Scapini F, McLachlan A (2008) Sandy beach ecosystems: key features, sampling issues, management challenges and climate change impacts. Mar Ecol 29(1):70–90
Shim WJ, Oh JR, Kahng SH, Shim JH, Lee SH (1999) Horizontal distribution of butyltins in surface sediments from an enclosed bay system, Korea. Environ Pollut 106:351–357
Silva TF, Azevedo DA, Neto FRA (2007) Polycyclic aromatic hydrocarbons in fishes and sediments from the Guanabara Bay. Brazil. Env Forensics 8(3):257–264
Simoneit BRT (2005) A review of current applications of mass spectrometry for biomarker/molecular tracer elucidations. Mass Spectro Rev 24:719–765
Sousa A, Laranjeiro F, Takahashi S, Tanabe S, Barroso CM (2009) Imposex and organotin prevalence in a European post-legislative scenario: temporal trends from 2003 to 2008. Chemosphere 77:566–573
Sprovieri M, Feo ML, Prevedello L, Salvagio Manta D, Sammartino S, Tamburrino S, Marsella E (2007) Heavy metals, polycyclic aromatic hydrocarbons and polychlorinated biphenyls in surface sediments of the Naples harbour (southern Italy). Chemosphere 67:998–1009
Srogi K (2007) Monitoring of environmental exposure to polycyclic aromatic hydrocarbons: a review. Env Chem Lett 5:169–195
Suzdalev S, Gulbinskas S, Blažauskas N (2015) Distribution of tributyltin in surface sediments from transitional marine-lagoon system of the south-eastern Baltic Sea, Lithuania. Env Sci Pollut Res 22:2634–2642
Town RM, Filella M (2002) Implications of natural organic matter binding heterogeneity on understanding lead (II) complexation in aquatic systems. Sci Total Env 300:143–154
Undap SL, Nirmala K, Miki S, Inoue S, Qiu X, Honda M, Shimasaki Y, Oshima Y (2013) High tributyltin contamination in sediments from ports in Indonesia and Northern Kyushu, Japan. J Fac Agric Kyushu Univ 58(1):131–135
Ünlü S, Sari E, Apak R, Balci N, Koldemir B (2013) Distribution and source identification of polycyclic aromatic hydrocarbons in sediments of the Ambarlı Port area, Sea of Marmara, Turkey. Global Adv Res J Env Sci Toxicol 2(6):144–151
Veiga P, Besteiro C, Rubal M (2010) Meiofauna communities in exposed sandy beaches on the Galician coast (NW Spain), six months after the Prestige oil spill: the role of polycyclic aromatic hydrocarbons (PAHs). Sci Mar 72(4):385–394
Waite ME, Waldock MJ, Thain JE, Smith DJ, Milton SJ (1991) Reductions in TBT concentrations in UK estuaries following legislation in 1986 and 1987. Mar Env Res 32:89–111
Yunker MB, Macdonald RW, Vingarzan R, Mitchell RH, Goyette D, Sylvestre S (2002) PAHs in the Fraser River basin: a critical appraisal of PAH ratios as indicators of PAH source and composition. Org Geochem 33:489–515
Acknowledgments
M. V. Laitano was sponsored by Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) (doctoral fellowship), Í.B. Castro by CAPES (802-16/2012) and FAPERGS (0885/12-1), P. G. Costa by FAPERGS (12/1426-8) and CAPES (23038.028239/2009-69), G. Fillmann by CNPq (PQ 314335/2009-9 and 312341/2013-0) and M. Cledón by CONICET (PIP 0798) and UNMdP (15/E627). The authors are especially grateful to Prof. Landis Hare (INRS, Canada) for English corrections and two anonymous reviewers who improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Laitano, M.V., Castro, Í.B., Costa, P.G. et al. Butyltin and PAH Contamination of Mar del Plata Port (Argentina) Sediments and Their Influence on Adjacent Coastal Regions. Bull Environ Contam Toxicol 95, 513–520 (2015). https://doi.org/10.1007/s00128-015-1637-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-015-1637-y