Abstract
Purpose
Prehospital guidelines stratify and manage patients with Glasgow Coma Scale (GCS) less than nine and any sign of head injury as affected by severe traumatic brain injury (STBI). We hypothesized that this group of patients is so inhomogeneous that uniform treatment guidelines cannot be advocated.
Methods
Patients (2005–2012) with prehospital GCS below nine and abbreviated injury scale head and neck above two were identified from trauma registry. Patients with acute lethal injuries, isolated neck injuries, extubated within 24 h or transferred interhospitally were excluded. Patients were dichotomized based on the worst prehospital GCS (recorded before sedatives) into two groups: GCS 3–5 and GCS 6–8. These were statistically compared using univariate analysis.
Results
The GCS 3–5 group (99 patients) when compared with the GCS 6–8 group (49 patients) had shorter prehospital times (63 vs. 79 min; p < 0.05), more frequent episodes of both hypoxia (30.3% vs. 7.7%; p < 0.05) and hypotension (26.7% vs. 6.4%; p < 0.05), more often required craniectomy (15.1% vs. 4.0%; p = 0.05) and higher mortality (33.3% vs. 2%; p < 0.05). In the GCS 3–5 group, prehospital endotracheal intubation was attempted more often (57.5% vs. 28.6%, p < 0.05) and was more often successful (39.3% vs. 10.2%; p = 0.05). Length of stay in ICU did not differ.
Conclusions
STBI patients are fundamentally different based on whether their initial GCS falls into 3–5 or 6–8 category. Recommendations from trials investigating trauma patients with GCS less than nine as one group should be translated with caution to clinical practice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Severe traumatic brain injury (STBI) is a major cause of mortality and morbidity [1]. Prehospital management is crucial to minimize secondary brain insult from hypoxia and hypotension [2]. Prehospital guidelines label all trauma patients with Glasgow Coma Scale (GCS) less than nine and sign of head injury as patients with STBI and recommend treating them all in a similar fashion. For example, prehospital guidelines in patients with STBI recommend airway protection by any means including prehospital endotracheal intubation (ETI) [3,4,5,6]. In patients with STBI, immediate ETI is the standard of care in Emergency Department (ED), but the role of prehospital ETI with or without rapid sequence intubation (RSI) by paramedics remains controversial [7,8,9,10]. The poor predictive value of prehospital GCS 3–8 investigated as one group could possibly explain the absence of a survival benefit in STBI patients who receive prehospital ETI [11]. “GCS below nine”, although a simple and straightforward criterion, might not be adequate to discern alone which patients should receive a specific and invasive treatment, such as prehospital ETI [11,12,13]. This assumption could sound trivial, but the current literature even in highest level of evidence communications does not seem to address this idiosyncrasy.
The aim of this study was to investigate the differences in outcome of trauma patients with prehospital GCS below nine and radiologically proven STBI. We compared the subgroup of patients with lower GCS (3–5) and the subgroup with higher GCS (6–8). We hypothesized that “patients with prehospital GCS below nine” do not form a homogeneous group.
Methods
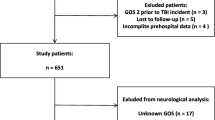
The protocol of this study was approved by the Hunter New England Human Research Ethics Committee. Being a retrospective study, the local Human Research Ethics Committee waived the need for the patient consent to participate, allowed review of patient records and allowed the use of the collected data once de-identified. The Level 1 Trauma Centre institutional trauma registry was queried for all patients with prehospital GCS less than nine and abbreviated injury scale head and neck (AIS H/N) higher than two from January 2005 to December 2012. The prehospital GCS reported in the trauma registry was the lowest recorded before sedative administration. Patients with penetrating mechanism, those who required interhospital transfers, or those who were minors were not included (Fig. 1). Patients with acute lethal injuries (who died prehospitally or were palliated within 24 h from admission), those with isolated neck injury, those who suffered hanging and those who never required ETI during hospitalization or were extubated during within 24 h from admission (intoxicated patients) were subsequently excluded from further analysis. The study population was dichotomized into two groups according to the lowest recorded prehospital GCS (lower GCS group and higher GCS group being 3–5 and 6–8, respectively) and then compared.
Trauma registry and patient hospital and prehospital records were investigated to obtain the following data points: demographics, mechanism of injury, injury severity score (ISS), AIS of main body regions, worst prehospital vital signs, prehospital airway management, time to ED, neurosurgical interventions and haemostatic procedures, days in intensive care (ICU) and outcome (alive at discharge versus deceased in hospital). Hypoxia was defined as an oxygen saturation of less than 90% and hypotension was defined as systolic blood pressure of less than 90 mm Hg.
Categorical data were presented as a percentage (over observed events) and compared with Fisher’s exact test. Continuous data were first tested for normality using the Shapiro–Wilk test, and while nonparametric variables were presented as median and interquartile range and analysed using Mann–Whitney U test, the continuous parametric data were presented as mean and standard deviation and compared using Student t test. A p value of less than 0.05 was considered to be statistically significant. Stata release 11 software was used for statistical analysis.
Results
Out of 11,287 patients requiring trauma team activation, 246 patients fitted inclusion criteria (Fig. 1). Ninety-eight (39%) patients were excluded from further analysis: 31 (13%) never required ETI, 23 (9%) were extubated in less than 24 h, 33 (13%) died or were palliated within 24 h for acute lethal injuries [9 (4%) due to age and/or comorbidities, 14 (6%) for radiological or clinical diagnosis of unsurvivable STBI and 10 (4%) as a direct consequence of exsanguination]. The study population was constituted of 148 patients with potentially survivable STBI and did not differ from the overall group of patients, but for male predominance (p < 0.05) and lower mortality (p < 0.05) (Table 1). The group with GCS 3–5 consisted of 99 patients and the group with GCS 6–8 consisted of 49 patients. When these two groups were compared, demographics, ISS and AIS H/N were statistically similar (Table 2). Additionally, median AIS for face, chest, abdomen and lower limbs was low and statistically similar (0 vs 0, 2 vs 2, 0 vs 0, 0 vs 0, 0 vs 0, respectively). The number of patients with severe extra cranial injures requiring massive transfusion protocol or haemostatic procures such us laparotomy and angioembolization was also low and evenly distributed among groups (Table 2). This confirms that the selection process captured a study population with mainly isolated STBI as cause for the low prehospital GCS. Patients with GCS 3–5 had shorter prehospital times (p < 0.05), more frequent episodes of hypoxia (p < 0.05) and more frequent episodes of hypotension (p < 0.05). Prehospital ETI was attempted more often in patients with GCS 3–5 (p < 0.05) and was more often successful (p = 0.05). Interestingly, both groups showed a similar prehospital ETI failure rate at about 18%. The group of patients with low prehospital GCS had similar rate of intracerebral pressure monitor and external ventricular drain insertions but underwent craniectomy more often (p < 0.05). Length of stay in ICU did not differ between groups (14.2 versus 11.9 days; p = 0.5), even when only survivors were included (19.4 versus 11.9 days; p = 0.1). Patients within the GCS 3–5 group were burdened by much higher mortality (33.3% versus 2%; p < 0.05). The distribution of mortalities among patients with different GCS is reported in Fig. 2. The mortality gradually decreases as GCS increases and only one death was observed in the patients with initial GCS of 6–8.
Discussion
The GCS was developed almost 50 years ago [14], and it is universally accepted as the most pragmatic scoring system to promptly evaluate brain functionality. It was not designed to be used prehospitally, nor before resuscitation, but has been shown to predict mortality better than other vital signs and anatomical injury severity scores [12, 13]. Today, prehospital “GCS below nine” is routinely used in both research settings and management guidelines to define, stratify and manage patients with STBI [2,3,4,5,6,7,8,9,10,11,12,13]. In this study, we have evaluated the population of trauma patients with mostly isolated STBI and demonstrated that these patients do not form a homogenous group. They have dissimilar vital signs, dissimilar critical events dissimilar needs in terms of airway management and surgical procedures and dissimilar outcomes in terms of mortality. Therefore, the common strategy (in both clinical and research setting) of pooling patients with GCS below nine in a single group and offering them the same therapeutic options might underestimate their clinical diversity.
This oversimplification has crucial implications in both the management of the individual patient and during the recruitment of patients in clinical trials. The obvious risk is diluting the effect of an otherwise clinically sensible intervention randomizing patients who would not benefit or (even worse) might be harmed by it. For example, according to the findings of this study, prehospital ETI might not be necessary in patients with GCS 5–8 as they can maintain oxygenation without invasive airway management. This subgroup of patients may even be harmed by prehospital ETI, as is well known to increase prehospital time, hypoxemic event and ICP [7].
Reviewing the PubMed indexed English literature (between 2007 and 2018) for randomized controlled trials (RCT) on prehospital management of STBI, we identified five RCT on patients with initial GCS less nine (Table 3). These studies investigated: prehospital ETI [15], transport mode [16], prehospital therapeutic hypothermia [17], prehospital hypertonic saline [18] and prehospital progesterone [19]. Most of these studies encompassing some of the highest level of evidence in our current prehospital STBI management did not correct, nor report the variation in GCS variability and none used GCS disparity for stratification during the randomization process (Table 3). Interestingly, when reported, GCS disparity was high and with about 50% of patients in the GCS 5–8 group [15].
Based on our study finding, it is not unluckily that many of these RCT could have had different results and subsequent implications in trauma management. If RCT cannot be rerun with ease, it would certainly be relevant to rerun the analysis after adjusting for GCS disparity.
This study has several limitations that need to be acknowledged. It has a retrospective design (despite a prospectively maintained trauma database and accurate data collection from patient inhospital and prehospital notes). Secondly, GCS as all vital signs varies over time and analysis of variance was not performed. Thirdly GCS components were not available, nor analysed. Finally, outcome data were limited to mortality and hospital resource utilization surrogates.
This investigation has, however, a few strengths: it represents a snapshot of a peculiar rural/metropolitan setting in which patients with severe injuries are distant from definitive care, have long prehospital times and are managed by paramedics. We have analysed the clinical history of quite a large cohort of patients with prehospital GCS below nine and have further evaluated the outcome of those with potentially survivable basically isolated STBI. We have observed crucial and statistically significant clinical differences among these patients by simply dichotomizing the whole cohort in two equal groups. This simple dichotomy may work well for prehospital care providers, who in the field need simple pragmatic rules to managed acutely ill patients in a timely fashion.
Oversimplification is always treacherous, and we advocate at least dichotomizing the GCS as per this study and evaluate other information (such hypoxia, loss of gag reflex, signs of intoxication, time to definitive care) before treatment administration or trial inclusion in patient with low prehospital GCS.
Conclusions
Patients with signs of head injury and prehospital GCS below nine appear to be far too heterogeneous to be similarly treated, similarly benchmarked and similarly included in therapeutic trials. Simply stratifying these patients in two groups based on their GCS could be a simple strategy to better predict which patients would benefit from interventions. Further research should consider the present findings and at least dichotomize in two different subgroups patients with prehospital GCS below nine.
Abbreviations
- AIS H/N:
-
Abbreviated injury score head and neck
- GCS:
-
Glasgow Coma Scale
- ED:
-
Emergency department
- ETI:
-
Endotracheal intubation
- EVD:
-
External ventricular drain
- ICP:
-
Intracerebral pressure monitor
- ICU:
-
Intensive care unit
- ISS:
-
Injury severity score
- STBI:
-
Severe traumatic brain injury
References
Evans JA, van Wessem KJ, McDougall D, Lee KA, Lyons T, Balogh ZJ. Epidemiology of traumatic deaths: comprehensive population-based assessment. World J Surg. 2010;34:158–63.
Franschman G, Peerdeman SM, Andriessen TM, Greuters S, Toor AE, Vos PE, et al. Effect of secondary prehospital risk factors on outcome in severe traumatic brain injury in the context of fast access to trauma care. J Trauma. 2011;71:826–32.
Kerby JD, Cusick MV. Prehospital emergency trauma care and management. Surg Clin N Am. 2012;9:823–41.
Battjadia N, Carney N, Crocco T, Fallat ME, Hennes HMA, Jagoda AS, et al. Guidelines for prehospital management of traumatic brain injury, 2nd edn. Prehosp Emerg Care. 2008;12(Suppl 1):S1–52.
Hoogmartens O, Heselmans A, Van de Velde S, Castrén M, Sjölin H, Sabbe M, Aertgeerts B, Ramaekers D. Evidence-based prehospital management of severe traumatic brain injury: a comparative analysis of current clinical practice guidelines. Prehosp Emerg Care. 2014;18:265–73.
Mayglothling J, Duane TM, Gibbs M, McCunn M, Legome E, Eastman AL, et al. Emergency tracheal intubation immediately following traumatic injury: an Eastern Association for the surgery of trauma practice management guideline. J Trauma Acute Care Surg. 2012;73:S333–40.
Yeh DD, Velmahos GC. Prehospital intubation for traumatic brain injury: do it correctly, or not at all. ANZ J Surg. 2012;82:484–5.
Karamanos E, Talving P, Skiada D, Osby M, Inaba K, Lam L, et al. Is prehospital endotracheal intubation associated with improved outcomes in isolated severe head injury? A matched cohort analysis. Prehosp Disaster Med. 2014;29:32–6.
Haltmeier T, Schnüriger B, Benjamin E, Maeder MB, Künzler M, Siboni S, et al. Isolated blunt severe traumatic brain injury in Bern, Switzerland, and the USA: a matched cohort study. J Trauma Acute Care Surg. 2016;80:296–301.
Bochicchio GV, Ilahi O, Joshi M, Bochicchio K, Scalea TM. Endotracheal intubation in the field does not improve outcome in trauma patients who present without an acutely lethal traumatic brain injury. J Trauma Acute Care Surg. 2016;80:296–301.
Gomez D, Byrne JP, Alali AS, Xiong W, Hoeft C, Neal M, Subacius H, Nathens AB. Inclusion of highest glasgow coma scale motor component score in mortality risk adjustment for benchmarking of trauma center performance. J Am Coll Surg. 2017;225:755–62.
Reith FCM, Lingsma HF, Gabbe BJ, Lecky FE, Roberts I, Maas AIR. Differential effects of the Glasgow Coma Scale Score and its components: an analysis of 54,069 patients with traumatic brain injury. Injury. 2017;48:1932–43.
Murray GD, Brennan PM, Teasdale GM. Simplifying the use of prognostic information in traumatic brain injury. J Neurosurg. 2018;128:1612–34.
Teasdale G, Jennett B. Assessment of coma and impaired consciousness. A practical scale. Lancet. 1974;2:81–4.
Bernard SA, Nguyen V, Cameron P, Masci K, Fitzgerald M, Cooper DJ, Walker T, Std BP, Myles P, Murray L, David Taylor, Smith K, Patrick I, Edington J, Bacon A, Rosenfeld JV, Judson R. Prehospital rapid sequence intubation improves functional outcome for patients with severe traumatic brain injury: a randomized controlled trial. Ann Surg. 2010;252:959–65.
Garner AA, Mann KP, Fearnside M, Poynter E, Gebski V. The head injury retrieval trial (HIRT): a single-centre randomised controlled trial of physician prehospital management of severe blunt head injury compared with management by paramedics only. Emerg Med J. 2015;32:869–75.
Nichol A, Gantner D, Presneill J, Murray L, Trapani T, Bernard S, Cameron P, Capellier G, Forbes A, McArthur C, Newby L, Rashford S, Rosenfeld JV, Smith T, Stephenson M, Varma D, Walker T, Webb S, Cooper DJ. Protocol for a multicentre randomised controlled trial of early and sustained prophylactic hypothermia in the management of traumatic brain injury. Crit Care Resusc. 2015;17:92–100.
Bulger EM, May S, Brasel KJ, Schreiber M, Kerby JD, Tisherman SA, Newgard C, Slutsky A, Coimbra R, Emerson S, Minei JP, Bardarson B, Kudenchuk P, Baker A, Christenson J, Idris A, Davis D, Fabian TC, Aufderheide TP, Callaway C, Williams C, Banek J, Vaillancourt C, van Heest R, Sopko G, Hata JS, Hoyt DB, ROC Investigators. Out-of-hospital hypertonic resuscitation following severe traumatic brain injury: a randomized controlled trial. JAMA. 2010;304:1455–64.
Wright DW, Yeatts SD, Silbergleit R, Palesch YY, Hertzberg VS, Frankel M, Goldstein FC, Caveney AF, Howlett-Smith H, Bengelink EM, Manley GT, Merck LH, Janis LS, Barsan WG, NETT Investigators. Very early administration of progesterone for acute traumatic brain injury. N Engl J Med. 2014;371:2457–66.
Funding
None.
Author information
Authors and Affiliations
Contributions
CB contributed to data interpretation and writing. DK performed data collection, data analysis and data interpretation and writing. KK helped in data collection and data interpretation. SN performed literature search and data collection. ZJB contributed to study design, writing and critical revision. All authors have read and approved the manuscript
Corresponding author
Ethics declarations
Conflict of interest
Cino Bendinelli, Dominic Ku, Kate Louise King, Shane Nebauer, Zsolt Janos Balogh declare that they have no conflict of interest.
Ethics approval and consent to participate
The protocol was approved by the Hunter New England Human Research Ethics Committee (09/12/16/5.01). Being a retrospective study with de-identified data, the need for patient approval to participate was waived.
Availability of data and material
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Rights and permissions
About this article
Cite this article
Bendinelli, C., Ku, D., King, K.L. et al. Trauma patients with prehospital Glasgow Coma Scale less than nine: not a homogenous group. Eur J Trauma Emerg Surg 46, 873–878 (2020). https://doi.org/10.1007/s00068-019-01139-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00068-019-01139-9