Abstract
Based on the previous reports on α-glucosidase inhibitory activity of benzimidazole class, we intend to evaluate further this class as potential inhibitors of α-glucosidase enzyme. Thus, in the current study synthesis of 5-bromo-2-aryl benzimidazole derivatives 1–25 was carried out. All the synthetic compounds were characterized by different spectroscopic techniques EIMS, HRMS, 1H-NMR, and 13C-NMR. Molecular docking was also performed on the selected compounds 1, 4, 7, and 17 having varying substitution pattern in order to understand the molecular interaction of molecules with the active site of the enzyme. All compounds were evaluated for their in vitro α-glucosidase inhibitory activities. Twenty-three compounds out of twenty-five showed excellent to moderate activity in the range of IC50 = 12.4–103.2 μM. Inhibitory results were compared with the standard drug acarbose (IC50 = 38.25 ± 0.12 μM). Compounds 1 (IC50 = 37.82 ± 0.08 μM), 9 (IC50 = 37.76 ± 0.05 μM), 12 (IC50 = 24.96 ± 0.09 μM), 16 (IC50 = 21.15 ± 0.08 μM) and 17 (IC50 = 8.34 ± 0.02 μM) showed excellent inhibition as compared to standard drug acarbose (IC50 = 38.25 ± 0.12 μM). Especially, 17 (IC50 = 8.34 ± 0.02 μM) was found to be five-fold more active than the standard.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Benzimidazole is a bicyclic and heterocyclic ring structure consisting of a fused benzene and imidazole ring system (Brink and Folkers, 1949). Some benzimidazole derivatives are abundant in marine natural products, like in marine sponge (Calcul et al., 2003). All seven positions are available for substitution in benzimidazole, but the most biological active analogs are 1, 2, 5, di- or tri-substituted analogs. Many drugs possess benzimidazole moiety as the core part of their structures, for example, omeprazole, mebendazole, albendazole, and astemizole. This indicates the importance of benzimidazole as a privileged pharmacophore (Dinparast et al., 2016; Castillo et al., 2016; Song et al., 2015; Gao et al., 2015; Mariappan et al., 2015; Mavrova et al., 2015; Błaszczak-Świątkiewicz et al., 2014; Desai et al., 2014; Zhang et al., 2012; Guo et al., 2008; Sur et al., 2005; Olbe et al., 2003).
Literature search shows that benzimidazole and its derivatives possess a wide spectrum of biological activities such as antimicrobial, antiviral, antitumor, antioxidant, antiinflammatory, antihypertensive, anticoagulant, antidiabetic, antiallergic, antihistaminic, antitubercular, anti-HIV, antihelmentic, antidepressant, and analgesic activity (Bansal and Silakari, 2012; Nakano et al., 2000; Achar et al., 2010; Kazimierczuk et al., 2002; Ozden et al., 2005; Walia et al., 2011).
Diabetes mellitus is responsible for about 5% of the deaths of the global population. Type 2 diabetes mellitus is the most common type of disease, in which α-glucosidase enzyme catalyzes the carbohydrates and converts it into absorbable monosaccharide in the small intestine (Gao et al., 2007). α-Glucosidase hydrolyzes the α-glucosidal bond of linear and branched isomaltose oligosaccharides and releases α-D-glucose, which is the main cause of hyperglycemia (Van de Laar, 2008). Inhibition of this enzyme is one of the simplest ways to treat type 2 diabetes mellitus by delaying the intestinal glucose absorption process (Rhabasa-Lhoret et al., 2004). Acarbose, miglitol, and voglibose are inhibitors of α-glucosidase, which are being clinically used for the treatment of type 2 diabetes mellitus (Meneilly et al., 2000; Lesley and Caroline, 2000) and can be used as antidiabetic, anti-HIV, anti-obesity, and anticancer agents (Gallienne et al., 2006; Groopman, 1990; Zitzmann et al., 1999).
However, these drugs are 50 % less effective than other antidiabetic agents such as metformin and sulfonylurea, as well as have some associated side effects such as diarrhea, flatulence, and abdominal discomfort (Ag, 1994). Therefore, it is a restrictive aspect to use the drug alone and thus it is often used in combination with other antidiabetic drugs to improve the efficacy. Hence it is an utmost important task to develop safer medication for diabetes.
Our group has explored several classes of heterocyclic compounds including benzimidazole for their potential therapeutic effects (Khan et al., 1999, 2000, 2002, 2003, 2013; Saify et al., 1999; Zaidi et al., 2001; Zawawi et al., 2015; Rahim et al., 2015; Taha et al., 2015) and also explored many classes of compounds for their α-glucosidase inhibitory activities (Niaz et al., 2015; Taha et al., 2015; Rahim et al., 2015; Kashtoh et al., 2014; Khan et al., 2014).
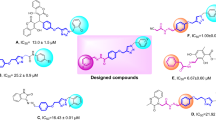
There are only few reports available regarding the α-glucosidase inhibitory activity of benzimidazole class members such as benzimidazole pthalimide containing amino acid (structure A) (Mobinikhaledi et al., 2015) and aryl substituted benzimidazole (structures B and C) (Kumar et al., 2010) (Fig. 1). Therefore, we decided to further explore this class regarding α-glucosidase inhibitory activity in order to get the more potent inhibitor.
In the light of previous reports, we synthesized 5-bromo-benzimidazole analogs 1–25 (general structure D) in order to identify some more molecules having α-glucosidase inhibitory potential with less or no cytotoxicity. To the best of our knowledge, structures of compounds 1, 2, 4, 6, 9 (Dandegaonker and Shastri, 1965), 11, 12, 16, 17 (Cui et al., 2011), 18, 19, and 21 (Dandegaonker and Shastri, 1965) are previously known, whereas rest of the molecules are new.
Results and discussion
Chemistry
Benzimidazoles 1–25 were synthesized by treating commercially available 4-bromo-1,2-benzenediamine with different aromatic aldehydes in N,N-dimethylformamide in the presence of a catalytic amount of sodium metabisulfite (Na2S2O5) (Scheme 1). 1,2-Diamino groups of 4-bromo-1,2-benzenediamine undergo cyclization reaction with the aromatic aldehydes to afford desired benzimidazole moieties. The reaction mixture was refluxed for 4 h to afford the products in the form of precipitates, which were collected via filtration and crystallized in ethanol to get the pure products in high yields. Synthesized derivatives were characterized by different spectroscopic techniques such as EIMS, HRMS, 1H-NMR, and 13C-NMR.
Biological activities
5-Bromo-2-aryl benzimidazole derivatives 1–25 were evaluated for their α-glucosidase inhibitory potential (Table 1). All compounds were found to be active and showed their potential in terms of IC50 values within the range of 8.34–174.62 μM when compared with the standard drug acarbose (IC50 = 38.25 ± 0.12 μM), only two molecules 24 and 25 showed no activity. Compounds 1 (IC50 = 37.82 ± 0.08 μM), 9 (IC50 = 37.76 ± 0.05 μM), 12 (IC50 = 24.96 ± 0.09 μM), 16 (IC50 = 21.15 ± 0.08 μM), 17 (IC50 = 8.34 ± 0.02 μM) showed excellent and compounds 3 (IC50 = 54.62 ± 0.07 μM) and 5 (IC50 = 61.34 ± 0.08 μM) showed good inhibitory potential as compared to standard acarbose (Table 1). However, other analogs exhibited moderate inhibitory potential.

Limited structure-activity relationship studies revealed that the activity of this series of compound mainly depends upon the substitutions on the phenyl part of benzimidazole moiety at position 2, their nature and respective positions of substituents. Compound 17 (IC50 = 8.34 ± 0.02 μM) having 4-hydroxyl substitution was found to be five-fold more active than the standard (IC50 = 38.25 ± 0.12 μM), analog 16 having 3-bromo substitution, residue 12 having 4-butoxy substitution, molecule 9 having 2-hydroxyl substitution and moiety 1 having 2-hydroxy, and 4-methoxy substitution on the phenyl part at position 2 of benzimidazole showed excellent inhibitory potential as compared to standard acarbose. In order to understand the mechanism of α-glucosidase inhibition, mode of binding inside the binding pocket of enzyme of these benzimidazoles and to confirm the assay results, molecular docking studies were carried out.
Molecular docking
Docking studies were carried out using MOE-Dock as docking software implemented in molecular operating environment (MOE) (www.chemchomp.com). The docking scores demonstrated that these compounds showed interactions with active site residues of the enzyme. Docking score is the binding free energy calculated by the GBVI/WSA scoring function, which is the score of the last stage showing the overall fitness of compound in the pocket. For all scoring functions, lower scores indicate more favorable poses. The unit for all scoring functions is kcal/mol. The docking conformation of the most active compound 17 in the series showed six interactions with important active site residues (Fig. 2a). The hydroxyl group at the phenyl ring established two hydrogen bonds with His111 and Gln181, respectively. The phenyl ring of the compound formed arene–arene and arene–cation interactions with active site residues Phe177 and Arg439, respectively. Furthermore, the imidazole ring of the compound formed arene–arene and arene–cation interactions with Phe300 and Arg439, respectively. The presence of electron donating group OH at position 4 of the phenyl ring increases the electron density; as a result more interactions were observed for this compound. This strong bonding network might be one of the reasons for compound 17 to be the most active in the series. Like compound 17, good interactions were observed for compounds 1, 3, 5, 9, 12, and 16. All these compounds have electron donating moiety at phenyl ring and thus have increased electron density that might be responsible for the good interactions between these compounds and active site residues. The lower activities of compounds 1, 3, 5, and 12 as compare to compound 17 might be due to the presence of methoxy or chlorine moieties at the phenyl ring that produce steric hindrance between these compounds and active site residues of the enzyme as indicated by the lower docking scores of these compound. For example, in case of compound 1, only four interactions were observed between the compound and active site residues of the enzyme (Fig. 2b). The oxygen atom of the methoxy group of compound 1 forms a hydrogen bond with His239, the phenyl ring of the compound forms arene–arene interaction with Phe157, whereas the imidazole ring of the compound establishes two arene–cation interactions with Arg312. In the case of least-active compounds 15, 7, 2, and 4 mild interactions were observed. The mild interactions observed in case of these compounds might be due to the presence of more steric hindrance on the phenyl ring of these compounds as compared to compound 17. For example, in the case of compound 7, only two interactions were observed. The oxygen atom of methoxy group of compound 7 established hydrogen bond with active site residue Arg312, while the imidazole ring of the compound formed arene–arene interaction with active site residue Phe177 (Fig. 2c). Similarly, the docking conformation of compound 4 showed that oxygen atom of the methoxy moiety of the compound formed hydrogen bond with active site residue His348, whereas the phenyl and imidazole rings of the compound established arene–cation interactions with Arg439 and Arg312, respectively (Fig. 2d). Overall the docking results showed that the electron donating moieties on the phenyl ring of these compounds promote the interaction with the active site residues and thus increase the activity. However, increasing the number of different moieties on the phenyl ring increases the steric hindrance and thus decreases the number of interactions, resulting in lowering the activity. To test the adopted protocols and to compare the molecular docking study of the synthetic compounds, the standard inhibitor acarbose was docked into the active site of our developed α-glucosidase model. The acarbose fitness in the binding pocket and interaction with the important active site residues is shown in Fig. 3, which reflects a good correlation.
Conclusion
5-Bromo-2-aryl benzimidazole derivatives 1–25 were screened for α-glucosidase inhibitory potential. Compounds 1 (IC50 = 37.82 ± 0.08 μM), 9 (IC50 = 37.76 ± 0.05 μM), 12 (IC50 = 24.96 ± 0.09 μM), 16 (IC50 = 21.15 ± 0.08 μM), and 17 (IC50 = 8.34 ± 0.02 μM) showed potent α-glucosidase inhibitory activity as compared to standard acarbose (IC50 = 38.25 ± 0.12 μM). Molecular docking studies were carried out to identify their mode of binding, which revealed that further chemical modifications on these molecules could have resulted in lead molecules with high degree of inhibitory activity and selectivity towards α-glucosidase enzyme.
Materials and methods
Reagents were purchased from Sigma-Aldrich, USA. All reagents and solvents were of analytical grade and used as received. Thin layer chromatography was performed on pre-coated silica gel, GF-254. Spots were visualized under ultraviolet light at 254 and 366 nm. Mass spectra were recorded under electron impact (EI) on MAT 312 and MAT 113D mass spectrometers. The 1H-NMR were recorded on a Bruker AM spectrometer, operating at 300, 400, and 500 MHz. The chemical shift values are presented in ppm (δ) relative to tetramethylsilane (TMS) as an internal standard and the coupling constants (J) are in Hz.
In vitro α-glucosidase inhibition assay
α-Glucosidase inhibitory potential of all the synthetic benzimidazoles was measured by the reported method (Rahim et al., 2015). Typically, α-glucosidase activity was measured in phosphate buffer 50 mM of pH 6.8 that contains 5 % v/v dimethylsulfoxide, and PNP glycoside was used as a substrate. The inhibitors were pre-incubated with enzyme for half an hour at 37 °C. Then substrate was added and the enzymatic reaction was performed for 60 min at 37 °C. Absorbance was measured spectrophotometrically at 400 nm. The assay was carried in triplicate at five different concentrations around the IC50 values that were roughly calculated in the first turn of the experiments, and the mean values were adopted.
General experimental procedure for the syntheses of 5-bromo-2-aryl benzimidazole derivatives 1–25
4-Bromo-1,2-diaminobenzene (1 mmol) and different substituted aromatic aldehydes (1 mmol) in N,N-dimethylformamide (10 mL) were taken into a 100 mL round-bottomed flask. Catalytic amount of sodium metabisulfite (Na2S2O5) was added into the reaction mixture and refluxed for 4 h. Reaction progress was carefully monitored by thin layer chromatography. After completion of reaction, it was added into chilled distilled water (100 mL). Precipitates were formed and filtered to afford products. Crude products were crystallized from ethanol to get pure products in high yields.
2-(5-Bromo-1H-benzo[d]imidazol-2-yl)-5-methoxyphenol (1)
Yield: 82 %; 1H-NMR (300 MHz, DMSO-d 6 ): δ 7.88 (d, 1H, J 6′,5′ = 9.3 Hz, H-6′), 7.79 (d, 1H, J 4,6 = 1.8 Hz, H-4), 7.56 (d, 1H, J 7,6 = 8.7 Hz, H-7), 7.4 (dd, 1H, J 6,4 = 1.8, J 6,7 = 8.4 Hz, H-6), 6.59 (m, 2H, H-3′, H-5′), 3.85 (s, 3H, OCH3); 13C-NMR (75 MHz, DMSO-d 6 ): δ 162.2 (C, C-4′), 156.1 (C, C-2′), 152.7 (C, C-2) , 141.3 (C, C-8), 140.6 (C, C-9), 129.8 (CH, C-6′), 126.1 (CH, C-6), 118.8 (CH, C-4), 117.6 (C, C-5), 117.4 (CH, C-7), 110.5 (C, C-1′), 107.3 (CH, C-5′), 104.4 (CH, C-3′), 55.9 (CH3, OCH3); EIMS: m/z (rel. abund. %), 318 (M+, 100), 320 (M+2, 96), 289 (14), 277 (21), 196 (5), 168 (7), 90 (4); HRMS (EI) calcd. for C14H11BrN2O2: m/z = 318.0004, found 318.0010.
5-Bromo-2-(4-bromo-2-fluorophenyl)-1H-benzo[d]imidazole (2)
Yield: 85 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 8.16 (d, 1H, J 7,6 = 8.4 Hz, H-7), 7.85 (dd, 2H, J 6,4/5′,3′ = 1.6, J 6,7/5′,6′ = 9.2 Hz, H-6, H-5′), 7.64 (d, 1H, J 3′,5′ = 2 Hz, H-3′), (d, 1H, J 4,6 = 1.6 Hz, H-4), 7.4 (m, 1H, H-6′); 13C-NMR (75 MHz, DMSO-d 6 ): δ 160.4 (C, C-2′), 152.7 (C, C-2), 141.3 (C, C-8), 140.6 (C, C-9), 131.2 (CH, C-6′), 127.6 (CH, C-5′), 126.4 (CH, C-6), 124.6 (C, C-4′), 122.3 (C, C-1′), 121.1 (CH, C-3′), 118.6 (CH, C-4), 117.6 (C, C-5), 117.3 (CH, C-7); EIMS: m/z (rel. abund. %), 368 (M+, 94), 370 (M+2, 100), 372 (M+4, 70), 291 (20), 228 (49), 154 (40), 127 (11); HRMS (EI) calcd. for C13H7Br2FN2: m/z = 367.8960, found 367.8968.
5-Bromo-2-(2-bromo-4-methoxyphenyl)-1H-benzo[d]imidazole (3)
Yield: 75 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 8.4 (d, 1H, J 4,6 = 2 Hz, H-4), 8.19 (dd, 1H, J 6,4 = J 6,5 = 8.8 Hz, H-6), 7.75 (d, 1H, J 3′,5′ = 1.6 Hz, H-3′), 7.54 (d, 1H, J 7,6 = 8.4 Hz, H-7), 7.35 (dd, 1H, J 5′,3′ = 2, J 5′,6′ = 8.8 Hz, H-5′), 7.27 (d, 1H, J 6′,5′ = 8.8 Hz, H-6′), 3.99 (s, 3H, OCH3); 13C-NMR (75 MHz, DMSO-d 6 ): δ 158.5 (C, C-4′), 152.7 (C, C-2), 141.3 (C, C-8), 140.6 (C, C-9), 130.8 (C, C-1′), 130.6 (CH, C-6′), 126.1 (CH, C-6), 121.3 (C, C-2′), 118.8 (CH, C-4), 118.3 (CH, C-3′), 117.7 (C, C-5), 117.4 (CH, C-7), 113.7 (CH, C-5′), 55.7 (CH3, OCH3); EIMS: m/z (rel. abund. %), 380 (M+, 51), 382 (M+2, 100), 383 (M+4, 45), 367 (16), 352 (49), 339 (40), 303 (11), 288 (20); HRMS (EI) calcd. for C14H10 Br2N2O: m/z = 379.9160, found 379.9166.
5-Bromo-2-(4-ethoxy-3-methoxyphenyl)-1H-benzo[d]imidazole (4)
Yield: 76 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 7.80 (d, 1H, J 4,6 = 2 Hz, H-4), 7.74 (dd, 1H, J 6′,2′ = 2, J 6′,5′ = 8.8 Hz, H-6′), 7.72 (d, 1H, J 2′,6′ = 1.6 Hz, H-2′), 7.51 (d, 1H, J 7,6 = 8.4 Hz, H-7), 7.32 (dd, 1H, J 6,4 = 1.6, J 6,7 = 8.4 Hz, H-6), 7.09 (d, 1H, J 5′,6′ = 8 Hz, H-5′), 4.16 (m, 2H, CH2), 3.92 (s, 3H, OCH3), 1.42 (t, 3H, CH3); 13C-NMR (75 MHz, DMSO-d 6 ): δ 152.8 (C, C-2), 150.5 (C, C-4′), 150.3 (C, C-3′), 141.0 (C, C-8), 140.6 (C, C-9), 126.3 (CH, C-6), 123.1 (C, C-1′), 122.0 (CH, C-6′), 118.8 (CH, C-4), 117.6 (C, C-5), 117.3 (CH, C-7), 111.8 (CH, C-1′) , 111.3 (CH, C-5′), 64.7 (CH2, OCH2), 56.4 (CH3, OCH3) 14.7 (CH3, OCH2CH3); EIMS: m/z (rel. abund. %), 346 (M+, 100), 348 (M+2, 93), 317 (47), 289 (15), 260 (5), 192 (2); HRMS (EI) calcd. for C16H15BrN2O2: m/z = 346.0317, found 346.0310.
2-(5-Bromo-1H-benzo[d]imidazol-2-yl)-4,6-dichlorophenol (5)
Yield: 72 %; 1H-NMR: (300 MHz, DMSO-d 6): δ 7.97 (d, 1H, J 4,6 = 2.4 Hz, H-4), 7.86 (bd.s, 1H, H-6′), 7.65 (d, 1H, J 7,6 = 8.7 Hz, H-7), 7.54 (d, 1H, J 4′,6′ = 2.4 Hz, H-4′), 7.48 (dd, 1H, J 6,4 = 1.8, J 6,7 = 8.4 Hz, H-6); 13C-NMR (75 MHz, DMSO-d 6 ): δ 154.4 (C, C-2′), 152.7 (C, C-2), 141.3 (C, C-8), 140.6 (C, C-9), 131.7 (CH, C-4′), 127.4 (C, C-5′), 126.8 (CH, C-6′), 126.2 (CH, C-6), 126.0 (C, C-3′), 121.3 (C, C-1′), 118.6 (CH, C-4), 117.7 (C, C-5), 117.3 (CH, C-7); EIMS: m/z (rel. abund. %), 356 (M+, 78), 358 (M+2, 100), 360 (M+4, 58), 295 (19), 277 (5), 249 (5), 213 (4), 179 (7), 90 (4), 63 (6); HRMS (EI) calcd. for C13H7BrCl2N2O: m/z = 355.9119, found 355.9113.
2-Bromo-4-(5-bromo-1H-benzo[d]imidazol-2-yl)phenol (6)
Yield: 71 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 11.05 (s, 1H, NH), 8.32 (d, 1H, J 4,6 = 2 Hz, H-4), 8.01 (dd, 1H, J 6′,2′ = 1.6, J 6′,5′ = 8.4 Hz, H-6′), 7.79 (d, 1H, J 2′,6′ = 0.8 Hz, H-2′), 7.56 (d, 1H, J 7,6 = 8.4 Hz, H-7), 7.04 (dd, 1H, J 6,4 = 1.6, J 6,7 = 8.4 Hz, H-6), 7.13 (d, 1H, J 5′,6′ = 8.8 Hz, H-5′); 13C-NMR (75 MHz, DMSO-d 6 ): δ 156.7 (C, C-4′), 152.8 (C, C-2), 141.3 (C, C-8), 140.5 (C, C-9), 133.1 (CH, C-2′), 129.6 (CH, C-6′), 126.3(CH, C-6), 123.8 (C, C-1′), 118.8 (CH, C-4), 118.4 (CH, C-5′), 117.6 (C, C-5), 117.3 (CH, C-7), 114.2 (C, C-3′); EIMS: m/z (rel. abund. %), 366 (M+, 100), 368 (M+2, 100), 370 (M+4, 100), 239 (2), 287 (31), 259 (13), 208 (25), 179 (18); HRMS (EI) calcd. for C13H8 Br2N2O: m/z = 365.9003, found 365.9007.
5-Bromo-2-(2-chloro-3,4-dimethoxyphenyl)-1H-benzo[d]imidazole (7)
Yield: 56 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 7.91 (d, 1H, J 6′,5′ = 8.8 Hz, H-6′), 7.81 (d, 1H, J 4,6 = 1.6 Hz, H-4), 7.60 (d, 1H, J 7,6 = 8.4 Hz, H-7), 7.38 (dd, 1H, J 6,4 = 1.6, J 6,7 = 8.4 Hz, H-6), 7.22 (d, 1H, J 5′,6′ = 8.8 Hz, H-5′), 3.98 (s, 3H, OCH3), 3.86 (s, 3H, OCH3); 13C-NMR (75 MHz, DMSO-d 6 ): δ 152.8(C, C-2), 151.0 (C, C-4′), 150.8 (C, C-3′), 141.2 (C, C-8), 140.5 (C, C-9), 131.6 (C, C-1′), 126.1 (CH, C-6), 122.3 (CH, C-6′) 118.7 (CH, C-4), 118.5 (C, C-5), 117.6 (CH, C-7), 117.5 (C, C-2′), 109.2 (CH, C-5′), 56.3 (2CH3, OCH3); EIMS: m/z (rel. abund. %), 366 (M+, 100), 368 (M+2, 100), 370 (M+4, 98), 353 (31), 325 (55), 310 (23), 288 (16), 244 (30) 184 (17), 166 (10); HRMS (EI) calcd. for C15H12 BrClN2O2: m/z = 365.9771, found 365.9777.
5-Bromo-2-(4-bromo-3,5-dimethoxyphenyl)-1H-benzo[d]imidazole (8)
Yield: 42 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 7.80 (d, 1H, J 4,6 = 1.6 Hz, H-4), 7.58 (d, 1H, J 7,6 = 9.2 Hz, H-7), 7.55 (s, 2H, H-2′, H-6′), 7.39 (dd, 1H, J 6,4 = 2, J 6,7 = 8.8 Hz, H-6), 4.00 (s, 6H, 2OCH3); 13C-NMR (75 MHz, DMSO-d 6 ): δ 158.4 (C, C-3′), 158.4 (C, C-5′), 152.7 (C, C-2), 141.3 (C, C-8), 140.6 (C, C-9), 131.4 (C, C-1′), 126.1 (CH, C-6), 118.9 (CH, C-4), 117.6 (C, C-2), 117.4 (CH, C-7), 105.6 (CH, C-2′) , 105.6 (CH, C-6′), 99.6 (C, C-4′), 55.0 (CH3, OCH3), 55.0 (CH3, OCH3); EIMS: m/z (rel. abund. %), 410 (M+, 79), 412 (M+2, 100), 414 (M+4, 80), 381 (8), 352 (4), 331 (7), 301 (17), 273 (21), 247 (9), 206 (11); HRMS (EI) calcd. for C15H12Br2N2O2: m/z = 409.9266, found 409.9261.
2-(5-Bromo-1H-benzo[d]imidazol-2-yl)phenol (9)
Yield: 76 %; 1H-NMR: (300 MHz, DMSO-d 6): δ 7.80 (d, 1H, J 4,6 = 1.5 Hz, H-4), 7.73 (m, 1H, H-5′), 7.68 (d, 1H, J 6′,5′ = 7.8 Hz, H-6′), 7.59 (d, 1H, J 7,6 = 8.7 Hz, H-7), 7.39 (m, 2H, H-6, H-4′), 7.02 (m, 1H, H-3′); 13C-NMR (75 MHz, DMSO-d 6 ): δ 154.2 (C, C-2′), 152.8 (C, C-2), 141.3 (C, C-8), 140.6 (C, C-9), 131.8 (CH, C-6′), 130.3 (CH, C-4′), 126.3 (CH, C-6), 121.9 (CH, C-5′), 118.6 (C1, C-1′), 118.4 (CH, C-4), 117.6 (C, C-5), 117.6 (CH, C-3′), 117.3 (CH, C-7); EIMS: m/z (rel. abund. %), 286 (M+, 100), 288 (M+2, 89), 236 (10), 192 (7), 157 (10); HRMS (EI) calcd. for C13H9BrN2O: m/z = 287.9898, found 287.9894.
2-(3-(Benzyloxy)-4-methoxyphenyl)-5-bromo-1H-benzo[d]imidazole (10)
Yield: 63 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 7.88 (d, 1H, J 4,6 = 1.6 Hz, H-4), 7.81 (d, 1H, J 2′,6′ = 1.2 Hz, H-2′), 7.79 (dd, 1H, J 6,4 = 1.6, J 6,7 = 8.4 Hz, H-6), 7.60 (d, 1H, J 7,6 = 8.8 Hz, H-7), 7.51 (d, 2H, J 2′′,3′′ = J 6′′,5′′ = 7.2 Hz, H-2′′, H-6′′), 7.44 (t, 3H, J 3′′(2′′,4′′) = J 4′′(3′′,5′′) =J 5′′(3′′,4′′) = 7.2 Hz, H-3′′, H-4′′, H-5′′), 7.37 (d, 1H, J 6′,5′ = 7.2 Hz, H-6′), 7.23 (d, 1H, J 5′,6′ = 8.8 Hz, H-5′), 5.19 (s, 2H, CH2), 3.39 (s, 3H, OCH3); 13C-NMR (75 MHz, DMSO-d 6 ): δ 152.8 (C, C-2), 149.6 (C, C-4′), 148.5 (C, C-3′), 141.3 (C, C-8), 140.6 (C, C-9), 136.5 (CH2, OCH2), 128.8 (CH, C-3′′), 128.6 (CH, C-5′′), 127.7 (CH, C-4′′), 127.2 (CH, C-6′′), 127.0 (CH, C-2′′), 126.1 (CH, C-6), 123.8 (C, C-1′), 122.5 (CH, C-6′), 118.5 (CH, C-4), 117.6 (C, C-5), 117.3 (CH, C-7), 112.2 (CH, C-2′), 111.1 (CH, C-5′), 71.2 (CH3, OCH3); EIMS: m/z (rel. abund. %), 391 (M+, 33), 393 (M+2, 42), 317 (58), 291 (4), 246 (2); HRMS (EI) calcd. for C21H17BrN2O2: m/z = 392.0524, found 392.0529.
5-Bromo-2-(3-(trifluoromethyl)phenyl)-1H-benzo[d]imidazole (11)
Yield: 68 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 7.95 (d, 1H, J 6′,5′ = 7.6 Hz, H-6′), 7.84 (m, 4H, H-4, H-2′, H-4′, H-5′), 7.59 (d, 1H, J 7,6 = 8.8 Hz, H-7), 7.39 (dd, 1H, J 6,4 = 1.6, J 6,7 =8.8 Hz, H-6); 13C-NMR (75 MHz, DMSO-d 6 ): δ 152.8 (C, C-2), 141.3 (C, C-8), 140.6 (C, C-9), 137.7 (C, C-2′), 135.3 (CH, C-6′), 131.4 (C, C-3′), 129.6 (CH, C-5′), 126.3 (CH, C-6), 125.4 (CH, C-2′), 125.0 (CH, C-4′), 124.2 (C, CF3), 118.8 (CH, C-4), 117.6 (C, C-5), 117.4 (CH, C-7); EIMS: m/z (rel. abund. %), 340 (M+, 99), 342 (M+2, 99), 322 (100), 301 (29), 241 (44), 152 (14); HRMS (EI) calcd. for C14H8BrF3N2: m/z = 339.9823, found 339.9828.
5-Bromo-2-(4-butoxyphenyl)-1H-benzo[d]imidazole (12)
Yield: 51 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 8.13 (d, 2H, J 2′,6′ = J 6′,5′ = 8.8 Hz, H-2′, H-6′), 7.79 (s, 1H, H-4), 7.68 (d, 1H, J 7,6 = 8.4 Hz, H-7), 7.41 (d, 1H, J 6,7 = 8.4 Hz, H-6), 7.16 (d, 2H, J 3′,2′ = J 5′,6′ = 8.4 Hz, H-3′, H-5′), 4.09 (t, 2H, OCH2), 1.75 (m, 2H, CH2), 1.48 (m, 2H, CH2), 0.96 (t, 2H, CH3); 13C-NMR (75 MHz, DMSO-d 6 ): δ 159.5 (C, C-4′), 152.8 (C, C-2), 141.2 (C, C-8), 140.6 (C, C-9), 129.8 (CH, C-2′), 129.8 (CH, C-6′), 126.0 (CH, C-6), 118.9 (CH, C-4), 117.4 (C, C-4), 117.3 (CH, C-7), 114.8 (CH, C-3′), 114.8 (CH, C-5′), 112.2 (C, C-1′), 68.6 (CH2, OCH2), 31.7 (CH2, O-CH2CH2), 19.1 (CH2, O-CH2CH2CH2), 14.2 (CH2, OCH2CH2CH2CH3); EIMS: m/z (rel. abund. %), 344 (M+, 55), 346 (M+2, 56), 319 (3), 289 (100), 260 (6), 209 (6); HRMS (EI) calcd. for C17H17BrN2O: m/z = 344.0524, found 344.0528.
2-(5-Bromo-1H-benzo[d]imidazol-2-yl)-4,6-di-tert-butylphenol (13)
Yield: 65 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 7.91 (d, 1H, J 6′,4′ = 2 Hz, H-6′), 7.74 (m, 2H, H-4, H-7), 7.43 (d, 1H, J 6,7 = 8 Hz, H-6), 7.37 (d, 1H, J 4′,6′ = 2 Hz, H-6′), 1.42 (s, 9H, 3CH3), 1.34 (s, 9H, 3CH3); 13C-NMR (75 MHz, DMSO-d 6 ): δ 152.8 (C, C-2), 145.4 (C, C-2′), 142.5 (C, C-5′), 141.2 (C, C-8), 140.6 (C, C-9), 138.3 (C, C-3′), 126.3(C, C-6), 126.1 (CH, C-4′), 124.8 (CH, C-6′), 118.6 (CH, C-4), 117.4 (C, C-1′), 117.6 (C, C-5), 117.4 (CH, C-7), 34.9 (C, C(CH3)3), 34.7 (C, C(CH3)3), 31.5 (CH3, C(CH3)3), 31.5 (CH3, C(CH3)3), 31.5 (CH3, C(CH3)3), 31.4 (CH3, C(CH3)3), 31.4 (CH3, C(CH3)3), 31.4 (CH3, C(CH3)3); EIMS: m/z (rel. abund. %), 400 (M+, 40), 402 (M+2, 34), 387 (100), 359 (25), 343 (21), 329 (21), 250 (3); HRMS (EI) calcd. for C21H25BrN2O: m/z = 400.1150, found 400.1154.
5-Bromo-2-(2-fluoro-4-methoxyphenyl)-1H-benzo[d]imidazole (14)
Yield: 84 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 8.15 (m, 1H, H-3′), 7.82 (d, 1H J 4,6 = 1.2 Hz, H-4), 7.60 (d, 1H, J 7,6 = 8.4 Hz, H-7), 7.42 (dd, 1H, J 6,4 = 1.6, J 6,7 = 8.8 Hz, H-6), 7.13 (dd, 1H, J 6′,3′ = 2, J 6′,5′ = 13.6 Hz, H-6′), 7.03 (dd, 1H, J 5′,3′ = 2, J 5′,6′ = 8.8 Hz, H-5′), 3.87 (s, 3H, OCH3); 13C-NMR (75 MHz, DMSO-d 6 ): δ 159.7 (C, C-4′), 159.1 (C, C-2′), 152.8 (C, C-2), 141.0 (C, C-8), 140.6 (C, C-8), 130.2 (CH, C-6′), 126.3(CH, C-6), 118.6 (CH, C-4), 117.6 (C, C-5), 117.4 (CH, C-7), 115.7 (C, C-1′), 110.3 (CH, C-5′), 102.8 (CH, C-3′), 55.6 (CH3, OCH3); EIMS: m/z (rel. abund. %), 320 (M+, 98), 322 (M+2, 100), 307 (36), 279 (15), 259 (11), 241 (3); HRMS (EI) calcd. for C14H10BrFN2O: m/z = 319.9961, found 319.9966.
5-Bromo-2-(3-bromo-4-fluorophenyl)-1H-benzo[d]imidazole (15)
Yield: 88 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 8.49 (dd, 1H, J 6′,2′ = 2, J 6′,5′ = 6.4 Hz, H-6′), 8.22 (m, 1H, H-5′), 7.81 (s, 1H, H-2′), 7.62 (m, 2H, H-4, H-7), 7.39 (dd, 1H, J 6,4 = 1.6, J 6,7 = 8.8 Hz, H-6); 13C-NMR (75 MHz, DMSO-d 6 ): δ 165.3 (C, C-4′), 152.7 (C, C-2), 141.3 (C, C-8), 140.8 (C, C-9), 134.6 (CH, C-2′), 128.3 (CH, C-6′), 126.7 (C, C-1′), 126.1(CH, C-6), 118.9 (CH, C-4), 118.3 (CH, C-5′), 117.6 (C, C-5), 117.3 (CH, C-7), 110.4 (CH, C-3′); EIMS: m/z (rel. abund. %), 368 (M+, 71), 370 (M+2, 100), 372 (M+4, 68), 289 (25), 262 (3), 210 (34), 187 (9); HRMS (EI) calcd. for C13H7Br2FN2: m/z = 367.8960, found 367.8965.
5-Bromo-2-(3-bromophenyl)-1H-benzo[d]imidazole (16)
Yield: 76 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 8.36 (s, 1H, H-4), 8.18 (d, 1H, J 6′,5′ =7.6 Hz, H-6′), 7.84 (s, 1H, H-2′), 7.76 (d, 1H, J 4′,5′ = 8 Hz, H-4′), 7.62 (d, 1H, J 7,6 = 8.8 Hz, H-7), 7.57 (t, 1H, J 5′,4′ = J 5′,6′ = 8 Hz, H-5′), 7.42 (d, 1H, J 6,7 = 8.4 Hz, H-6); 13C-NMR (75 MHz, DMSO-d 6 ): δ 152.7 (C, C-2), 141.0 (C, C-8), 140.6 (C, C-9), 131.8 (CH, C-2′), 131.5 (CH, C-5′), 131.2 (C, C-1′), 128.2 (CH, C-4′), 126.6 (CH, C-6′), 126.2 (CH, C-6), 122.0 (CH, C-3′), 118.6 (CH, C-4), 117.6 (C, C-5), 117.3 (CH, C-7); EIMS: m/z (rel. abund. %), 350 (M+, 87), 352 (M+2, 100), 354 (M+4, 94), 290 (2), 271 (25), 246 (2), 192 (45), 176 (7); HRMS (EI) calcd. for C13H8Br2N2: m/z = 349.9054, found 349.9058.
4-(5-Bromo-1H-benzo[d]imidazol-2-yl)phenol (17)
Yield: 0.11 g (71 %); 1H-NMR: (400 MHz, DMSO-d 6): δ 8.07 (dd, 2H, J 2′,6′/6′,2′ = 1.6, J 2′,3′/6′,5′ = 6.8 Hz, H-2′, H-6′), 7.71 (d, 1H, J 4,6 = 1.6 Hz, H-4), 7.49 (d, 1H, J 7,6 = 8.8 Hz, H-7), 7.31 (dd, 1H, J 6,4 = 2 J 6,7 = 8.4 Hz, H-6), 6.99 (d, 2H, J 3′,2′ = J 5′,6′ = 8.8 Hz, H-3′, H-5′); 13C-NMR (75 MHz, DMSO-d 6 ): δ 158.6 (C, C-4′), 152.7 (C, C-2), 141.3 (C, C-8), 140.6 (C, C-9), 130.5 (CH, C-2′), 130.5 (CH, C-6′), 126.3 (CH, C-6), 118.8 (CH, C-4), 117.6 (C, C-5), 117.4 (CH, C-7), 116.6 (CH, C-3′), 116.6 (CH, C-5′), 113.2 (C, C-1′); EIMS: m/z (rel. abund. %), 288 (M+, 100), 290 (M+2, 91), 261 (4), 209 (9), 182 (8), 170 (2), 144 (4); HRMS (EI) calcd. for C13H9BrN2O: m/z = 287.9898, found 287.9892.
5-Bromo-2-(naphthalen-2-yl)-1H-benzo[d]imidazole (18)
Yield: 65 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 8.72 (s, 1H, H-4), 8.36 (dd, 1H, J 8′,2′ = 1.6, J 8′,7′ = 8.8 Hz, H-8′), 8.06 (d, 1H, J 7,6 = 8.4 Hz, H-7), 8.04 (m, 1H, H-3′), 7.99 (m,1H, H-6′), 7.81 (d, 1H, J 2′,8′ = 1.6 Hz, H-2′), 7.81 (m, 3H, H-4′, H-5′, H-7′), 7.39 (dd, 1H, J 6,4 = 2 J 6,7 = 8.4 Hz, H-6); 13C-NMR (75 MHz, DMSO-d 6 ): δ 152.8 (C, C-2), 141.2 (C, C-8), 140.6 (C, C-9), 133.8 (C, C-1′), 133.7 (C, C-9′), 133.2 (C, C-10′), 131.8 (CH, C-8′), 128.3 (CH, C-4′), 128.3 (CH, C-7′), 126.3 (CH, C-6), 126.1 (CH, C-5′), 126.1 (CH, C-6′), 125.8 (CH, C-2′), 124.6 (CH, C-8′), 118.6 (CH, C-4), 117.6 (C, C-5), 117.4 (CH, C-7); EIMS: m/z (rel. abund. %), 322 (M+, 100), 324 (M+2, 92), 242 (22), 216 (3), 189 (2), 153 (7); HRMS (EI) calcd. for C17H11BrN2: m/z = 322.0106, found 322.0101.
5-Bromo-2-(4-ethoxyphenyl)-1H-benzo[d]imidazole (19)
Yield: 70 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 8.14 (d, 2H, J 2′,3′ = J 6′,5′ = 8.8 Hz H-2′, H-6′), 7.72 (d, 1H, J 4,6 = 1.6 Hz, H-4), 7.51 (d, 1H, J 7,6 = 8.4 Hz, H-7), 7.32 (dd, 1H, J 6,4 = 2 J 6,7 = 8.4 Hz, H-6), 7.08 (d, 2H, J 3′,2′ = J 5′,6′ = 8.8 Hz, H-3′, H-5′), 4.17 (m, 2H, OCH2), 1.41 (t, 3H, CH3); 13C-NMR (75 MHz, DMSO-d 6 ): δ 159.3 (C, C-4′), 152.8 (C, C-2), 141.2 (C, C-8), 140.6 (C, C-9), 129.8 (CH, C-2′), 129.8 (CH, C-6′), 126.3 (CH, C-6), 118.6 (CH, C-4), 117.7 (C, C-5), 117.3 (CH, C-7), 114.8 (CH, C-3′), 114.8 (CH, C-5′), 112.4 (C, C-1′), 64.5 (CH2, OCH2CH3), 14.9 (CH3, OCH2CH3); EIMS: m/z (rel. abund. %), 316 (M+, 100), 318 (M+2, 100), 287 (100), 259 (98), 237 (22), 208 (67), 192 (23), 180 (91), 153 (30); HRMS (EI) calcd. for C15H13BrN2O: m/z = 316.0211, found 316.0217.
2-(4-(Benzyloxy)phenyl)-5-bromo-1H-benzo[d]imidazole (20)
Yield: 69 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 8.16 (d, 2H, J 2′,3′ = J 6′,5′ = 9.2 Hz, H-2′, H-6′), 7.72 (d, 1H, J 4,6 = 1.2 Hz, H-4), 7.52 (m, 3H, H-2′′, H-4′′, H-6′′), 7.43 (t, 2H, J 3′′(2′′,4′′) = J 5′′(4′′,6′′) =7.2 Hz, H-3′′, H-5′′), 7.36 (d, 1H, J 7,6 = 7.2 Hz, H-7), 7.32 (dd, 1H, J 6,4 = 1.6, J 6,7 = 8.4 Hz, H-6), 7.19 (d, 2H, J 3′,2′ = J 5′,6′ = 9.2 Hz, H-3′, H-5′), 5.22 (s, 2H, OCH2); 13C-NMR (75 MHz, DMSO-d 6 ): δ 159.2 (C, C-4′), 152.7 (C, C-2), 141.0 (C, C-8), 140.6 (C, C-9), 136.6 (C, C-1′′), 130.2 (CH, C-2′), 130.2 (CH, C-6′), 128.7 (CH, C-3′′), 128.7 (CH, C-5′′), 127.7 (CH, C-4′′), 127.0 (CH, C-2′′), 127.0 (CH, C-6′′), 126.3 (CH, C-6), 118.8 (CH, C-4), 117.6 (C, C-5), 117.5 (CH, C-7), 114.9 (CH, C-3′), 114.9 (CH, C-5′), 113.2 (C, C-1′), 70.7 (CH2, OCH2C6H6); EIMS: m/z (rel. abund. %), 378 (M+, 16), 380 (M+2, 17), 248 (49), 218 (53), 201 (20), 189 (100), 161 (35), 135 (76); HRMS (EI) calcd. for C20H15BrN2O: m/z = 378.0368, found 378.0362.
4-(5-Bromo-1H-benzo[d]imidazol-2-yl)-N,N-dimethylaniline (21)
Yield: 59 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 7.99 (d, 2H, J 2′,3′ = J 6′,5′ = 9.2 Hz, H-2′, H-6′), 7.76 (s, 1H, H-4), 7.54 (d, 1H, J 7,6 = 8.4 Hz, H-7), 7.42 (d, 1H, J 6,7 = 8.8 Hz, H-6), 6.89 (d, 2H, J 3′,2′ = J 5′,6′ = 9.2 Hz, H-3′, H-5′), 3.03 (s, 6H, N(CH3)2); 13C-NMR (75 MHz, DMSO-d 6 ): δ 155.4 (C, C-4′), 152.8 (C, C-2), 141.0 (C, C-8), 140.6 (C, C-9), 128.3 (CH, C-2′), 128.3 (CH, C-6′), 126.4 (CH, C-6), 118.6 (CH, C-4), 117.6 (C, C-5), 117.3 (CH, C-7), 112.6 (CH, C-3′), 112.6 (CH, C-5′), 115.7 (C, C-1′), 41.2 (CH3, N(CH3)2), 41.2 (CH3, N(CH3)2); EIMS: m/z (rel. abund. %), 315 (M+, 100), 317 (M+2, 96), 301 (13), 273 (3), 236 (11), 221 (4); HRMS (EI) calcd. for C15H14BrN3: m/z = 315.0371, found 315.0376.
5-Bromo-2-(2-bromo-4,5-dimethoxyphenyl)-1H-benzo[d]imidazole (22)
Yield: 49 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 7.83 (d, 1H, J 4,6 = 1.6 Hz, H-4), 7.61 (s, 1H, H-3′), 7.59 (d, 1H, J 7,6 = 2.8 Hz, H-7), 7.4 (dd, 1H, J 6,4 = 2, J 6,7 = 8.8 Hz, H-6), 3.93 (s, 3H, OCH3), 3.90 (s, 3H, OCH3); 13C-NMR (75 MHz, DMSO-d 6 ): δ 152.8 (C, C-2), 150.3 (C, C-4′), 149.2 (C, C-5′), 141.0 (C, C-8), 140.6 (C, C-9), 133.0 (C, C-1′), 126.3 (CH, C-6), 118.6 (CH, C-4), 117.7 (C, C-5), 117.5 (CH, C-7), 115.5 (CH, C-3′), 114.4 (CH, C-6′), 113.6 (C, C-2′), 56.2 (CH3, O CH3), 56.1 (CH3, O CH3); EIMS: m/z (rel. abund. %), 410 (M+, 49), 412 (M+2, 100), 414 (M+4, 51), 381 (24), 366 (19), 315 (88), 273 (23), 236 (14); HRMS (EI) calcd. for C15H12Br2N2O2: m/z = 409.9266, found 409.9260.
2-(Anthracen-9-yl)-5-bromo-1H-benzo[d]imidazole (23)
Yield: 72 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 8.78 (s, 1H, H-6′), 8.19 (d, 2H, J 2′,3′ = J 10′,9′ = 8.4 Hz, H-2′, H-10′), 7.92 (s, 1H, H-4), 7.80 (d, 2H, J 5′,4′ = J 7′,8′ = 8.8 Hz, H-5′, H-7′), 7.71 (d, 1H, J 7,6 = 8.4 Hz, H-7), 7.57 (m, 5H, H-6, H-3′, H-4′, H-8′, H-9′); 13C-NMR (75 MHz, DMSO-d 6 ): δ 152.8 (C, C-2), 141.2 (C, C-8), 140.6 (C, C-9), 134.2 (C, C-1′), 132.1 (C, C-11′), 132.1 (C, C-13′), 130.5 (C, C-12′), 130.5 (C, C-14′), 129.7 (CH, C-6′), 128.1 (CH, C-5′), 128.1 (CH, C-7′), 126.4 (CH, C-6), 125.6 (CH, C-4′), 125.6 (CH, C-8′), 125.8 (CH, C-3′), 125.8 (CH, C-9′), 124.1 (CH, C-2′), 124.1 (CH, C-10′), 118.6 (CH, C-4), 117.6 (C, C-5), 117.3 (CH, C-7); EIMS: m/z (rel. abund. %), 371 (M+, 84), 373 (M+2, 100), 313 (54), 292 (45), 264 (5), 232 (15); HRMS (EI) calcd. for C21H13BrN2: m/z = 372.0262, found 372.0267.
4-(5-Bromo-1H-benzo[d]imidazol-2-yl)-2-methoxyphenyl acetate (24)
Yield: 63 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 7.93 (d, 1H, J 4,6 = 2 Hz, H-4), 7.79 (m, 2H, H-2′, H-6′), 7.57 (d, 1H, J 7,6 = 8.4 Hz, H-7), 7.37 (dd, 1H, J 6,4 = 2, J 6,7 = 8.4 Hz, H-6), 7.23 (d, 1H, J 5′,6′ = 8 Hz, H-5′), 3.94 (s, 3H, O=C-CH3), 2.27 (s, 3H, CH3); 13C-NMR (75 MHz, DMSO-d 6 ): δ 169.1 (C, O=C), 152.7 (C, C-2), 151.3 (C, C-3′), 141.7 (C, C-4′), 141.2 (C, C-8), 140.6, (C, C-9), 128.3 (C, C-1′), 126.1 (CH, C-6), 123.0 (CH, C-5′), 122.2 (CH, C-6′), 118.6 (CH, C-4), 117.6 (C, C-5), 117.4 (CH, C-7), 126.2 (CH, C-6), 111.6 (CH, C-2′), 55.9 (CH3, O=C-CH3), 20.2 (CH3, OCH3); EIMS: m/z (rel. abund. %), 360 (M+, 9), 362 (M+2, 12), 318 (100), 305 (5), 290 (7), 275 (9), 239 (6), 196 (5); HRMS (EI) calcd. for C16H13BrN2O3: m/z = 360.0110, found 360.0117.
2-Bromo-6-(5-bromo-1H-benzo[d]imidazol-2-yl)-4-chlorophenol (25)
yield: 72 %; 1H-NMR: (400 MHz, DMSO-d 6): δ 10.33 (s, 1H, NH), 7.88 (m, 2H, H-4, H-6′), 7.62 (d, 1H, J 7,6 = 8.8 Hz, H-7), 7.38 (d, 1H, J 6,7 = 8.4 Hz, H-6), 6.50 (s, 1H, H-4′), 6.48 (s, 1H, OH); 13C-NMR (75 MHz, DMSO-d 6 ): δ 152.9 (C, C-2), 152.7 (C, C-2′), 141.3 (C, C-8), 140.6 (C, C-9), 133.7 (CH, C-4′), 129.5 (C, C-5′), 127.7 (CH, C-6′), 126.3 (CH, C-6), 121.8 (C, C-1′), 118.6 (CH, C-4), 117.5 (C, C-5), 117.3 (CH, C-7), 115.6 (C, C-3′); EIMS: m/z (rel. abund. %), 400 (M+, 74), 402 (M+2, 76), 385 (100), 357 (54), 345 (25), 329 (11); HRMS (EI) calcd. for C13H7Br2ClN2O: m/z = 399.8614, found 399.8610.
References
Achar K, Hosamani KM, Seetharamareddy HR (2010) In-vivo analgesic and anti-inflammatory activities of newly synthesized benzimidazole derivatives. Eur J Med Chem 45:2048–2054
Ag H (1994) Pharmacology of α‐glucosidase inhibition. Eur J Clin Invest 1:3–10
Bansal Y, Silakari O (2012) The therapeutic journey of benzimidazole: a review. Bioorg Med Chem 20:6208–6236
Błaszczak-Świątkiewicz K, Olszewska P, Mikiciuk-Olasik E (2014) Biological approach of anticancer activity of new benzimidazole derivatives. Pharma Rep 66:100–106
Brink NG, Folkers K (1949) Vitamin B12. VI. 5,6-Dimethylbenzimidazole, a degradation product of vitamin B12. J Am Chem Soc 71:2951–2951
Calcul L, Longeon A, Mourabit AA, Guyot M, Bourguet-Kondracki ML (2003) Novel alkaloids of the aaptamine class from an Indonesian marine sponge of the genus Xestospongia. Tetrahedron 59:6539–6544
Castillo I, Suwalsky M, Gallardo MJ, Troncoso V, Sánchez-Eguía BN, Santiago-Osorio E, Aguiñiga I, González-Ugarte AK (2016) Structural and functional effects of benzimidazole/thioether copper complexes with antitumor activity on cell membranes and molecular models. J Inorg Biochem 156:98–104
Cui M, Ono M, Kimura H, Kawashima H, Liu BL, Saji H (2011) Radioiodinated benzimidazole derivatives as single photon emission computed tomography probes for imaging of β-amyloid plaques in Alzheimer’s disease. Nucl Med Biol 38:313–320
Dandegaonker SH, Shastri D (1965) Bromobenzimidazoles. Monatsh Chem 96:614–24
Desai NC, Shihory NR, Kotadiya GM, Desai P (2014) Synthesis, antibacterial and antitubercular activities of benzimidazole bearing substituted 2-pyridone motifs. Eur J Med Chem 82:480–489
Dinparast L, Valizadeh H, Bahadori MB, Soltani S, Asghari B, Rashidi MR (2016) Design, synthesis, α-glucosidase inhibitory activity, molecular docking and QSAR studies of benzimidazole derivatives. J Mol Struct 1114:84–94
Gallienne E, Gefflaut T, Bolte J, Lemaire M (2006) Synthesis of new nitrogen analogues of salacinol and deoxynojirimycin and their evaluation as glycosidase inhibitors. J Org Chem 71:894–902
Gao C, Li B, Zhang B, Sun Q, Li L, Li X, Chen C, Tan C, Liu H, Jiang Y (2015) Synthesis and biological evaluation of benzimidazole acridine derivatives as potential DNA-binding and apoptosis-inducing agents. Bioorg Med Chem 23:1800–1807
Gao H, Huang YN, Xu PY, Kawabata J (2007) Inhibitory effect on α-glucosidase by the fruits of Terminalia chebula Retz. Food Chem 105:628–634
Groopman JE (1990) Current advances in the diagnosis and treatment of AIDS: An introduction. Rev Infect Dis 12:908–911
Guo XZ, Shi L, Wang R, Liu XX, Li BG, Lu XX (2008) Synthesis and biological activities of novel nonpeptide angiotensin II receptor antagonists based on benzimidazole derivatives bearing a heterocyclic ring. Bioorg Med Chem 16:10301–10310
Kashtoh H, Hussain S, Khan A, Saad SM, Khan JAJ, Khan KM, Perveen S, Choudhary MI (2014) Oxadiazoles and thiadiazole: Novel α-glucosidase inhibitors. Bioorg Med Chem 22:5454–5465
Kazimierczuk Z, Upcroft JA, Upcroft P, Gorska A, Starosciak B, Laudy A (2002) Synthesis, antiprotozoal and antibacterial activity of nitro and halogeno-substituted benzimidazole derivatives. Acta Biochem Pol 49:185–196. English edition
Khan KM, Rahat S, Choudhary MI, Rahman Atta-ur, Ghani U, Perveen S, Khatoon S, Dar A, Malik A (2002) Synthesis and biological screening of 2-[4-substituted phenyl] 4-(2-cyanoethyl)-4(H)-1,3,4-oxadiazine-5(6H)-ones and their intermediates. Helv Chem Acta 85:559–570
Khan M, Yousaf M, Wadood A, Junaid M, Ashraf M, Alam U, Ali M, Arshad M, Hussain Z, Khan KM (2014) Discovery of novel oxindole derivatives as potent α-glucosidase inhibitors. Bioorg Med Chem 22:3441–3448
Khan KM, Saify ZS, Begum S, Noor F, Khan MZ, Hayat S, Choudhary MI, Perveen S, Ullah Zia (2003) Synthesis and biological screening of 7-hydroxy-4-methyl-2H-chromen-2-one, 7-hydroxy-4,5-dimethyl-2H-chromen-2-one and their some derivatives. Nat Prod Res 17:115–125
Khan KM, Saify ZS, Hayat S, Khan MZ, Noor F, Choudhary MI, Ullah Zia, Perveen S (2002) Synthesis and antioxidant and insecticidal activities of coumarin derivatives. J Chem Soc Pak 24:226–231
Khan KM, Saify ZS, Khan ZA, Ahmed M, Saeed M, Abdel-Jalil RJ, Grubler G, Voelter W (1999) Syntheses of selected quaternary phenacylbromopyridinium compounds and their biological evaluation. Z Naturforsch 54b:1210–1218
Khan KM, Saify ZS, Khan ZA, Ahmed M, Saeed M, Schick M, Kohlbau HJ, Voelter W (2000) Syntheses and cytotoxic, antimicrobial, antifungal and cardiovascular activity of new quinoline derivatives. Arzneim Forsch/Drug Res 50:915–924
Khan KM, Saleem M, Perveen S, Khan M (2013) Benzimidazoles: a new class of carbonic anhydrase inhibitors. J Chem Soc Pak 35:901–904
Kumar JA, Tiwari AK, Ali AZ, Madhusudhana K, Reddy BS, Ramakrishna S, Raju BC (2010) New antihyperglycemic, α-glucosidase inhibitory, and cytotoxic derivatives of benzimidazoles. J Enzyme Inhib Med Chem 25:80–86
Lesley JS, Spencer CM (2000) Miglitol. Drugs 59:521–549
Mariappan G, Hazarika R, Alam F, Karki R, Patangia U, Nath S (2015) Synthesis and biological evaluation of 2-substituted benzimidazole derivatives. Arabian J Chem 8:715–719
Mavrova AT, Yancheva D, Anastassova N, Anichina K, Zvezdanovic J, Djordjevic A, Markovic D, Smelcerovic A (2015) Synthesis, electronic properties, antioxidant and antibacterial activity of some new benzimidazoles. Bioorg Med Chem 23:6317–6326
Meneilly GS, Ryan EA, Radziuk J, Lau DC, Yale JF, Morais J (2000) Effect of acarbose on insulin sensitivity in elderly patients with diabetes. Diabetes Care 23:1162–1167
Mobinikhaledi A, Asghari B, Jabbarpour M (2015) Design and synthesis of new benzimidazole and pyrimidine derivatives as α-glucosidase inhibitor. Iranian J Pharm Res 14:723–731
Nakano H, Inoue T, Kawasaki N, Miyataka H, Matsumoto H, Taguchi T, Inagaki N, Nagai H, Satoh T (2000) Synthesis and biological activities of novel antiallergic agents with 5-lipoxygenase inhibiting action. Bioorg Med Chem 8:373–380
Niaz H, Kashtoh H, Khan AJ, Khan A, Wahab Atia-tul, Alam MT, Khan KM, Perveen S, Choudhary MI (2015) Synthesis of diethyl 4-substituted-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylates as a new series of inhibitors against yeast α-glucosidase. Eur J Med Chem 95:199–209
Olbe L, Carlsson E, Lindberg P (2003) A proton-pump inhibitor expedition: the case histories of omeprazole and esomeprazole. Nat Rev Drug Disc 2:132–139
Ozden S, Atabey D, Yıldız S, Goker H (2005) Synthesis and potent antimicrobial activity of some novel methyl or ethyl-yl-benzimidazole-5-carboxylates derivatives carrying amide or amidine groups. Bioorg Med Chem 13:1587–1597
Rahim F, Malik F, Ullah H, Wadood A, Khan F, Javid MT, Taha M, Rehman W, Rehman AshfaqUr, Khan KM (2015) Isatin based schiff bases as inhibitors of α-glucosidase: synthesis, characterization, in vitro evaluation and molecular docking studies. Bioorg Chem 60:42–48
Rahim F, Ullah K, Ullah H, Wadood A, Taha M, Rehman AshfaqUr, Uddin I, Ashraf M, Shaukat A, Rehman W, Hussain S, Khan KM (2015) Triazinoindole analogs as potent inhibitors of α-glucosidase: synthesis, biological evaluation and molecular docking studies. Bioorg Chem 58:81–87
Rhabasa-Lhoret R, Chiasson JL, Defronzo RA, Ferrannini E, Keen H, Zimmet P (2004) α-glucosidase inhibitors, International Textbook of Diabetes Mellitus, 1st edn.. John Wiley, UK
Saify ZS, Khan KM, Haider SM, Zeeshan Shah STA, Saeed M, Shekhani MS, Voelter W (1999) Syntheses and evaluation of the analgesic activity of some 4-acetyl-4- phenylepiperidine and 4-hydroxy-4-phenylepiperidine derivatives. Zeitsch Naturforsch 54b:1327–1336
Song WJ, Lin QY, Jiang WJ, Du FY, Qi QY, Wei Q (2015) Synthesis, interaction with DNA and antiproliferative activities of two novel Cu(II) complexes with norcantharidin andbenzimidazole derivatives. . Spectrochim Acta Part A 137:122–128
Sur D, Saha D, Manna B, Rajendran K, Bhattacharya S (2005) Periodic deworming with albendazole and its impact on growth status and diarrhoeal incidence among children in an urban slum of India. Trans Royal Soc Tropic Med Hyg 99:261–267
Taha M, Ismail NH, Lalani S, Fatmi MQ, Wahab Atia-tul, Siddiqui S, Khan KM, Imran S, Choudhary MI (2015) Synthesis of novel inhibitors of α-glucosidase based on the benzothiazole skeleton containing benzohydrazide moiety and their molecular docking studies. Eur J Med Chem 92:387–400
van de Laar FA (2008) α-Glucosidase inhibitors in the early treatment of type 2 diabetes, Vasc. Health Risk Manag 4:1189–1195
Walia R, Hedaitullah M, Naaz SF, Iqbal K, Lamba HS (2011) Benzimidazole derivatives, An overview. Int J Res Pharm Chem 1:2231–2781
Zaidi JH, Naeem F, Iqbal R, Choudhary MI, Khan KM, Shah STA, Hayat S, Voelter W (2001) Synthesis and bioactivities of naturally occurring anthraquinones: isochrysophanol, isozyganein, ω-hydroxyisochrysophanol and morindaparvin. Z Naturforsch 56b:689–696
Zawawi NKNA, Taha M, Ahmat N, Wadood A, Ismail NH, Rahim F, Ali M, Abdullah N, Khan KM (2015) Novel 2,5-disubtituted-1,3,4-oxadiazoles with benzimidazole backbone: a new class of β-glucuronidase inhibitors and in silico studies. Bioorg Med Chem 23:3119–3125
Zhang J, Wang JL, Zhou ZM, Li ZH, Xue WZ, Xu D, Hao LP, Han XF, Fei F, Liu T, Hua A (2012) Design, synthesis and biological activity of 6-substituted carbamoyl benzimidazoles as new nonpeptidic angiotensin II AT1receptor antagonists. Bioorg Med Chem 20:4208–4216
Zitzmann N, Mehta AS, Carrouée S, Butters TD, Platt FM, McCauley J, Blumberg BS, Dwek RA, Block TM (1999) Imino sugars inhibit the formation and secretion of bovine viral diarrhea virus, a pestivirus model of hepatitis C virus: Implications for the development of broad spectrum anti-hepatitis virus agents. Proc Nat Ac Sci 96:11878–11882
Acknowledgments
The authors are thankful to the Higher Education Commission (HEC) Pakistan, for their financial support to Research Project No. 20-2073 under National Research Program for Universities.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Rights and permissions
About this article
Cite this article
Arshad, T., Khan, K.M., Rasool, N. et al. Syntheses, in vitro evaluation and molecular docking studies of 5-bromo-2-aryl benzimidazoles as α-glucosidase inhibitors. Med Chem Res 25, 2058–2069 (2016). https://doi.org/10.1007/s00044-016-1614-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-016-1614-y