Abstract
Mosses are a dominant ground cover in a wide array of ecosystems, especially in those developing under cold-stressed environments such as arctic and alpine ice-melting glacial forelands. Consequently, mosses may influence the performance and distribution of other plants. Here, we assessed the nature of interactions between vascular plants and cushion-forming mosses in three alpine communities in the northern Patagonian Andes. We recorded species richness, plant abundance and cover of vascular plants within and outside moss cushions, measuring also patch area and moss layer depth. To determine the effect of mosses on vascular plant assemblages, we calculated the relative interaction index (RII) in terms of richness, abundance and cover of all vascular plants, and of individual species. Moss-cushion patches showed higher species richness, plant abundance and cover in comparison with bare ground areas. Overall, the diversity of vascular plants increased with both moss-cushion area and layer depth. Species-specific RII values revealed that the effects of moss cushions on neighboring vascular plants were predominantly positive for all three plant communities surveyed. These results highlight the role of mosses as nurse plants in alpine ice-melting glacial forelands and, thus, as ecosystem engineers that can be key in maintaining vascular plant diversity.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Biotic interactions are key drivers of plant population dynamics and community composition. Particularly, in severe environments, the importance of facilitation in sustaining plant diversity is widely acknowledged (Brooker et al. 2008). As opposed to competition, plant–plant facilitative interactions occur when one species increases the performance of another plant by, for instance, the modification of the local abiotic or biotic environment (Bertness and Callaway 1994; Brooker and Callaghan 1998). Although facilitation and competition are processes that can occur between neighboring plants within a single community, facilitation is more frequent in harsh environments, shifting to competition as abiotic conditions become more benign (Bertness and Callaway 1994; Callaway et al. 2002). However, plant–plant interactions can also depend on intrinsic traits of the interacting species, both of the beneficiary and of the benefactor (e.g., Gomez-Aparicio 2009; Navas and Violle 2009; Schöb et al. 2013; Bonanomi et al. 2015). In particular, cushion-like plant species are known to be particularly effective “nurse plants” in alpine and arctic environments (Butterfield et al. 2013; Cavieres et al. 2014). Furthermore, most of the current knowledge about plant–plant interactions is based on interactions occurring between vascular plants, highlighting the need to explore the role of plants belonging to other taxonomic groups as possible ecosystem engineers that determine plant community structure, composition, and diversity.
Mosses are a dominant ground cover in a wide array of ecosystems, especially in those developing in cold-stressed environments, such as the Arctic tundra and Antarctic fellfields, where they typically adopt a cushion-like form. Consequently, mosses may affect the performance and distribution of small-sized and/or short-lived vascular plants through both abiotic and biotic pathways. Recent studies have addressed the facilitation of vascular plants by bryophytes (Groeneveld et al. 2007; Gornall et al. 2011; Casanova-Katny and Cavieres 2012; Rayburn et al. 2012; Sand-Jensen et al. 2015). Moreover, mosses in the polar tundra can act as ecosystem engineers given their influence on microclimate and soil processes (Malmer et al. 2003; Gornall et al. 2007). Indeed, like many vascular plants with a cushion growth form, mosses can provide better microhabitats than the surrounding environment by increasing soil moisture and nutrients, reducing frost damage experienced by seedlings and plants roots, and buffering the extreme temperatures that impact the surrounding bare ground (e.g., Smith 1988; Carlsson and Callaghan 1991; Groeneveld and Rochefort 2005; Gornall et al. 2007; Groeneveld et al. 2007; Startsev et al. 2007; Roberts et al. 2009). In addition, the physical structure of mosses, while favoring seed trapping, can provide safe sites that protect seeds against predation (Jeschke and Kiehl 2008). Therefore, the microenvironment provided by mosses can enhance the establishment, growth and/or survival of other plant species (Carlsson and Callaghan 1991; Groeneveld et al. 2007; Casanova-Katny and Cavieres 2012).
On the other hand, negative interactions between mosses and vascular plants have also been reported (e.g., Hobbie et al. 1999; Bret-Harte et al. 2004; Jeschke and Kiehl 2008; Gornall et al. 2011; Doxford et al. 2013). In fact, several studies indicate that competition between mosses and vascular plants is the prevalent type of interaction in the Antarctic fellfield (Block et al. 2009; Krna et al. 2009). Whereas moss growth can be suppressed through leaf litter deposition and shading from associated vascular plants (Malmer et al. 2003; Van der Wal et al. 2005; Startsev et al. 2008), mosses can hamper seedling emergence and plant growth of vascular plants via moss ground cover effects (Bai and Romo 1995, Donath and Eckstein 2010, Hobbie et al. 1999; Gornall et al. 2011; Doxford et al. 2013). Furthermore, it has been shown that moss layer depth is crucial in determining the sign and magnitude of these plant–plant interaction (Van der Wal and Brooker 2004; Jeschke and Kiehl 2008; Gornall et al. 2011) since shallow and deep mosses tend to affect positively and negatively, respectively, the growth of plant species of the Arctic tundra. Particularly, it has been reported that deep mosses can decrease local soil temperature and nutrient availability, creating more stressful microhabitats (Van der Wal and Brooker 2004; Gornall et al. 2007, 2011). In sum, mosses can exert both positive and negative effects on vascular plants.
Most studies on plant–plant interactions involving mosses have been conducted in polar ecosystems. Alpine environments can be as stressful as the Arctic tundra (Bliss 1962; Körner 2003), as both are characterized by extreme cold and poor soils with low nutrient availability. However, alpine ecosystems are subjected to much larger daily variations of temperature and solar radiation during the growing season and also to a greater impact from ultraviolet radiation than arctic ecosystems (Bliss 1962; Gardes and Dahlberg 1996). Therefore, the nature of moss–vascular plant interactions between alpine and arctic ecosystems can differ, although, to the best of our knowledge, no study on moss–plant interactions has been conducted so far in an alpine environment.
In this study, we investigated the nature of moss—vascular plant interactions in three alpine communities in the Andes of North Patagonia. Specifically, we addressed the following questions: (1) Are moss-cushion patches richer in plant species than bare ground areas of similar size? (2) Is diversity of vascular plants related to moss-cushion area and depth? (3) What is the nature and strength of vascular plant responses to cushion mosses at the community and species levels? (4) How effective are mosses as nurse plants in comparison with angiosperm-cushion species?
Methods
Site and species studied
The study was conducted in three high-Andean communities dominated by mosses in the North Patagonian Andes during January and February of 2018. The three moss-dominated communities studied were located on the (1) east-facing slope of Cerro López at 1850 m (41°6ʹS, 71°33ʹW), (2) east-facing slope of Cerro Hielo Azul at 1820 m (41°52ʹS, 71°39ʹW), and (3) southeast-facing slope of Cerro Catedral at 1840 m (41°11ʹS, 71°30ʹW). The region of the North Patagonian Andes is strongly influenced by moisture-carrying westerlies, with most precipitation falling in winter. All the communities included in this study occur on ice-influenced large plateaus where plant cover is extremely low (from 3 to 5%) due to the severe abiotic conditions, resulting in a landscape mainly covered by stones, rock crevices, soil and mosses (Fig. 1). In these sites, moss cushions establish on ice-melting glacial forelands, more specifically, on a plateau surrounding a lagoon fed by rain and the thaw of a nearby glacier (Hielo Azul), or by the melting of nearby semi-permanent ice patches and snow packs (López and Catedral). Accordingly, all sites are environmentally characterized by freeze–thaw action, cryogenic processes, and large water and temperature variations, typical of the high-cold permafrost characterizing several alpine ecosystems (Wang et al. 2006). Mean annual temperature is ca. 4.5 °C with an annual precipitation of approximately 1054 mm at López, 937 mm at Hielo Azul, and 942 mm at Catedral (Worldclim, http://www.worldclim.org; Hijmans et al. 2005), mainly falling during winter in the form of snow, leaving mounds of ice and snow during summer.
In these alpine communities, we studied the nurse effect of moss patches that grow as cushions of varied shapes and sizes (Fig. 1). At least three moss species occur at the López community, Philonotis esquelensis Matteri (Bartramiaceae), Bryum pseudotriquetrum (Hedw.) G. Gaertn., B. Mey & Scherb. (Bryaceae), and Pohlia wahlenbergii (F. Weber & D. Mohr) A.L. Andrews (Mielichhoferiaceae). The most abundant moss species with cushion-like form at this community was P. esquelensis. Philonotis is commonly found growing on rocks or soil, usually associated with wet sites such as stream banks or seeps, from sea level to 4700 m, well above tree line (Gradstein et al. 2001; Jimenez and Suárez 2017; Jimenez et al. 2014, 2016). At the Hielo Azul community, two moss species were found, Bryum pseudotriquetrum and Pohlia wahlenbergii; however, B. pseudotriquetrum was the dominant species. The distributions of B. pseudotriquetrum and P. wahlenbergii also encompass all kind of moist or wet habitats from wet meadows, swamps, stream banks, rivers, and lake shores to mountain meadows near springs, streams and flushes, among others (Erzberger and Schröder 2013; Suárez and Schiavone 2011). At the Catedral community, only one moss species was found, Bartramia patens Brid. (Bartramiaceae), an endemic species of South America with incursions into the Antarctic continent (Larraín 2007). At each community, we sampled the most abundant moss species (i.e., Philonotis esquelensis at López, Bryum pseudotriquetrum at Hielo Azul, and Bartramia patens at Catedral).
Vegetation sampling
At each community, a total of 50 moss cushions were haphazardly selected, pairing each moss cushion with an adjacent non-cushion or ‘open area’ microsite about 50 cm distant in a random direction. This short distance ensures that the non-cushion area was similar in substrate and overall micro-environmental conditions. To obtain comparable data sampling in the surrounding open area, a wire hoop was shaped to match the size of the sampled cushion which was then placed on the ground (e.g., Cavieres et al. 2014). Vascular plant species abundance and plant cover were recorded at both cushions and matching open-area plots. For the whole vascular plant assemblage and for each individual species, abundance was estimated as number of plant individuals and plant cover measured visually as a percentage of the sampled area. Even though plant cover is generally related to the number of plant individuals, it can be used as an estimation of occupancy (Damgaard et al. 2011). Given that cushions were usually roughly elliptical, microsites were defined as elliptical plots and, thus, the longer and shorter axes of each cushion were measured as an approximate estimation of their area. We also measured cushion depth as the distance between the ground level and the maximum height of the cushion, recorded at the cushion center. To evaluate the comparative efficiency of mosses as nurse plants, vascular plant species growing within several angiosperm-cushion species from different mountain systems in the same region were also recorded using the same sampling protocol as we did with the cushion mosses. Particularly, we identified 15 angiosperm-cushion species performing as nurses in several genera and families in a total of 21 high-Andean communities occurring between 1600 and 2000 m spread among seven mountains, estimating absolute and relative plant species richness associated with each of these species (Online Resource, Table S1). Besides absolute richness, similarly to the measurements performed in moss cushions, we estimated relative richness from counts of plant species growing in 50 paired angiosperm-cushion and open-area patches (see below).
Statistical analyses
Assessment of differences in species richness between cushions and open-area plots requires sampling efforts large enough to fully capture the species assemblages associated with each microhabitat (Colwell 2013). Therefore, rarefaction curves were constructed with Mao Tau estimations for mean species richness per sampling class for both cushion and open-area plots at the three communities, using the software EstimateS (version 9.1.0, Colwell 2013). Additionally, we estimated asymptotic species richness using non-parametric estimators (Gotelli and Colwell 2011). For incidence data, the most proper estimator of species richness is the Chao-2 estimator (Colwell 2013), which takes into account the rare species and the total number of species observed in the sample to estimate the expected total species richness (Colwell 2013). Accordingly, we calculated Chao-2 values at both microsites (moss cushions and open areas) for the three communities.
Generalized linear mixed-effect models (glmer function in lme4 R package, Bates et al. 2015) were used to test for the effect of microsite (moss cushion vs. open area) on vascular plant species richness, and assemblage-wide plant abundance and cover, using patch area as a covariable. Paired plots and community were included as random effects. We estimated the Pearson correlation between cushion measurements, area and depth to determine to what extent these factors are associated. The influence of moss-cushion depth on vascular species richness, plant abundance and cover was analyzed with generalized linear mixed-effect models, using cushion depth as a fixed factor and community as a random effect. Response count variables (i.e., species richness and plant abundance) were examined for over-dispersion; plant species richness was analyzed considering a Poisson distribution (Zuur et al. 2009), whereas a quasi-Poisson model was used for plant abundance with the function glmmPQL of the MASS package. Instead, plant cover was analyzed assuming a normal distribution. Predicted values with 95% confidence intervals of all models were computed and plotted with the function ggpredict of the ggeffects package. All analyses were performed with the R software (R Development Core Team 2017).
To assess the effect of cushion mosses on vascular plant diversity, abundance, and cover, we calculated the relative interaction index (RII; Armas et al. 2004) at both the whole assemblage and species level. The RII is calculated as (x within cushion–x in open area)/(x within cushion + x in open area), where x represents any performance variable of interest. The RII varies from − 1 to 1 and in the case of abundance, for instance, RII = 1 (i.e., complete facilitation) if all individuals occur within cushions; RII = − 1 if all individuals are found growing in the open areas; or RII = 0 if individuals are equally distributed between cushion and open-area plots. At the community level, RII values were assessed by comparing species richness, total plant abundance, and total plant cover in each paired moss–open-area plot. One sample t tests with the Bonferroni correction (Rice 1989) were used to examine if community RII values (RIIrichness, RIIabundance, and RIIcover), at each site, were significantly different from zero, allowing to ascertain if overall moss–plant interactions are positive, negative or neutral (Yang et al. 2017). At the species level, the RII was estimated for each vascular plant species using species-specific records of plant abundance and cover (e.g., Anthelme et al. 2012).
Finally, to assess how effective were mosses as nurse plants compared to angiosperm-cushion plants, a ranking was performed by ordering angiosperm- and moss-cushion plant species according to the mean species richness found growing within and to the estimated RIIrichness value, which was calculated using the number of species found inside and outside cushion plant patches and which correct for differences in the richness of the overall species pool found at the different communities (Butterfield et al. 2013; Cavieres et al. 2014).
Results
A total of 56 vascular plant species were found in the three surveyed moss-dominated communities, with the most frequent species being Cardamine cordata, Cerastium arvense, Luzula racemosa, Poa obvallata, Epilobium australe and Trisetum barbinode. The community at Hielo Azul had the highest richness, with a total of 36 species of vascular plants, and the community at López had the lowest richness, harboring 22 species only. Species richness increased at a faster rate with the number of moss patches sampled than with the number of bare ground patches outside mosses for the three sampled communities (Fig. 2). Among the sampled communities, cumulative species richness recorded in moss cushions ranged from 22 to 33, whereas species number in open-ground plots ranged from 12 to 21 species (Table 1, Fig. 2). Cushion mosses also sustained higher species diversity than bare ground at the community level, as indicated by differences in the asymptotic Chao-2 estimates (Table 1).
Rarefaction curves for plant species number sampled growing within moss cushions and paired open-area plots (i.e., non-cushion plant vegetation present). The Mao Tau estimators and their respective standard deviations are shown for each of the three communities at a López, b Hielo Azul, and c Catedral
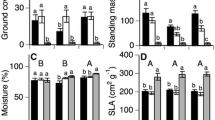
At the patch level, there were significantly higher species number, plant abundance and cover of vascular plants associated with moss cushions (Table 2) than in paired open-area plots in all studied communities (Fig. 3). Both moss-cushion area and depth were highly variable (Figs. 4, 5) and only weakly correlated (r = 0.15, n = 150, P = 0.06). Patch area was a significant predictor for all plant community estimates (Table 2). Particularly, species richness, plant abundance and cover increased with plot area, yet this increase was only observed for the moss-cushion patches and not for the paired open-area plots (Fig. 4). All these variables also tended to significantly increase with the depth of moss-cushion patches (species richness, X2 = 22.37, P < 0.0001, n = 50; plant abundance, X2 = 81.25, P < 0.0001, n = 50; plant cover, X2 = 6.42, P = 0.011, n = 50; Fig. 5). Therefore, large and deep moss cushions were those that sustained the highest plant diversity, abundance and cover at the patch level.
Relationships between patch size (m2) and number of vascular plant species (i.e., species richness), individuals (i.e., abundance), and total cover (as a percentage) for moss-cushions (left panels) and open-area patches (right panels). Model predicted values (solid lines) and 95% confidence intervals (dashed lines) are shown
Relationships between moss-cushion layer depth (cm), measured as the maximum cushion height at the patch middle, and a number of vascular plant species (i.e., species richness), b total number of plant individuals (i.e., abundance), and c total plant cover (as a percentage). Model predicted values (solid lines) and 95% confidence intervals (dashed lines) are shown
As a consequence of the higher diversity of vascular plants associated with mosses than with open-area patches, the RII values of the vascular plant assemblage were significantly positive in terms of species richness, abundance and cover at the three sites (Table 3). Accordingly, species-specific RII-values, either based on the number of individuals or species cover, were predominantly positive (Online Resource, Fig. S1). Based on our results, we estimated that about 90% of the vascular plant species surveyed at López and Catedral, and 75% at Hielo Azul, were facilitated by mosses. Furthermore, about 80% of all species encountered only once in any of the communities studied were exclusively associated with moss cushions (Online Resource, Fig. S1). As a consequence, moss cushion plants appear to be key elements in sustaining diversity at the patch and community levels in the cold-stressed environments surveyed.
We identified a total of 15 angiosperm-cushion species performing as potential nurse plants among the additional 21 high-Andean communities sampled in the region (Fig. 6). Mean plant species richness associated with the three sampled moss-cushion species was both in absolute and relative terms (i.e., as estimated by the RII) comparable to that associated with angiosperm-cushion species (Fig. 6). In fact, the mean number of species growing inside the dominant moss cushions at the Hielo Azul community (Bryum pseudotriquetrum) and the Catedral community (Bartramia patens) ranked among the highest, whereas at López the number of species inside the moss Philonotis esquelensis ranked at an intermediate position (Fig. 6a). When species richness associated with cushion plants was estimated in relative terms (i.e., based on the RII index), moss-cushion species were even better nurses than more than half of the angiosperm-cushion species (Fig. 6b).
Species richness ranking among cushion plant species. The mean (± 1 SE) is shown for the a absolute species richness measured as the number of species found within cushions, and b relative species richness estimated by the Relative Interaction Index (RII) as the difference in species richness inside and outside cushions, for three moss-cushion and fifteen angiosperm-cushion species. Bry pse (Bryum pseudotriquetrum), Mul lep (Mulinum leptacanthum), Bar pat (Bartramia patens), Azo lyc (Azorella lycopodioides), Bel chi (Belloa chilensis), Azo mad (Azorella madreporica), Ore gla (Oreopolus glacialis), Aca ant (Acaena antarctica), Sen cri (Senecio crithmoides), Azo mon (Azorella monantha), Mul spi (Mulinum spinosum), Bac mag (Baccharis magellanica), Phi esq (Philonotis esquelensis), Mul ech (Mulinum echinus), Aca mac (Acaena macrocephala), Nas pyg (Nassauvia pygmaea), Gau pum (Gaultheria pumila), Oxa ery (Oxalis erythrorhiza)
Discussion
In this study, we evaluated the effect of cushion-forming mosses on vascular plant diversity in three alpine communities of ice-melting glacial forelands of the North Patagonian Andes. In particular, moss-cushion patches harbored higher species richness, plant abundance and cover of vascular plants in comparison with bare ground areas. Furthermore, vascular plant richness, abundance and cover increased with both moss-cushion area and depth. Overall, our results suggest that the effect of moss-cushions on interacting vascular plants was predominantly positive at both the whole plant assemblage and individual species levels. To the best of our knowledge, this study represents the first report of mosses as nurse plants in alpine communities.
Alpine plants, in general, are capable of producing abundant seeds and seedlings (Marcante et al. 2013); however, germination, establishment and initial growth are considered the most hazardous life-cycle stages (Erschbamer et al. 2008; Marcante et al. 2013). Indeed, occurrence of vascular plants outside moss cushions was rather restricted (Fig. 3), suggesting that in these high-Andean communities the foremost obstacle is possibly the lack of safe sites for initial establishment and seedling survival. Instead, occurrence of diverse assemblages of vascular plants was always associated with moss cushions, indicating that these moss patches provide favorable niches that facilitate the recruitment of many species of vascular plants that otherwise would not occur in these wet- and cold-stressed plant communities.
Whereas several studies have focused on the effect of mosses upon specific-species performance (e.g., Gornall et al. 2011; Casanova-Katny and Cavieres 2012), few have evaluated their effects on whole species assemblages (Van der Wal and Brooker 2004). Our community approach allowed us to find a high dependence of rare vascular plants species on cushion-forming mosses. In fact, 80% of the rarest species occurred only in moss cushions and, consequently, these species would be potentially excluded from the studied community in the absence or removal of mosses.
Several biological features of mosses make them extraordinary colonizers in extreme ecosystems (Belkina and Mavlyudov 2011; Dickson and Johnson 2014; Belkina and Vilnet 2015) such as mosses’ tolerance to extremely low temperatures, ability to retain water, and establish and grow on limited and loose soil without being nursed by other species (Belkina and Mavlyudov 2011; Belkina and Vilnet 2015). Consequently, mosses are common pioneer species (Bruun et al. 2006), facilitating the colonization of less stress-tolerant plants (Sohlberg and Bliss 1984), the reason why they are important in triggering ecological successions (Jones and Henry 2003; Johnstone et al. 2010; Turetsky et al. 2010; Garibotti et al. 2011; Arróniz-Crespo et al. 2014; Dickson and Johnson 2014). Hence, mosses might play a significant role as pioneer species in ice-driven high-Andean communities, with later successional stages involving small-sized and/or short-lived vascular plants occurring within already-established moss cushions.
Facilitation can be particularly important during early successional stages (Niederfriniger Schlag and Erschbamer 2000), particularly in severe environments (Choler et al. 2001; Callaway et al. 2002; Cavieres et al. 2005). In fact, one outstanding finding of our results was the almost complete dominance of positive interactions between mosses and vascular plants, either regarding overall vascular plant diversity or species-specific recruitment within moss patches. This is consistent with the prevalence of facilitative over competitive interactions in harsh environments (Bertness and Callaway 1994; Callaway et al. 2002). Nonetheless, a gradient of environmental severity might systematically alter the nature of moss–vascular plant interactions and, thus, in less stressful environments these interactions could shift from positive to negative (Choler et al. 2001; Maestre et al. 2009; Anthelme et al. 2012; but see Cavieres et al. 2006; De Bello et al. 2011; Dvorsky et al. 2013). However, alpine moss cushions are limited to these peculiar cold-stressed wet environments (Gavini, personal observation), making the evaluation of this hypothesis unfeasible in this case. Moreover, at all three mountains mosses occurred in the same altitudinal belt (~ 1800 m.a.s.l.) making it impossible to undertake comparisons among communities across altitudes to assess the altitudinal version of the stress-gradient hypothesis (Cavieres et al. 2006; Anthelme et al. 2012; Schöb et al. 2013). In any event, moss-cushion facilitation could represent one of the main processes fostering ecological plant succession and maintaining plant diversity in high-Andean ice-melting glacial forelands.
Although we did not assess the microhabitat properties of the non-vascular nurse species surveyed, it is known that mosses can enhance the establishment of other species by modifying the local environment. In particular, in addition to acting as seed traps (e.g., Groeneveld et al. 2007), mosses can increase soil moisture and nutrients, buffer extreme temperatures, reduce frost damage and provide seed and seedlings safe sites (e.g., Startsev et al. 2007; Gornall et al. 2007; Groeneveld et al. 2007; Jeschke and Kiehl 2008; Roberts et al. 2009). Measurements of abiotic variables and experimental field studies involving, for instance, moss removal (Hobbie et al. 1999; Bret-Harte et al. 2004; Casanova-Katny and Cavieres 2012; Doxford et al. 2013) would be needed to identify the mechanisms by which these moss species are facilitators. However, given that the studied sites are settled close to ice-melting lagoons, where plant physiological drought could be rare, an increase in soil moisture may not be the mechanism underlying moss-cushion facilitation. Instead, the importance of mosses in insulating plants from extreme temperatures could be key in understanding the facilitating role of mosses, as explained below.
Contrary to the widespread moss-facilitative effects reported here, negative interplays between mosses and vascular plants have also been reported. Among negative effects, mosses may hamper seedling emergence and plant growth of vascular plants via ground cover effects (Bai and Romo 1995; Hobbie et al. 1999; Donath and Eckstein 2010; Gornall et al. 2011; Doxford et al. 2013). Yet, the sign and intensity of plant–plant interactions are often related to nurse traits (Michalet et al. 2011, 2014), being moss-cushion depth a key factor in the particular case of moss-plant interactions (Van der Wal and Brooker 2004; Jeschke and Kiehl 2008; Gornall et al. 2011). In our sites, moss-cushion depth, indeed, affected the number of associated vascular plants, but unlike the findings of these other studies (Gornall et al. 2011), moss depth influenced positively rather than negatively the establishment of other plants.
Deep mosses can potentially increase the severity of micro-environmental conditions due to a reduction in soil temperature, nutrient availability, and growing season length, hence hampering the growth of associated plants (Van der Wal and Brooker 2004; Gornall et al. 2007, 2011). Indeed, negative relationships between moss depth and graminoid biomass and/or survival have been reported in several tundra studies (e.g., Olofsson et al. 2001, 2004; Van der Wal and Brooker 2004; Jeschke and Kiehl 2008; Gornall et al. 2011). In contrast, in the alpine communities studied, the positive relationships between moss depth and species abundance and diversity suggest that there must be increasing benefits associated with deep mosses that must overcome the costs for the nursed species. These contrasting results are probably explained by the climatic differences between alpine and arctic ecosystems. Particularly, during the growing season, arctic environments are subject to less solar and ultraviolet radiation, and lower diurnal temperatures than alpine environments (Bliss 1962; Gardes and Dahlberg 1996). Consequently, thick moss layers in the Arctic can insulate the cold and poorly drained soils caused by underlying permafrost, thus constraining the growth of accompanying vascular plants (Beringer et al. 2001; Van der Wal and Brooker 2004). Instead, deep mosses in alpine environments of lower latitudes, where permafrost is generally absent, can better buffer the high day temperatures and irradiance that can restrict plant growth (Bliss 1962) favouring higher vascular plant diversity than shallow mosses. Besides, in lieu with these less stressful micro-environmental conditions, deeper cushions might be fostering seed trapping and retention by reducing wind speed and enhancing seed germination through reduced competition for underground space (e.g., root growth) and/or by increasing water availability. In addition, vascular plant diversity increased with the area of the moss cushion independently of its depth. These results indicate that the overall size of this ameliorated microhabitat fosters recruitment of a higher diversity of plants. Alternatively, based on the positive relationship between size and age in cushion plant species (Molau 1997; Morris and Doak 1998), larger moss cushions might also be older and, therefore, may have had more time to accumulate more species than younger small-sized cushion patches (Nuñez et al. 1999).
Across arctic and alpine ecosystems, a cushion-like architecture represents a frequent life-form that evolved independently in many different plant lineages (Aubert et al. 2014). This fact evidences an evolutionary convergence of the cushion life-form among phylogenetically unrelated taxa (Körner 2003), even between vascular and non-vascular plants. Interestingly, there is also a convergence in the ecological role accomplished by plants from highly unrelated lineages with cushion life-growth form as ecosystem engineers, increasing plant diversity at the patch and community levels (Anthelme et al. 2012; Butterfield et al. 2013; Cavieres et al. 2014). Here, we show that the nurse effect of mosses resembles the pattern exhibited by several angiosperm-cushion plant species in the North Patagonian Andes (e.g., Nuñez et al. 1999). Yet, the variation observed between the different moss and angiosperm-cushion plant species could be due not only to the morphological traits of the species involved (e.g., Aubert et al. 2014), but also to differences in the environmental severity and climate of the sites they inhabit. In particular, plant–plant interactions, but more importantly overall plant diversity, are likely to differ between wet and more arid alpine environments (Michalet et al. 2014). This could potentially explain why the communities at Hielo Azul and Catedral exhibited higher species richness than the one at López, since the latter is subject to greater precipitation. In the southern Andes, environmental conditions of alpine areas are generally more favorable on the relatively more arid mountains, where snow cover is less and the growing season lasts longer (Ferreyra et al. 1998). Ultimately, this study suggests that in extreme wet-cold environments, such as those studied here, mosses may be important nurse plants, as angiosperms are in dry-cold alpine environments.
In conclusion, patches vegetated with mosses host higher plant diversity than the surrounding open areas, indicating that moss cushions facilitate the recruitment of vascular plants. This study highlights the importance of mosses as potential nurse plants in alpine environments and, thus, their role as facilitators and reservoirs of biodiversity. Accordingly, these findings contribute to the relatively recent growing literature regarding moss–vascular plant interactions. The recognition of mosses as ecosystem engineers in the severe environments of ice-melting glacial forelands may allow a better understanding of how community assemblage and species diversity are structured and preserved in these extreme cold and wet alpine ecosystems.
References
Anthelme F, Buendia B, Mazoyer C, Dangles O (2012) Unexpected mechanisms sustain the stress-gradient hypothesis in a tropical alpine environment. J Veg Sci 23:62–72
Armas C, Ordiales R, Pugnaire F (2004) Measuring plant interactions: a new comparative index. Ecology 85:2682–2686
Arróniz-Crespo M, Perez-Ortega S, De los Ríos A, Green TGA, Ochoa-Hueso R et al (2014) Bryophyte-Cyanobacteria associations during primary succession in recently deglaciated areas of Tierra del Fuego (Chile). PLoS One 9(5):e96081
Aubert S, Boucher F, Lavergne S, Renaud J, Choler P (2014) 1914–2014: a revised worldwide catalogue of cushion plants 100 years after Hauri and Schröter. Alpine Bot 124(1):59–70
Bai Y, Romo JT (1995) Seedling emergence of Artemisia frigida in relation to hydration-dehydration cycles and seedbed characteristics. J Arid Environ 30:57–65
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48
Belkina OA, Mavlyudov BR (2011) Mosses on glaciers of Spitsbergen. Bot Zhurn 96(5):582–596
Belkina OA, Vilnet AA (2015) Some aspects of the moss population development on the Svalbard glaciers. Czech Polar Rep 5(2):160–175
Beringer J, Lynch AH, Chapin FS, Mack M, Bonan GB (2001) The representation of arctic soils in the land surface model: the importance of mosses. J Clim 14:3324–3335
Bertness MD, Callaway RM (1994) Positive interactions in communities. Trends Ecol Evol 9:191–193
Bliss LC (1962) Adaptations of arctic and alpine plants to environmental conditions. Arctic 15:117–144
Block W, Smith RIL, Kennedy AD (2009) Strategies of survival and resource exploitation in the Antarctic fellfield ecosystem. Biol Rev 84:449–484
Bonanomi G, Stinca A, Chirico GB, Ciaschetti G, Saracino A, Incerti G (2015) Cushion plant morphology controls biogenic capability and facilitation effects of Silene acaulis along an elevation gradient. Funct Ecol 30:1216–1226
Bret-Harte MS, García EA, Sacre VM, Whorley JR, Wagner JL, Lippert SC, Scarano FR, Chapin FS (2004) Plant and soil responses to neighbour removal and fertilization in Alaskan tussock tundra. J Ecol 92:635–647
Brooker RW, Callaghan TV (1998) The balance between positive and negative plant interactions and its relationship to environmental gradients: a model. Oikos 81:196–207
Brooker RW, Maestre FT, Callaway RM, Lortie CJ, Cavieres LA, Kunstler G, Liancourt P, Tielbörger K, Travis MJT et al (2008) Facilitation in plant communities: the past, the present and the future. J Ecol 96:18–34
Bruun HH, Moen J, Virtanen R, Grytnes J, Oksanen L, Angerbjörn A (2006) Effects of altitude and topography on species richness of vascular plants, bryophytes and lichens in alpine communities. J Veg Sci 17:37–46
Butterfield BJ, Cavieres LA, Callaway RM, Cook BJ, Kikvidze Z, Lortie CJ, Michalet R et al (2013) Alpine cushion plants inhibit the loss of phylogenetic diversity in severe environments. Ecol Lett 16:478–486
Callaway RM, Brooker RW, Choler P, Kikvidze Z, Lortie CJ, Newingham B, Aschehoug ET, Armas C, Kikodze D, Cook BJ (2002) Positive interactions among alpine plants increase with stress. Nature 417:844–848
Carlsson BA, Callaghan TV (1991) Positive plant interactions in tundra vegetation and the importance of shelter. J Ecol 79:973–983
Casanova-Katny A, Cavieres LA (2012) Antarctic moss carpets facilitate growth of Deschampsia antarctica, but not its survival. Polar Biol 35:1869–1878
Cavieres LA, Quiroz CL, Molina-Montenegro MA, Muñoz AA, Pauchard A (2005) Nurse effect of the native cushion plant Azorella monantha on the invasive non-native Taraxacum officinale in the high-Andes of central Chile. Perspect Plant Ecol Syst 7:217–226
Cavieres LA, Badano EI, Sierra-Almeida A, Gómez-González S, Molina-Montenegro MA (2006) Positive interactions between alpine plant species and the nurse cushion plant Laretia acaulis do not increase with elevation in the Andes of central Chile. New Phytol 169:59–69
Cavieres LA, Brooker RW, Butterfield BJ et al (2014) Facilitative plant interactions and climate simultaneously drive alpine plant diversity. Ecol Lett 17:193–202
Choler P, Michalet R, Callaway RM (2001) Facilitation and competition on gradients in alpine plant communities. Ecology 82:3295–3308
Colwell RK (2013) EstimateS: statistical estimation of species richness and shared species from samples. Version 9.1.0. (Software and User’s Guide). http://purl.oclc.org/estimates
Damgaard C, Merlin A, Mesléard F, Bonis A (2011) The demography of space occupancy: measuring plant colonisation and survival probabilities using repeated pin-point measurements. Methods Ecol Evol 2:110–115
De Bello F, Dolezal J, Dvorsky M, Chlumska Z, Rehakova K, Klimesova J, Klimes L (2011) Cushions of Thylacospermum caespitosum (Caryophyllaceae) do not facilitate other plants under extreme altitude and dry conditions in the north-west Himalayas. Ann Bot 108:567–573
Dickson JH, Johnson RE (2014) Mosses and the beginnings of plant succession on the Walker Glacier, southeastern Alaska. Lindbergia 37:60–65
Donath TW, Eckstein RL (2010) Effects of bryophytes and grass litter on seedling emergence vary by vertical seed position and seed size. Plant Ecol 207:257–268
Doxford SW, Ooi MKJ, Freckleton RP (2013) Spatial and temporal variability in positive and negative plant–bryophyte interactions along a latitudinal gradient. J Ecol 101:465–474
Dvorsky M, Dolezal J, Kopecky M, Chlumska Z, Janatkova K, Altman J et al (2013) Testing the Stress-Gradient Hypothesis at the roof of the world: effects of the cushion plant Thylacospermum caespitosum on species assemblages. PLoS One 8:e53514
Erschbamer B, Niederfriniger Schlag R, Winkler E (2008) Colonization processes on a central Alpine glacier foreland. J Veg Sci 19:855–862
Erzberger P, Schröder W (2013) The genus Bryum (Bryaceae, Musci) in Hungary. Studia bot hung 44:5–192
Ferreyra M, Cingolani A, Ezcurra C, Bran D (1998) High-Andean vegetation and environmental gradients in northwestern Patagonia, Argentina. J Veg Sci 9(3):307–316
Gardes M, Dahlberg A (1996) Mycorrhizal diversity in arctic and alpine tundra: an open question. New Phytol 133:147–157
Garibotti IA, Pissolito CI, Villalba R (2011) Vegetation development on deglaciated rock outcrops from Glaciar Frías, Argentina. Arct Antarct Alp Res 43:35–45
Gomez-Aparicio L (2009) The role of plant interactions in the restoration of degraded ecosystems: a meta-analysis across life-forms and ecosystems. J Ecol 97:1202–1214
Gornall JL, Jonsdottir IS, Woodin SJ, Van der Wal R (2007) Arctic mosses govern below-ground environment and ecosystem processes. Oecologia 153:931–941
Gornall JL, Woodin SJ, Jonsdottir IS, Van der Wal R (2011) Balancing positive and negative plant interactions: how mosses structure vascular plant communities. Oecologia 166:769–782
Gotelli NJ, Colwell RK (2011) Estimating species richness. In: Magurran AE, McGill BJ (eds) Frontiers in measuring biodiversity. Oxford University Press, New York, pp 39–54
Gradstein SR, Churchill SP, Salazar-Allen N (2001) Guide to the bryophytes of tropical America. Memoirs of the New York Botanical Garden, vol 86
Groeneveld EVG, Rochefort L (2005) Polytrichum strictum as a solution to frost heaving in disturbed ecosystems: a case study with milled Peatlands. Restor Ecol 13:74–82
Groeneveld EVG, Masse A, Rochefort L (2007) Polytrichum strictum as a nurse-plant in Peatland restoration. Restor Ecol 15:709–719
Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A (2005) Very high resolution interpolated climate surfaces for global land areas. Int J Clim 25:1965–1978
Hobbie SE, Shevtsova A, Chapin FS (1999) Plant responses to species removal and experimental warming in Alaskan tussock tundra. Oikos 84:417–434
Jeschke M, Kiehl K (2008) Effects of a dense moss layer on germination and establishment of vascular plants in newly created calcareous grasslands. Flora 203:557–566
Jimenez MS, Suárez GM (2017) Philonotis pomangium (Bartramiaceae, Bryophyta): an addition to the Bryoflora of Peru. Bol Soc Argent Bot 52(2):341–350
Jimenez MS, Suárez GM, Schiavone MM (2014) Fruiting material of Philonotis esquelensis Matteri (Bartramiaceae, Bryophyta) discovered in Chile. Bol Soc Argent Bot 49(4):463–471
Jimenez MS, Suárez GM, Larraín J (2016) Rediscovery and lectotypification of Philonotis brevifolia Herzog. (Bartramiaceae, Bryophyta), a neglected species from Chile. Cryptogam Bryol 37(2):1–6
Johnstone JF, Chapin FS III, Hollingsworth TN, Me Mack, Romanovsky V, Turetsky M (2010) Fire, climate change, and forest resilience in interior Alaska. Can J For Res 40(7):1302–1312
Jones GA, Henry GHR (2003) Primary plant succession on recently deglaciated terrain in the Canadian High Arctic. J Biogeogr 30(2):277–296
Körner C (2003) Alpine plant life, 2nd edn. Springer, Berlin
Krna MA, Day TA, Ruhland CT (2009) Effects of neighboring plants on the growth and reproduction of Deschampsia antarctica in Antarctic tundra. Polar Biol 32:1487–1494
Larraín J (2007) Adiciones a la flora de musgos de la isla grande de chiloe, Chile. Gayana Bot 64(1):7–23
Maestre FT, Callaway RM, Valladares F, Lortie CJ (2009) Refining the stress-gradient hypothesis for competition and facilitation in plant communities. J Ecol 97:199–205
Malmer N, Albinsson C, Svensson BM, Wallen B (2003) Interferences between Sphagnum and vascular plants: effects on plant community structure and peat formation. Oikos 100:469–482
Marcante S, Kiebacher T, Erschbamer B (2013) Reproductive responses of glacier foreland species to simulated climate change. Coll Phytosociol 29:361–373
Michalet R, Xiao S, Touzard B, Smith DS, Cavieres LA, Callaway RM, Whitham TG (2011) Phenotypic variation in nurse traits and community feedbacks define an alpine community. Ecol Lett 14:433–443
Michalet R, Schob C, Lortie CJ, Brooker RW, Callaway RM (2014) Partitioning net interactions among plants along altitudinal gradients to study community responses to climate change. Funct Ecol 28:75–86
Molau U (1997) Age-related growth and reproduction in Diapensia lapponica, an arctic-alpine cushion plant. Nord J Bot 17:225–234
Morris WF, Doak DF (1998) Life history of the long-lived gynodioecious cushion plant Silene acaulis (Caryophyllaceae), inferred from size-based population projection matrices. Am J Bot 85:784–793
Navas ML, Violle C (2009) Plant traits related to competition: how do they shape the functional diversity of communities? Community Ecol 10:131–137
Niederfriniger Schlag R, Erschbamer B (2000) Germination and establishment of seedlings on a glacier foreland in the Central Alps, Austria. Arct Antarct Alp Res 32:270–277
Nuñez C, Aizen M, Ezcurra C (1999) Species associations and nurse plant effects in patches of high-Andean vegetation. J Veg Sci 10:357–364
Olofsson J, Kitti H, Rautiainen P, Stark S, Oksanen L (2001) Effects of summer grazing by reindeer on composition of vegetation, productivity and nitrogen cycling. Ecography 24:13–24
Olofsson J, Stark S, Oksanen L (2004) Reindeer influence on ecosystem processes in the tundra. Oikos 105:386–396
R Development Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org
Rayburn AP, Davidson JB, White HM (2012) Possible effects of moss on distribution and performance of a threatened endemic primrose. West N Am Nat 72:84–92
Rice WR (1989) Analyzing tables of statistical tests. Evolution 43:223–225
Roberts P, Newsham KK, Bardgett RD, Farrar JF, Jones DL (2009) Vegetation cover regulates the quantity, quality and temporal dynamics of dissolved organic carbon and nitrogen in Antarctic soils. Polar Biol 32:999–1008
Sand-Jensen K, Hammer KJ, Madsen-Østerbye M, Dencker T, Kragh T (2015) Positive interactions between moss cushions and vascular plant cover improve water economy on Öland’s alvar, Sweden. Botany 93(3):141–150
Schöb C, Armas C, Guler M, Prieto I, Pugnaire FI (2013) Variability in functional traits mediates plant interactions along stress gradients. J Ecol 101:753–762
Smith RIL (1988) Recording bryophyte microclimate in remote and severe environments. In: Glime JM (ed) Methods in bryology. Hattori Botanical Laboratory, Nichinan, pp 275–284
Sohlberg EH, Bliss LC (1984) Microscale pattern of vascular plant distribution in two High Arctic plant communities. Can J Bot 62:2033–2042
Startsev NA, LieVers VJ, McNabb DH (2007) Effects of feathermoss removal, thinning and fertilization on lodgepole pine growth, soil microclimate and stand nitrogen dynamics. For Ecol Manag 240:79–86
Startsev N, LieVers VJ, Landhausser SM (2008) Effects of leaf litter on the growth of boreal feather mosses: implication for forest floor development. J Veg Sci 19:253–260
Suárez GM, Schiavone MM (2011) Pohlia section Pohlia (Bryaceae) in Central and South America. Nova Hedwigia 92(3–4):453–477
Turetsky MR, Mack MC, Hollingsworth TN, Harden JW (2010) The role of mosses in ecosystem succession and function in Alaska’s boreal forest. Can J For Res 40:1237–1264
Van der Wal R, Brooker RW (2004) Mosses mediate grazer impacts on grass abundance in arctic ecosystems. Funct Ecol 18:77–86
Van der Wal R, Pearce ISK, Brooker RW (2005) Mosses and the struggle for light in a nitrogen-polluted world. Oecologia 142:159–168
Wang GX, Li YS, Wu QB, Wang Y (2006) Impacts of permafrost changes on alpine ecosystem in Qinghai-Tibet Plateau. Sci China Ser D Earth Sci 49(11):1156–1169
Yang Y, Chen JG, Schöb C, Sun H (2017) Size-mediated interaction between a cushion species and other non-cushion species at high elevations of the Hengduan mountains, SW China. Front Plant Sci 8:465
Zuur AF, Leno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology. Springer, New York
Acknowledgements
The authors thank two anonymous reviewers and A. Sáez for useful comments and suggestions. We are grateful to the Administración de Parques Nacionales, Argentina, which provided permission to work at the study sites. This research was supported by the National Research Council of Argentina (CONICET) doctoral scholarship to S.S.G. G.M.S, C.E. and M.A.A are career researchers of CONICET.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
SSG and MAA conceived the study. SSG collected field data. GMS identified the bryophytes. CE contributed materials and taxonomy expertise. SSG performed the analyses and wrote the first draft of the manuscript. All authors assisted with the writing of the manuscript and provided comments and edits.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
35_2019_222_MOESM1_ESM.pdf
Additional supporting information in the online version of this article (see Supplementary Material) contains: the list of additional studied sites assessing the effect of angiosperm-cushions plants, and the individual responses of vascular plant species to mosses (PDF 1747 kb)
Rights and permissions
About this article
Cite this article
Gavini, S.S., Suárez, G.M., Ezcurra, C. et al. Facilitation of vascular plants by cushion mosses in high-Andean communities. Alp Botany 129, 137–148 (2019). https://doi.org/10.1007/s00035-019-00222-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00035-019-00222-6