Abstract
Understanding the mechanisms structuring metacommunities is a fundamental question in community ecology. Ostracods are an ideal study group for metacommunity studies, due to their environmental sensitivity, high dispersal capacity and increased use as biological proxy in (paleo-)ecology studies. We studied the contribution of environmental and spatial processes in the structure of non-marine ostracod metacommunities in Northern and Southern Patagonia. A total of 37 ostracod species were found at the 105 environments studied. According to variation partitioning results, the metacommunities in Northern Patagonia were influenced by a combination of species sorting (environmental control, including non-spatial and spatial components) and spatial effects (e.g. dispersal limitation), while species sorting (environmental control, due to non-spatial components) was the principal mechanism affecting Southern Patagonia. Discrepancies between the degree of environmental and spatial control affecting ostracod metacommunity structure was associated with differences in study area extension and in sampled site distances, more than differences in Ostracoda response. Instead, the spatial extent did not influence total beta diversity and its components between the studied areas. Our study expands the understanding of the drivers affecting the non-marine ostracod metacommunities in Patagonia, Argentina, and also the variables affecting the structure of the ostracod assemblages. Increased knowledge in these aspects has important implications for (paleo-)ecology studies, allowing reliable interpretation in biological assessment programs and paleoenvironmental interpretation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A metacommunity can be defined as “a set of local communities that are linked by dispersal of multiple potentially interacting species” (Leibold et al. 2004). Currently, understanding the underlying drivers that shape a metacommunity is a crucial component of community ecology. In this context, and depending on the relative importance of environmental and spatial processes, four main models of metacommunity dynamics have been identified: species sorting, patch dynamics, mass effects, and neutral (Leibold et al. 2004). In species sorting, variation in local environmental features prevails, and species occupy suitable sites along environmental gradients. Although adequate dispersal rates are necessary so that species can track variation in environmental conditions among localities (Leibold et al. 2004), dispersal-related processes are the basis in patch dynamics, mass-effects and neutrality models (Alahuhta et al. 2014). The patch dynamic model is characterized by low rates of dispersal across a homogeneous landscape, in which communities are majority structured by competition colonization dynamics. In the mass-effects model, high dispersal rates allow species inhabiting adjacent localities regardless of their environmental conditions. The neutral perspective assumes that the species are ecologically equivalent and the community assemblage is driven by random speciation, extinction, migration and immigration (independently of the abiotic environment and species traits) (Leibold et al. 2004; Heino et al. 2015). Different authors have indicated that these models do not have clear boundaries, and the different mechanisms may have greater or lesser importance depending on spatial scale (Leibold et al. 2004; Cottenie 2005). In addition, it has been recently suggested that these perspectives should break down and that community studies should focus on the relative influence of spatial and environmental factors, rather than on the identification between the four paradigms (Rosati et al. 2016). In this context, metacommunity studies should focus on the relative influence of species sorting (i.e., environmental factors ‘filter’ species presence and abundance, which only occurs at environmentally suitable sites) and dispersal (i.e., a spatial process that affects a local community irrespective of local environmental conditions) (Heino et al. 2015).
The relevance of the environmental and the spatial influence is affected by the spatial extent; however, there is limited knowledge about the relative importance of these processes at different scales (He et al. 2020). Spatial extent is a key factor in metacommunity studies, having a profound effect on community inferences, so that one should expect different dispersal effects at different spatial extensions, and observations made at one scale cannot readily be extrapolated to others (Mykrä et al. 2007; Heino et al. 2015). Viana and Chase (2019) postulate that “if spatial scale influences conclusions about the relative importance of different assembly mechanisms, it would be critical to recognize, and account for this relationship”. In general, landscapes with a broader spatial extent tend to be associated with a larger influence of dispersal limitation, because suitable sites can be more separated than in landscapes with smaller spatial extents (Soininen 2016). According to He and coworkers (2020), it could be expected high dispersal rates in small spatial areas or in well-connected systems, leading to a strong spatial signal and a low influence of environmental control. At intermediate spatial scales, a metacommunity would show an increased relation between environment and community composition. Moderate spatial extensions enable more environmental heterogeneity and a weaker dispersal rate, so the species could track variation in environmental heterogeneity showing a strong environmental signal. Finally, at large spatial extents or in regions with major dispersal barriers, dispersal limitation leads to a strong spatial signal, as species are no longer able to effectively track variation in local environmental conditions (Heino et al. 2015; He et al. 2020). Related to the spatial extent, there is the difficult to define strict limits of a metacommunity. Defining when a spatial extent is too large to consider it ‘a metacommunity’ remains an open question, and defining the strict limits to their boundaries is difficult (Heino 2013). In this context, metacommunity studies ideally should consider various spatial ranges (Heino 2013).
On the other hand, spatial scale also can influence beta diversity patterns (i.e., heterogeny in community composition among sites), due to differential contributions of local and regional processes (Fu et al. 2019). Beta diversity can be partitioned into two different components: nestedness and turnover. Nestedness occurs when the sites with a smaller number of species represent subsets of sites with higher richness, reflecting a non-random process of species loss (or gain) along an environmental gradient. The turnover component implies the replacement of some taxa by others due to environmental filtering, or spatial and historical constraints (Baselga 2010). Both nestedness and turnover components contribute to the total compositional changes among communities; however, they imply distinct ecological processes. In this sense, the distinction of the turnover and nestedness components of beta diversity can enhance our understanding of the mechanisms that shape biodiversity patterns (Fu et al. 2019).
Ostracods are abundant in freshwater environments, being present in almost every aquatic system, including temporary environments, streams, estuaries, deep lakes and semi-terrestrial habitats (Mesquita-Joanes et al. 2012; Coviaga et al. 2018a; Cusminsky et al. 2020). The presence and relative abundance of these microcrustaceans are influenced by the physical and chemical features of host waters, such as electrical conductivity, temperature, energy level and dissolved oxygen concentration (Yassini and Jones 1995; Holmes 2001; Mezquita et al. 2005). Ostracods are passive dispersers with a relatively high dispersal rate, being able to identify abiotic environment changes by habitat tracking, reaching suitable patches more often and, therefore, being less affected by spatial structure than low-dispersal organisms (Padial et al. 2014; Michelson et al. 2016). Previous results showed that environmental factors (species-sorting mechanism) contribute significantly in structuring the entire ostracod metacommunity (Cottenie 2005; Michelson et al. 2016; Castillo-Escrivà et al. 2017; de Campos et al. 2018). In contrast, other studies found that both environment and space significantly explain the assemblage variation of ostracod communities (Escrivà et al. 2015; Zhai et al. 2015; Rossatti et al. 2016). In Patagonia, environmental studies on ostracods have been often conducted on regional scale (e.g. Schwalb et al. 2002; Cusminsky et al. 2005; 2020; Ramón Mercau et al. 2012; Coviaga 2016; Coviaga et al. 2015, 2018a; Ramos et al. 2015; 2017); nevertheless, the influence of space itself remains uncertain. In this sense, due to their environmental sensitivity and high dispersal capacity, non-marine ostracods are an ideal study group for evaluating the spatial influence on metacommunity structure and composition (Michelson et al. 2016). Additionally, given that ostracods are increasingly used as biological and paleoecological indicators, understanding the influence of environmental and spatial processes on their metacommunity structure has important implications for trustworthy biological and paleoenvironmental interpretations (Castillo-Escrivà et al. 2016).
The aim of this study is to compare the relative contribution of environmental and spatial processes in the assemblage structure and composition of non-marine ostracod metacommunities in two different areas of Argentina, Northern and Southern Patagonia, and separate the unique and shared effects of these two categories of variables. We hypothesized that (H1) Ostracoda metacommunities are mainly influenced by environmental parameters and that (H2) the spatial processes are more relevant in the larger study area.
Study region and methods
Study area
Argentine Patagonia is located in the southern part of South America extending southward of Río Colorado, between 36° S to 55° S, and covering 28.5% of Argentine continental territory (790,000 km2) (Rabassa 2008). This region presents a large latitudinal and longitudinal extension, which guarantees a wide range of climatic conditions and a broad environmental heterogeneity. In this context, studied sites were located in four ecoregions: Andean-Patagonian forest, Patagonian steppe, Monte, and Espinal; defined according to climate, geomorphology, soils and vegetation (Burkart et al. 1999). In the Andean-Patagonian forest, the presence of the Andean mountains provides a glacial to fluvial geomorphology. In this ecoregion, the climate is wet (800 to 3500 mm/year of precipitation), temperate to cool (5.4 to 9 °C mean annual temperature), and the soils are rich in organic matter (Burkart et al. 1999). The Patagonian steppe ecoregion is located to the east of the Andean-Patagonian forest, representing about 85% of Patagonia territory. This large ecoregion is characterized by a tableland relief, forming numerous landscape steps from the Andean piedmont to the Atlantic Ocean coast (Paruelo et al. 1998). Their soils are poorly developed, with a low organic matter contents and scrubby vegetation (Paruelo et al. 2005). The climate of the Patagonian steppe is cool temperate, with a mean annual temperature range from 10 to 14 °C in the north to 5 to 8 °C to the south of the region, and dry, with low precipitations (200 mm/year) (Burkart et al. 1999). The Monte ecoregion comprises a relief of steppes and plains; with soils poorly developed and a vegetation mainly represented by scrub steppe. Also, it is characterized by a climate temperate (mean annual temperature range from 10 to 14 °C) and dry (precipitation between 300 and 800 mm/year). The Espinal ecoregion presents a relief with drained plains, gentle slopes and poorly developed soils. The climate is warm (temperature between 15 and 16 °C) and semiarid (precipitation from 300 to 600 mm/year, temperature between 15 and 16 °C) (Menendez and La Roca 2006).
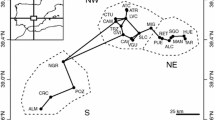
In this study, two different areas were considered: Northern (59 sites) and Southern Patagonia (46 sites) (Fig. 1). Additionally, sampling sites were selected to represent a heterogeneous gradient of freshwater habitat types: permanent, semi-permanent and temporary pond as well as lentic, spring and stream environments.
Field sampling and sample processing
A total of 105 permanent, ephemeral, pond and spring freshwater bodies of Patagonia were sampled in the course of this and other investigations (Schwalb et al. 2002; Coviaga et al. 2015; 2018a; Ramón Mercau et al. 2012; Ramón Mercau and Laprida 2016; Pérez et al. 2019) (see Online Resource 1, Table S1 for more details). At each site, ostracods were collected from surface sediment and/or from the water–sediment interface. Sediment samples were taken by scraping over the sediment surface with a plastic bag, using an Ekman grab sampler from a rubber boat or with a bolapipe dredge. The water–sediment interface samples were recovered using a hand net (D frame 200-µm mesh aperture) along a 1–6-m long transect, or with 12-ml glass bottles with a silicon septum. Samples were fixed with ethanol (70%). Electrical conductivity (µScm−1) was simultaneously measured in situ. For more methodologic details, see Schwalb et al. 2002; Coviaga et al. 2015, 2018a; Ramón Mercau et al. 2012; Ramón Mercau and Laprida 2016; and Pérez et al. 2019.
All adult ostracod individuals were sorted under a stereomicroscope. Representative specimens were measured and dissected under stereoscopic microscopes (Olympus SZ30 and SZ61 and Nikon SMZ-645). Taxonomic identification was done based on carapaces, valves and appendages following Van Morkhoven (1963), Martens (1990), Cusminsky and Whatley (1996), Meisch (2000), Cusminsky et al. (2005), Karanovic (2012), Coviaga et al. (2018b) and Pérez et al. (2019).
Explanatory variables
The effects of two types of explanatory variables on ostracod distribution and abundance were evaluated, environmental and spatial variables. The environmental group included electrical conductivity and climatic variables. We have chosen electrical conductivity because it is known to strongly affect the distribution and abundance of Patagonian non-marine ostracods, and this has been the only variable exhaustively recorded in studies of ostracods in the region (Cusminsky et al. 2005, 2020; Ramón-Mercau et al. 2012; Coviaga et al. 2018a).
The historical (1970–2000) climate data for South America was obtained from WorldClim data set version 2.1 (Fick and Hijmans 2017). For this data set, we selected the wind speed and the 19 bioclimatic variables derived from the monthly temperature and rainfall; representing annual trends, seasonality and extreme or limiting environmental factors. The variables were obtained at the spatial resolution of 30 s (approximately 1 × 1 km per pixel), which is an adequate coarse resolution where climate influences species distributions (Pearson and Dawson 2003).
Spatial variables comprised the second group of explanatory variables and were generated as Moran’s Eigenvector Maps (MEMs) from two different geographical coordinates matrices (one for Northern Patagonia and the other for Southern Patagonia) (Dray et al. 2012; Legendre and Legendre 2012), using the R package ‘adespatial’ (Dray et al. 2018). MEMs are a set of orthogonal (linearly independent) eigenvectors computed from a multiscale analysis that represent the spatial relationships among study sites and can be used to detect complex patterns of spatial variation at different scales (Dray et al. 2012; Legendre and Legendre 2012; Castillo-Escrivà et al. 2017). In Southern Patagonia, spatial structure was not detected (i.e., we did not detect a significant positive correlation between the relative species abundance and the MEMs, i.e., the geographic variation). In order to determine whether this was a result of an irregular sampling, the correction suggested by Brind'Amour (the complete-grid approach in cases of irregular sampling) was applied to compute the MEMs (Brind'Amour et al. 2018). Different types of spatial connectivity criteria (Gabriel graph, relative neighborhood graph and minimum spanning tree) and weight (binary without weights, linear weighting function) were tested for the two matrices, preserving only the eigenvectors with positive autocorrelation (Dray et al. 2018). For Northern Patagonia sites, a relative neighborhood graph connection network with a linear weighting function generated the spatial weighting matrix that computed the eigenvector subset with the highest adjusted R2. In Southern Patagonia, a spatial weighting matrix based on a relative neighborhood graph connection network with a binary without weights assessed the eigenvector subset with the highest adjusted R2. A forward selection procedure with double-stopping criterion was implemented to select only the significant subset of MEMs (R package ‘adespatial’; Dray 2018). For these MEMs, smaller eigenvectors were associated with broader-scale patterns (i.e., at a watershed level, linked to biological constraints as climate, dispersion and colonization), whereas higher eigenvectors were associated with finer-scale variations (i.e., at a microhabitat level, related to biotic interactions and/or environmental filtering) (Gascón et al. 2012; Benito et al. 2018).
Data analysis
Correlation between electrical conductivity, wind speed and the 19 bioclimatic variables was checked running a Spearman analysis using the package ‘corrplot’ (Wei and Simko 2017) for species matrices of Northern and Southern Patagonia; and only uncorrelated data (r < 0.7) were kept. Assumption of normality was previously evaluated using the Lilliefors test, with the package ‘nortest’ (Gross and Ligges 2015).
The similarity between Northern and Southern Patagonia species assemblages was tested by the Jaccard/Tanimoto similarity index, using the ‘jaccard’ package (Chung et al. 2019).
Because ostracod samples were obtained with different methods and were of different sizes, we used the relative species abundance as response variable in our analyses to allow for comparison between samples. These relative abundances were Hellinger-transformed (Peres-Neto et al. 2006), which is the appropriate method for matrices with abundant zeros (Legendre and Gallagher 2001). We also tested the response data (i.e., relative species abundance) for linear trends and detrended them if the trend surface was significant to remove the effect of a linear gradient. Eliminating the linear spatial trend in the data (i.e., the directional component) allows us to detect possible patterns at finer scales (Borcard and Legendre 2002; Borcard et al. 2004; Dray et al. 2006). The ‘vegan’ package was used for Hellinger transformation. From explanatory data, only conductivity values were log-transformed prior to ordinations.
Exclusive and shared effects of environmental and spatial variables on species composition were determined using a partial redundancy analysis (pRDA; Legendre and Legendre 2012), with adjusted canonical R2 values (Legendre and Legendre 2012). A forward selection with 999 permutations was used to identify variables that significantly explained (p < 0.05) ostracod abundance and distribution (ter Braak and Prentice 1988; ter Braak and Šmilauer 2012). For this analysis, the correction method proposed by Clappe et al. (2018) was used to assess the importance of spatialized and non-spatialized environmental drivers. In nature, the environment and space are usually intercorrelated, making it difficult to distinguish their particular influences. Therefore, our conclusions would not be the same if the shared fraction were fully or majority linked to the environment, or conversely if the shared effect could be a consequence of dispersal-related processes (Rosati et al. 2016). In this sense, Clappe’s method allows us to separate the spatialized (i.e., spatially autocorrelated) contribution of the environment that is due to spurious correlations from the contributions of the environment to species sorting. As such, this variation partitioning approach allowed us to determine the variation explained by the non-spatially structured environmental (E) fraction, the pure spatial (S) factors, the overlap between environment and space (E \(\cap\) S; spatialized environmental variation) and the unexplained fraction (U).
Both for Northern and Southern Patagonia, the total beta diversity and their two components (turnover and nestedness) were calculated using the species beta diversity partitioning proposed by Baselga (2010). This approach was based in three dissimilarity coefficients: (1) Sørensen coefficient, a measure of total beta diversity, (2) Simpson coefficient, a measure of turnover, and (3) a coefficient assessing nestedness richness differences (Baselga 2010).
Statistical procedures were performed with R version 3.5.2 (R Core Team 2018).
Results
A total of 37 ostracod species were found in the 105 environments studied. Both regions shared 13 species, while 14 species were recorded exclusively in Northern Patagonia and 10 in Southern Patagonia (Table 1). The Jaccard/Tanimoto similarity index showed a very small similarity (uncentered = 0.31, centered = −0.17, p = 0.007) between the community composition of Northern and Southern Patagonia. The most frequently occurring species were Ilyocypris ramirezi and Cypridopsis silvestrii, occurring at 40 and 21 sites in Northern and Southern Patagonia, respectively. Five species (Limnocythere cusminskyae, Darwinula stevensoni, Typhlocypris sp., Cypridinae indet. 1, Isocypris beauchampi) were found only at single sites.
The uncorrelated subset of environmental variables for Northern Patagonia sites were compose by mean annual temperature (MAT), isothermality (ISO), minimum temperature of the coldest month (MTCM), mean temperature of the driest quarter (MTDQ), annual precipitation (AP), precipitation of the warmest quarter (PWQ), wind speed (wind) and electrical conductivity. Of these, environmental RDA results indicated that four variables explained significantly the Ostracoda variability: AP, electrical conductivity, MAT and MTCM. Variance partitioning analysis for Northern Patagonia showed that the spatial variables explained the highest portion of ostracod variability (14.0%), followed by the spatialized environmental variables (6.8%) and the environmental variables (4.3%) (Fig. 2). For Southern Patagonia, the uncorrelated variables were mean annual temperature (MAT), isothermality (ISO), mean temperature of the wettest quarter (MTWQ), annual precipitation (AP), wind speed (wind) and electrical conductivity. Three of these variables (MAT, electrical conductivity and wind) explain significantly the ostracod assemblage composition. The variation partitioning analysis displayed that the pure environmental portion explained the highest portion of variability (14.6%), followed by the overlap fraction of space and environment (5.0%). The spatial variables do not explain a significant proportion of Southern Patagonian ostracod composition (Fig. 2).
Venn diagrams of the variation partitioning analysis, showing the fractions of ostracod metacommunity structure explained by the shared and pure effects of environmental (E) and spatial (S) variables. The explained variation (%) is based on adjusted R2 values (** denotes p < 0.005). U represents the unexplained fraction
Both Northern and Southern Patagonia ostracod metacommunities showed similar results of beta diversity, with intermediate levels of total beta diversity and a variation in community composition almost entirely due to species turnover with a negligible portion attributable to the nestedness component (Table 2).
Discussion
Assessing the relative importance of environmental and spatial processes on community assembly is one of the key approaches for enhancing our basic understanding of metacommunity dynamics (Heino 2011; García-Girón et al. 2019). In this context, this is the first study to evaluate the relative importance of environmental and spatial factors at broadscale distributional patterns of freshwater ostracods in Patagonia.
Variance partitioning analysis partly supported our hypotheses (H1 and H2), indicating that both spatial and environmental (non-spatially and spatially structured) variables contributed equally and significantly in structuring ostracod metacommunities in Northern Patagonia, whereas only the environmental variables significantly influenced the ostracod assemblage in Southern Patagonia. Previous studies carried out on ostracods are not conclusive, showing different effects of the space over their metacommunities (Gansfort et al. 2020). Existing investigations on this group have shown a high pure spatial effect over their metacommunities (as in our Northern Patagonia results, and in e.g. Escrivà et al. 2015; Zhai et al. 2015; Rosati et al. 2016), whereas in other surveys it has been found that the relative effect of the space was low and not significant (as in our Southern Patagonia results, and in e.g. Michelson et al. 2016; Castillo-Escrivà et al. 2017; de Campos et al. 2018). Probably, much of the variation between these results is due to differences in habitat connectivity, spatial extent, and environmental heterogeneity (Gansfort et al. 2020).
Our results suggest that both species sorting and spatial effects contributed to the structure of ostracod communities of the North, while only the species-sorting mechanisms seem to be structuring the Ostracoda metacommunity of South Patagonia. Species sorting has long been recognized as a key mechanism underlying metacommunity structure, in which community composition is driven by environmental conditions (Leibold et al. 2004). Ideally, one should estimate the contribution of the total environmental variation containing both its non-spatial and spatial components (Clappe et al. 2018). In Northern Patagonia, most of the environmental variables were spatially autocorrelated (fraction [ab]). Hence, the correction proposed by Clappe and coworkers (2018) allowed us to consider this fraction as part of the environmental contribution to species distribution, instead of only considering the fraction [a] as an indicator of species sorting as in previous studies. In Southern Patagonia, because the complete-grid approach was applied to compute the MEMs, it was not possible to apply the method proposed by Clappe et al. (2018) and therefore separate the effects of spurious correlation from the spatial structure of the environment in the fraction [ab]. Probably the environmental contribution be superior to that considered in this study, given that fraction [ab] is greater than [a]. With respect to the spatial influence in structuring the ostracod metacommunities in Patagonia, our results showed that in the North, the pure space effects were stronger. This suggests that despite dispersal ostracod adaptations (e.g. small propagules, drought-resistant eggs, parthenogenesis reproduction), the dispersion may be limited within this studied area (Escrivà et al. 2015; Zhai et al. 2015; Castillo-Escrivà et al. 2016; Rosati et al. 2016). In Southern Patagonia, the pure fraction attributed to the space was not significant, suggesting that the dispersal of ostracods was high enough so all species can reach all lakes, and therefore the abiotic environment of each lake is the primary determinant of species composition in that lake (Michelson et al. 2016).
The discrepancies between the degree of environmental control and spatial structuring in the studied regions could be due to differences in the extension of study areas (almost twice larger in Northern than Southern Patagonia), and in relation with this, due to differences in the distance between sampled sites (average distance 247 km and 119 km, and maximum distance 715 km and 556 km in Northern and Southern Patagonia, respectively). In this sense, a higher spatial influence is expected at a broader spatial scale (Heino et al. 2015), due to the importance of a dispersal-related processes is likely to increase with increasing spatial extent in a metacommunity context (Leibold et al. 2014; Cottenie 2005). In agreement, studies including different spatial scales on meiofauna metacommunities showed that species-sorting relevance was scale-dependent, with less significance at larger scales, which were dominated by dispersal processes (Gansfort et al. 2020). Additionally, we cannot rule out that the high spatial structure significance in Northern Patagonia could be an artifact of unmeasured environmental variables or an imbalance between the number of environmental and spatial variables (more than double than environmental variables). It has been suggested that the degree of environmental influence is positively correlated with the number of environmental variables included (Soininen 2014).
Conductivity and mean annual temperature (MAT) contributed significantly to the structure of ostracod assemblages in both north and south regions. The significant influence of electrical conductivity on species composition of ostracod assemblages has been repeatedly demonstrated across various aquatic habitats (Mesquita-Joanes et al. 2012; Zhai et al. 2015; Gansfort et al. 2020). Previous studies in Patagonia have shown that conductivity has special relevance in the distribution, abundance and life cycle of Patagonian ostracods (Schwalb et al. 2002; Cusminsky et al. 2005; 2020; Ramón Mercau et al. 2012; Coviaga 2016; Coviaga et al. 2015, 2018a). In line with this, ostracods are widely used as a bioproxy for conductivity paleo-reconstructions (Whatley and Cusminsky 1999; Cusminsky et al. 2011; Rodriguez-Lazaro and Ruiz-Muñoz 2012; Coviaga et al. 2017; Coviaga et al. 2018c; Borromei et al. 2018; Ramos et al. 2019). Similarly, water temperature is a key factor for distribution, survival, growth and reproduction of ostracods (Mesquita-Joanes et al. 2012 and references cited therein). The temperature relevance on ostracods has been shown in different studies carried out in Patagonia (Schwalb et al. 2002; Cusminsky et al. 2005, 2020, 2018a; Ramón Mercau et al. 2012; Coviaga 2016; Coviaga et al. 2015). Likewise, the minimum temperature of the coldest month (MTCM) explains significantly the Northern Patagonia ostracod distribution. This index represents the minimum monthly temperature occurrence over a given year or averaged span of years, information which is useful when analyzing species distributions that are affected by cold temperatures (O'Donell and Ignizio 2012). Previous studies showed that the minimum air temperature of the coldest month predicted the Ostracoda potential distribution in the south of South America (Conceição et al. 2020). In addition, some species recorded in Northern Patagonia were associated with cold waters (Coviaga et al. 2018a). Annual precipitation (AP) also influences the ostracod metacommunity of North Patagonia and the distribution of total water inputs, which is useful when ascertaining the importance of water availability (temporal vs. permanent) to the species distribution (O'Donell and Ignizio 2012). In most areas of Patagonia, with arid and semiarid characteristics, water is a scarce resource and thus limits the presence of waterbodies and distribution of ostracod habitats. Wind significantly explained the distribution of Southern Patagonian ostracods. This is considered an important vector for dispersal and, therefore, can be a significant driver of metapopulation dynamics (Pinceel et al. 2020 and references therein). It has been shown that wind can transport desiccation-resistant eggs of aquatic organisms over distances of 100 km (Rivas et al. 2019), so wind could allow ostracod (individuals and resistant eggs) dispersal over regional distances (Meisch 2000 and references therein). In particular in Patagonia, wind is a key factor, as it is the only region which is permanently affected by the westerly wind belt that influences the regional biota composition and distribution (Villa Martínez and Moreno 2007). In this sense, Ramos and coworkers (2022) have recently shown that wind speed was the main climatic predictor of Patagonian ostracod abundance, resulting in higher abundances in windy areas.
Our variation partitioning results showed that a considerable percentage of the variation was not explained by the environmental or by the spatial variables studied. This is relatively common in metacommunity studies (Alahuhta et al. 2014; Bispo et al. 2017), especially for those with a large numbers of sites and species (Zhai et al. 2015). We suggest that the fraction of unexplained variation could be related to some important variables unmeasured and biotic interactions ignored that affect the ostracod biota (Bispo et al. 2017; Lindholm et al. 2018). Moreover, we have to consider possible effects of the sampling design, as we sampled the ostracod communities only once, which reduced the probability of obtaining complete species lists for sites (Zhai et al. 2015). On the other hand, the unexplained variation related to stochasticity (probably inherent in communities) can also be responsible for the structuring.
Regarding the ostracod metacommunity composition, our beta diversity analysis showed similar results in Northern and Southern Patagonia. In both regions, we found intermediate levels of total beta diversity, which could be almost entirely attributed to species turnover. This suggests that total beta diversity and associated components would not be influenced by the spatial scale between the studied areas. Hill and coworkers (2018) found an increase of the turnover component and total beta diversity with study extent; however, they explain that such finding must be interpreted with care, as their data set included a few smaller-scale studies, and multiple site beta-diversity metrics did not vary with scale. On the other hand, the higher contribution of the turnover component in total beta diversity has been verified in different studies. Hill et al. (2018) found that turnover is typically much higher than the nestedness component (in some data sets even approaching zero, like in this study), and this suggests that dissimilarity among sites was mainly driven by variation in community composition rather than differences in taxonomic richness (Hill et al. 2017). Particularly in Patagonia, Epele and coworkers (2019) identified species turnover as the most important component influencing the freshwater invertebrate’s beta diversity from isolated ponds.
Non-marine ostracods are increasingly used as biological indicators in paleolimnology and paleoclimatology studies, as well as in pollution monitoring and management and environmental change studies, being of utmost importance enhancing our knowledge about their ecology and taxonomy (e.g. Külköylüoğlu 2004; Castillo-Escrivà et al. 2016; Michelson et al. 2016, and references therein; Coviaga et al. 2018a). For their use as bio- and paleo-indicators, a direct relationship between the environmental variables and the metacommunity structure is assumed (Michelson et al. 2016). Thus, spatial processes could lead to misinterpretations of the relationships between analyzed environmental variables and observed community structure and diversity (Castillo-Escrivà et al. 2016). In this context, studies combining the environmental and spatial roles, like this, are fundamental for a better understanding of the main processes structuring the metacommunities, and therefore for a trustworthy interpretation in biological assessment programs and paleoenvironmental interpretations (Castillo-Escrivà et al. 2016).
Conclusion
This represents the first study evaluating the relative importance of environmental and spatial factors at broadscale distributional patterns of freshwater ostracods in Patagonia. Understanding the influence of environmental and spatial processes on the metacommunities’ structure has important implications for both ecology and applied paleoecology. Our results showed that the structures of Northern Patagonia metacommunity were influenced by a combination of species sorting (environmental control) and spatial effects (dispersal process). On the other hand, the Southern Patagonia metacommunity was structured only by species-sorting mechanisms. The differences between environmental and spatial roles in structuring both Northern and Southern Patagonia Ostracoda metacommunities could be due to discrepancy in the extension of study areas, more than differences in Ostracoda response. Conversely, the spatial scale differences did not affect the beta diversity of non-marine ostracod metacommunities from Northern and Southern Patagonia. The strong and consistent relationships between community structure and the environment in Patagonia suggest that we can use these organisms as bio- and paleo-indicators, recognizing a priori how is influencing the space in the metacommunities.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Alahuhta J, Johnson LB, Olker J, Heino J (2014) Species sorting determines variation in the community composition of common and rare macrophytes at various spatial extents. Ecol Complex 20:61–68. https://doi.org/10.1016/j.ecocom.2014.08.003
Baselga A (2010) Partitioning the turnover and nestedness components of beta diversity. Glob Ecol Biogeogr 19:134–143. https://doi.org/10.1111/j.1466-8238.2009.00490.x
Benito X, Fritz SC, Steinitz-Kannan M, Vélez MI, Mcglue MM (2018) Lake regionalization and diatom metacommunity structuring in tropical South America. Ecol Evol 8:7865–7878. https://doi.org/10.1002/ece3.4305
Bispo PdC, Balzter H, Malhi Y, Slik JWF, dos Santos JR, Rennó CD, Espírito-Santo FD, Aragão LEOC, Ximenes AC, Bispo PdC (2017) Drivers of metacommunity structure diverge for common and rare Amazonian tree species. PLoS ONE 12(11):0188300. https://doi.org/10.1371/journal
Borcard D, Legendre P (2002) All-scale spatial analysis of ecological data by means of principal coordinates of neighbour matrices. Ecol Modell 153:51–68
Borcard D, Legendre P, Avois-Jacquet C, Toumisto H (2004) Dissecting the spatial structure of ecological data at multiple scales. Ecology 85:1826–1832
Brind’Amour A, Mahévas S, Legendre P, Bellanger L (2018) Application of Moran Eigenvector Maps (MEM) to irregular sampling designs. Spat Stat 26:56–68. https://doi.org/10.1016/j.spasta.2018.05.004
Burkart R, Bárbaro NO, Sánchez RO, Gómez DA (1999). Eco- regiones de la Argentina. Programa Desarrollo Institucional Ambiental.
Castillo-Escrivà A, Valls L, Rochera C, Camacho A, Mesquita-Joanes F (2016) Spatial and environmental analysis of an ostracod metacommunity from endorheic lakes. Aquat Sci 78:707–716. https://doi.org/10.1007/s00027-015-0462-z
Castillo-Escrivà A, Valls L, Rochera C, Camacho A, Mesquita-Joanes F (2017) Metacommunity dynamics of Ostracoda in temporary lakes: overall strong niche effects except at the onset of the flooding period. Limnologica 62:104–110. https://doi.org/10.1016/j.limno.2016.11.005
Chung NC, Miasojedow BZ, Startek M, Gambin A (2019) Jaccard/Tanimoto similarity test and estimation methods for biological presence-absence data. BMC Bioinform 20:1–11. https://doi.org/10.1186/s12859-019-3118-5
Clappe S, Dray S, Peres-Neto PR (2018) Beyond neutrality: disentangling the effects of species sorting and spurious correlations in community analysis. Ecology 99:1737–1747. https://doi.org/10.1002/ecy.2376
Cottenie K (2005) Integrating environmental and spatial processes in ecological community dynamics. Ecol Lett 8:1175–1182. https://doi.org/10.1111/j.1461-0248.2005.00820.x
Coviaga C, Cusminsky G, Baccalá N, Pérez AP (2015) Dynamics of ostracod populations from shallow lakes of Patagonia: life history insights. J Nat Hist 49:1023–1045. https://doi.org/10.1080/00222933.2014.981310
Coviaga CA, Rizzo A, Pérez AP, Daga R, Poiré D, Cusminsky G, Ribeiro Guevara S (2017) Reconstruction of the hydrologic history of a shallow Patagonian steppe lake during the past 700 yr, using chemical, geologic, and biological proxies. Quat Res 87:208–226. https://doi.org/10.1017/qua.2016.19
Coviaga CA, Cusminsky GC, Pérez AP (2018a) Ecology of freshwater ostracods from Northern Patagonia and their potential application in paleo-environmental reconstructions. Hydrobiologia 816:3–20. https://doi.org/10.1007/s10750-017-3127-1
Coviaga CA, Pérez AP, Ramos LY, Alvear P, Cusminsky GC (2018b) On two species of Riocypis (Crustacea, Ostracoda) from Northern Patagonia and their relation to Eucypris fontana; implications in paleo-environmental reconstructions. Can J Zool 96:801–817. https://doi.org/10.1139/cjz-2017-0233
Coviaga C, Cusminsky G, Pérez AP, Schwalb A, Markgraf V, Ariztegui D (2018c) Paleoenvironmental changes during the last 3000 years in Lake Cari-Laufquen (Northern Patagonia, Argentina), inferred from ostracod paleoecology, petrophysical, sedimentological and geochemical data. The Holocene 28:1881–1893
Coviaga C (2016) Ostrácodos lacustres actuales de Patagonia Norte y su correspondencia con secuencias holocénicas. PhD Thesis, Universidad Nacional del Comahue.
Cusminsky GC, Whatley R (1996) Quaternary non-marine ostracods from lake beds in northern Patagonia. Revista Española De Paleontología 11:143–154
Cusminsky GC, Pérez AP, Schwalb A, Whatley R (2005) Recent lacustrine ostracods from Patagonia, Argentina. Revista Española De Paleontología 37:431–450
Cusminsky G, Schwalb A, Pérez AP, Pineda D, Viehberg F, Whatley R, Markgraf V, Gilli A, Ariztegui D, Anselmetti FS (2011) Late quaternary environmental changes in Patagonia as inferred from lacustrine fossil and extant ostracods. Biol J Linn Soc 103:397–408. https://doi.org/10.1111/j.1095-8312.2011.01650.x
Cusminsky G, Coviaga C, Ramos L, Pérez AP, Schwalb A, Markgraf V, Ariztegui D, Viehberg F, Alperín M (2020) Characterizing ecoregions in Argentinian Patagonia using extant continental ostracods. Anais Acad Brasil Ci. 92:20190459. https://doi.org/10.1590/0001-3765202020190459
de Campos R, Lansac-Tôha FM, de Oliveira DA, Conceição E, Martens K, Higuti J (2018) Factors affecting the metacommunity structure of periphytic ostracods (Crustacea, Ostracoda): a deconstruction approach based on biological traits. Aquat Sci 80:16. https://doi.org/10.1007/s00027-018-0567-2
de Oliveira DA, Conceição E, Mantovano T, de Campos R, Fernando RT, Martens K, Bailly D, Higuti J (2020) Mapping the observed and modelled intracontinental distribution of non-marine ostracods from South America. Hydrobiologia 847:1663–1687. https://doi.org/10.1007/s10750-019-04136-6
Dray S, Legendre P, Peres-Neto PR (2006) Spatial modelling: a comprehensive framework for principal coordinate analysis of neighbor matrices (PCNM). Ecol Modell 196:483–493. https://doi.org/10.1016/j.ecolmodel.2006.02.015
Dray S, Pélissier R, Couteron P, Fortin MJ, Legendre P, Peres-Neto PR, Bellier E, Bivand R, Blanchet FG, De Cáceres M, Dufour AB, Heegaard E, Jombart T, Munoz F, Oksanen J, Thioulouse J, Wagner HH (2012) Community ecology in the age of multivariate multiscale spatial analysis. Ecol Monogr 82:257–275. https://doi.org/10.1890/11-1183.1
Dray S, Blanchet G, Bocard D, Clappe S, Guenard G, Jombart T, Larocque G, Legendre P, Madi N, Wagner HH (2018) Package ‘adespatial’. Retrieved from January 7, 2021 from https://cran.r-project.org/web/packages/adespatial/
Epele LB, Brand C, Miserendino ML (2019) Environment Ecological drivers of alpha and beta diversity of freshwater invertebrates in arid and semiarid Patagonia (Argentina). Sci Total Environ 678:62–73. https://doi.org/10.1016/j.scitotenv.2019.04.3920048-9697
Escrivà A, Poquet JM, Mesquita-Joanes F (2015) Effects of environmental and spatial variables on lotic ostracod metacommunity structure in the Iberian Peninsula. Inland Waters 5:283–294. https://doi.org/10.5268/IW-5.3.771
Fick SE, Hijmans RJ (2017) WorldClim 2: new 1-km spatial resolution climate surfaces for global land areas. Int J Climatol 37:4302–4315. https://doi.org/10.1002/joc.5086
Fu A, Yuan G, Jeppesen E, Ge D, Li W, Zou D, Huang Z, Wu A, Liu Q (2019) Local and regional drivers of turnover and nestedness components of species and functional beta diversity in lake macrophyte communities in China. Sci Total Environ 687:206–217. https://doi.org/10.1016/j.scitotenv.2019.06.092
Gansfort B, Fontaneto D, Zhai M (2020) Meiofauna as a model to test paradigms of ecological metacommunity theory. Hydrobiologia 847:2645–2663. https://doi.org/10.1007/s10750-020-04185-2
Gascón S, Machado M, Sala J, Cancela de Fonseca L, Cristo M, Boix D (2012) Spatial characteristics and species niche attributes modulate the response by aquatic passive dispersers to habitat degradation. Mar Fresh Res 63:232–245. https://doi.org/10.1071/MF11160
Gross J, Ligges U (2015) nortest: Tests for Normality. R package version 1.0–4. Available from: https://CRAN.R-project.org/package=nortest
He S, Soininnen G, Deng G, Wang c (2020) Metacommunity structure of stream insects across three hierarchical spatial scales metacommunity structure of stream insects across three hierarchical spatial scales. Ecol Evol 10: 2874–2884
Heino J (2011) A macroecological perspective of diversity patterns in the freshwater realm. Freshw Biol 56:1703–1722. https://doi.org/10.1111/j.1365-2427.2011.02610.x
Heino J, Melo AS, Siqueira T, Soininen J, Valanko S, Bini LM (2015) Metacommunity organisation, spatial extent and dispersal in aquatic systems: patterns, processes and prospects. Freshw Biol 60:845–869. https://doi.org/10.1111/fwb.12533
Holmes JA (2001) Ostracoda. In: Smol SP, Birks HJB, Last WM (eds) Tracking environmental change using lake sediments, vol 4. Zoological. Kluwer Academic Publishers, Dordrecht, The Netherlands, pp 125–151
Karanovic I (2012) Recent freshwater ostracods of the world. Crustacea, Ostracoda, Podocopida. Springer, Berlin.
Külköylüoğlu O (2004) On the usage of ostracods (Crustacea) as bioindicator species in different aquatic habitats in the Bolu region, Turkey. Ecol Indic 4:139–147. https://doi.org/10.1016/j.ecolind.2004.01.004
Legendre P, Gallagher ED (2001) Ecologically meaningful transformations for ordination of species data. Oecologia 129:271–280. https://doi.org/10.1007/s004420100716
Legendre P, Legendre L (2012) Numerical Ecology, 3rd edn. Elsevier, Oxford
Leibold MA, Holyoak M, Mouquet N, Amarasekare P, Chase JM, Hoopes MF, Holt RD, Shurin JB, Law R, Tilman D, Loreau M, Gonzalez A (2004) The metacommunity concept: a framework for multi-scale community ecology. Ecol Lett 7:601–613. https://doi.org/10.1111/j.1461-0248.2004.00608.x
Lindholm M, Grönroos M, Hjort J, Karjalainen SM, Tokola L, Heino J (2018) Different species trait groups of stream diatoms show divergent responses to spatial and environmental factors in a subarctic drainage basin. Hydrobiologia 816:213–230. https://doi.org/10.1007/s10750-018-3585-0
Martens K (1990) Revision of African Limnocythere s.s. Brady, 1867 (Crustacea, Ostracoda), with special reference to the Rift Valley Lakes; morphology, taxonomy, evolution and (palaeo-) ecology. Arch Hydrobiol 83:453–524
Meisch C (2000) Freshwater Ostracoda of Western and Central Europe. Spektrum, Heidelberg
Menéndez JL, La Rocca SM (2006). Primer Inventario Nacional de Bosques Nativos. Segunda Etapa, Inventario de campo de la Región del Espinal, Distritos Caldén y Ñandubay. Secretaría de Ambiente y Desarrollo Sustentable de la Nación.
Mesquita-Joanes F, Smith AJ, Viehberg FA (2012) The Ecology of Ostracoda Across Levels of Biological Organisation from Individual to Ecosystem: A Review of Recent Developments and Future Potential. In: Horne DJ (ed) Developments in Quaternary Sciences. Ostracoda as Proxies for Quaternary Climate Change, Elsevier Amsterdam
Mezquita F, Roca JR, Reed JM, Wansard G (2005) Quantifying species-environment relationships in non- marine Ostracoda for ecological and palaeoecological studies: examples using Iberian data. Palaeogeogr, Palaeoclimatol, Palaeoecol 225:93–117. https://doi.org/10.1016/j.palaeo.2004.02.052
Michelson AV, Park Boush LE, Pan JJ (2016) Discerning patterns of diversity from biogeographical distributions: testing models of metacommunity dynamics using non- marine ostracodes from San Salvador Island, Bahamas. Hydrobiologia 766:305–319. https://doi.org/10.1007/s10750-015-2464-1
Mykrä H, Heino J, Muotka T (2007) Scale-related patterns in the spatial and environmental components of stream macroinvertebrate assemblage variation. Glob Ecol Biogeogr 16:149–159. https://doi.org/10.1111/j.1466-8238.2006.00272.x
O’Donnell MS, Ignizio DA (2012) Bioclimatic predictors for supporting ecological applications in the conterminous United States. US Geo Sur Data Ser 691(10):4–9
Padial A, Ceschin F, Declerck SAJ, De Meester L, Bonecker CC, Lansac-Tôha FA, Rodrigues L, Rodrigues LC, Train S, Velho LFM, Bini LM (2014) Dispersal ability determines the role of environmental, spatial and temporal drivers of metacommunity structure. PLoS ONE 9:1–8. https://doi.org/10.1371/journal.pone.0111227
Paruelo JM, Beltrán A, Jobbágy E, Sala O, Golluscio R (1998) The climate of Patagonia: general patterns and controls on biotic process. Ecol Austral 8:85–101
Paruelo JM, Golluscio RA, Jobbágy EG, Canevari M, Aguiar MR (2005) Situación ambiental en la estepa patagónica. In: Brown A, Martinez Ortiz U, Acerbi M (eds). La situación ambiental argentina. Fundación Vida Silvestre Argentina
Pearson RG, Dawson TP (2003) Predicting the impacts of climate change on the distribution of species: are bioclimatic envelope models useful? Glob Ecol Biogeogr 12:361–371. https://doi.org/10.1046/j.1466-822X.2003.00042.x
Peres-Neto PR, Legendre P, Dray S, Borcard D (2006) Variation partitioning of species data matrices: estimation and comparison of fractions. Ecology 87:2614–2625. https://doi.org/10.1890/0012-9658(2006)87[2614:VPOSDM]2.0.CO;2
Pérez AP, Coviaga CA, Ramos LY, Lancelotti J, Alperin M, Cusminsky GC (2019) Taxonomic revision of Cypridopsis silvestrii comb. nov. (Ostracoda, Crustacea) from Patagonia, Argentina with morphometric analysis of their intraspecific shape variability and sexual dimorphism. Zootaxa 4563:83–102. https://doi.org/10.11646/zootaxa.4563.1.4
Pinceel T, Vanschoenwinkel B, Weckx M, Brendonck L (2020) An empirical test of the impact of drying events and physical disturbance on wind erosion of zooplankton egg banks in temporary ponds. Aquat Ecol 54:137–144. https://doi.org/10.1007/s10452-019-09731-2
R Core Team (2018) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rabassa J (2008) Late cenozoic glaciations in patagonia and tierra del fuego. In: Rabassa J (ed) The late cenozoic of patagonia and tierra del fuego: developments in quaternary science, vol 11. Elsevier, Amsterdam, pp 151–204
Ramón Mercau J, Laprida C (2016) An ostracod-based calibration function for electrical conductivity reconstruction in lacustrine environments in Patagonia, Southern South America. Ecol Indic 69:522–532. https://doi.org/10.1016/j.ecolind.2016.05.026
Ramón Mercau J, Laprida C, Massaferro J, Rogora M, Tartari G, Maidana NI (2012) Patagonian ostracods as indicators of climate related hydrological variables: implications for paleoenvironmental reconstructions in Southern South America. Hydrobiologia 694:235–251. https://doi.org/10.1007/s10750-012-1192-z
Ramos LY, Cusminsky GC, Schwalb A, Alperin M (2017) Morphotypes of the lacustrine ostracod Limnocythere rionegroensis Cusminsky and Whatley from Patagonia, Argentina, shaped by aquatic environments. Hydrobiologia 786:137–148. https://doi.org/10.1007/s10750-016-2870-z
Ramos L, Alperin M, Schwalb A, Markgraf V, Ariztegui D, Cusminsky G (2019) Changes in ostracod assemblages and morphologies during lake-level variations of Lago Cardiel (49S), Patagonia, Argentina, over the last 156 ka. Boreas. https://doi.org/10.1111/bor.12371
Ramos L, Epele LB, Grech MG, Manzo LM, Macchi PA, Cusminsky GC (2022) Modelling influences of local and climatic factors on the occurrence and abundance of non-marine ostracods (Crustacea: Ostracoda) across Patagonia (Argentina). Hydrobiologia 849:229–244. https://doi.org/10.1007/s10750-021-04722-7
Rivas JA Jr, Schröder T, Gill TE, Wallace RL, Walsh EJ (2019) Anemochory of diapausing stages of microinvertebrates in North American drylands. Freshwa Biol 64:1303–1314. https://doi.org/10.1111/fwb.13306
Rosati M, Rossetti G, Cantonati M, Pieri V, Roca JR, Mesquita-Joanes F (2016) Are aquatic assemblages from small water bodies more stochastic in dryer climates? An analysis of ostracod spring metacommunities. Hydrobiologia 793:199–212. https://doi.org/10.1007/s10750-016-2938-9
Schwalb A, Burns SJ, Cusminsky G, Kelts K, Markgraf V (2002) Assemblage diversity and isotopic signals of modern ostracodes and host waters from Patagonia, Argentina. Palaeogeogr, Palaeoclimatol, Palaeoecol 187:323–339
Soininen J (2014) A quantitative analysis of species sorting across organisms and ecosystems. Ecology 95:3284–3292. https://doi.org/10.1890/13-2228.1
Soininen J (2016) Spatial structure in ecological communities – a quantitative analysis. Oikos 125:160–166. https://doi.org/10.1111/oik.02241
ter Braak CJF, Prentice IC (1988) A theory of gradient analysis. Adv Ecol Res 18:271–317
ter Braak CJF, Šmilauer P (2012) Canoco reference manual and user’s guide: software for ordination, version 50. Microcomputer Power, New York, Ithaca USA
Wei T, Simko V (2017) R package "corrplot": Visualization of a Correlation Matrix (Version 0.84). Available from https://github.com/taiyun/corrplot
Whatley RC, Cusminsky GC (1999) Lacustrine ostracoda and late Quaternary palaeoenvironments from the lake Cari-Laufquen region, Rio Negro province, Argentina. Paleogeogr, Paleoclimatol, Paleoecol 151:229–239
Yassini I, Jones BG (1995) Foraminiferida and Ostracoda from estuarine and shelf environments on the southeastern coast of Australia. University of Wollongong Press, Wollongong
Zhai M, Nováček O, Výravský D, Syrovátka V, Bojková J, Helešic J (2015) Environmental and spatial control of ostracod assemblages in the Western Carpathian spring fens. Hydrobiologia 745:225–239. https://doi.org/10.1007/s10750-014-2104-1
Acknowledgements
The authors wish to express their gratitude to the workers and owners of properties we visited during samplings. The authors also thank Dr. Daniel Ariztegui, Dr. Julio Lancelotti and members of Maca Tobiano Project for the fieldwork assistance. This study was supported by the National Council of Science and Technology of Argentina (CONICET); the National Agency of Scientific and Technologic Promotion and the National University of Comahue
Funding
This study was supported by the National Council of Science and Technology of Argentina, CONICET (PIP 00819, PIP 0021, PIP 11220200100224); the National Agency of Scientific and Technologic Promotion (PICT 2010–0082, PICT 2014–1271, PICT 2019–02408, PICT-2020-SERIEA-02515) and the National University of Comahue (UNCo 04/B166, UNCo 04/B194 and UNCo B/237).
Author information
Authors and Affiliations
Contributions
CC and APP conceived and designed the study. CC, APP, LR and GC participated in the field sampling. LZ assisted with climate and spatial data. PEG, CC and APP realized the statistical analyses. GC and APP obtained the funding for this study. CC wrote the manuscript, with substantial help from APP and GC interpretations. All authors discussed the results and commented jointly on the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent for publication
All the authors consent to publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
27_2023_981_MOESM1_ESM.pdf
Supplementary file1 Geographical localization and references of the studied sites. NP: Northern Patagonia, SP: Southern Patagonia. (PDF 232 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Coviaga, C.A., Pérez, A.P., Ramos, L.Y. et al. Assessing environmental and spatial drivers of non-marine ostracod metacommunities structure in Northern and Southern Patagonian environments. Aquat Sci 85, 82 (2023). https://doi.org/10.1007/s00027-023-00981-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00027-023-00981-9