Abstract
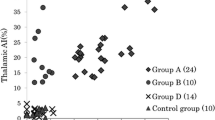
Objective: Crossed cerebellar diaschisis (CCD) is defined as a depression of blood flow and oxidative metabolism of glucose in the cerebellum contralateral to a supratentorial brain lesion, as detected with positron emission tomography (PET) and single photon emission computed tomography. We examined whetherl-[methyl-11C]methionine (MET) uptake is affected in CCD.Methods: In 12 patients with a unilateral supratentorial brain tumor, we evaluated the uptake of 2-deoxy-2-[18F]fluoroe-d-glucose (FDG) and MET in the cerebellar hemispheres by means of PET. Asymmetry index (AI) was defined as a difference in the average count between the ipsilateral and contralateral cerebellar hemispheres divided by the average count in both cerebellar hemispheres. Patients with AI of FDG PET more than 0.1 and those with AI equal to 0.1 or less than 0.1 were classified as CCD-positive and CCD-negative, respectively.Results: Six patients were CCD-positive and others were CCD-negative in the FDG PET study. Between CCD-positive and CCD-negative patients, mean AI of MET was not significantly different (0.017±0.023 and 0.014±0.039, respectively).Conclusions: Different from glucose metabolism, cerebellar MET uptake was not affected in CCD. The present study may indicate that cerebellar MET uptake is independent of suppression of cerebellar neuronal activity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Baron JC, Bousser MG, Comar D, Castaigne P. “Crossed cerebellar diaschisis” in human supratentorial brain infarction.Trans Am Neurol Assoc 1980; 105: 459–461.
Patronas NJ, Di Chiro G, Smith BH, De La Paz R, Brooks RA, Milam HL, et al. Depressed cerebellar glucose metabolism in supratentorial tumors.Brain Res 1984; 291: 93–101.
Alavi A, Mirot A, Newberg A, Alves W, Gosfield T, Berlin J, et al. Fluorine-18-FDG evaluation of crossed cerebellar diaschisis in head injury.J Nucl Med 1997: 38: 1717–1720.
Stubgen JP. Crossed cerebellar diaschisis related to recurrent focal seizures.Epilepsia 1995; 36: 316–318.
Gold L, Lauritzen M. Neuronal deactivation explains decreased cerebellar blood flow in response to focal cerebral ischemia or suppressed neocortical function.Proc Natl Acad Sci USA 2002; 99: 7699–7704.
Yamauchi H, Fukuyama H, Kimura J. Hemodynamic and metabolic changes in crossed cerebellar hypoperfusion.Stroke 1992; 23: 855–860.
Ito H, Kanno I, Shimosegawa E, Tamura H, Okane K, Hatazawa J. Hemodynamic changes during neural deactivation in human brain: a positron emission tomography study of crossed cerebellar diaschisis.Ann Nucl Med 2002; 16: 249–254.
Comar D, Cartron JC, Maziere M, Marazano C. Labelling and metabolism of methionine-methyl-11C.Eur J Nucl Med 1976; 1: 11–14.
Grange E, Gharib A, Lepetit P, Guillard J, Sarda N, Bobilliier P. Brain protein synthesis in the conscious rat usingl-[35S]methionine relationship of methionine specific activity between plasma and precursor compartment and evaluation of methionine metabolic pathways.J Neurochem 1992; 59: 1437–1443.
Shamoto H, Chugani HT. Glucose metabolism in the human cerebellum: an analysis of crossed cerebellar diaschisis in children with unilateral cerebral injury.J Child Neurol 1997; 12: 407–414.
Meneghetti G, Vorstrup S, Mickey B, Lindewald H, Lassen NA. Crossed cerebellar diaschisis in ischemic stroke: a study of regional cerebral blood flow by133Xe inhalation and single photon emission computerized tomography.J Cereb Blood Flow Metab 1984; 4: 235–240.
Niimura K, Chugani DC, Muzik O, Chugani HT. Cerebellar reorganization following cortical injury in humans: effects of lesion size and age.Neurology 1999; 52: 792–797.
Kim SE, Choi CW, Chung JK, Roth JH, Lee MC, Koh CS. Crossed cerebellar diaschisis in cerebral infarction: technetium-99m-HMPAO SPECT and MRI.J Nucl Med 1997; 38: 14–19.
Sokoloff L. Relation between physiological function and energy metabolism in the central nervous system.J Neurochem 1977; 29: 13–26.
Yarowsky P, Kadekaro M, Sokoloff L. Frequency-dependent activation of glucose utilization in the superior cervical sympathetic trunk.Proc Natl Acad Sci USA 1993; 80: 4179–4183.
Kennedy C, Miyaoka M, Suda S, Macko K, Jarvis C, Mishkin M, et al. Local metabolic responses in brain accompanying motor activity.Trans Am Neurol Assoc 1980; 105: 13–17.
Shahbazian FM, Jacobs M, Lajtha A. Regional and cellular differences in rat brain protein synthesisin vivo and in slices during development.Int J Devel Neurosci 1986; 4: 209–215.
Kennedy C, Suda S, Smith CB, Miyaoka M, Ito M, Sokoloff L. Changes in protein synthesis underlying functional plasticity in immature monkey visual system.Proc Natl Acad Sci USA 1981; 78: 3950–3953.
Lajtha A, Dunlop D, Banay-Schwartz M. Cerebral protein turnover: aspects and problems. InPET studies on amino acid Metabolism and protein synthesis (Mazoyer BM, Heiss WD, Comar D, eds), Dordrecht Kluwer Academic Publishers, 1993: 1–17.
Salmon E, Gregorire MC, Delfiore G, Lemaire C, Degueldre C, Frank G, et al. Combined study of cerebral glucose metabolism and [11C]methionine accumulation in probable Alzheimer's disease using positron emission tomography.J Cereb Blood Flow Metab 1996; 16: 399–408.
Nakagawa M, Kuwabara Y, Sasaki M, Koga H, Chen T, Kaneko K, et al.11C-methionine uptake in cerebrovacular disease: A comparison with18F-FDG PET and99mTc-HMPAO SPECT.Ann Nucl Med 2002; 16: 207–211.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kajimoto, K., Oku, N., Kimura, Y. et al. Crossed cerebellar diaschisis: a positron emission tomography study withl-[methyl-11C]methionine and 2-deoxy-2-[18F]fluoro-d-glucose. Ann Nucl Med 21, 109–113 (2007). https://doi.org/10.1007/BF03033988
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03033988