Abstract
Purpose of Review
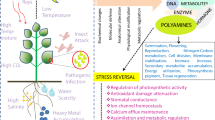
Environmental stress is increasingly wearing down crop productivity. As a consequence, a major priority of plant research is to get deeper insights on tolerance mechanisms, providing solutions with the generation of stress-tolerant cultivars. To this goal, a common target of genetic modification is the polyamine signaling pathway. Its association with a protective role against stressful stimuli is widely acknowledged; however, the nature of this function is highly complex. In consequence, this review aims to present up-to-date evidence in regard with metabolic and physiological role of polyamines, protecting plants during severe stressful events (such as drought and soil salinization).
Recent Findings
The most recent evidence from stress physiology research highlight polyamines as key players in signaling responses involved in central metabolism, sugar and lipid homeostasis, maintenance, and induction of antioxidant capacity as well as osmotic regulation. Nevertheless, a number of questions remain open, such as the extent of their roles and whether they represent hub metabolic molecules. Recent advances on polyamine metabolism are therefore summarized in relation to salt and drought stress tolerance and its possible implication on the generation of tolerant crops.
Summary
This review highlights recent findings related to polyamine protective role during drought and salt stress. A clear synergy is established between these amine compounds, ABA, and reactive oxygen and nitrogen species. However, the involvement of these amine compounds on stress physiology goes far beyond the modulation of nitro-oxidative homeostasis and ABA signaling, with an increasing body of evidence demonstrating that polyamines are emerging metabolic hubs of plant stress signaling.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction: Polyamines, Nitrogenous Molecules Related to Several Processes
One of the biggest challenges of modern science is to apply the fundamental discoveries from basic research in providing solutions aiming at increasing food crop yield. Moreover, the increment of the world population has added greater pressure on the demand [1]. Global climate change is expected to intensify the severity and frequency of water stress events and soil salinization worldwide, causing decrease on growth rate and pollen viability, which severely affect crop production [2, 3]. As a result, plant stress physiology research has been widely directed towards the investigation of genetic determinisms underlying stress tolerance of higher plants as well as macroalgae, greatly due to their importance to cover global nutritional needs. In regard with this research area, polyamines (PAs) are relevant molecules [4, 5].
PAs are aliphatic organic compounds with variable hydrocarbon chains and two or more amino groups [6]. PAs have been detected in all cells across all kingdoms [7]. PA biological roles were initially thought to be structural. Their polycationic nature enables the binding and stabilization of anionic macromolecules in the cell. However, later studies revealed that PAs are also involved in fundamental cellular processes such as cell division, differentiation, transcriptional regulation, and translation [8].
Genetic analyses indicate that the most abundant PAs in plants (putrescine (Put), spermidine (Spd), and spermine (Spm)) are required for plant stress tolerance [7, 9, 10]. In addition, these PAs are important sinks of assimilated nitrogen [11]. However, other quantitatively less abundant PAs have shown unique roles in PA functions and stress signaling. Recently, it has been shown that an isomer of Spm (thermospermine (T-Spm)) found in higher plants [7] is not a minor polyamine [12] and it is involved in tolerance responses [13]. In addition, cadaverine (Cad), a formally recognized minor PA [14], is involved in different PA-related signaling processes, modulating plant development and response to external stimulus [15].
Recent and extensive reviews present compelling evidence regarding the participation of PAs in abiotic and biotic stress responses in plants [5, 8]. In reference to salt and drought stress tolerance responses, there is no doubt about the implication of PAs in the physiological processes that leads to plant resilience. However, the nature of the signaling mechanisms and the number of interactions between PAs and other molecules (such as sugars, nitric oxide (NO), ABA, and H2O2) are not only far from being fully elucidated but are also increasing in complexity. Currently, plant tolerance to salt and drought stresses can be achieved by either transgenic approaches regulating PA homeostasis or exogenous addition of individual PAs through the activation of priming phenomena [16]. This is due to the implication of PAs in important pathways of central metabolism and/or modulation of several functions in plant organs such as roots and leaves. Thus, it is important to summarize recent findings regarding the involvement of each PA in drought and salt stress responses.
Brief and Essential Insights in PA Metabolism: Homeostasis Mechanisms
Metabolic studies indicate that the intracellular levels of PAs in higher plants are mostly regulated by anabolic and catabolic processes, as well as by their conjugation to hydroxycinnamic acids (HCAA) [9] and transport by specific membrane transporters [17].
PA biosynthesis is initiated by the synthesis of the diamine Put. In plants, Put is derived from arginine (Arg) through multiple sequential steps catalyzed by the enzymes arginine decarboxylase (ADC), agmatine iminohydrolase (AIH), and N-carbamoylPut amidohydrolase (CPA). Higher PAs Spd, Spm, and its isomer T-Spm are synthesized by the action of aminopropyltransferases (APT) Spd synthase (SPDS), Spm synthase (SPMS), and tSpm synthase (tSPMS, ACL5) which catalyze the addition of aminopropyl groups to Put or Spd, producing Spd, Spm, or T-Spm, respectively. Decarboxylated S-adenosylmethionine (dSAM), a product of S-adenosylmethionine (SAM) decarboxylation by SAM decarboxylase (SAMDC), is the donor of the aminopropyl groups required for PA biosynthesis [6, 18, 19]. On the other hand, there is less information about Cad biosynthesis in plants. It is known that it is mainly synthesized from lysine by lysine decarboxylase (LDC) and homoagmatine or homoarginine to a lesser extent [14, 20].
Amine oxidases (AO) involved in oxidation of PAs are classified in copper-containing amine oxidases (CuAO) and FAD-dependent polyamine oxidases (PAO) [8, 10] releasing in all cases hydrogen peroxide (H2O2). CuAO enzymes preferentially oxidize the primary amino groups of Put and Cad and show less affinity for Spd and Spm [11] and participate in PA final catabolism in peroxisomes and the apoplast [21, 22]. PAOs catalyze the oxidation of higher PAs Spd, Spm, and T-Spm at their secondary amino groups [23]. Classification of PAOs is dependent on whether they catalyze the terminal oxidation of PAs or mediate the PA back-conversion, which is related to the position of the carbon in which PAO is able to oxidize the PA molecule. First group of PAO oxidize Spd and Spm, producing hydrogen peroxide, diaminopropane, 4-aminobutanal (from Spd), and 3-(aminopropyl)-4-aminobutanal (from Spm) [11]. The second group oxidize Spd and Spm (and/or their acetylated derivatives) producing Put and Spd, respectively [24, 25].
In plants, PAs occur in free amine forms but also conjugate to hydroxycinnamic acids to form hydroxycinnamic acid amides (HCAA), in a process catalyzed by N-acyltransferases. Recently, several proteins with Spd N-acyltransferase activity have been reported in Arabidopsis supporting the role of HCAA in pollen tube development [26]. Several years ago, PA conjugates were considered to be inactive forms. However, evidence in the last decade indicates that they are essential for development and biotic interactions [8]. Indeed, an emerging area of research is the characterization of PA conjugates and their pathways involved [8].
PA transport also plays a pivotal role in the regulation of PA cell homeostasis [17]. To date, few PA transporters have been identified in plants, with special attention to rice [27] and Arabidopsis [17]. Recent discovery of PA transporters by genetic analyses has involved the analysis of oxidative tolerance or PA uptake capacity [28]. This investigation opened a new branch of PA research. Interestingly, over the last few years, it was found that transporter AtOCT1 is implied in Cad efflux process [29] and also that Cad flux can be regulated by overexpression of genes encoding transporters (OCT1 included) due to Spm accumulation, proposing a new role for Spm in the regulation of intracellular Cad concentration, which is also redox-dependent [30]. Research on PA transporters will provide deeper insights into the molecular function of PAs and its homeostasis and better understanding into the PA antioxidative properties [17].
Essentially, the PA pool is dynamic and fluctuates over time. PAs undergo rapid exchange through what has been referred to as the “PA cycle,” in which different cycles of biosynthesis and back-conversion lead to the release of H2O2 [9]. This mechanism has been proposed to induce plant stress tolerance [23] and stress signaling affecting multiple cellular compartments [15, 31–34]. In agreement with this, recent findings in plants with impaired production of Spd and Spm by SAMDC depletion suggested that SAMDC-mediated higher PA biosynthesis is a putative modulator of the trade-off between stress tolerance and plant growth and developmental traits [34]. Furthermore, Cad has been demonstrated to affect the accumulation of higher PAs in plant tissues [15]; therefore, its contribution to plant stress response cannot be assessed on its own. In any event, it seems reasonable to argue that PA homeostasis modulated by SAMDC, APTs, and AOs is not a simple PA biosynthesis/degradation process.
Interactions of PAs with ROS, NO, ABA, and Other Metabolites
A common acceptance in current research regarding plant stress physiology relates with a number of essential interactions between PAs and signaling molecules such as H2O2, NO, and ABA [35–38]. PAs have been related to ROS through H2O2 production via their catabolism pathway [19, 22, 39]. Nonetheless, their cross-talk appears to be far more complex. ROS derived from oxidation of PAs are vital to trigger stress response signaling. However, the size and rate of its accumulation determine cell fate, which means that ROS should not exceed specific thresholds; if so, it shifts the balance between gene expression modification to programmed cell death [40]. Recent findings established that Spd homeostasis by AtPAO3 is involved in ROS production other than H2O2. The ratio of O2 −/ H2O2 showed to be an important signal in transcription [31] and might be a mediator of PA contribution to plant adaptation under suboptimal conditions [41].
On the other hand, it is widely accepted by PA researchers that PA metabolism and NO biosynthesis are tightly interconnected. NO biosynthesis is stimulated by Spd and Spm in the root tip and primary leaves of Arabidopsis seedlings [42]. A recent study in citrus demonstrated the tissue-specific modulation of PAO expression upon nitro-oxidative stress, after which pretreated plants show increased tolerance to salinity, suggesting that PAs may mediate the link between oxidative and nitrosative signaling [43] and are able to reprogram the nitro-oxidative status as well as the proteome of salinity-stressed plants [44]. It is also known that Spm and Spd modulate the arginine-linked NO synthase and nitrate reductase pathways [45]. NO is filling gaps between several physiological effects of PAs and the amelioration of stress [31, 46, 47], which is essential for further understanding of stress physiology events.
With regard to tolerance mechanisms and stomatal closure, the role of ABA is undeniable, especially during stress response [48, 49]. Interestingly, SPMS is an ABA-inducible gene [50], which is associated with Spm protective role found in Arabidopsis during water stress [51]. However, Put accumulation is also related with increased drought tolerance [52]. Transcriptional upregulation by drought of PA biosynthetic enzymes AtADC, AtSPDS, and AtSPMS and resulting increases in PA content are ABA-dependent [53]. Similar trends were recorded in ADC expression patterns in response to salinity [54] and cold stresses [55]. Conversely, Put was found to promote ABA biosynthesis in response to drought in Lotus tenuis [56]; therefore, a cross-regulation between ABA and Put is not excluded. In support to this, transcriptomic analyses of transgenic SAMDC1 or SPMS overexpressing Arabidopsis plants exhibiting higher Spm levels revealed the transcriptional upregulation of the ABA biosynthesis gene NCED, ABA accumulation, and induction of genes involved in water deprivation and defense responses, including several transcription factors [57]. The relationship between PAs and ABA is significant, and overlapping functions between Put and Spm in regard with ABA have been documented [58].
Another important component under consideration is the role of PAs in central metabolic networks. Recent reports suggest the interplay between PAs and primary metabolism during exposure to stress and/or plant development [59–64]. Put, proline (Pro), and GABA are derived from glutamate (Glu), a hub molecule of nitrogen metabolism [59]. Glu signaling impacts amino acid metabolism, which is directly linked to carbon mobilization pathways. In this regard, GABA has been suggested to mediate C/N balance [65, 66] via Glu receptors [67]. PA and GABA accumulation has been reported under both optimum and stressful conditions [68]. Similarly, a metabolic interaction has been detected between PA and sugar metabolism [69]. This is intriguing since sugar signaling is known to be important in the stress response [62, 70, 71]. Apart from known metabolic connections previously reported, PAs have also been implicated in the modulation of metabolite levels for which no metabolic connections are known. Put over-producing Arabidopsis plants revealed the differential regulation of stress-responsive, hormone, and signaling-related genes, involved in the biosynthesis of hormones such as auxin, ethylene, and ABA among others [57]. Overexpression of SPDS induced the transcription of various putative stress-related genes in chilling-stressed transgenic Arabidopsis plants compared with wild-type ones [72]. In addition, transgenic Arabidopsis plants accumulating Spm exhibited modified transcript levels of genes involved in the biosynthesis of jasmonates, salicylic acid and ABA, mitogen-activated protein kinases, receptor-like kinases, and genes linked with calcium regulation [57]. In that sense, recent findings from our group revealed that lack of Spm degradation mechanisms by knocking out PAO4 expression in Arabidopsis leads to an increase in metabolites of central sugar and lipid metabolism such as pyruvate or myo-inositol as well as an enhancement of the antioxidant capacity [61]. In support of this, it was also shown that T-Spm accumulation triggers metabolic and transcriptional reprogramming, enhancing among others, osmoprotectants such as proline, sugars, polyols, and TCA cycle intermediates [64]. In addition, it has been demonstrated that T-Spm modifies the expression of auxin-related genes [33]. Regarding Cad, this PA serves as a critical precursor for the synthesis of secondary metabolites and specific alkaloids involved in defense in Leguminosae as well as several other plants [20]. The identification of PA-regulated downstream targets and the discovery of the interplay between PAs and other stress-responsive molecules have opened new possibilities to investigate the function of individual PAs at molecular level [73]. Therefore, the progressive investigation about the involvement of PAs in several pathways of central metabolism is essential to further understand the role of these molecules under optimum and stress conditions.
Current PA-Related Research Towards Drought and Salt Stress Protection
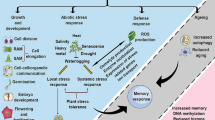
PAs provide tolerance against several stresses including water and salt stress, through different mechanisms and in relevant species of agronomic use such as maize, wheat, rice, tomato, potato, grape, cacao, or lettuce among others [74–83]. This might be the result of their accumulation in the cell providing direct protection against stress or because of PA degradation products which act as signal molecules triggering stress protection. The following sections summarize recent findings regarding PAs and their protective roles against drought and salinity (see Table 1).
PA Implications in Drought Tolerance
The adaptation of plants to drought involves a series of complex physiological processes and alterations that can take place over the short or long-term (e.g., stomatal closure limiting carbon assimilation) [105]. After drought imposition, plants must coordinate processes to alleviate both cell hyperosmolarity and oxidative stress as well as osmotic imbalance [106]; therefore, physiological alterations at several levels of metabolism must be expected. PA application increases the levels of osmoprotectant molecules and alleviates oxidative damage, which is caused by the stressor and it is additionally involved in other important plant tolerance metabolic processes. Furthermore, a link between PAs and well-known stress-core molecules such as reactive nitrogen and sulfur species has been described.
Exogenous Spm treatment in mung bean conferred tolerance to separate or combined drought/heat stress [84] and salt stress [94] by upregulating the gene expression of important essential antioxidant enzymes (e.g., SOD, CAT) and enhancing the glyoxalase system, which reduced methyglyoxal toxicity, a stress-induced compound [84]. A similar trend was also reported in tomato [85]. Furthermore, recent reports on white clover showed that exogenous application of Spd improves drought tolerance by increasing the level of soluble carbohydrates (WSC) such as sucrose, fructose, and sorbitol as well as dehydrin synthesis [86]. Inhibition of PA biosynthesis reduced cytosolic Ca2+ and antioxidant enzyme activities, while these effects were mitigated by the external application of Spd. Besides this, Spd enhanced the activation of NR and NOS which are the two most known enzymatic sources of NO in the cell. Both NO and Spd enhanced drought stress tolerance by triggering the signaling cascade to enhance the levels of enzymatic antioxidative machinery in a NO-mediated process [87]. Interestingly, it has been recently reported that NO also modulates ABA signaling in Arabidopsis during drought imposition, demonstrating that stomatal closure is not only ABA-dependent but also NO-dependent [88]. Remarkably, PA content increased after H2S treatment thus providing important evidence that another reactive species interacts with PAs, in this case by controlling PA levels under drought conditions. More specifically, it was demonstrated that H2S significantly enhances the tolerance of Spinacia oleracea to drought stress, by changing the transcript levels of genes associated with sugar and PA biosynthesis, which in turn promoted higher sugar and PA accumulation [90]. Recently, it was also shown that drought alleviation by H2S in wheat involves the ABA signaling pathway [91], thereby adding further support to the considerable evidence about the critical role of H2S in physiological and metabolic processes in plants [89, 107].
In addition, some reports have attempted to unravel the involvement of PAs in species- or tissue-specific processes during drought stress condition. A pioneering study on Triticale, a synthetic hybrid of wheat and rye, demonstrated that drought stress tolerance on this species at late developmental stages was linked with a gradual increase in cell wall-bound PAs and a decline in free PAs, thus suggesting a possible role of cell wall strengthening by PAs under abiotic stress conditions [93]. In regard with wheat, it has been found that higher PAs under drought conditions are not only involved in tolerance to stress but also in grain filling [92], which is severely inhibited during water deprivation. More specifically, it was demonstrated that particularly in grains, Spd and Spm significantly increased ABA and zeatin concentrations and decreased the ethylene evolution rate, which promoted wheat grain filling under water deficit conditions, suggesting that this process mediated by hormone interactions is PA-related [92].
PAs and Salt Stress Tolerance
Among the various abiotic stress factors, soil salinization is one of the main harmful factors limiting crop productivity, especially on species highly sensitive to salinity such as cereals [3]. It has been reported that more than 6 % of global land area and 20 % of irrigated lands are affected by this kind of stress, often simultaneously with soil alkalinization, severely reducing plant growth due to concomitant nutrient limitation, ion toxicity, high-osmotic, and oxidative stress [95, 108].
PAs are commonly applied exogenously at a range of concentrations on many plant species, in order to protect plants upon salt stress imposition. Recent findings in cucumber seedlings under salt stress suggested that exogenous Put addition alleviates stress by regulating protein production at transcriptional and translational levels. Interestingly, Put application caused an increase in the levels of endogenous PAs, mainly Spd and Spm, which in turn enhanced fatty acid mobilization that leads to a stabilized photosynthetic apparatus [97]. In addition, omics analyses revealed that Put was able to restore root growth in cucumber seedlings grown under salt stress by enhancing the expression of genes encoding proteins that are involved in defense responses and carbohydrate and amino acid metabolism [98]. Similar findings were also observed after exogenous Spd treatment [99]. Interestingly, another important class of protein which was enhanced by Spd application was proteins related to maintenance of C/N balance [99].
Counteracting positive results on salt stress alleviation obtained by Put application, it has been found that GABA derived from Put degradation ameliorates the deleterious effect of salt stress [100, 101]. However, positive regulation of PA biosynthetic pathway has been reported after GABA exogenous addition to muskmelon seedlings under ionic/osmotic stress [102]. Overall, the implication of Put on salt stress responses is clear, with GABA potentially playing a key role promoting synergistic dynamics with PA levels.
Regarding higher PAs, exogenous application of Spd in zoysiagrass exposed to salt stress caused enhancement of PA biosynthetic pathway and antioxidative enzymatic machinery [103]. Proteomic analysis of tomato seedlings exposed to Spd exogenous pretreatment followed by salt/alkaline stress imposition demonstrated that the protective effect of Spd was mediated by the enhancement of five major categories of proteins involved in energy, sugar, and amino acid metabolism [95]. Later on, a more specific protective effect of Spd was demonstrated on chlorophyll and D1 protein content, which collaborates with the maintenance of photosynthetic apparatus and continuation of photosynthesis rate, thus facilitating the growth of tomato seedlings under salt/alkaline stress [96].
A recent study on Medicago plants under salt stress reported an Spm protective effect due to interactions with epibrassinolide, a plant hormone highly involved in salt stress response. Exogenous addition of epibrassinolide induced PA biosynthetic pathway and concomitant Spm increase caused amelioration of oxidative damage and modulation of Pro levels [104]. In agreement with this, recent findings reported the accumulation of Pro, sugars, and other osmoprotectants, in atpao5 mutants impaired in T-Spm oxidation [64].
The involvement of sugars, amino acid, and oxidative metabolism in the PA signaling pathway after stress imposition has been established. However, further research is imperative in order to fully elucidate the complex physiological responses observed.
Conclusions and Future Perspectives
Drought and salinity lead to yield penalties in modern agriculture. A lot of studies have been conducted to understand and solve the complexity of the processes that occur in plants during stress imposition. In this context, several molecules have been described to play a pivotal role in plant tolerance against stresses, with PAs shaping up as important players. PAs are molecules involved in several fundamental cellular processes, and their production is enhanced under drought and salinity. Remarkably, not only the most abundant PAs (Put, Spd, Spm) are playing a role in stress tolerance but also the quantitatively less abundant ones such as T-Spm and Cad. Different approaches, such as transgenic technologies in the PA pathway and exogenous application of PAs, have been employed to unravel the effects of PAs on drought and salinity. Clearly, there is involvement of PAs in central metabolism and in triggering several mechanisms related to plant stress tolerance (such as through osmoprotection and by inducing the antioxidant machinery). However, a more detailed description on the mechanistic involvement of PAs and the interaction with other molecules to accelerate plants’ tolerance is far from being elucidated because of the complexity of their roles. The dynamic nature of these metabolites, which is related to rapid degradation/interconversion from one to another in the PA cycle, and the interactions between PAs and signaling molecules, such as hormones and reactive species, provide solid evidence on their role in plant stress tolerance. For these reasons, research in that area is ongoing and must to be continued with state-of-the-art approaches such as systems biology technologies, in order to get a better understanding of the contribution of PAs on plant stress tolerance.
References
Pathak MR, Teixeira da Silva JA, Wani SH. Polyamines in response to abiotic stress tolerance through transgenic approaches. GM Crops Food. 2014;5:87–96.
Pottosin I, Shabala S. Polyamines control of cation transport across plant membranes: implications for ion homeostasis and abiotic stress signaling. Front Plant Sci. 2014;5:154.
Das P, Nutan KK, Singla-Pareek SL, Pareek A. Understanding salinity responses and adopting “omics-based” approaches to generate salinity tolerant cultivars of rice. Front Plant Sci. 2015;6:712.
Schweikert K, Burritt DJ. Polyamines in macroalgae: advances and future perspectives. J Phycol. 2015;51:838–49.
Tavladoraki P, Cona A, Angelini R. Copper-containing amine oxidases and FAD-dependent polyamine oxidases are key players in plant tissue differentiation and organ development. Front Plant Sci. 2016;7
Takahashi T, Kakehi JI. Polyamines: ubiquitous polycations with unique roles in growth and stress responses. Ann Bot. 2010;105:1–6.
Moschou PN, Paschalidis KA, Roubelakis-Angelakis KA. Plant polyamine catabolism: the state of the art. Plant Signal Behav. 2008;3:1061–6.
Tiburcio AF, Altabella T, Bitrián M, Alcázar R. The roles of polyamines during the lifespan of plants: from development to stress. Planta. 2014;240:1–18.
Alcázar R, Altabella T, Marco F, Bortolotti C, Reymond M, Koncz C, et al. Polyamines: molecules with regulatory functions in plant abiotic stress tolerance. Planta. 2010;231:1237–49.
Angelini R, Cona A, Federico R, Fincato P, Tavladoraki P, Tisi A. Plant amine oxidases “on the move”: an update. Plant Physiol Biochem. 2010;48:560–4.
Moschou PN, Wu J, Cona A, Tavladoraki P, Angelini R, Roubelakis-Angelakis KA. The polyamines and their catabolic products are significant players in the turnover of nitrogenous molecules in plants. J Exp Bot. 2012;63:5003–15.
Takano A, Kakehi JI, Takahashi T. Thermospermine is not a minor polyamine in the plant kingdom. Plant Cell Physiol. 2012;53:606–16.
Kim DW, Watanabe K, Murayama C, Izawa S, Niitsu M, Michael AJ, et al. Polyamine oxidase 5 regulates Arabidopsis thaliana growth through a thermospermine oxidase activity. Plant Physiol. 2014;165:1575–90.
Fujihara S, Abe H, Yoneyama T. A new polyamine 4-aminobutylcadaverine. Occurrence and its biosynthesis in root nodules of adzuki bean plant Vigna angularis. J Biol Chem. 1995:9932–8.
Liu T, Dobashi H, Kim DW, Sagor GHM, Niitsu M, Berberich T, et al. Arabidopsis mutant plants with diverse defects in polyamine metabolism show unequal sensitivity to exogenous cadaverine probably based on their spermine content. Physiol Mol Biol Plants. 2014;20:151–9.
Savvides A, Ali S, Tester M, Fotopoulos V. Chemical priming against multiple abiotic stresses: mission possible? Trends Plant Sci. 2016;21:329–40.
Fujita M, Shinozaki K. Identification of polyamine transporters in plants: paraquat transport provides crucial clues. Plant Cell Physiol. 2014;55:855–61.
Alcázar R, Marco F, Cuevas JC, Patron M, Ferrando A, Carrasco P, et al. Involvement of polyamines in plant response to abiotic stress. Biotechnol Lett. 2006;28:1867–76.
Moschou PN, Sanmartin M, Andriopoulou AH, Rojo E, Sanchez-Serrano JJ, Roubelakis-Angelakis KA. Bridging the gap between plant and mammalian polyamine catabolism: a novel peroxisomal polyamine oxidase responsible for a full back-conversion pathway in Arabidopsis. Plant Physiol. 2008;147:1845–57.
Bunsupa S, Katayama K, Ikeura E, Oikawa A, Toyooka K, Saito K, et al. Lysine decarboxylase catalyzes the first step of quinolizidine alkaloid biosynthesis and coevolved with alkaloid production in Leguminosae. Plant Cell. 2012;24:1202–16.
Planas-Portell J, Gallart M, Tiburcio AF, Altabella T. Copper-containing amine oxidases contribute to terminal polyamine oxidation in peroxisomes and apoplast of Arabidopsis thaliana. BMC Plant Biol. 2013;13:109.
Wimalasekera R, Villar C, Begum T, Scherer GFE. COPPER AMINE OXIDASE1 (CuAO1) of Arabidopsis thaliana contributes to abscisic acid-and polyamine-induced nitric oxide biosynthesis and abscisic acid signal transduction. Mol Plant. 2011;4:663–78.
Tavladoraki P, Cona A, Federico R, Tempera G, Viceconte N, Saccoccio S, et al. Polyamine catabolism: target for antiproliferative therapies in animals and stress tolerance strategies in plants. Amino Acids. 2012;42:411–26.
Fincato P, Moschou PN, Ahou A, Angelini R, Roubelakis-Angelakis KA, Federico R, et al. The members of Arabidopsis thaliana PAO gene family exhibit distinct tissue- and organ-specific expression pattern during seedling growth and flower development. Amino Acids. 2012;42:831–41.
Takahashi Y, Cong R, Sagor GHM, Niitsu M, Berberich T, Kusano T. Characterization of five polyamine oxidase isoforms in Arabidopsis thaliana. Plant Cell Rep. 2010;29:955–65.
Fuell C, Elliott K a, Hanfrey CC, Franceschetti M, Michael AJ. Polyamine biosynthetic diversity in plants and algae. Plant Physiol Biochem. 2010;48:513–20.
Mulangi V, Phuntumart V, Aouida M, Ramotar D, Morris P. Functional analysis of OsPUT1, a rice polyamine uptake transporter. Planta. 2012;235:1–11.
Fujita M, Fujita Y, Iuchi S, Yamada K, Kobayashi Y, Urano K, et al. Natural variation in a polyamine transporter determines paraquat tolerance in Arabidopsis. Proc Natl Acad Sci. 2012;109:6343–7.
Strohm AK, Vaughn LM, Masson PH. Natural variation in the expression of ORGANIC CATION TRANSPORTER 1 affects root length responses to cadaverine in Arabidopsis. J Exp Bot. 2015;66:853–62.
Sagor GHM, Berberich T, Kojima S, Niitsu M, Kusano T. Spermine modulates the expression of two probable polyamine transporter genes and determines growth responses to cadaverine in Arabidopsis. Plant Cell Rep. 2016;35:1247–57. This study shows evidence that Spm is involved in PA transport modulation and Cad sensitivity
Andronis E a, Moschou PN, Toumi I, Roubelakis-Angelakis KA. Peroxisomal polyamine oxidase and NADPH-oxidase cross-talk for ROS homeostasis which affects respiration rate in Arabidopsis thaliana. Front Plant Sci. 2014;5:132.
Ahou A, Martignago D, Alabdallah O, Tavazza R, Stano P, Macone A, et al. A plant spermine oxidase/dehydrogenase regulated by the proteasome and polyamines. J Exp Bot. 2014;65:1585–603.
Tong W, Yoshimoto K, Kakehi JI, Motose H, Niitsu M, Takahashi T. Thermospermine modulates expression of auxin-related genes in Arabidopsis. Front Plant Sci. 2014;5:94.
Mellidou I, Moschou PN, Ioannidis NE, Pankou C, Gėmes K, Valassakis C, et al. Silencing S-adenosyl-L-methionine decarboxylase (SAMDC) in Nicotiana tabacum points at a polyamine-dependent trade-off between growth and tolerance responses. Front Plant Sci. 2016;7:1–17.
Saxena I, Srikanth S, Chen Z. Cross talk between H2O2 and interacting signal molecules under plant stress response. Front Plant Sci. 2016;7:570.
Gupta K, Sengupta A, Chakraborty M, Gupta B. Hydrogen peroxide and polyamines act as double edged swords in plant abiotic stress responses. Front Plant Sci. 2016;7:1343.
Niu L, Liao W. Hydrogen peroxide signaling in plant development and abiotic responses: crosstalk with nitric oxide and calcium. Front Plant Sci. 2016;7:230.
Filippou P, Antoniou C, Fotopoulos V. The nitric oxide donor sodium nitroprusside regulates polyamine and proline metabolism in leaves of Medicago truncatula plants. Free Radic Biol Med. 2013;56:172–83.
Kamada-Nobusada T, Hayashi M, Fukazawa M, Sakakibara H, Nishimura M. A putative peroxisomal polyamine oxidase, AtPAO4, is involved in polyamine catabolism in Arabidopsis thaliana. Plant Cell Physiol. 2008;49:1272–82.
Moschou PN, Paschalidis KA, Delis ID, Andriopoulou AH, Lagiotis GD, Yakoumakis DI, et al. Spermidine exodus and oxidation in the apoplast induced by abiotic stress is responsible for H2O2 signatures that direct tolerance responses in tobacco. Plant Cell. 2008;20:1708–24.
Pál M, Szalai G, Janda T. Speculation: polyamines are important in abiotic stress signaling. Plant Sci. 2015;237:16–23.
Tun NN, Santa-Catarina C, Begum T, Silveira V, Handro W, Segal Floh EI, et al. Polyamines induce rapid biosynthesis of nitric oxide (NO) in Arabidopsis thaliana seedlings. Plant Cell Physiol. 2006;47:346–54.
Tanou G, Filippou P, Belghazi M, Job D, Diamantidis G, Fotopoulos V, et al. Oxidative and nitrosative-based signaling and associated post-translational modifications orchestrate the acclimation of citrus plants to salinity stress. Plant J. 2012;72:585–99.
Tanou G, Ziogas V, Belghazi M, Christou A, Filippou P, Job D, et al. Polyamines reprogram oxidative and nitrosative status and the proteome of citrus plants exposed to salinity stress. Plant Cell Environ. 2014;37:864–85. This report extensively demonstrates the involvement of PAs in nitro-oxidative homeostasis during salt stress
Shi H, Chan Z. Improvement of plant abiotic stress tolerance through modulation of the polyamine pathway. J Integr Plant Biol. 2014;56:114–21.
Wimalasekera R, Tebartz F, Scherer GFE. Polyamines, polyamine oxidases and nitric oxide in development, abiotic and biotic stresses. Plant Sci. 2011;181:593–603.
Molassiotis A, Fotopoulos V. Oxidative and nitrosative signaling in plants: two branches in the same tree? Plant Signal Behav. 2011;6:210–4.
Nakashima K, Yamaguchi-Shinozaki K. ABA signaling in stress-response and seed development. Plant Cell Rep. 2013;32:959–70.
Wei L, Wang L, Yang Y, Wang P, Guo T, Kang G. Abscisic acid enhances tolerance of wheat seedlings to drought and regulates transcript levels of genes encoding ascorbate-glutathione biosynthesis. Front Plant Sci. 2015;6:458.
Rambla JL, Vera-Sirera F, Blázquez MA, Carbonell J, Granell A. Quantitation of biogenic tetraamines in Arabidopsis thaliana. Anal Biochem. 2010;397:208–11.
Yamaguchi K, Takahashi Y, Berberich T, Imai A, Takahashi T, Michael AJ, et al. A protective role for the polyamine spermine against drought stress in Arabidopsis. Biochem Biophys Res Commun. 2007;352:486–90.
Alcázar R, Planas-Portell J, Saxena T, Zarza X, Bortolotti C, Cuevas J, et al. Putrescine accumulation confers drought tolerance in transgenic Arabidopsis plants over-expressing the homologous arginine decarboxylase 2 gene. Plant Physiol Biochem. 2010;48:547–52.
Alcázar R, Cuevas JC, Patron M, Altabella T, Tiburcio AF. Abscisic acid modulates polyamine metabolism under water stress in Arabidopsis thaliana. Physiol Plant. 2006;128:448–55.
Urano K, Yoshiba Y, Nanjo T, Ito T, Yamaguchi-Shinozaki K, Shinozaki K. Arabidopsis stress-inducible gene for arginine decarboxylase AtADC2 is required for accumulation of putrescine in salt tolerance. Biochem Biophys Res Commun. 2004;313:369–75.
Cuevas JC, López-Cobollo R, Alcázar R, Zarza X, Koncz C, Altabella T, et al. Putrescine is involved in Arabidopsis freezing tolerance and cold acclimation by regulating abscisic acid levels in response to low temperature. Plant Physiol. 2008;148:1094–105.
Espasandin FD, Maiale SJ, Calzadilla P, Ruiz OA, Sansberro PA. Transcriptional regulation of 9-cis-epoxycarotenoid dioxygenase (NCED) gene by putrescine accumulation positively modulates ABA synthesis and drought tolerance in Lotus tenuis plants. Plant Physiol Biochemist. 2014;76:29–35.
Marco F, Alcázar R, Tiburcio AF, Carrasco P. Interactions between polyamines and abiotic stress pathway responses unraveled by transcriptome analysis of polyamine overproducers. Omics J Integr Biol. 2011;15:775–81.
Minocha R, Majumdar R, Minocha SC. Polyamines and abiotic stress in plants: a complex relationship. Front Plant Sci. 2014;5:175.
Mattoo AK, Minocha SC, Minocha R, Handa AK. Polyamines and cellular metabolism in plants: transgenic approaches reveal different responses to diamine putrescine versus higher polyamines spermidine and spermine. Amino Acids. 2010;38:405–13.
Page AF, Cseke LJ, Minocha R, Turlapati SA, Podila GK, Ulanov A, et al. Genetic manipulation of putrescine biosynthesis reprograms the cellular transcriptome and the metabolome. BMC Plant Biol. 2016;16:113.
Sequera-Mutiozabal MI, Erban A, Kopka J, Atanasov KE, Bastida J, Fotopoulos V, et al. Global metabolic profiling of Arabidopsis polyamine oxidase 4 (AtPAO4) loss-of-function mutants exhibiting delayed dark-induced senescence. Front Plant Sci. 2016;7:173. This study demonstrates the metabolic connection between Spm and central metabolites of sugar and lipid metabolism (e.g. pyruvate and myo-Inositol)
Nahar K, Hasanuzzaman M, Alam MM, Fujita M. Exogenous spermidine alleviates low temperature injury in mung bean (Vigna radiata L.) seedlings by modulating ascorbate-glutathione and glyoxalase pathway. Int J Mol Sci. 2015;16:30117–32.
Majumdar R, Barchi B, Turlapati SA, Gagne M, Minocha R, Long S, et al. Glutamate, ornithine, arginine, proline, and polyamine metabolic interactions: the pathway is regulated at the post-transcriptional level. Front Plant Sci. 2016;7:78.
Zarza X, Atanasov KE, Marco F, Arbona V, Carrasco P, Kopka J, et al. Polyamine oxidase 5 loss-of-function mutations in Arabidopsis thaliana trigger metabolic and transcriptional reprogramming and promote salt stress tolerance. Plant, Cell Environ. 2016; (in press) 1–16. This study provides evidence that T-SPM is involved in plant tolerance to salt stress.
Bouché N, Lacombe B, Fromm H. GABA signaling: a conserved and ubiquitous mechanism. Trends Cell Biol. 2003;13:607–10.
Bouché N, Fromm H. GABA in plants: just a metabolite? Trends Plant Sci. 2004;9:110–5.
Kang J, Turano FJ. The putative glutamate receptor 1.1 (AtGLR1.1) functions as a regulator of carbon and nitrogen metabolism in Arabidopsis thaliana. Proc Natl Acad Sci U S A. 2003;100:6872–7.
Shelp BJ, Bozzo GG, Trobacher CP, Zarei A, Deyman KL, Brikis CJ. Hypothesis/review: contribution of putrescine to 4-aminobutyrate (GABA) production in response to abiotic stress. Plant Sci. 2012;193–194:130–5.
Handa AK, Mattoo AK. Differential and functional interactions emphasize the multiple roles of polyamines in plants. Plant Physiol Biochem. 2010;48:540–6.
Van den Ende W. Sugars take a central position in plant growth, development and, stress responses. A focus on apical dominance. Front Plant Sci. 2014;5:1–3.
Krasensky J, Broyart C, Rabanal F, Jonak C. The redox-sensitive chloroplast trehalose-6-phosphate phosphatase AtTPPD regulates salt stress tolerance. Antioxid Redox Signal. 2014;21:1–16.
Kasukabe Y, He LX, Nada K, Misawa S, Ihara I, Tachibana S. Overexpression of spermidine synthase enhances tolerance to multiple environmental stresses and up-regulates the expression of various stress regulated genes in transgenic Arabidopsis thaliana. Plant Cell Physiol. 2004;45:712–22.
Kusano T, Suzuki H, editors. Polyamines: a universal molecular nexus for growth, survival, and specialized metabolism. Tokio: Springer; 2015.
Agudelo-Romero P, Ali K, Choi YH, Sousa L, Verpoorte R, Tiburcio AF, et al. Plant physiology and biochemistry perturbation of polyamine catabolism affects grape ripening of Vitis vinifera cv. Trincadeira. Plant Physiol Biochem. 2014;74:141–55.
Bae H, Kim SH, Kim MS, Sicher RC, Lary D, Strem MD, et al. The drought response of Theobroma cacao (cacao) and the regulation of genes involved in polyamine biosynthesis by drought and other stresses. Plant Physiol Biochem. 2008;46:174–88.
Hatmi S, Gruau C, Trotel-Aziz P, Villaume S, Rabenoelina F, Baillieul F, et al. Drought stress tolerance in grapevine involves activation of polyamine oxidation contributing to improved immune response and low susceptibility to Botrytis cinerea. J Exp Bot. 2014;66:775–87.
An ZF, Li CY, Zhang LX. Alva a. K. Role of polyamines and phospholipase D in maize (Zea mays L.) response to drought stress. South African J Bot. 2012;83:145–50.
Mao X, Zhang H, Tian S, Chang X, Jing R. TaSnRK2.4, an SNF1-type serine/threonine protein kinase of wheat (Triticum aestivum L.), confers enhanced multistress tolerance in Arabidopsis. J Exp Bot. 2010;61:683–96.
Kim SH, Kim SH, Palaniyandi SA, Yang SH, Suh J-W. Expression of potato S-adenosyl-l-methionine synthase (SbSAMS) gene altered developmental characteristics and stress responses in transgenic Arabidopsis plants. Plant Physiol Biochem. 2015;87:84–91.
Mehta RA, Cassol T, Li N, Ali N, Handa AK, Mattoo AK. Engineered polyamine accumulation in tomato enhances phytonutrient content, juice quality, and vine life. Nat Biotechnol. 2002;20:613–8.
Soudek P, Ursu M, Petrová Š, Vaněk T. Improving crop tolerance to heavy metal stress by polyamine application. Food Chem. 2016;213:223–9.
Montesinos-Pereira D, Barrameda-Medina Y, Romero L, Ruiz JM, Sánchez-Rodríguez E. Genotype differences in the metabolism of proline and polyamines under moderate drought in tomato plants. Plant Biol. 2014;16:1050–7.
Rouphael Y, Colla G, Bernardo L, Kane D, Trevisan M, Lucini L. Zinc excess triggered polyamines accumulation in lettuce root metabolome, as compared to osmotic stress under high salinity. Front Plant Sci. 2016;7:1–10.
Nahar K, Hasanuzzaman M, Alam MM, Rahman A, Mahmud J-A, Suzuki T, et al. Insights into spermine-induced combined high temperature and drought tolerance in mung bean: osmoregulation and roles of antioxidant and glyoxalase system. Protoplasma. 2016; (in press) doi: 10.1007/s00709-016-0965-z.
Romero L, Ruiz JM. Accumulation of free polyamines enhances the antioxidant response in fruits of grafted tomato plants under water stress. J Plant Physiol. 2016;190:72–8.
Li Z, Jing W, Peng Y, Zhang XQ, Ma X, Huang LK, et al. Spermine alleviates drought stress in white clover with different resistance by influencing carbohydrate metabolism and dehydrins synthesis. PLoS One. 2015;10:e0120708.
Peng D, Wang X, Li Z, Zhang Y, Peng Y, Li Y, et al. NO is involved in spermidine-induced drought tolerance in white clover via activation of antioxidant enzymes and genes. Protoplasma. 2016;253:1243–54. This study provides evidence that cross-talk signaling between NO and Spd is essential to induce anti-oxidant machinery towards protection against water stress
Wang P, Du Y, Hou Y-J, Zhao Y, Hsu C-C, Yuan F, et al. Nitric oxide negatively regulates abscisic acid signaling in guard cells by S-nitrosylation of OST1. Proc Natl Acad Sci. 2015;112:613–8.
Xie Y, Mao Y, Zhang W, Lai D, Wang Q, Shen W. Reactive oxygen species-dependent nitric oxide production contributes to hydrogen-promoted stomatal closure in Arabidopsis. Plant Physiol. 2014;165:759–73.
Chen J, Shang Y-T, Wang W-H, Chen X-Y, He E-M, Zheng H-L, et al. Hydrogen sulfide-mediated polyamines and sugar changes are involved in hydrogen sulfide-induced drought tolerance in Spinacia oleracea seedlings. Front Plant Sci. 2016;7:1–18.
Ma D, Ding H, Wang C, Qin H, Han Q, Hou J, et al. Alleviation of drought stress by hydrogen sulfide is partially related to the abscisic acid signaling pathway in wheat. PLoS One. 2016;11:e0163082.
Liu Y, Liang H, Lv X, Liu D, Wen X, Liao Y. Effect of polyamines on the grain filling of wheat under drought stress. Plant Physiol Biochem. 2016;100:113–29.
Hura T, Dziurka M, Hura K, Ostrowska A, Dziurka K. Free and cell wall-bound polyamines under long-term water stress applied at different growth stages of ×Triticosecale Wittm. PLoS One. 2015;10:e0135002.
Nahar K, Hasanuzzaman M, Rahman A, Alam MM, Mahmud J-A, Suzuki T, et al. Polyamines confer salt tolerance in mung bean (Vigna radiata L.) by reducing sodium uptake, improving nutrient homeostasis, antioxidant defense, and methylglyoxal detoxification systems. Front Plant Sci. 2016;7:1104.
Zhang Y, Zhang H, Zou ZR, Liu Y, Hu XH. Deciphering the protective role of spermidine against saline-alkaline stress at physiological and proteomic levels in tomato. Phytochemistry. 2015;110:13–21. This study demonstrates the alleviating effect of Spd against salt stress effects by inducing detoxification mechanisms
Hu L, Xiang L, Li S, Zou Z, Hu XH. Beneficial role of spermidine in chlorophyll metabolism and D1 protein content in tomato seedlings under salinity-alkalinity stress. Physiol Plant. 2016;156:468–77.
Shu S, Yuan Y, Chen J, Sun J, Zhang W, Tang Y, et al. The role of putrescine in the regulation of proteins and fatty acids of thylakoid membranes under salt stress. Sci. Rep. 2015;5:14390. This report shows that Put is able to modulate fatty acid mobilization in chloroplasts during salt stress, exerting a protective role for the plant
Yuan Y, Zhong M, Shu S, Du N, Sun J, Guo S. Proteomic and physiological analyses reveal putrescine responses in roots of cucumber stressed by NaCl. Front Plant Sci. 2016;7:1035.
Du J, Shu S, An Y, Zhou H, Guo S, Sun J. Influence of exogenous spermidine on carbon–nitrogen metabolism under Ca(NO3)2 stress in cucumber root. Plant Growth Regul. 2016;129:79–91.
Xing SG, Jun YB, Hau ZW, Liang LY. Higher accumulation of γ-aminobutyric acid induced by salt stress through stimulating the activity of diamine oxidases in Glycine max (L.) Merr. roots. Plant Physiol Biochem. 2007;45:560–6.
Zarei A, Trobacher CP, Shelp BJ. Arabidopsis aldehyde dehydrogenase 10 family members confer salt tolerance through putrescine-derived 4-aminobutyrate (GABA) production. Sci Rep. 2016;6:35115.
Hu X, Xu Z, Xu W, Li J, Zhao N, Zhou Y. Application of γ-aminobutyric acid demonstrates a protective role of polyamine and GABA metabolism in muskmelon seedlings under Ca(NO3)2 stress. Plant Physiol Biochem. 2015;92:1–10.
Li S, Jin H, Zhang Q. The effect of exogenous spermidine concentration on polyamine metabolism and salt tolerance in zoysiagrass (Zoysia japonica Steud) subjected to short-term salinity stress. Front Plant Sci. 2016;7
López-Gómez M, Hidalgo-Castellanos J, Lluch C, Herrera-Cervera JA. 24-Epibrassinolide ameliorates salt stress effects in the symbiosis Medicago truncatula-Sinorhizobium meliloti and regulates the nodulation in cross-talk with polyamines. Plant Physiol Biochem. 2016;108:212–21.
Chaves MM, Pereira JS, Maroco J, Rodrigues ML, Ricardo CPP, Osório ML, et al. How plants cope with water stress in the field? Photosynthesis and growth. Ann Bot. 2002;89:907–16.
Rangan P, Subramani R, Kumar R, Singh AK, Singh R. Recent advances in polyamine metabolism and abiotic stress tolerance. Biomed Res Int. 2014;2014:239621.
García-Mata C, Lamattina L. Hydrogen sulphide, a novel gasotransmitter involved in guard cell signalling. New Phytol. 2010;188:977–84.
Kaur N, Dhawan M, Sharma I, Pati PK. Interdependency of reactive oxygen species generating and scavenging system in salt sensitive and salt tolerant cultivars of rice. BMC Plant Biol. 2016;16:1–13.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Miren Sequera-Mutiozabal, Chrystalla Anotoniou, Antonio F. Tiburcio, Rubén Alcázar, and Vasileios Fotopoulos each declare no potential conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Enhancing Agricultural Production
Rights and permissions
About this article
Cite this article
Sequera-Mutiozabal, M., Antoniou, C., Tiburcio, A.F. et al. Polyamines: Emerging Hubs Promoting Drought and Salt Stress Tolerance in Plants. Curr Mol Bio Rep 3, 28–36 (2017). https://doi.org/10.1007/s40610-017-0052-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40610-017-0052-z