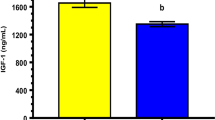
Abstract
The objective of this study is to determine the optimal conditions for human semen incubation treated with exogenous platelet activating factor (ePAF) for intra-uterine insemination (IUI). This prospective study was carried out on 32 infertile men and each semen sample was processed with the ISolate Sperm Separation Medium, washed with sperm washing medium (SWM) and resuspended either in SWM alone (control samples), or with ePAF 0.1, 0.5, and 1.0 μM. Each concentration was subsequently incubated and evaluated at 5, 15, 30, and 60 min. The motility parameters were evaluated by the computer-aided sperm analysis (C.A.S.A.) system. Curvilinear velocity, straight line velocity, average path velocity, rapid and progressive motility significantly increased compared to control samples at an ePAF concentration of 0.1 μM (with at least 15 min of incubation). The best results were obtained with ePAF concentrations of: 0.1 μM (60 min of incubation) and 0.5 μM (30–60 min of incubation). In conclusion, results are enhanced when ePAF is added to standard semen preparation for IUI. An ePAF concentration of 0.1 μM, with an incubation time of 15 min, can be used for semen samples with normal motility. Whilst, for semen samples with poor motility, the ePAF concentration is best increased to 0.5 μM and/or the incubation time prolonged to 60 min.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ishii S, Nagase T, Shimizu T. Platelet-activating factor receptor. Prostaglandins Other Lipid Mediat 2002, 68-69: 599–609.
Minhas BS, Kumar R, Ricker DD, Robertson JL, Dodson MG. The presence of platelet-activating factor-like activity in human spermatozoa. Fertil Steril 1991, 55: 372–6.
Roudebush WE, Purnell ET. Platelet-activating factor content in human spermatozoa and pregnancy outcome. Fertil Steril 2000, 74: 257–60.
Lapetina EG. Platelet-activating factor stimulates the phosphatidylinositol cycle. Appearance of phosphatidic acid is associated with the release of serotonin in horse platelets. J Biol Chem 1982, 257: 7314–7.
Ahmed A, Sage SO, Plevin R, Shoaibi MA, Sharkey AM, Smith SK. Functional platelet-activating factor receptors linked to inositol lipidic hydrolysis, calcium mobilization and tyrosine kinase activity in the human endometrial HEC-1B cell line. J Reprod Fertil 1994, 101: 459–66.
Roudebush WE, Wild MD, Maguire EH. Expression of the platelet-activating factor receptor in human spermatozoa: differences in messenger ribonucleic acid content and protein distribution between normal and abnormal spermatozoa. Fertil Steril 2000, 73: 967–71.
Sathananthan AH, Ratnam SS, Ng SC, Tarin JJ, Gianaroli L, Trounson A. The sperm centriole: its inheritange, replication and perpetuation in early human embryos. Hum Reprod 1996, 11: 345–56.
Sengoku K, Tamate K, Takaoka Y, Ishikawa M. Effects of platelet activating factor on human sperm function in vitro. Hum Reprod 1993, 8: 1443–7.
Krausz C, Gervasi G, Forti G, Baldi E. Effect of platelet-activating factor on motility and acrosome reaction of human spermatozoa. Hum Reprod 1994, 9: 471–6.
Levine AS, Kort HI, Toledo AA, Roudebush WE. A review of the effect of platelet-activating factor on male reproduction and sperm function. J Androl 2002, 23: 471–6.
Roudebush WE, Massey JB, Elsner CW, Shapiro DB, Mitchell-Leef D, Kort HI. The significance of platelet-activating factor and fertility in the male primate: a review. J Med Primatol 2005, 34: 20–4.
Roudebush WE, Toledo AA, Kort HI, Mitchell-Leef D, Elsner CW, Massey JB. Platelet-activating factor significantly enhances intrauterine insemination pregnancy rates in non-male factor infertility. Fertil Steril 2004, 82: 52–6.
Grigoriou O, Makrakis E, Konidaris S, et al. Effect of sperm treatment with exogenous platelet-activating factor on the outcome of intrauterine insemination. Fertil Steril 2005, 83: 618–21.
Yogev L, Gamzu R, Botchan A, Hauser R, Paz G, Yavetz H. Zona pellucida binding improvement effect of different sperm preparation techniques is not related to changes in sperm motility characterization. Fertil Steril 2000, 73: 1120–5.
Krause W. The significance of computer-assisted semen analysis (CASA) for diagnosis in andrology and fertility prognosis. Int J Androl 1995, 18 (Suppl 2): 32–5.
Ricker DD, Minhas BS, Kumar R, Robertson JL, Dodson MG. The effects of platelet-activating factor on the motility of human spermatozoa. Fertil Steril 1989, 52: 655–8.
Hellstrom WJG, Wang R, Sikka SC. Platelet activating factor stimulates motion parameters of cryopreserved human sperm. Fertil Steril 1991, 56: 768–70.
World Health Organization. WHO Laboratory Manual for the Examination of Human Semen and Sperm — Cervical Mucus Interaction. 4th edition. Cambridge: Cambridge University Press World Health Organization, 1999.
Mack SO, Wolf DP, Tash JS. Quantitation of specific parameters of motility in large numbers of human sperm by digital image processing. Biol Reprod 1988, 38: 270–81.
Mortimer ST. A critical review of the physiological importance and analysis of sperm movement in mammals. Hum Reprod Update 1997, 3: 403–39.
Moohan JM, Lindsay KS. Spermatozoa selected by a discontinuous Percoll density gradient exhibit better motion characteristic, more hyperactivation and longer survival than direct swim-up. Fertil Steril 1995, 64: 160–5.
Shibahara H, Obara H, Ayustawati, et al. Prediction of pregnancy by intrauterine insemination using C.A.S.A. estimates and strict criteria in patients with male factor infertility. Int J Androl 2004, 27: 63–8.
Liu DY, Clarke GN, Baker HWG. Relationship between sperm motility assessed with the Hamilton-Thorn motility analyzer and fertilization rates in vitro. J Androl 1991, 4: 231–9.
Barratt CL, Tomlinson MJ, Cooke ID. Prognostic significance of computerized motility analysis for in vivo fertility. Fertil Steril 1993, 60: 520–5.
Irvine DS, Macleod IC, Templeton AA, Masterton A, Taylor A. A prospective clinical study of the relationship between the computer-assisted assessment of human semen quality and the achievement of pregnancy in vivo. Hum Reprod 1994, 9: 2324–34.
Mortimer ST, Mortimer D. Kinematics of human spermatozoa incubated under capacitating conditions. J Androl 1990, 11: 195–203.
Mortimer ST, Swan MA. Effect of image sampling frequency on establishing and smoothing-independent kinematic values of capacitating human sperm. Hum Reprod 1999, 14: 997–1004.
Pacey AA, Ladbrook MB, Barratt CLR, Cooke ID. The potential shortcomings of measuring hyperactivated motility by computeraided sperm analysis when sperm motion is multiphasic. Hum Reprod Update 1997, 3: 185–93.
Chantler E, Abraham-Peskir J, Roberts C. Consistent presence of two normally distributed sperm subpopulations within normozoospermic human semen: a kinematic study. Int J Androl 2004, 27: 350–9.
McCann CT, Chantler E. Properties of sperm separated using Percoll and IxaPrep density gradients. A comparisons made using CASA, longevity, morphology and the acrosome reaction. Int J Androl 2000, 23: 205–9.
Ford WC, McLaughlin EA, Prior SM, Rees JM, Wardle PG, Hull MG. The yield, motility and performance in the hamster egg test of human spermatozoa prepared from cryopreserved semen by four different methods. Hum Reprod 1992, 7: 654–9.
Jaroudi KA, Carver-Ward JA, Hamilton CJ, Sieck UV, Sheth KV. Percoll semen preparation enhances human oocyte fertilization in male-factor infertility as shown by a randomized cross-over study. Hum Reprod 1993, 8: 1438–42.
De Geyter C, De Geyter M, Koppers B, Nieschlag E. Diagnostic accuracy of computer-assisted sperm motion analysis. Hum Reprod 1998, 13: 2512–20.
Zhu J, Massey JB, Mitchell-Leef D, Elsner CW, Kort HI, Roudebush WE. Platelet-activating factor acetylhydrolase activity affects sperm motility and serves as a decapacitation factor. Fertil Steril 2006, 85: 391–4.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grassi, G., Cappello, N., Gheorghe, M.F. et al. Exogenous platelet-activating factor improves the motility of human spermatozoa evaluated with C.A.S.A.: Optimal concentration and incubation time. J Endocrinol Invest 33, 684–690 (2010). https://doi.org/10.1007/BF03346670
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03346670