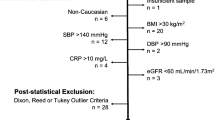
Abstract
Reference intervals in clinical chemistry are commonly based on results of measurements in reference western population or are taken from the western literature. Reference Values are thought to aid physicians to interpret results of measurements and, should be representative of a defined group of individuals. This group should be as similar as possible to the patients under investigation. The reference population in this, study has been recruited from the individuals attending the Health Plan Clinic who fulfill the defined inclusion and exclusion criteria as well as defined partition criteria. The samples were sorted based on the decision by the physician. The emerging group of individuals was considered as a reference population for the hospital patients and the results of measurements in this study was evaluated statistically, to stress on the urgent need to establish the in-house reference values. The reference limits are defined as the central 95 percentile of the population after eliminating the outliers. The lower, reference limit is the 2.5 percentile while the upper reference limit constituted the 97.5 percentile for the population.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- AST:
-
Aspartate Aminotransferase
- ALT:
-
Alanine Aminotranferase
- ALP:
-
Alkaline Phosphatase
- γ-GT:
-
Gamma—Glutamyl Transferase
References
Burtis, C.A. and Ashwood, E.R. (1999) Establishment and use of Reference Values. In:Tietz Textbook of Clinical Chemistry. Chapter 14, 3rd edition W.B. Saunders Company, Philadelphia U.S.A. 336–356.
Grasbeck, R. and Alstrom, T. (Eds.) (1981)Reference Values in laboratory medicine: The current state of Art Chichester, England, John Wiley and Sons, Ltd.
Solberg, H.E. and Stamm, D. (1991) Approved recommendation on the theory of Reference Values. Part 4 control of analytical variation in the production, transfer and application of Reference Values. Eur. J. Clin. Chem. Clin. Biochem. 29, 531–535.
Harris, E.K. and Boyd, J.C. (1995)Statistical bases of Reference Values in laboratory medicine New York: Marcel Dekker.
Nano, V., Slami, T. T., Kairisto, V., Kouri, T., Rajamaki, A., Uusipaikka, E.,et al (1991)Pediatric values for hematological parameters from diagnosis-selected hospital patient population. IX European Congress of Clinical Chemistry, Krakow, Poland, Sep. 8–14.
National Committee for Clinical Laboratory Standards: How to define, determine and utilize reference intervals in the Clinical laboratory: Proposed guidelines (2000) 2 ed., NCCLS Document C28-A2, Villanova, PA, NCCLS.
Kingsley, G.R. (1942) The direct biuret method for determination of serum proteins as applied to photoelectric and visual colorimetry. J. Lab. Clin. Med 27, 840–845.
Silverman, L.M. and Christenson, R.H. (1993) Amino acids and proteins. In:Tietz Textbook of Clinical Chemistry. 2nd ed. C.A. Burtis and E.R. Ashwood (Eds.). Philadelphia, W.B. Saunders, 696–698.
Doumas, B.T. and Peters, T., Jr. (1997) Serum and Urine albumin: A progress report on their measurement and clinical significance. Clin. Chem. Acta. 258, 3–20.
Peters, T. Jr. (1996)All about albumin: Serum Albumin- Biochemistry, genetics and Medical Applications. Washington D.C., American Association of Clinical Chemistry Press.
Doumas, B.T., Perry, B.W., Sasse, E.A.,et al. (1973) Standardisation in bilirubin assays: Evaluations of selected methods and stability of bilirubin solutions. Clin. Chem. 19, 984–993.
Saris, N.E. (1978) Revised IFCC method for aspartate aminotransferase. Clin. Chem. 24, 720–721.
Bergmeyer, H.U., Scheibe, P. and Wahlefeld, A.W. (1978) Optimization of methods for aspartate aminotransferase and alanine aminotransferase. Clin. Chem. 24, 1.
Bowers, G.N. and McComb, R.B. (1966) A continuous spectrophotometric method for measuring the activity of serum alkaline phosphatase. Clin. Chem. 12, 70.
Shaw, L.M., Stromme, J.H., London, J.L. and Theodorsen, L. (1983) IFCC methods for the dtermination of enzymes part 4. IFCC method for gamma glutamyl transferase [(gammaglutamyl)-peptide; amino acid gamma glutamyl transferase, EC 2.3.2.2.]. Clin. Chem. Acta. 15F–338F.
Whitehead, T.P., Robinson, D., Hale, A.C. and Bailey, A.R. (1994) Clinical Chemistry and haematology: adult Reference Values. London: BUPA Medical Research and Development Ltd.
Anders, Kallner, Elisabeth, Gustavsson and Eva, Hendig. (2000) Can age and sex related reference intervals be derived for non-healthy and non-diseased individuals from results of measurements in Primary Health Care? Clin. Chem. Lab. Med. 38, 7, 633–654.
Gyorgy, Abel and Darryl, E. Palmer-Toy (2002) Heme Synthesis and catabolism. In:Clinical Laboratory Medicine. Kenneth, D. McClatchey. (Ed.). 2nd Ed. Lippincott williams and Wilkins. 414–416.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Furruqh, S., Anitha, D. & Venkatesh, T. Estimation of reference values in liver function test in health plan individuals of an urban south Indian population. Indian J Clin Biochem 19, 72–79 (2004). https://doi.org/10.1007/BF02894260
Issue Date:
DOI: https://doi.org/10.1007/BF02894260