Abstract
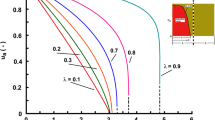
Four diffusion models for the dynamic adsorber, i.e. LDF model, single diffusivity diffusion model, two diffusivity diffusion model for beds packed with bidisperse and/or zeolite-type particles, were considered. The third moments for the four diffusion models were obtained. Relations between the system parameters involved in each model were derived by matching mean, vanance or the third moment between diffusion models. The two relations from either variance or the third moment matching were examined to investigate which one is superior when model simplification is required, by comparing the time domain elution curves for the single and the two diffusivity diffusion models. For the symmetric elution curves, relation from the variance matching is much better as expected, than the relation matching the third moment which measures skewness about mean. As the elution curves become highly asymmetric, eluting shortly after injection and exhibiting long tailing due to both the small intraparticle diffusivities and small space time in the adsorber, either relation failed to satisfactorily simplify the two diffusivity diffusion model. Contrary to the expectation that the third moment matching would work better in the asymmetric curves due to the nature of the third moment, variance matching still gives slightly better results. Relation from the variance, instead of the third moment, matching is strongly recommended for model simplification due to its simplicity in formula.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dang, N. and Gibilaro, L., “Numerical Inversion of Laplace Transforms by a Simple Curve Fitting Technique”,Chem. Eng. J.,8, 157(1974).
Dogu, G. and Smith, J. M., “Rate Parameters from Dynamic Experiments with Single Catalysts Pellets”,Chem. Eng. Sci,31, 123 (1976).
Glueckauf, E. and Coates, J. E., “Theory of Chromatography”,J. Chem. Soc, 1315(1947).
Hashimoto, N. and Smith, J. M., “Macropore Diffusion in Molecular Sieve Pellets by Chromatography”,Ind. Eng. Chem. Fundam.,12(3), 353(1973).
Hashimoto, N. and Smith, J. M., “Diffusion in Bidisperse Porous Catalyst Pellets”,Ind. Eng. Chem. Fundam.,13(2), 115(1974).
Hsu, L. K. P. and Haynes, H. W., “Effective Diffusivity by the Gas Chromatography Technique: Analysis and Application to Measurements of Diffusion of Various Hydrocarbons in Zeolite NaY”,AIChE J.,27(1), 81 (1981).
Kim, D. H., “Single Effective Diffusivities for Dynamic Adsorption in Bidisperse Adsorbents”,AIChE J.,36(2), 302 (1990).
Raghavan, N. S. and Ruthven, D. M., “Simulation of Chromatographic Response in Columns Packed with Bidisperse Structured Particles”,Chem. Eng. Sci.,40(5), 699(1985).
Ruthven, D., “Principles of Adsorption and Adsorption Processes”, John Wiley and Sons, New York (1984).
Schneider, P. and Smith, J. M., “Adsorption Rate Constants from Chromatography”,AIChE J.,14(5), 762(1968).
Suzuki, M. and Smith, J. M., “Dynamics of Diffusion and Adsorption in a Single Catalyst Pellet”,AIChE J.,18(2), 326(1972).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Song, DI. The second vs. the third moment matching between diffusion models for dynamic adsorber. Korean J. Chem. Eng. 13, 60–66 (1996). https://doi.org/10.1007/BF02705890
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02705890