Abstract
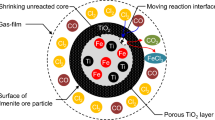
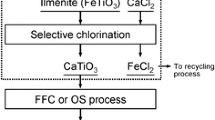
The intrinsic kinetics of the selective chlorination of iron from ilmenite ore using carbon monoxide as the reducing agent were studied in a shallow fluidized bed. Experiments on the effects of chlorination temperature, carbon monoxide and chlorine gas partial pressures, and particle size were conducted in the absence of mass- and heat-transfer influences. Results indicate that the kinetics in the temperature range 923 to 1123 K are represented by the following pore-blocking rate law: λ[ exp (XFe/λ) − 1 ] = 33.7 exp (− E/RT)p 0.52co 0.322 t where E is 37.2 kJ/mol and p and t are in atm (=101.3 kPa) and minutes, respectively. The partial pressure of carbon monoxide was found to affect the chlorination rate more strongly than that of chlorine. A reaction mechanism in which iron in ilmenite reacts with chlorine before the liberated oxygen is removed by carbon monoxide is proposed.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
J.B. Rosenbaum:Light Met., 1982, pp. 1123-34.
N.H. Orr:Light Met., 1982, pp. 1149-56.
R. Powell:Titanium Oxide and Titanium Tetrachloride, Noyes Development Co., Park Ridge, NJ, 1968, pp. 1–6.
J. Barksdale:Titanium: Its Occurrence, Chemistry, and Technology, Ronald Press Co., New York, NY, 1966, pp. 10–46 and 213-23.
H.M. Harris, A.W. Henderson, and T.T. Campbell: U.S. Bureau of Mines, R.I. 8165, 1976, 19 pp.
L.K. Doraiswamy, H.C. Bijawat, and M.V. Kunte:Chem. Eng. Prog., 1959, vol. 55 (10), pp. 80–88.
B.P. Judd and E.R. Palmer:Proc. Australas. Inst. Min. Metall., 1973, vol. 247, pp. 23–33.
M.H. Tikkanan, T. Tyynela, and E. Vuoristo:Metall. Soc. Conf., [Proc] 1964, vol. 24, pp. 269–82.
G.W. Elger, J.B. Wright, J.E. Tress, H.E. Bell, and R.R. Jordan: U.S. Bureau of Mines, R.I. 9002, 1986, 24 pp.
G.W. Elger and W.A. Stickney: U.S. Bureau of Mines, Technical Progress Report-37, 1971, 9 pp.
C.D. Shiah: U.S. Patent No. 3,252,787, 1966.
S. Yamada:Ind. Miner., 1976, vol. 1, pp. 33–40.
V.G. Neurgaonkar, A.N. Gorkan, and K. Joseph:J. Chem. Technol. Biotechnol., 1986, vol. 36, pp. 27–30.
C.M. Lakeshmanan, H.E. Moelescher, and B. Chennakesavan:Chem. Eng. Sci., 1968, vol. 20, pp. 1107–13.
A.S. Athavale and V.A. Altekar:Ind. Eng. Chem. Process Des. Dev., 1971, vol. 10 (4), pp. 523–30.
K.I. Rhee and H.Y. Sohn:Metall. Trans. B, 1990, vol. 21B, pp. 331–40.
K.I. Rhee and H.Y. Sohn:Metall. Trans. B, 1990, vol. 21B, pp. 341–47.
I.M. Kolthoff, E.B. Sandell, E.J. Meehan, and S. Bruckenstein:Quantitative Chemical Analysis, 4th ed., 1969, pp. 752-53 and 838.
R.A. Robie, B.S. Hemingway, and J.R. Fisher: U.S. Geological Survey Bulletin 1452, 1979.
L.B. Pankratz, J.M. Stuve, and N.A. Gokcen: Bureau of Mines Bulletin 677, 1984, pp. 145-78.
D.J. Milne and R.D. Holliday:Ind. Eng. Chem. Process Des. Dev., 1975, vol. 14 (4), pp. 442–52.
J.F. Richardson and J. Szekely:Trans. Inst. Chem. Eng., 1961, vol. 39, pp. 212–21.
K.I. Rhee: Ph.D. Dissertation, University of Utah, Salt Lake City, UT, 1988.
Z.I. Latina and A.A. Furman:Zh. Prikl. Khim., 1970, vol. 43 (4), pp. 830–34.
A.B. Bezukladninov and V.A. Pronichkin:Zh. Prikl. Khim., 1972, vol. 45 (6), pp. 1221–24.
B. Yu:Acta Metall. Sin., 1982, vol. 18 (2), pp. 164–75.
D.G. James:Trans. Inst. Min. Metall., 1973, vol. 82, pp. C186-C192.
A. Fuwa, E. Kimura, and S. Fukushima:J. Jpn. Inst. Met., 1980, vol. 44 (8), pp. 839–45.
Author information
Authors and Affiliations
Additional information
Formerly Graduate Student at the Department of Metallurgical Engineering, University of Utah
Rights and permissions
About this article
Cite this article
Rhee, K.I., Sohn, H.Y. The selective chlorination of iron from llmenite ore by CO-Cl2 mixtures: Part I. intrinsic kinetics. Metall Trans B 21, 321–330 (1990). https://doi.org/10.1007/BF02664200
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02664200