Abstract
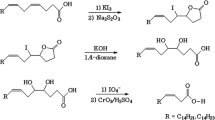
Incubation of [1-14C]linoleic acid with an enzyme preparation obtained from the red algaLithothamnion corallioides Crouan resulted in the formation of 11-hydroxy-9(Z),12(Z)-octadecadienoic acid as well as smaller amounts of 9-hydroxy-10(E),12(Z)-octadecadienoic acid, 13-hydroxy-9(Z),11(E)-octadecadienoic acid and 11-keto-9(Z),12(Z)-octadecadienoic acid. Steric analysis showed that the 11-hydroxyoctadecadienoic acid had the (R) configuration. The 9- and 13-hydroxyoctadecadienoic acids were not optically pure, but were due to mixtures of 75% (R) and 25% (S) enantiomers (9-hydroxyoctadecadienoate), and 24% (R) and 76% (S) enantiomers (13-hydroxy-octadecadienoate). 11-Hydroxyoctadecadienoic acid was unstable at acidic pH. In acidified water, equal parts of 9(R,S)-hydroxy-10(E),12(Z)-octadecadienoate and 13(R,S)-hydroxy-9(Z),11(E)-octadecadienoate, plus smaller amounts of the corresponding (E),(E) isomers were produced. In aprotic solvents, acid treatment resulted in dehydration and in the formation of equal amounts of 8,10,12- and 9,11,13-octadecatrienoates. The enzymatic conversion of linoleic acid into the hydroxyoctadecadienoic acids and the ketooctadecadienoic acid was oxygen-dependent; however, inhibitor experiments indicated that neither lipoxygenase nor cytochrome P-450 were involved in the conversion. This conclusion was supported by experiments with18O2 and H2 18O, which demonstrated that the hydroxyl oxygen of the hydroxy-octadecadienoic acids and the keto oxygen of the 11-ketooctadecadienoic acid were derived from water and not from molecular oxygen.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- ETYA:
-
5,8,11,14-eicosatetraynoic acid
- GC/MS:
-
gas chromatography/mass spectrometry
- GLC:
-
gas-liquid chromatography
- 12(R),13(S)-diHETE:
-
12(R),13(S)-dihydroxy-5(Z),8(Z)10(E),14(Z)-eicosatetraenoic acid
- 5-HETE:
-
5-hydroxy-6(E),8(Z),11(Z),14(Z)-eicosatetraenoic acid
- 11-HETE:
-
11-hydroxy-5(Z),8(Z),12(E),14(Z)-eicosatetraenoic acid
- 12-HETE:
-
12-hydroxy-5(Z),8(Z),10(E),14(Z)-eicosatetraenoic acid
- 15-HETE:
-
15-hydroxy-5(Z),8(Z),11(Z),13(E)-eicosatetraenoic acid
- 9-HOD:
-
9-hydroxy-10(E),12(Z)-octadecadienoic acid
- 13-HOD:
-
13-hydroxy-9(Z),11(E)-octadecadienoic acid
- 12(S)-HPETE:
-
12(S)-hydroperoxy-5(Z),8(Z),10(E),14(Z)-eicosatetraenoic acid
- MC, (−)-menthoxycarbonyl:
-
Me3Si, trimethylsilyl
- NDGA:
-
nordihydroguaiaretic acid
- TLC:
-
thin-layer chromatography
References
Gerwick, W.H., Moghaddam, M.F., and Hamberg, M. (1991)Arch. Biochem. Biophys. 290, 436–444.
Gerwick, W.H., and Bernart, M.W. (1992) inAdvances in Marine Biotechnology: Pharmaceutical and Bioactive Natural Products (Zaborsky, O.R., and Attaway, D.H., eds.), Vol. 1, Plenum Press, New York, in press.
Moghaddam, M.F., and Gerwick, W.H. (1991)J. Nat.Prod. 54, 1619–1624.
Guerriero, A., D'Ambrosio, M., and Pietra, F. (1990)Helv. Chim. Acta 73, 2183–2189.
Hamberg, M. (1989)Lipids 24, 249–255.
Hamberg, M. (1987)Biochim. Biophys. Acta 920, 76–84.
Bradford, M.M. (1976)Anal. Biochem. 72, 248–254.
Hamberg, M., and Fahlstadius, P.(1992)Plant Physiol., in press.
Hamberg, M. (1971)Anal. Biochem. 43, 515–526.
Hamberg, M. (1991)Lipids 26, 407–415.
Crombie, L., and Jacklin, A.G. (1957)J. Chem. Soc., 1632-1646.
Samuelsson, B., Dahlén, S.-E., Lindgren, J.-Å, Rouzer, C.A., and Serhan, C.N. (1987)Science 237, 1171–1176.
Gardner, H.W. (1991)Biochim. Biophys. Acta 1084, 221–239.
Smith, W.L., and Marnett, L.J. (1991)Biochim. Biophys. Acta 1083, 1–17.
Capdevila, J., Chacos, N., Werringloer, J., Prough, R.A., and Estabrook, R.W. (1981)Proc. Natl. Acad. Sci. USA 78, 5362–5366.
Croteau, R., and Kolattukudy, P.E. (1975)Arch. Biochem. Biophys. 170, 61–72.
Salaün, J.-P., Benveniste, I., Reichhart, D., and Durst, F. (1978)Eur. J. Biochem. 90, 155–159.
Hamberg, M., and Hamberg, G. (1990)Arch. Biochem. Biophys. 283, 409–416.
Blée, E., and Schuber, F. (1990)J. Biol. Chem. 265, 12887–12894.
Porter, N.A., Ziegler, C.B., Khouri, F.F., and Roberts, D.H. (1985)J. Org. Chem. 50, 2252–2258.
Badami, R.C., and Patil, K.B. (1981)Prog. Lipid. Res. 19, 119–153.
Author information
Authors and Affiliations
Additional information
The term “oxylipin” was introduced recently (ref. 1) as an encompassing term for oxygenated compounds which are formed from fatty acids by reaction(s) involving at least one step of mono- or dixoygenase-catalyzed oxygenation.
About this article
Cite this article
Hámberg, M., Gerwick, W.H. & Åsen, P.A. Linoleic acid metabolism in the red algaLithothamnion corallioides: Biosynthesis of 11(R)-hydroxy-9(Z),12(Z)-octadecadienoic acid. Lipids 27, 487–493 (1992). https://doi.org/10.1007/BF02536128
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02536128