Abstract
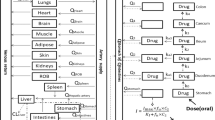
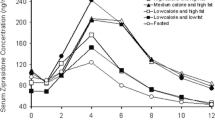
Investigation of the underlying mechanism leading to inter- and intrasubject variations in the plasma concentration-time profiles of drugs (1) can considerably benefit rational drug therapy. The significant effect of gastric emptying on the rate and extent of celiprolol absorption and its role with respect to double-peak formation was demonstrated in the present study. In four dogs racemic celiprolol was dosed perorally in a crossover design during four different phases of the fasted-state gastric cycle and gastric motility was recorded simultaneously using a manometric measurement system. Intravenous doses were also given to obtain disposition and bioavailability parameters. The blood samples were assayed by a stereoselective HPLC method (2). The time to onset of the active phase of the gastric cycle showed an excellent correlation with the time to celiprolol peak concentration. Furthermore, bioavailability was increased when celiprolol was administered during the active phase. Double peaks were observed when the first active phase was relatively short, suggesting that a portion of the drug remained in the stomach until the next active phase. Population pharmacokinetic modeling of the data with a two-compartment open model with two lag times incorporating the motility data confirmed the effect of time to gastric empyting on the variability of the oral pharmacokinetics of celiprolol. The fasted-state motility phases determine the rate and extent of celiprolol absorption and influence the occurrence of double peaks. Peak plasma levels of celiprolol exhibit less variability if lag times, and therefore gastric emptying times, are taken into consideration.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
R. J. Milne and M. T. Buckley. Celiprolol: An undated review of its pharmacokinetic properties and therapeutic efficacy in cardiovascular diseases.Drugs 41:941–969 (1991).
C. Hartmann, D. Krauss, H. Spahn, and E. Mutschler. Simultaneously determination of (R)- and (S)-celiprolol in human plasma and urine: High performance liquid chromatographic assay on a chiral stationary phase with fluorimetric detection.J. Chromatog. 496:387–396 (1989).
C. Hartmann. Enantiomerenanalytik und-kinetik des β-Adrenozeptorenblockers Celiprolol. Ph.D. thesis, Department of Pharmacology, University of Frankfurt, Germany 1989.
R. D. Smith, and P. S. Wolf. Celiprolol. In A. Sciabine (ed.)New Drugs Annual: Cardiovascular Drugs, Vol. 2, Raven, New York 1984.
J. R. Norris, E. H. Lee, D. Muirhead, and S. W. Saunders. A pharmacokinetic evaluation of celiprolol in healthy elderly volunteers.J. Cardiovasc. Pharmacol. 8:S91-S92 (1986).
R. F. Bergstrom, D. R. Kay, and J. G. Wagner. The pharmacokinetics of penicillamine in a female mongrel dog.J. Pharmacokin. Biopharm. 9:603–621 (1981).
C. G. Regårdh, A. Heggelund, K. Kylberg-Hanssen, and P. Lundborg. Pharmacokinetics of pafenolol after iv and oral administration of three separate doses of different strength to man.Biopharm. Drug Disp. 11:607–617 (1990).
H. Lennernäs, and C. G. Regårdh. Pharmacokinetics of pafenolol in the rat: A suitable model for studying absorption mechanisms of a drug exhibiting unusual absorption properties in man.Biopharm. Drug Disp. 11:619–631 (1990).
T. Yamagushi, C. Ikeda, and Y. Sekine. Intestinal absorption of a β-adrenergic blocking agent nadolol I. Comparison of absorption behaviour of nadolol with those of other β-blocking agents in rats.Chem. Pharm. Bull. 34:3362–3369 (1986).
F. S. Caruso, H. D. Doshan, P. H. Hernandez, R. Costello, W. Applin, and E. S. Neiss. Celiprolol: Pharmacokinetics and duration of pharmacodynamic activity.Br. J. Clin. Pract. 40:12–16 (1985).
J. Dreyfuss, D. L. Griffith, S. M. Singhvi, J. M. Shaw, and J. J. Ross Jr. Pharmacokinetics of nadolol, a beta-receptor antagonist: Administration of therapeutic single-and multiple-dosage regimens to hypertensive patients.J. Clin. Pharmacol. 19:712–720 (1979).
T. Yamagushi, C. Ikeda, and Y. Sekine. Intestinal absorption of a β-adrenergic blocking agent nadolol II. Mechanism of the inhibitory effect on the intestinal absorption of nadolol by sodium cholate in rats.Chem. Pharm. Bull. 34:3836–3843 (1986).
H. Lennernäs and C. G. Regårdh. Evidence for an interactions between the β-blocker pafenolol and bile salts in the intestinal lumen of the rat leading to dose-dependent oral absorption and double peaks in the plasma concentration-time profile.Pharm. Res. 10:879–883 (1993).
H. Ogata, N. Aoyagi, K. Kaniwa, T. Shibazaki, A. Ejima, N. Takasugi, E. Mafune, T. Hayashi, and K. Suwa. Bioavailability of nalidixic acid from uncoated tablets in humans-Part 1: Correlation with the dissolution rates of tabletsInt. J. Clin. Pharmacol. Ther. Toxicol. 22:175–183 (1984).
J. A. Clements, R. C. Heading, W. S. Nimmo, and L. F. Prescott. Kinetics of acetaminophen absorption and gastric emptying in man.Clin. Pharmacol. Ther. 24:420–431 (1978).
R. Oberle, and G. L. Amidon. The influence of variable gastric emptying and intestinal transit rates on the plasma level curve of cimetidine; an explanation for the double peak phenomenon.J. Pharmacokin. Biopharm. 15:529–544 (1987).
G. Mullersmann, V. P. Gotz, W. L. Russel, and H. Derendorf. Lack of clinically significantin vivo andin vitro interaction between ranitidine and sucralfate.J. Pharm. Sci. 75:995–998 (1986).
A. Grahnen, M. Hammarlund, and T. Lundquist. Implications of intraindividual variability in bioavailability studies of furosemide.Eur. J. Pharmacol. 27:595–602 (1984).
J. B. Dressman, R. R. Beradi, G. H. Elta, T. M. Gray, P. A. Montgomery, H. S. Lau, K. L. Pelekoudas, H. S. Szpunar, and J. G. Wagner. Absorption of flurbiprofen in the fed and fasted state.Pharm. Res. 9:901–907 (1992).
H. Lennernäs, L. Renberg, K. J. Hoffmann C. G. Regårdh. Presystemic elimination of the beta-blocker pafenolol in the rat after oral and intraperitoneal administration and identification of a main metabolite in both rats and humans.Drug Metab. Dispos. 21:435–440 (1993).
Y. Plusquellec, G. Campistron, S. Staveris, J. Barre, L. Jung, J. P. Tillement, and G. Houin. A double-peak phenomenon in the pharmacokinetics of veralipride after oral administration: a double-site model for drug absorption.J. Pharmacokin. Biopharm. 15:225–239 (1987).
B. Liebmann, R. Ding, E. Weber, E. Mutschler, H. Spahn-Langguth, and P. Langguth.Biopharm. Drug. Disp., submitted. (1994).
B. Voith, H. Spahn-Langguth, R. Paliege, H. Knauf, and E. Mutschler. Etacrynic acid: Evidence for active metabolites.Naunyn-Schmiedenberg's Arch. Pharmakol. 349(Suppl):594 (Abstract) (1994).
A. F. Parr, R. M. Beihn, R. M. Franz, G. J. Szpunar, and M. Jay. Correlation of ibuprofen bioavailability with gastrointestinal transit by scintigraphic monitoring of171Er-labeled sustained release tablets.Pharm. Res. 4:486–491 (1987).
H. Spahn-Langguth, G. Hahn, W. Moehrke, and P. Langguth. Variabilities and discontinuities in tranylcypromine input profiles following oral dosage.Eur. J. Pharm. Sci. 2:160 (1994).
C. Hartmann, M. Fröhlich, D. Krauss, H. Spahn, H. Knauf, and E. Mutschler. Comparative enantioselective pharmacokinetic studies of celiprolol in healthy volunteers and patients with impaired renal function.Eur. J. Clin. Pharmacol. 38:573–576 (1990).
J. Karlson, S. M. Kuo, J. Ziemniak, and P. Artursson. Transport of celiprolol across human intestinal epithelial (Caco-2) cells: Mediation of secretion by multiple transporters including P-glycoprotein.Br. J. Pharmacol. 110:1009–1016 (1993).
A. J. Boeckmann, L. B. Sheiner, and S. L. Beal.NONMEM Users Guide-Park V, Introductory Guide, NONMEM project group, C255, University of California at San Francisco, 1991.
C. F. Code, and J. A. Marlett. The interdigestive myo-electric complex of the stomach and small bowel of dogs.J. Physiol. 246:289–309 (1975).
P. Kerlin, and S. Phillips. Variability of motility of the ileum and jejunum in healthy volunteers.Gastroenterology 82:694–700 (1982).
P. Langguth, K. M. Lee, H. Spahn-Langguth, and G. L. Amidon. Variable gastric emptying and discontinuities in drug absorption profiles: Dependence of rates and extent of cimetidine absorption on motility phases and pH.Biopharm. Drug Disp. 15:719–746 (1994).
T. Yamagushi, C. Ikeda, and Y. Sekine. Intestinal absorption of a β-adrenergic blocking agent nadolol II. Mechanism of the inhibitory effect on the intestinal absorption of nadolol by sodium cholate in rats.Chem. Pharm. Bull. 34:3836–3843 (1986).
H. Lenneräs, and C. G. Regårdh. Evidence for an interaction between the β-blocker pafenolol and bile salts in the intestinal lumen of the rat leading to dose-dependent oral absorption and double peaks in the plasma concentration-time profile.Pharm. Res. 10:879–883 (1993).
Y. Hayashi, I. Lee, and G. L. Amidon. Effects of gastrointestinal motility and pH variation on oral absorption of cimetidine in dogs.Pharm. Res. 10(Suppl):S371 (1993).
S. M. Kou, B. R. Whitby, P. Artursson, and J. A. Ziemniak. The contribution of intestinal secretion to the dose-dependent absorption of celiprolol.Pharm. Res. 11:743–749 (1994).
U. Wetterich, E. Mutschler, H. Spahn-Langguth, and P. Langguth. Evidence for intestinal secretion of the β-adrenoceptor antagonist talinolol: Data from humans and studies with Caco-2 cells.Naunyn-Schmiedenberg's Arch. Pharmakol. (Suppl.) Abstract accepted.
Author information
Authors and Affiliations
Additional information
This work was supported by FDA-01462 and in part by the Dr. Robert Pfleger-Stiftung (Bamberg, Germany).
Rights and permissions
About this article
Cite this article
Lipka, E., Lee, ID., Langguth, P. et al. Celiprolol double-peak occurrence and gastric motility: Nonlinear mixed effects modeling of bioavailability data obtained in dogs. Journal of Pharmacokinetics and Biopharmaceutics 23, 267–286 (1995). https://doi.org/10.1007/BF02354285
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02354285