Abstract
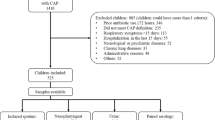
Viral and bacterial antigen and antibody assays were prospectively applied to study the microbial actiology of community-acquired pneumonia in 195 hospitalised children during a surveillance period of 12 months. A viral infection alone was indicated in 37 (19%), a bacterial infection alone in 30 (15%) and a mixed viral-bacterial infection in 32 (16%) patients. Thus, 46% of the 69 patients with viral infection and 52% of the 62 patients with bacterial infection had a mixed viral and bacterial aetiology. Respiratory syncytial virus (RSV) was identified in 52 patients andStreptococcus pneumoniae in 41 patients. The next common agents in order were non-classifiedHaemophilus influenzae (17 cases), adenoviruses (10 cases) andChlamydia species (8 cases). The diagnosis of an RSV infection was based on detecting viral antigen in nasopharyngeal secretions in 79% of the cases. Pneumococcal infections were in most cases identified by antibody assays; in 39% they were indicated by demonstrating pneumococcal antigen in acute phase serum. An alveolar infiltrate was present in 53 (27%) and an interstitial infiltrate in 108 (55%) of the 195 patients. The remaining 34 patients had probable pneumonia. C-reactive protein (CRP), erythrocyte sedimentation rate and total white blood cell count were elevated in 25%, 40% and 36% of the patients, respectively, CRP was more often elevated in patients with bacterial infection alone than in those with viral or mixed viral-bacterial infections. No other correlation was seen between the radiological or laboratory findings and serologically identified viral, bacterial or mixed viralbacterial infections. By using a comprehensive serological panel, the causative agent could be found in over 50% of patients with pneumonia. We conclude that RSV and pneumococcus are the two most common organisms causing pneumonia in children. Our results suggest that mixed viral-bacterial aetiology is common in lower respiratory tract infections affecting children.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- AOM:
-
acute otitis media
- CF:
-
complement fixation
- EIA:
-
enzyme immunoassay
- IF:
-
immunofluorescence
- NPS:
-
nasopharyngeal secretion
- PNC:
-
pneumococcal
- PV:
-
parainfluenza
- RSV:
-
respiratory syncytial virus
References
Aldous MB, Wang S, Foy HM, Grayston JT (1990)Chlamydia pneumoniae, strain TWAR, infection in Seattle children and their families, 1965–1979. In: Bowie et al (eds) Chlamydial Infections, Cambridge University Press
Arstila P, Halonen P (1988) Direct antigen detection. In: Lennette EH, Halonen P, Murphy FA (eds) Laboratory diagnosis of infectious disease; principles and practice, vol. II, viral, rickettsial, and chlamydial diseases. Springer, Berlin Heidelberg New York, pp 60–75
Chang M, Rodriquez W, Mohla C (1982) Chlamydia trachomatis in otitis media in children. Pediatr Infect Dis 1:95–97
Claesson BA, Trollfors B, Brolin J, et al (1989) Etiology community-acquired pneumonia in children based on antibody responses to bacterial and viral antigens. Pediatr Infect Dis J 8:856–862
Coch WM (1987) Bronchitis and pneumonia in ambulatory patients. Pediatr Infect Dis J 6:137–140
Degre M (1986) Interaction between viral and bacterial infections in the respiratory tract. Scand J Infect Dis 49:140–145
Halonen P, Obert G, Hierholzer JC (1985) Direct detection of viral antigens in respiratory infections by immunoassays: a four year experience and new development. In: Maza LM de la, Peterson EM (eds) Medical virology IV. Lawrence Erlbaum Associates Publishers, Hillsdale, pp 65–85
Hammerschlag M, Hammerschlag P, Russel EA (1980) The role of Chlamydia trachomatis in middle ear effusions in children. Pediatrics 66:615–617
Hawkes RA (1979) General principles underlying laboratory diagnosis of viral infections. In: Lennette EH, Schmidt NJ (eds) Diagnostic procedures for viral, rickettsial and chlamydial infections, 5th edn. American Public Health Association, Washington DC, pp 35–42
Hietala J, Uhari M, Tuokko H (1988) Antigen detection in the diagnosis of viral infections. Scand J Infect Dis 20:595–599
Hietala J, Uhari M, Tuokko H, Leinonen M (1989) Mixed viral and bacterial infections are common in children. Pediatr Infect Dis J 8:683–686
Isaacs D (1989) Problems in determining the etiology of community-acquired childhood pneumonia. Pediatr Infect Dis J 8:143–148
Jalonen E, Paton J, Koskela M, Kerttula Y, Leinonen M (1989) Measurement of antibody responses to pneumolysin —a promising diagnosis of pneumococcal pneumonia. J Infect 19:127–134
Kalin M, Lindberg AA (1983) Diagnosis of pneumococcal pneumonia: a comparison between microscopic examination of expectorates, antigen detection and cultural procedures. Scand J Infect Dis 15:247–255
Kalin M, Lindberg AA, Olausson EH (1982) Diagnosis of pneumococcal pneumonia by coagglutination and counterimmunoelectrophoresis of sputum samples. Eur J Clin Microbiol 1:91–96
Kanclercki K, Blomqvist S, Granström Möllby R (1988) Serum antibodies to pneumolysin in patients with pneumonia. J Clin Microbiol 26:96–100
Kerttula Y, Leinonen M, Koskela M, et al (1987) The aetiology of pneumonia. Application of bacterial serology and basic laboratory methods. J Infect 14:21–30
Korppi M, Halonen P, Kleemola M, Launiala K (1986) Viral findings in children under the age of two years with expiratory difficulties. Acta Paediatr Scand 75:457–464
Korppi M, Halonen P, Kleemola M, Launiala K (1988) The role of parainfluenza viruses in inspiratory difficulties in children. Acta Paediatr Scand 77:105–111
Korppi M, Leinonen M, Mäkelä PH, Launiala K (1989) Bacterial coinfection in children hospitalized with respiratory syncytial virus infections. Pediatr Infect Dis J 8:687–692
Korppi M, Leinonen M, Mäkelä PH, Launiala K (1990) Bacterial involvement in parainfluenza virus infection in children. Scand J Infect Dis 22:307–312
Korppi M, Leinonen M, Mäkelä PH, Launiala K (1991) Mixed infection is common in children with respiratory adenovirus infection. Acta Paediatr Scand 80:413–417
Koskela M (1987) Serum antibodies to pneumococcal C-polysaccharide in children: a response to acute pneumococcal otitis media or to vaccination. Pediatr Infect Dis J 6:519–526
Koskela M, Leinonen M, Luotonen J (1982) Serum antibody response to pneumococcal otitis media. Pediatr Infect Dis 1:245–252
Koskela M, Leinonen M, Häivä V-M, Timonen M, Mäkelä PH (1986) First and second dose antibody responses to pneumococcal polysaccaride vaccine in infants. Pediatr Infect Dis 5:45–50
Lancet: Editorial (1982) How does influenza pave the way for bacteria? Lancet I:485–486
Lehtomäki K, Leinonen M, Takala A, Hovi T, Herva E, Koskela M (1988) Etiological diagnosis of pneumonia in military conscripts by combined use of bacterial culture and serological methods. Eur J Clin Microbiol Infect Dis 7:348–354
Leinonen MK (1980) Detection of pneumococcal capsular polysaccharide antigens by latex agglutination, counterimmunoelectrophoresis, and radioimmunoassay in middle ear exudates in acute otitis media. J Clin Microbiol 11:135–140
Leinonen M, Luotonen J, Herva E, Valkonen K, Mäkelä PH (1981) Preliminary serologic evidence for a pathogenic role ofBranhamella catarrhalis. J Infect Dis 144:570–574
Leinonen M, Säkkinen A, Kalliokoski R, Luotonen J, Timonen M, Mäkelä PH (1986) Antibody response to 14-valent pneumococcal capsular polysaccharide vaccine in preschool age children. Pediatr Infect Dis 5:39–44
Loda FA, Collier AM, Glezen WP, Strangert K, Clyde WA, Denny FW (1975) Occurrence ofDiplococcus pneumoniae in the upper respiratory tract of children. J Pediatr 87:1087–1093
Meurman O, Ruuskanen O, Sarkkinen H, Hänninen P, Halonen P (1984) Immunoglobulin class-specific antibody response in respiratory syncytial virus infection measured by enzyme immunoassay. J Med Virol 14:67–72
Nohynek H, Eskola J, Laine E, Halonen P, Ruutu P, Saikku P, Kleemola M, Leinonen M (1991) The etiology of hospitaltreated acute lower respiratory tract infection in children. Am J Dis Child 145:618–622
Ogawa H, Hashigucki K, Kazuyama Y (1990) Isolation of chlamydia trachomatis from the middle ear aspirates of otitis media. Acta Otolaryngol (Stockholm) 110:105–109
Payne R (1984) Aetiology of pneumonia in children in Goroka Hospital, Papua New Guinea. Lancet II:537–541
Peter G (1988) The child with pneumonia: diagnostic and therapeutic considerations. Pediatr Infect Dis J 7:453–456
Puolakkainen M, Saikku P, Leinonen M, Nurminen M, Väänanen P, Mäkelä PH (1984) Chlamydial pneumonitis and its serodiagnosis in infants. J Infect Dis 149:598–604
Putto A, Ruuskanen O, Meurman O, et al (1986) C-reactive protein in the evaluation of febrile illness. Arch Dis Child 61:24–29
Ramsey BW, Marcuse EK, Foy HM et al (1986) Use of bactetial antigen detection in the diagnosis of pediatric lower respiratory tract infections. Pediatrics 78:1–9
Rusconi F, Rancilio L, Assael BM, et al (1988) Counterimmunoelectrophoresis and latex particle agglutination in the etiologic diagnosis of presumed bacterial pneumonia in pediatric patients. Pediatr Infect Dis J 7:781–785
Saikku P, Ruutu P, Leinonen M, Panelius J, Tupasi TE, Grayston JT (1988) Acute lower-respiratory-tract infection associated with chlamydial TWAR antibody in Filipino children. J Infect Dis 158:1095–1097
Sarkkinen HK, Halonen PE, Arstila PP, Salmi AA (1981) Detection of respiratory syncytial, parainfluenza type 2, and adenovirus antigens by radioimmunoassay and enzyme immunoassay in nasopharyngeal specimens from children with acute respiratory disease. J Clin Microbiol 13:256–265
Sarkkinen HK, Halonen PE, Salmi AA (1981) Detection of influenza A virus by radioimmunoassay and enzyme immunoassay from nasopharyngeal specimens. J Med Virol 17:213–220
Sarkkinen HK, Halonen PE, Salmi AA (1981) Type specific detection of parainfluenza viruses by enzyme immunoassay and radioimmunoassay in nasopharyngeal specimens of patients with acute respiratory disease. J Gen Virol 56:49–57
Schultzer S, Coyle PK, Belman AL, Golighty MG, Drulle J (1990) Sequestration of antibodies toBorrelia burgdorferi in immune complexes in seronegative Lyme disease. Lancet 335: 312–315
Silverman M, Stratton D, Dialo A, et al (1977) Diagnosis of acute bacterial pneumonia in Nigerian Children. Arch Dis Child 52:925–931
Similä S, Linna O, Lanning P, Heikkinen E, Ala-Houhala M (1981) Chronic lung damage caused by adenovirus type 7: a ten-year follow-up study. Chest 80:127–131
Teele DW, Pelton SI, Grant MJA (1975) Bacteremia in febrile children under 2 years of age: Results of cultures of blood in 600 consecutive children seen in a “walk-in” clinic. J Pediatr 87:227–230
Trottier S, Stenberg K, Svanborg-Eden C (1989) Turnover of nontypableHaemophilus influenzae in the nasopharynges of healthy children. J Clin Microbiol 27:1275–1279
Turner RB, Hayden FG, Hendley JO (1983) Counterimmuno-electrophoresis of urine for diagnosis of bacterial pneumonia in pediatric patients. Pediatrics 71:780–783
Ukkonen P, Hovi T, Bonsdorff CH, Saikku P, Penttinen K (1984) Age-specific prevalence of complement-fixing antibodies to sixteen viral antigens: a computer analysis of 58.500 patients covering a period of eight years. J Med Virol 13:131–148
Wang SP, Grayston JT (1970) Immunologic relationship between genital TRIC, lymphogranuloma venereum, and related organisms in a new microtiter immuno-fluorence test. Am J Ophtalmol 70:367–374
Waris M, Ziegler T, Kiviranta M, Ruuskanen O (1990) Rapid detection of respiratory syncytial virus and influenza A virus in cell cultures by immunoperoxidase staining with monoclonal antibodies. J Clin Microbiol 28:1159–1162
Weinberg GA, Ghafoor A, Ishag Z, et al (1989) Clonal analysis ofHaemophilus influenzae isolated from children from Pakistan with lower respiratory tract infections. J Infect Dis 160:634–643
Yamasaki T, Nakada H, Sakurai N, Kuo CC, Wang SP, Grayston JT (1990) Transmission ofChlamydia pneumoniae in young children in Japanese family. J Infect Dis 162:1390–1392
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Korppi, M., Heiskanen-Kosma, T., Jalonen, E. et al. Aetiology of community-acquired pneumonia in children treated in hospital. Eur J Pediatr 152, 24–30 (1993). https://doi.org/10.1007/BF02072512
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02072512