Abstract
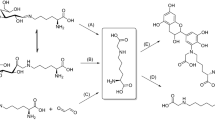
Deoxyosones are established key-intermediates in Maillard processes. Due to their dicarbonyl structure, they undergo condensation to form heterocyclic compounds with guanidine derivatives. In biological systems, guanidino functions are present in protein-bound arginine moieties as well as in creatine. The reactivity of such structures towards 3-deoxypentosulose is investigated withN-methyl- andN,N-dimethylguanidine as model substrates. Two diastereoisomers each are isolated from both reactions; they have been characterized unequivocally, respectively, as 4-(2,3-dihydroxypropyl)-2-N-methylamino-2-imidazolin-5-one and 4-hydroxy-5-(2,3-dihydroxypropyl)-2-(N,N-dimethylamino)-5H-imidazole. In aqueous medium as well as in the crystalline state, both diastereoisomer pairs exist in different tautomeric forms.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ledl F, Schleicher E (1990) Angew Chem 102: 597–626
Ledl F, Schleicher E (1990) Angew Chem Int Ed Engl 29: 565–594
Heyns K, Noack H (1962) Chem Ber 95: 720–227
Kramhöller B (1992) PhD thesis, Universität Stuttgart
Glomb M (1992) PhD thesis, Universität Stuttgart
Nishimura T, Kitajima K (1979) J Org Chem 44: 818–824
Duerksen-Hughes PJ, Williamson MM, Wilkinson KD (1989) Biochemistry 28: 8530–8536
Thornalley PJ (1994) Amino Acids 6: 15–23
Henle T, Walter AW, Haeßner R, Klostermeyer H (1994) Z Lebensm Unters Forsch 199: 55–58
Hayase F, Konishi Y, Hinuma H, Kato H In: Labuza TP, Reineccius GA, Monniev VM, O'Brien J, Baynes JW (eds) Maillard reactions in chemistry, food and health. The Royal Society of Chemistry, Cambridge, 1994, pp 347–351
Konishi Y, Hayase F, Kato H (1994) Biosci Biotech Biochem 58: 1953–1955
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sopio, R., Lederer, M. Reaction of 3-deoxypentosulose withN-methyl- andN,N-dimethylguanidine as model reagents for protein-bound arginine and for creatine. Z Lebensm Unters Forch 201, 381–386 (1995). https://doi.org/10.1007/BF01192738
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01192738