Abstract
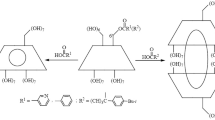
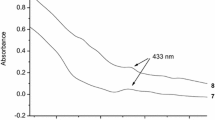
6N(N′-formyl-D-phenylalanyl)-deoxyamino-β-cyclodextrin and 6N(N′-formyl-L-phenylal-anyl)-deoxyamino-β-cyclodextrin (I andII) were prepared. These new hosts formed ‘intramolecular host-guest complexes’ and included naphthyl derivatives preferentially with a stoichiometry of 1:1. The inclusion behavior was different between I and II because the cavity shape formed with the CD cavity and the phenylalanine moiety of I was different from that of II.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
A. Croft and R. A. Bartsch:Tetrahedron 39, 1417 (1983).
I. Tabushi, Y. Kuroda and T. Mizutani:J. Am. Chem. Soc. 108, 4514 (l986).
Y. Yamamaoto and Y. Inoue:J. Carbohydr. Chem. 8, 29 (1989).
A. Ueno, F. Moriwaki, T. Osa, T. Ikeda, F. Toda and K. Hattori:Bull. Chem. Soc. Jpn. 59, 3109 (1986).
K. Takahashi, K. Hattori and F. Toda:Tetrahedron Lett. 25, 3331 (1984).
L. D. Melton and K. N. Slessor:Carbohydr. Res. 18, 29 (1971).
H. Kondo, H. Nakatani, and K. Hiromi:J. Biochem. (Tokyo) 79, 393 (1976).
Y. Inoue, H. Hoshi, M. Sakurai, and R. Chujo:J. Am. Chem. Soc. 107, 2319 (1985).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Takahashi, K., Ohtsuka, Y., Nakada, S. et al. Syntheses of 6N(N′-formyl-D-phenylalanyl)-deoxyamino-β-cyclodextrin and 6N(N′-formyl-L-phenylalanyl)deoxyamino-β-cyclodextrin and their inclusion behavior. J Incl Phenom Macrocycl Chem 10, 63–68 (1991). https://doi.org/10.1007/BF01041640
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01041640