Abstract
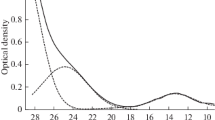
The effect of alkali metal cations in molten chloride baths on the behaviour of Ti(III) in these melts has been studied. With caesium chloride the percentage of total dissolved titanium present as Ti(II) after 72 hours at 700° C is 0.1%, whereas in lithium chloride it is 2%. In the presence of excess titanium metal the proportion of Ti(II) is always much higher. In LiCl it depends strongly on the area of titanium exposed and may reach 85%. In CsCl, on the other hand, this factor has no effect since a fine dispersion of powdery titanium appears at the very beginning of the reaction. The amount of Ti(II) remains close to 50%. In all the baths studied, the electrochemical reduction of Ti(III) at 700° C occurs in two steps: Ti(III) + e → Ti(II)E 1/2 = − 1.6V versus Cl−/Cl2 Ti(II) + 2e → Ti(O)E 1/2=−2.1V versus Cl−/Cl2. These results are different from those previously obtained in CsCl-LiCl (40–60 mol%) at 400° C, where reduction is a one-step process.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. Nardin and G. Lorthioir,J. Less-Common Metals 56 (1977) 269.
E. Chassaing, F. Basile and G. Lorthioir,ibid 68 (1979) 153.
R. MacMullin,J. Electrochem. Soc. 123 (1976) 359C.
P. R. Juckniess and D. R. Johnson (Dow Chemical Co.) US Patent 4118 291 (1978).
B. F. Markov and B. P. Podafa,Ukr. Khim. Z. 36 (1970) 881.
S. N. Flengas and P. Pint,Can. Metal. Quart. 8 (1969) 151.
S. Mellgren and W. Opie,J. Metals 9 (1957) 266.
F. R. Clayton, G. Mamantov and D. L. Manning,J. Electrochem. Soc. 120 (1973) 1193.
E. Chassaing, F. Basile and G. Lorthioir,J. Appl. Electrochem. 11 (1981) 193.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chassaing, E., Basile, F. & Lorthioir, G. Study of Ti(III) solutions in various molten alkali chlorides. I. Chemical and electrochemical investigation. J Appl Electrochem 11, 187–191 (1981). https://doi.org/10.1007/BF00610979
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00610979